Neuroendocrine neoplasms (NENs) first described as “karzinoide” by Dr. Oberndorfer in 1907, comprise a family of heterogeneous tumors which can range from well differentiated neuroendocrine tumors (NETs) to poorly differentiated neuroendocrine carcinomas (NECs). NENs emerge from the diffuse endocrine systemand therefore can be found throughout the body, but they most commonly occur in the gastrointestinal tract with a peak incidence around the 5th and 6th decade of life. Based on the latest Survival Epidemiology and End Results data, incidence of NENs is on the rise with almost 7 cases per 100,000 persons in the United States. NENs can be alternatively classified as functional or non-functional based on the release of specific hormones by the tumor cells (e.g., serotonin, vasoactive intestinal peptide, insulin, gastrin, somatostatin, glucagon) and the subsequent development of various secretory syndromes.
- NET
- neuroendocrine
- biomarkers
- small intestine NETs
- carcinoid heart disease
- NETest
1. Introduction
2. Biomarkers in the Diagnosis and Management of Small Intestine NETs (SI-NETs)
Biomarkers used in the setting of SI-NETS are extrapolated from the general field of GEP-NETs and other NETs and are divided in two main categories: mono-analyte and multi-analyte biomarkers [10] ( Figure 1 ). The former are specific molecules (protein, gene, RNA) whose levels in the blood or other bodily fluids can be used to predict the presence of disease as well as assess its degree of severity. This is true for traditional biomarkers like chromogranin A and 5-HIAA, as well as biomarkers used in other malignancies (e.g., CEA for adenocarcinoma). The main disadvantages of the single-analyte method include the high heterogeneity of NETs, the lack of secretory products in high proportion of NETs as well as the lack of standardization in measurement of biomarker concentrations, which subsequently lead to a range of sensitivities and specificities across different assays used [11]. To address the inherent limitations of mono-analyte assays, there has been a gradual shift towards the development of tests that are able to identify multiple genomic regulators (mRNA, miRNA, ctDNA) associated with a specific disease process, with NETest being the most heavily investigated multi-analyte assay in the field of NETs [12]. A brief overview of SI-NET biomarkers is presented on Table 1 .
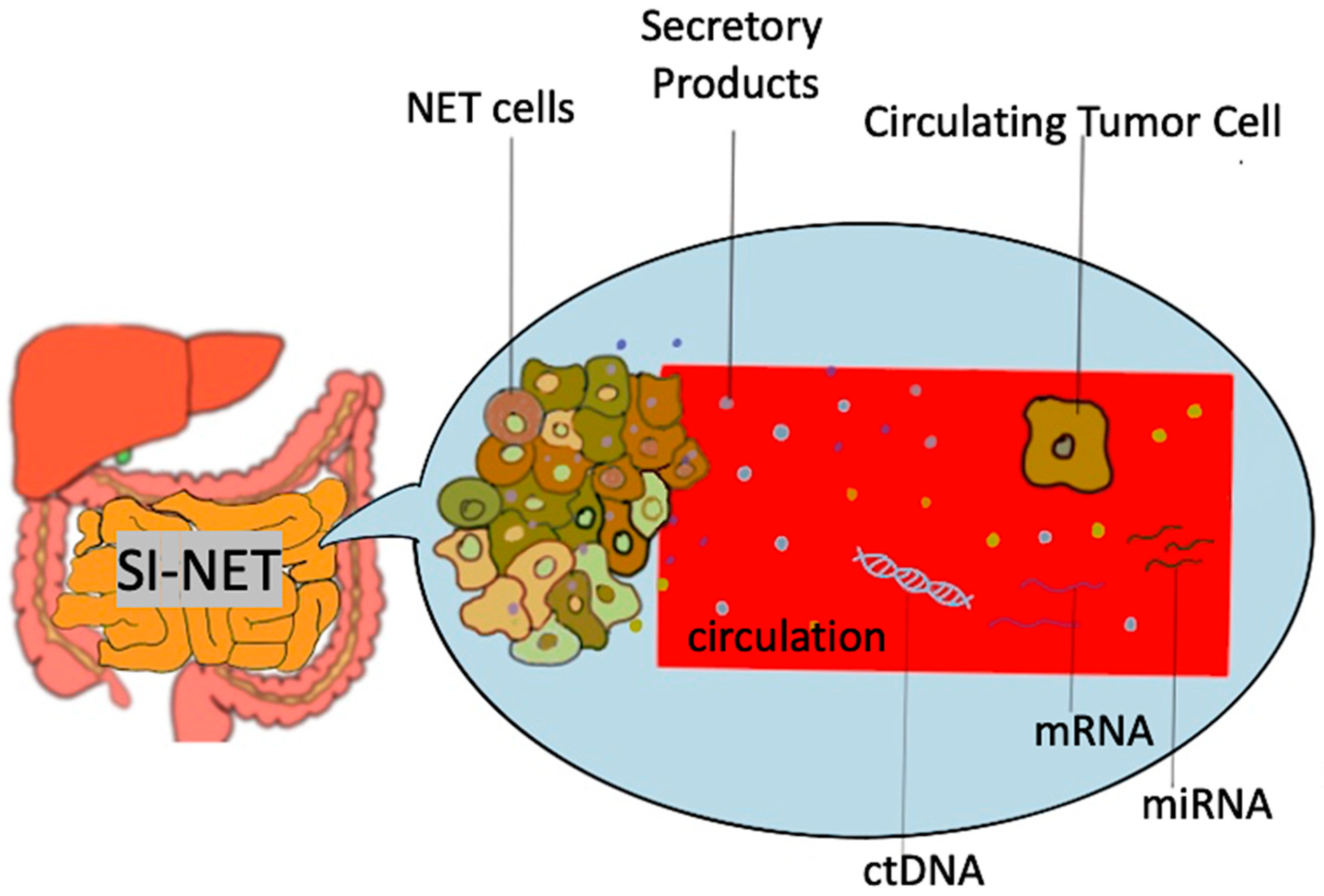
Figure 1. Schematic representation of biomarkers used in SI-NET. Secretory products include biomarkers such as Chromogranin A, 5-hydroxy-indole acetic acid, pancreastatin etc.; mRNA, messenger RNA, miRNA, microRNA, SI-NET, small intestine neuroendocrine tumor; ctDNA, circulating tumor DNA.
| Biomarker | Type | Utility in SI-NETs | Comments | ||
|---|---|---|---|---|---|
| Diagnostic | Predictive | Prognostic | |||
| CgA | Monoanalyte | + | − | + | Lab result variability, impairment by multiple diseases and medications. Diagnostic accuracy: 40–70% [5]. |
| 5-HIAA | Monoanalyte | + | − | + | Plasma 5HIAA comparable to 24 h urine 5HIAA. Useful diagnostic and prognostic aid in SI-NETs. |
| CgB | Monoanalyte | + | − | + | Like CgA but not impaired by renal disease and PPI use. |
| Pancreastatin | Monoanalyte | + | + | + | Diagnostic accuracy: 40–60% [5]. Prognostic accuracy may be superior to CgA. |
| Neuro-specific enolase | Monoanalyte | − | − | + | Minimal utility in SI-NET. |
| Neurokinin A | Monoanalyte | − | − | + | Minimal utility in SI-NET. |
| NETest | Multianalyte | + | + | + | Great diagnostic, predictive and prognostic value. Cost and availability remain a concern. |
| CTCs | Multianalyte | − | + | − | Not widely available, still experimental. No diagnostic value while prognostic value is unclear. |
| MiRNA | Multianalyte | + | + | + | Promising but not widely implemented. Limited available data. |
3. Conclusions
This entry is adapted from the peer-reviewed paper 10.3390/biology10100950
References
- Schottenfeld, D.; Beebe-Dimmer, J.L.; Vigneau, F.D. The Epidemiology and Pathogenesis of Neoplasia in the Small Intestine. Ann. Epidemiol. 2009, 19, 58–69.
- Modlin, I.M.; Kidd, M.; Latich, I.; Zikusoka, M.N.; Shapiro, M.D. Current Status of Gastrointestinal Carcinoids. Gastroenterol. 2005, 128, 1717–1751.
- Larouche, V.; Akirov, A.; AlShehri, S.; Ezzat, S. Management of Small Bowel Neuroendocrine Tumors. Cancers 2019, 11, 1395.
- Maxwell, E.J.; Howe, J.R. Imaging in neuroendocrine tumors: An update for the clinician. Int. J. Endocr. Oncol. 2015, 2, 159–168.
- Clift, A.K.; Kidd, M.; Bodei, L.; Toumpanakis, C.; Baum, R.P.; Oberg, K.; Modlin, I.M.; Frilling, A. Neuroendocrine Neoplasms of the Small Bowel and Pancreas. Neuroendocrinology 2020, 110, 444–476.
- Clement, D.; Ramage, J.; Srirajaskanthan, R. Update on Pathophysiology, Treatment, and Complications of Carcinoid Syndrome. J. Oncol. 2020, 2020, 8341426.
- Ratnayake, G.; Toumpanakis, C. Carcinoid syndrome and its sequelae. Curr. Opin. Endocr. Metab. Res. 2021, 18, 111–117.
- Davar, J.; Connolly, H.M.; Caplin, M.E.; Pavel, M.; Zacks, J.; Bhattacharyya, S.; Cuthbertson, D.J.; Dobson, R.; Grozinsky-Glasberg, S.; Steeds, R.P.; et al. Diagnosing and Managing Carcinoid Heart Disease in Patients with Neuroendocrine Tumors: An Expert Statement. J. Am. Coll. Cardiol. 2017, 69, 1288–1304.
- Laskaratos, F.-M.; Davar, J.; Toumpanakis, C. Carcinoid Heart Disease: A Review. Curr. Oncol. Rep. 2021, 23, 1–8.
- Modlin, I.M.; Oberg, K.; Taylor, A.; Drozdov, I.; Bodei, L.; Kidd, M. Neuroendocrine tumor biomarkers: Current status and perspec-tives. Neuroendocrinology 2014, 100, 265–277.
- Stridsberg, M.; Eriksson, B.; Oberg, K.; Janson, E.T. A comparison between three commercial kits for chromogranin A measurements. J. Endocrinol. 2003, 177, 337–341.
- Modlin, I.M.; Kidd, M.; Bodei, L.; Malczewska, A.; Kitz, A.; Oberg, K. Circulating biomarkers of gastroenteropancreatic and lung neuroendocrine neoplasms: “The times they are a changin. ” Curr. Opin. Endocr. Metab. Res. 2021, 18, 243–253.
