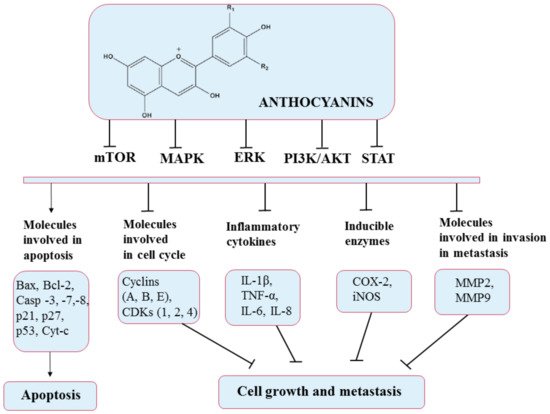
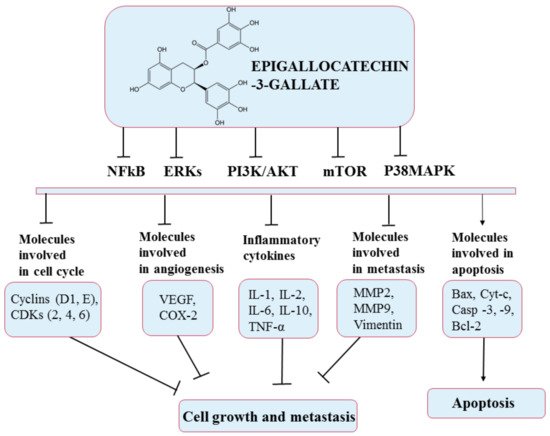
2. The Need to Encapsulate Polyphenols in Liposomes
Polyphenols have recently come to the attention of researchers from various fields such as pharmaceuticals, cosmetics, and food, due to their potential for prevention and protection against numerous diseases and their beneficial properties on an individual’s health. Many beneficial effects of polyphenols are attributed to their antioxidant, anti-inflammatory, antimicrobial, antimutagenic, anticarcinogenic, and digestion-stimulating properties [
5]. Over the years, the effects of polyphenols have been intensively studied in preventing a wide range of conditions, such as diabetes, obesity, cardiovascular disease, neurodegenerative disorders, and cancer [
6].
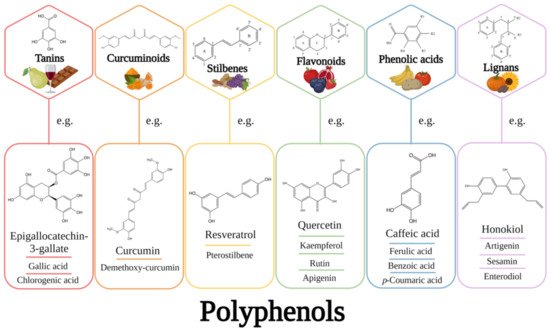
In everyday life, most people pursue a diet rich in polyphenols sourcing from various foods that contain them, such as green vegetables, fruits, soybeans, tea, beer, coffee, or red wine. However, there may be variations in their quantities consumed from one country to another in terms of their consumption. For example, in the United States, Spain, and Australia, estimated consumptions of flavonoids are about 190, 313, and 454 mg/day, respectively [
7]. However, because foods contain a variable amount of flavonoids due to their growing environment, storage, processing, or cooking, such data are generally considered approximations of food contents [
8].
Because vegetables, fruits, coffee, red wine, and tea are all rich in polyphenols, current research focuses on identifying those responsible for a particular pharmacological or chemopreventive effect, making considerable efforts to elucidate molecular mechanisms of action [
9].
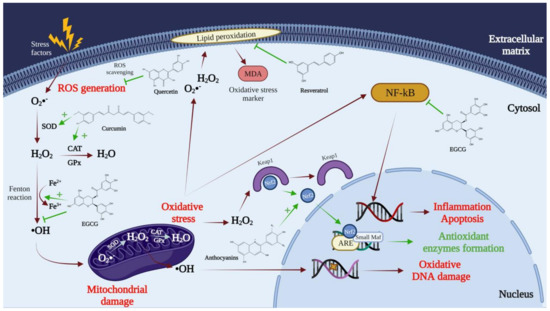
Most of the pathological conditions mentioned are related to oxidative stress caused by reactive oxygen and nitrogen species (
Figure 2). Thus, in particular, polyphenols represent the primary antioxidant agent in fruits, with superior efficacy to vitamin C [
10]. It has been determined that the substances with the most substantial antioxidant effect, which can neutralize free radicals by donating an electron or a hydrogen atom, are polyphenols. Therefore, polyphenols inhibit the generation of free radicals while reducing the oxidation rate by inhibiting the formation of active species and free-radical precursors or by deactivating them. Polyphenols usually perform as direct radical eliminators in lipid peroxidation chain reactions (chain breakers). They transfer an electron to the free radical, neutralizing it; after that, they become stable radicals (less reactive), and the chain reactions terminate [
11]. In addition to the functions presented above, polyphenols also act as metal chelators. They can cause the chelation of transition metals such as Fe
2+, and this process will directly reduce the rate of the Fenton reaction and prevent oxidation caused by highly reactive hydroxyl radicals [
12,
13]. It is also known that polyphenols can act as co-antioxidants and are part of the regeneration process of essential vitamins [
14]. These compounds can induce antioxidant enzymes such as glutathione peroxidase, superoxide dismutase, and catalase that decompose hydroperoxides, hydrogen peroxide, and superoxide anions. In contrast, they can also inhibit the expression of enzymes such as xanthine oxidase [
15].
Figure 2. The mechanisms via which polyphenols act on free radicals, reducing oxidative stress. SOD, superoxide dismutase; CAT, catalase; GPx, glutathione peroxidase; MDA, malondialdehyde; Nrf2, nuclear factor erythroid 2-related factor 2; ARE, antioxidant response elements; Small Maf, musculoaponeurotic fibrosarcoma proteins; Keap1, Kelch-like ECH-associated protein 1; NF-kB, nuclear factor kappa-light-chain-enhancer of activated B cells.
Through the mechanisms presented above, polyphenols are involved in critical activities that reduce oxidative stress. However, their roles at the cellular level are believed to be much more complicated, requiring further studies to elucidate them. Some polyphenols such as flavonoids are absorbed in the gastrointestinal tract, but their plasma concentration is low (1 µmol/L). The leading cause of this is the rapid metabolism of human tissues [
11].
According to research on polyphenols, they have been shown to have the capacity to diminish inflammation by inhibiting edema, suppress the development of tumors, possess proapoptotic and anti-angiogenic properties, modulate the immune system, improve capillary resistance by acting on blood vessel components, protect the cardiovascular system, and limit weight gain [
16].
Even though polyphenols have health benefits, the amount of polyphenols that can be administered orally is not enough to reach the concentration needed for systemic therapies to be effective. Characteristics such as low water solubility, poor absorption, and rapid metabolism play a role in decreasing the oral bioavailability of polyphenols [
9]. Although there are several definitions of the term bioavailability, the best of these expressions is the part of an ingested compound that can reach the systemic circulation and be distributed to the various targeted tissues where it can exert its biological action [
17]. Thus, only a small amount of the molecules administered orally are absorbed due to insufficient gastric residence time, low permeability, or low solubility. The absorption of polyphenols from food is always influenced by their chemical structure (degree of glycosylation or acylation), ability to conjugate with other phenols, molecular size, degree of polymerization, and water solubility. Therefore, the bioavailability of these compounds is low when they are administered orally, due to their poor solubility and rapid metabolism (polyphenols are metabolized extensively in tissues and by the colonic microbiota), as well as their membrane permeability and incompatibility with a process of passive diffusion [
18].
Another disadvantage of polyphenols is that they are very unstable and sensitive to environmental factors such as temperature, light, oxygen, acidic pH, and the enzymatic activity in the digestive system. These characteristics can decrease the concentration of polyphenols and even cause a total or partial loss of bioactivity [
2]. Thus, in order to have an efficient oral bioavailability, the hydrophilic part of the natural compound (which ensures its dissolution in the gastrointestinal fluids) must be in a balance with the lipophilic part (which has a role in crossing the lipid biomembranes) [
19].
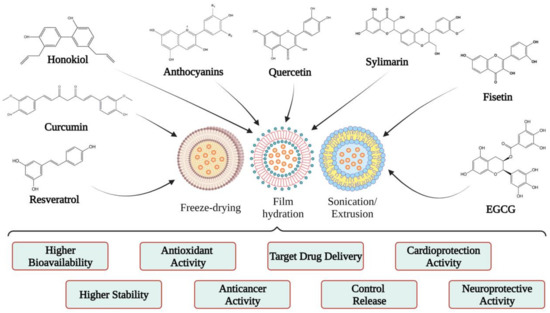
Over the years, attempts have been made to remedy these deficiencies by using various drug delivery systems to improve their bioavailability and therapeutic efficacy [
9]. Among the approaches that were evaluated, formulation with cyclodextrins [
20], simple emulsions, gels, and lipid nanocapsules [
21], nanoemulsion [
22], or liposomes [
23,
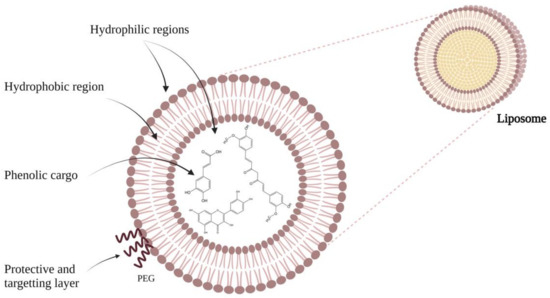
24] can be listed. Thus, researchers have managed to produce liposomes in which both hydrophilic and lipophilic substances can be incorporated, with high encapsulation efficiency and controlled drug release [
25]. An advantage of these compounds is their ability to change the membrane’s fluidity, thus achieving easier distribution of plant extracts to the target site. At the same time, the extract has a soluble character, thereby readily determining its location in the structure of liposomes since hydrophilic extracts are encapsulated in the aqueous phase, and amphiphilic and lipophilic compounds are found in the lipid layer of the liposome to reduce a material loss [
26,
27].
In addition to studies based on polyphenols as therapeutic agents in new therapies and with the development of drug delivery systems, the role of these compounds extends beyond therapeutic agents. They can also be used as primary component modules in drug delivery systems that are innovative. Polyphenols are not only incorporated in drug delivery systems to treat various illnesses, but also frequently employed as fundamental components of novel drug delivery systems due to their significant biological activity. Thus, considering the low toxicity and availability of natural substances, the production of novel drug delivery systems takes advantage of the physical and chemical characteristics of naturally active substances to achieve the design and assembly of drug delivery systems [
28].
Polyphenols’ intrinsic amphiphilic characteristic is, thus, critical for their use as functional components in novel drug delivery systems. Specifically, the hydroxy groups of polyphenols (as a hydrophilic component) are responsible for this, acting as donors or acceptors of hydrogen bonds, thus playing a role in the interactions between polyphenols and a wide variety of bioactive substances or carriers. In contrast, the aromatic benzene unit of polyphenols (as the hydrophobic component) allows other materials to be hydrophobic, which aids in the development of drug delivery methods [
28].
Therefore, due to the chemical structure of polyphenols, they can be combined with a wide variety of materials, such as metals, proteins, polymers, and small molecular compounds through hydrogen bonds, covalent bonds, metal coordination bonding, π–π stacking, and hydrophobic and electrostatic interactions. Polyphenols can not only successfully facilitate drug loading and delivery when combined with other bioactive materials or small molecules, but also safeguard the effective components and nanostructure of drug delivery systems [
28].