Aging is a biological process that occurs under normal conditions and in several chronic degenerative diseases. Bioactive natural peptides have been shown to improve the effects of aging in cell and animal models and in clinical trials. However, few reports delve into the enormous diversity of peptides from marine organisms.
- antiaging
- underused marine organism
- peptides
- mechanism
1. Introduction
In recent history, human life expectancy has continuously risen, and the proportion of the elderly population has sharply increased with the improvement of medical technology. Dysfunction of normal biological processes accompanies aging, and the acceleration of molecular damage and dysfunction in cells, tissues, and organs eventually leads to aging-related diseases, such as cardiovascular diseases, nutritional metabolic diseases, and cognitive disorders [1,2]. However, the aging process is adjustable, albeit irreversible, and scientific approaches can regulate the speed of aging and promote good health [3]. Therefore, development of efficient strategies to manage aging is critical for community health. In recent years, a growing body of work has aimed to test several hypotheses related to the molecular mechanisms that play critical roles in determining longevity. Among them, reactive oxygen species (ROS) inducing oxidative stress is considered one of the primary contributors to aging [4]. Hence, exploration of substances with antioxidant activity is an important strategy for the management of aging [5,6]. Many kinds of anti-aging drugs have been synthesized and tested in clinical trials, but many obstacles prevent drug development, including the adverse reactions of the human body to synthesized drugs. Therefore, natural active substances with high efficiency and low side effects are important alternatives for combating aging [6].
Recently, natural bioactive peptides have attracted special attention. Biologically active peptides with anticancer, anticoagulant, antidiabetic, antihypertensive, antimicrobial, antioxidant, and cholesterol-lowering properties have been reported in the biomedicine and pharmaceutical biotechnology literature [7]. Some of these compounds also exhibit notable antiaging activity in vitro or in vivo. Presently, potential bioactive peptides are mainly isolated and identified in different food products like milk, soy, rice, meat, fish, lobsters, crabs, and shrimp [8]. Some bioactive peptides are extremely safe, easily absorbed, and have no toxic side effects [9,10]. Moreover, because of their hypo-allergenicity characteristics, bioactive peptides have been explored as a method to treat patients with food allergies [11]. Therefore, bioactive peptides should be considered as excellent alternatives for anti-aging treatment.
Marine organisms contain high-quality functional protein or peptides with diverse molecular structures, and are an important source of new compounds, especially for China with its 18,000 km coastline, compared to terrestrial resources [12]. Presently, excluding various fish that are widely used as food materials, there is only limited use of marine protein resources from other species, such as sea cucumber, sea urchin, mussels, and several kinds of algae. Some marine bioactive peptides regulate free radical homeostasis in vitro and in vivo, and can have antiaging effects in cell and animal models and in human clinical trials [13,14,15,16]. Marine natural bioactive peptides have also been used in cosmeceutical skin products as antiaging agents [17,18,19].
2. Antiaging Activity of Peptides from the Ocean
ROS are highly reactive substances, and overproduction of ROS can result in DNA mutation, lipid, and protein dysfunction, and, ultimately, aging [20]. Thus, the main protective strategy to slow aging is to scavenge the excessive ROS by providing antioxidant agents, as well restoring the antioxidant defense system in cells. In the human body, the antioxidant defense system consists mainly of antioxidant peptides, including carnosine, glutathione, and antioxidant enzymes, including superoxide dismutase (SOD), glutathione oxidase (GHS-Px) and catalase (CAT).
3. The Mechanisms Underlying Antiaging Activity
Marine antiaging peptides have been shown to act in several different ways, including scavenging free radical capacities in vitro, inhibiting cell apoptosis, prolonging the life span of fruit flies and nematodes, and ameliorating D-galactose levels that induce aging in mice, and have shown promise as antiaging agents in human clinical trials.
Antioxidant enzymes are an important target of peptides in cells and organisms. The underlying mechanisms involved include regulation of the classical antioxidant gene Klotho and the Keap1/nuclear fac-tor erythroid 2-related factor 2- antioxidant responsive element (Nrf2-ARE) signaling pathway. In addition, marine peptides can regulate DAF-16/FOXO-SOD-3 expression, i.e., a forkhead transcription factor/forkhead transcription factors of the O class, which can enhance antioxidant enzyme activity.
The antiaging properties of the Leu-Cys-Gly-Glu-Cys peptide have been verified in healthy mouse models. After treatment with the peptides, mice display a healthier status based on a series of body indices, such as increased body weight and decreased liver and brain indices. Based on the analysis of liver and serum samples, the peptide was found to increase the activity of serum GSH-Px and SOD and decrease levels of MDA. Additionally, the transcription levels of Keap1 and Nrf2 were significantly downregulated in the liver and brain. These results confirmed that marine bioactive peptides can be used as potential regulators of aging in cells and mice, extending the earlier findings that showed it can scavenge free radicals in vitro. These results also highlight the importance of the Keap1/Nrf2-ARE pathway in antiaging [52].
As summarized in Table 1 , marine antioxidant peptides have potential as antiaging agents because they can protect cells from oxidative stress by directly scavenging excess free radicals. In addition, marine peptides can inhibit apoptosis via two pathways: the p53-Bax/Bcl-2 (BCL2-associated X protein/B lymphoblastic cells) pathway and the Mammalian target of rapamycin complex 1 (mTORC1)-mTOR-Bcl/Bax pathway.
Table 1. Marine antiaging peptides and their free radical scavenging activity in vitro.
| Source | Sequences | Activities (EC50) | Ref. |
|---|---|---|---|
| Microorganism | |||
| Chlorella vulgaris | favourzyme hydrolysates | Superoxide3 (0.323 mg/mL), Hydroxyl2 (0.139 mg/mL) |
[25] |
| Spirulina sp. | Thr-Met-Glu-Pro-Gly-Lys-Pro | Inhibition of ROS production | [26] |
| Dunaliella salina | Ile-Leu-Thr-Lys-Ala-Ala-Ile-Glu-Gly-Lys Ile-Ile-Tyr-Phe-Gln-Gly-Lys Asn-Asp-Pro-Ser-Thr-Val-Lys Thr-Val-Arg-Pro-Pro-Gln-Arg |
DPPH1 | [27] |
| Penicillium brevicompactum | N-cinnamoyl tripeptide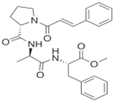 |
Hydroxyl2 (equivalent to that of quercetin at 0.1 mM) | [28] |
| Kocuria marina | Phe-Glu, Asp-Ile, Ser-Ser-Gln, Leu-Glu | DPPH1 (0.24 mg/mL) | [29] |
| Marine invertebrates | |||
| Neptunea arthritica cumingii | Tyr-Ser-Gln-Leu-Glu-Asn-Glu-Phe-Asp-Arg | DPPH1 (0.77 mM) | [30] |
| Tyr-Ile-Ala-Glu-Asp-Ala-Glu-Arg | DPPH1 (1.04 mM) | ||
| Tergillarca granosa | Glu-Met-Gly-Pro-Ala | DPPH1 (0.53 ± 0.02 mg/mL), Hydroxyl2 (0.47 ± 0.03 mg/mL), Superoxide3 (0.75 ± 0.04 mg/mL), ABTS4(0.96 ± 0.08 mg/mL), Inhibition of lipid peroxidation | [31] |
| Trp-Pro-Pro-Asp | DPPH1 (0.36 ± 0.02 mg/mL), Hydroxyl1 (0.38 ± 0.04 mg/mL), Superoxide3 (0.46 ± 0.05 mg/mL), ABTS4 (0.54 ± 0.03 mg/mL), Inhibition of lipid peroxidation | ||
| Brachionus rotundiformis | Leu-Leu-Gly-Pro-Gly-Leu-Thr-Asn-His-Ala, | DPPH1 (189.8 µM) | [32] |
| Asp-Leu-Gly-Leu-Gly-Leu-Pro-Gly-Ala-His | DPPH1 (167.7 µM) | ||
| Fish | |||
| Muscle of Scomberomorous niphonius | Pro-Glu-Leu-Asp-Trp | DPPH1 (1.53 mg/mL), Hydroxyl2 (1.12 mg/mL), Superoxide2 (0.85 mg/mL), Inhibition of lipid peroxidation, Protection of plasmid DNA | [33] |
| Trp-Pro-Asp-His-Trp | DPPH1 (0.70 mg/mL). Hydroxyl2 (0.38 mg/mL) Superoxide3 (0.49 mg/mL). Inhibition of lipid peroxidation, Protect plasmid DNA. | ||
| Phe-Gly-Tyr-Asp-Trp-Trp | DPPH1 (0.53 mg/mL), Hydroxyl2 (0.26 mg/mL), Superoxide3 (0.34 mg/mL). Inhibition of lipid peroxidation, | ||
| Tyr-Leu-His-Phe-Trp | DPPH1 (0.97 mg/mL), Hydroxyl2 (0.67 mg/mL), Superoxide3 (1.37 mg/mL), Inhibit lipid peroxidation. | ||
| Skin of Scomberomorous niphonius | Pro-Phe-Gly-Pro-Asp | DPPH1 (0.80 mg/mL), Hydroxyl2 (0.81 mg/mL), Superoxide3 (0.91 mg/mL, ABTS4 (0.86 mg/mL), FRAP and Inhibition of lipid peroxidation | [34] |
| Pro-Tyr-Gly-Ala-Lys-Gly | DPPH1 (3.02 mg/mL), Hydroxyl2 (0.66 mg/mL), Superoxide3 (0.80 mg/mL), ABTS4 (1.07 mg/mL), FRAP and inhibit lipid peroxidation | ||
| Tyr-Gly-Pro-Met | DPPH1 (0.72 mg/mL), Hydroxyl2 (0.88 mg/mL), Superoxide3 (0.73 mg/mL), ABTS4 (0.82 mg/mL), FRAP and inhibit lipid peroxidation | ||
| Cartilage of Dasyatis akajei | Ile-Glu-Glu-Glu-Gln | DPPH1 (4.61 mg/mL), Hydroxyl2 (0.77 mg/mL), Superoxide3 (0.08 mg/mL), ABTS4 (0.15 mg/mL). | [23] |
| Ile-Glu-Pro-His | DPPH1 (1.90 mg/mL,), Hydroxyl2 (0.46 mg/mL), Superoxide3 (0.17 mg/mL), ABTS4 (0.11 mg/mL), Lipid peroxidation inhibition activity. | ||
| Leu-Glu-Glu-Glu-Glu | DPPH1 (3.69 mg/mL), Hydroxyl2 (0.70 mg/mL), Superoxide3 (0.15 mg/mL), ABTS4 (0.19 mg/mL), Fe2+-chelating ability. | ||
| Val-Pro-Arg | DPPH1 (4.01 mg/mL), Hydroxyl2 (1.30 mg/mL), Superoxide3 (0.16 mg/mL), ABTS4 (0.18 mg/mL). | ||
| Head of Katsuwonus pelamis | Trp-Met-Gly-Pro-Tyr | DPPH1 (0.33 mg/mL), Hydroxyl2 (0.43 mg/mL), Superoxide3 (0.38 mg/mL), FRAP and lipid peroxidation inhibition. | [35] |
| Trp-Met-Phe-Asp-Trp | DPPH1 (0.31 mg/mL), Hydroxyl2 (0.30 mg/mL), Superoxide3 (0.56 mg/mL), FRAP and lipid peroxidation inhibition. | ||
| Glu-Met-Gly-Pro-Ala | DPPH1 (0.46 mg/mL), Hydroxyl2 (0.52 mg/mL), Superoxide3 (0.71 mg/mL), FRAP and lipid peroxidation inhibition. | ||
| Salmon gelatin | Gly-Gly-Pro-Ala-Gly-Pro-Ala-Val, Gly-Pro-Val-Ala, Pro-Pro and Gly-Phe | Oxygen radical absorbance capacity (ORAC, 540.94 ± 9.57 µmol TE/g d.w.) | [36] |
| Pacific cod skin gelatin | Leu-Leu-Met-Leu-Asp-Asn-Asp-Leu-Pro-Pro | Scavenging the intracellular ROS | [37] |
| Jumbo squid (Dosidicus gigas, squid) skin gelatin | Phe-Asp-Ser-Gly-Pro-Ala-Gly-Val-Leu Asn-Gly-Pro-Leu-Gin-Ala-Gly-Gln-Pro-Gly-Glu-Arg |
Inhibition of oxidant stress; Lipid peroxidation inhibition (>Vit. E). | [38] |
| Whole body of Parastromateus niger | Ala-Met-Thr-Gly-Leu-Glu-Ala | DPPH1 (54%), Metal chelating (78.6%) at 1 mg/mL | [24] |
| Smooth hound viscera (sharks) | Protein hydrate containing Gly, Glx, Lys, Asx, Arg, Pro and Ala | DPPH1, Inhibition of linoleic acid oxidation, Hydroxyl2. | [39] |
| Hoki (Johnius belengerii) frame | Glu-Ser-Thr-Val-Pro-Glu-Arg-Thr-His-Pro-Ala-Cys-Pro-Asp-Phe-Asn | DPPH1 (41.37 µM), Hydroxyl2 (17.77 µM), Peroxyl radical scavenging (18.99 µM), Superoxide3 (172.10 µM). | [40] |
| Limanda aspera frame | Arg-Pro-Asp-Phe-Asp-Leu-Glu-Pro-Pro-Tyr | Inhibition of linoleic acid autoxidation | [41] |
| Tuna backbone | Val-lys-Ala-Gly-Phe-Ala-Trp-Thr-Ala-Asn-Gln-Gln-Leu-Ser | Inhibited lipid peroxidation, Quenched free radicals (DPPH, hydroxyl and superoxide) | [42] |
| Frame of Theragra chalcogramma | Leu-Pro-His-Ser-Gly-Tyr | Hydroxyl2 (35% at 53.6 µM) | [43] |
Note: DPPH1-DPPH radical scavenging capacity, Hydroxyl2-Hydroxyl radical scavenging capacity, Superoxide3-Superoxide anion radical scavenging capacity, ABTS4-ABTS cation radical scavenging capacity.
As shown in Table 2 , recent investigations have focused on the impact of marine peptides on gut microbiota. The test substances have included protein hydrolysates, polypeptide fractions, and pure antioxidant peptides. Significant positive effects have been observed in many assays using peptides from microalgea, invertebrates, and byproducts of the fishing industry, such as the milt, roe and viscera of fish. Even glycosylated fish has been shown to have positive results. The balance of gut microbiota populations significantly improves not only in healthy mice or rats, but also in unhealthy ones subject to fatigue, alcohol-induced injury, and high-fat diet (HFD). In addition, a peptide combined with calcium significantly increases the abundance of beneficial gut microbes, including Firmicutes and Lactobacillus , in low-calcium diet-fed rats. Calcium absorption also increased in these animals.
Table 2. Regulation of gut microbiota by marine peptides.
| Marine Peptides | Test Animal | Improvement of Gut Microbiota | Ref. |
|---|---|---|---|
| Spirulina Phycocianin (Microalgae) | Mice | Increase the relative abundance of Bacteroidetes and Actinobacteria | [113] |
| Glycosylated fish protein | Mice | Increase the abundance of Allobaculum, Akkermansia, Lactobacillus animalis | [114] |
| Walleye Pollock skin | Mice | Upregulation relative abundance of Lactobacillus and Akkermansia, downregulation the abundance of bacteria associated with intestinal inflammation | [115] |
| Skin collagen peptide of Salmon salar and Tilapia nilotica | Male rats | Increased abundance of Lactobacillus | [116] |
| Herring milt hydrolysate (protein: 47–94%) | Mice | Maintain abundant of Lactobacillus decrease metabolites associated with obesity and inflammatory disease |
[117] |
| Peptides from tuna roe | Mice | Short-chain fatty acids production in feces and modulating gut microbiota composition | [52] |
| Abalone viscera | Alcohol induced injured mice | Increase in diversity index and the number of Bacilli (class), Lactobacillales (order), Lactobacillaceae (family), and Lactobacillus (genus) levels | [118] |
| Spirulina platensis protease hydrolyzate | High-fat diet (HFD)-fed rats | Enriched the abundance of gut beneficial bacteria | [119] |
| Chlorella pyrenoidosa protein hydrolysate-calcium chelate | Low-calcium diet-fed rats | Improving the abundances of Firmicutes and Lactobacillus | [120] |
| Oyster polypeptide (OP) fraction | Exhaustive fatigue mice | regulate the abundance of gut microbiota and maintain its balance | [121] |
4. Conclusions and Perspectives
The potential antiaging activities of natural peptides from underused marine organisms have been investigated in vitro, in cells, in animal models and in human clinical trials. These peptides act using the following molecular mechanisms: free radical scavenging, enhancing oxidase activity, protecting mitochondria, downregulating the apoptotic pathway, inhibiting MMP-1 expression, and restoring intestinal homeostasis. These results suggest that underused marine peptides have great potential as functional food and cosmetic ingredients for antiaging purposes. However, presently, there are three major outstanding issues. First, relevant research reports in the Web of Science database are few, and not enough animal assays have reported. We therefore focused on in vitro assays for this review. Second, in the limited literature, very few marine species have been explored, and vast marine protein resources are still underutilized. Third, apart from several applications in skin aging, there is a dearth of information about marine peptides in human clinical trials related to antiaging. This lack of literature makes it difficult to apply and transform these peptides for the market. To address these issues, further exploration of the abundant underused marine peptide sources is required.
This entry is adapted from the peer-reviewed paper 10.3390/md19090513
