Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Nutrition & Dietetics
Nutraceuticals possess several health benefits and functions; however, most nutraceuticals are prone to degradation in the gastrointestinal environment and have poor bioavailability. Application of a novel carrier system is of increasing importance to overcome obstacles and provide efficient applicability. Lipid-based nanocarriers provide a large surface-to-mass ratio, enhanced intestinal absorption by solubilization in the intestinal milieu, intestinal lymphatic transport, and altering enterocyte-based transport.
- bioavailability
- liposomes
- niosomes
- physicochemical stability
- solid lipid nanoparticles
- polyphenols
1. Introduction
The interest of public and health professionals has been drawn to the importance of nutraceuticals for the prevention of chronic diseases and has resulted in huge demand for nutraceutical products. The translation of nutraceutical compounds from lab to market is very tedious, which involves a long and extensive journey through basic research, formulation, complicated clinical trials, and regulatory approvals. According to the Global Opportunity Analysis and Industry Forecast, 2014–2022, the demand for functional foods (including nutraceuticals) and beverages has significantly increased in the past few years due to the increase in the cost of synthetic pharmaceutical active compounds. Further, the global nutraceutical market is expected to reach USD 302,306 million by 2022 from USD 184,092 million in 2015 with a CAGR of 7.04% from 2016 to 2022. The global nutraceutical product market is profitable due to the increase in awareness among consumers of certain nutraceuticals, such as omega-3-fatty-acid-fortified food and herbal extracts.
Oral delivery is the most convenient and widely acceptable route of administration due to ease of administration and cost-effectiveness in the formulation. The success of nutraceutical compounds is owing to numerous factors, such as therapeutic effect, physicochemical stability, and bioavailability. However, many bioactive compounds suffer from poor bioavailability due to poor solubility in the gastrointestinal tract, limited intestinal permeability, and hepatic first-pass metabolism. To reach the bloodstream, the bioactive compound should first dissolve in the gastrointestinal fluid and maintain its stability, but poorly water-soluble compounds (lipophilic bioactive compounds) result in poor absorption and low bioavailability due to the poor dissolution characteristics. Metabolism (intestinal and first-pass metabolism) is the second factor that restricts bioavailability to a significant extent [1]. Several approaches such as coacervation, liposome entrapment, inclusion complexation, cocrystallization, and emulsification have been evaluated for the improvement of the bioaccessibility and bioavailability of nutraceutical compounds. These technological and chemical modifications of the nutraceutical molecules are aimed at improving their solubility, the site of absorption, the design of colloidal systems (micelles and vesicles), and the use of nanosystems [2]. A nanotechnology (nanocarrier)-based delivery system is one of the major formulation research studies providing direct benefit to human health through clinical and commercial development [3]. These nanocarriers may lead to better half-life, precise release-completed short or extended durations, and identical site-specific targeted delivery of therapeutic compounds [4][5]. Different materials have been used for the construction of nanocarriers, including polymers, metals, and lipids. Among these, a lipid-based carrier is the most suitable technique in the food and nutraceutical sector because of its biocompatibility and biodegradability [6]. Although lipid nanocarriers cover a vast technique, only selected lipid nanocarriers were reported for its successful commercialization, including nanoemulsions, liposomes, solid lipid nanoparticles (SLNs), nanostructured lipid carriers (NLCs), niosomes, and self-emulsifying drug delivery systems (SEDDSs) [7]. A short summary of the selected lipid-based delivery system is presented in Table 1.
Table 1. Summary of the lipid-based delivery system.
| System | Definition | Advantages |
|---|---|---|
| Liposomes | Phospholipid bilayered vesicular systems having an aqueous core enclosed by one (unilamellar) or several (multilamellar) concentric phospholipid membranes. |
|
| Niosomes | Similar to liposomes, but bilayers for the niosomes are made up of nonionic surfactants. |
|
| Solid lipid nanoparticles (SLNs) | SLNs are matrix lipid particles formulated by replacing liquid lipid portions in emulsion formula with solid lipids. |
|
| Nanostructured lipid carriers (NLCs) | Second generation of lipid nanoparticles containing a mixture of solid and liquid lipids. |
|
| Self-emulsifying drug delivery system (SEDDS) | SEDDSs are isotropic mixtures of oil, surfactant, and cosurfactant that spontaneously form emulsion upon milk agitation. |
|
A wide range of essential bioactives/nutraceuticals in nature possess hydrophobic characteristics, including phenolic compounds, essential oils and fatty acids, carotenoids, and insoluble vitamins, and such hydrophobic bioactives are essential for human health. These poorly soluble characteristics of bioactive compounds eventually reduce bioavailability and sustainability [8]. A lipid-based carrier can be a favorable approach for protecting bioactive compounds in the gastrointestinal (GI) tract and enhancing their bioavailability. Thus, the object of this manuscript is to highlight the potential of lipid-based nanocarriers for the effective delivery of nutraceuticals. However, these lipid-based nanocarriers cover a wide range of techniques, including liposomes, ethosomes, Pickering emulsions, nanoemulsions, and solid lipid nanoparticles. This review was limited to the preparation methods, stability, and absorption mechanism of vesicular nanocarriers (liposomes and niosomes) and particulate systems (SLNs/NLCs).
2. Overview of Digestion
Food digestion is a complex process that involves the transit of food through the mouth, stomach, and small intestine. During digestion, the ingested food is broken down mainly by two processes: (i) mechanical breakdown, where solid foods of larger particles are broken down into smaller pieces, and (ii) enzymatic breakdown, where several digestive enzymes act on the food bolus during the gastrointestinal transit. The secretion of digestive enzymes initiates in the oral phase and continues in the stomach and intestine [9]. Solid and semisolid foods require considerable physical transformation during digestion.
The oral cavity is the first site of food digestion, involving mechanical grinding (physical disruption by chewing) and enzymatic hydrolysis by mixing with saliva. Salivary glands in the oral cavity secret saliva, which is near neutral pH with salivary amylase enzyme to hydrolyze starch molecules into glucose. During oral digestion, the food is broken down into small pieces, is mixed with saliva, and forms an oral bolus [10].
Gastric digestion: Oral bolus masticated with amylase is then transported to the gastric compartment through the esophagus by peristalsis motion. The stomach accommodates the bolus within four regions—cardiac, fundic, body, and pylorus—and converts masticated bolus into semisolid chyme [11]. Briefly, the proximal stomach (upper part of the stomach) acts as a receiving center/reservoir for the oral bolus/undigested food. Additionally, the distal part is responsible for the breaking of solid foods by grinding, propelling, and mixing the undigested food with gastric secretions for enzymatic breakdown. In a recent study, Sutter, et al. [12] reported that the rate of gastric contraction ranges between 2.6 and 3.8 times per minute, and the median contraction frequency is higher in women (3.2 times per minute) and lower in men (3.0 times per minute). Further, gastric secretion consists of hydrochloric acids, electrolyte salts, mucus, enzymes such as gastric lipase (digest fats) and pepsin (to digest protein), and various hormones. The pH of the stomach dynamically changes between 1.5 and 2.0 in the fasted state and 3.0 and 7.0 in the fed state [13]. The digested food leaves the stomach through the pylorus, which acts as a sieve and allows food with 1–2 mm size, and this process is called gastric emptying. Additionally, the food material that does not achieve <2 mm size is maintained in the distal part by the retropulsion mechanism and undergoes the grinding process until it achieves the desired size range.
Small intestine: Disintegrated food with <2 mm size will move to the small intestine through the pylorus for digestion and nutrient absorption. The small intestine comprises three compartments: duodenum, jejunum, and ileum. The duodenum possesses a short length (~30 cm) and is located immediately next to the stomach, where the digesta from acidic pH moves to neutral pH. In this region, the hydrolytic enzyme pancreatin secreted by the pancreas mixes with the digesta and further hydrolyzes the proteins, fats, and carbohydrates. Further, the bile acids introduced through the biliary tract into the small intestine emulsify the fat droplets and help in the digestion and absorption of fats and fat-soluble vitamins. Intestinal motility, including peristalsis and segmentation, plays a predominant role in mixing and transporting the digesta across the intestine [14].
In the case of lipid-based nanocarriers, it provides distinctive features, including a large surface-to-mass ratio, long-term stability, modification and conjugation capability, and encapsulation of nutraceutical molecules [15]. Lipid-based formulations can significantly enhance the intestinal absorption of poorly water-soluble compounds. Porter, et al. [16] reported three main mechanisms by which lipophilic excipients enhance the absorption:
-
Enhancing bioactive solubilization in the intestinal milieu: Dissolution is a prerequisite for the absorption of nutraceuticals from the small intestinal lumen into the enterocytes (intestinal absorptive cells). Lipid-based nanocarriers enhance the solubilization in the GI tract by providing the bioactive compounds in a solubilized form and preventing precipitation in the intestinal milieu.
-
Intestinal lymphatic transport: Highly lipophilic molecules can be transported to the systemic circulation through the intestinal lymphatic system. In normal cases, the portal vein is employed for the transport of molecules (through the first-pass metabolism) to the systemic circulation. Lipid-based systems recruit endogenous and exogenous lipid transport and stimulate intestinal lymphatic transport of coadministered lipophilic bioactive compounds, thereby protecting the molecules from the first-pass metabolism.
-
Altering enterocyte-based bioactive transport: Lipids and lipophilic excipients can alter the expression of intracellular lipid-binding proteins by interacting with apical membrane lipid transporters and modify the intracellular pooling of lipids within the enterocyte.
Various transfer mechanisms, such as paracellular absorption, M-cell-mediated transport, and endocytosis, are involved in the transportation of nanosized lipids across the intestinal membrane (Figure 1).
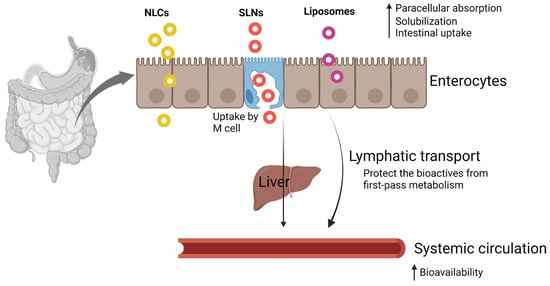
Figure 1. Transport mechanism of nanosized lipid-based delivery systems (created with Biorender.com).
Paracellular absorption: Orally administered nutraceuticals must reach the systemic circulation via absorption through the intestinal epithelial layer. There are four different transfer mechanisms: paracellular, transcellular, carrier-mediated, and receptor-mediated transport. Paracellular absorption refers to the transport of molecules between adjacent epithelial cells, but the rate-limiting step in this absorption process is the transport across the tight junction [17]. The intestinal epithelium is one of the major barriers to the absorption of hydrophilic bioactive molecules due to the nondiffusional properties of epithelial cells through lipid bilayer cell membranes to the bloodstream [18]. Ji, et al. [19] enhanced the solubility and bioavailability of curcumin by solid lipid nanoparticle formulation with TPGS and Brij78 (P-gp modulator). Intestinal effective permeability was significantly improved for curcumin SLNs, thereby increasing the relative bioavailability to 942.53% compared with the curcumin suspension.
M-cell-mediated transport: M-cells (also called as macrofold cells) are specialized intestinal epithelial cells found in the follicle-associated epithelium of intestinal Peyer’s patches of gut-associated lymphoid tissue and isolated lymphoid follicles responsible for the immune sensing of luminal bacteria [20][21]. The role of M-cells includes nutrient absorption from the intestine and delivery of intestinal microbial antigens to gut-associated lymphoid tissue for mucosal and systemic immune responses [21].
Endocytosis: All intestinal cells use endocytosis to take up nutrients from the external milieu by four separate mechanisms: clathrin-mediated endocytosis, caveolae, micropinocytosis, and phagocytosis. Clathrin- and caveolin-mediated endocytoses involve the receptor binding of macromolecules [22]. In the clathrin-mediated endocytosis, nanoparticles interact with receptors on the cytomembrane (clathrin-1) and form clathrin-coated vesicles (CCV) through the GTPase activity of dynamin. CCVs penetrate inside the cells through the energy supplied by actin, and the destination of the CCVs is decided by the receptor that is attached to the ligands of nanoparticles [23]. Macropinocytosis is a form of endocytosis that accompanies cell surface ruffling and is mediated by the actin cytoskeleton that internalizes the surrounding fluid into large vacuoles [24][25]. The diameter of the endocytotic vesicles can be larger than those of clathrin- and caveolin-mediated endocytoses. Chai, et al. [26] first revealed the transport mechanisms of solid lipid nanoparticles (SLNs) across intestinal epithelial cell monolayers (Caco-2 cells). Internalization of SLNs was mediated by the macropinocytosis pathway and clathrin- and caveolae (or lipid raft)-related routes. Further, transcytosis (transcellular transport) of SLNs did not damage the tight junctions of intestinal epithelial cells and ensures SLNs as a safe delivery system for oral administration of bioactive compounds.
Need for digestion studies: In recent years, there is an increasing awareness among consumers of the food they are consuming, including innovative functional food products and nutraceutical formulations. A thorough understanding of the digestion process from the particle breakdown at the oral phase to the nutrient absorption at the intestinal phase is necessary to design effective food products [27]. Figure 2 illustrates an overview of in vivo and in vitro digestion models.
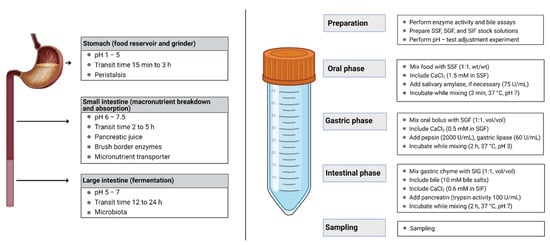
Figure 2. Region specificity and role of gastrointestinal tract during food digestion (left) and flow diagram of in vitro INFOGEST digestion protocol (right). This figure is reproduced from Li, et al. [28].
The evolution of food-grade delivery systems is showing promising results in improving the stability, bioavailability, and release characteristics of nutrients. These delivery systems are designed in such a way that the encapsulated nutrient should be released/exposed at the specific location in the GI tract, often by trigger factors, such as ionic strength, pH, or enzyme activity [29]. Testing the efficiency of such formulations is much necessary before introducing those products to the market. Digestion and absorption studies involving human volunteers (clinical trials) are still considered the ‘golden standard’ for addressing the response of a specific diet/formulation in the human gastrointestinal system. However, such in vivo studies are complex, labor-intensive, and expensive and undergo severe ethical restrictions [30]. In recent years, a considerable amount of effort has been devoted to developing in vitro models that provide an easy sampling technique and rapid digestion and simulate physiological factors such as pH, salt concentration, and digestive enzymes. Dynamic in vitro models can simulate experimental conditions similar to in vivo physiological conditions and widely accepted in food and pharmaceutical industries for their accuracy and reproducibility of data [31].
3. Vesicular Nanocarriers
Vesicular nanocarriers are colloidal carrier systems showing great promise in drug/nutraceutical delivery. Vesicular nanocarriers have been extensively investigated in the past few decades for pharmaceutical applications and revealed improved pharmacokinetics, solubility, stability, and biodistribution; decreased toxicities; controlled release; and site-specific delivery of therapeutic agents [32][33]. Vesicular systems are useful to encapsulate both hydrophilic (interior hydrophilic compartment) and lipophilic (in outer lipid layer) bioactive compounds. Vesicular nanocarriers are classified based on their principal compounds (see Figure 3). In this manuscript, only the application of liposomes and niosomes was reviewed due to their promising application in the food sector.
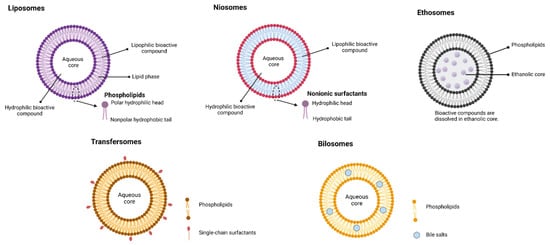
Figure 3. Classification of vesicular systems (created with Biorendor.com).
3.1. Liposomes
Liposomes are phospholipid bilayered vesicular systems having an aqueous core enclosed by one (unilamellar) or several (multilamellar) concentric phospholipid membranes [34][35]. Mozafari, et al. [36] proposed a definition for liposomes as ‘closed, continuous bilayered structures made mainly of lipid and/or phospholipid molecules’. To avoid confusion among other lipid-based delivery systems with liposomes, the US Food and Drug Administration (USFDA) clearly defined that liposomes are not emulsion systems or a drug–lipid complex [37].
Liposomes vary with composition, size, surface charge, and method of preparation. The main attractive feature in the application of liposomes is that they can accommodate both water-soluble (in the aqueous core region) and lipid-soluble (in lamellae) bioactive compounds [32]. Schematic representations of liposome preparation techniques are shown in Figure 4. These biodegradable and biocompatible compositions of liposomes make them excellent carriers of therapeutic agents. Conventional liposome preparation involves the following stages: drying of lipid/phospholipid ingredients from organic solvents, dispersion in aqueous media (it creates vesicles), purification of the resultant vesicles, and analysis of the final product. Thus, the increasing interest in lipid-based nutraceutical formulation can be correlated with the increasing number of clinical trials. Table 2 summarizes the recent clinical trials on lipid-based formulations. However, the traces of organic solvent residues may remain in the lipid and/or aqueous phase and result in toxicity. Advances in the field of encapsulation technology have brought a possible way to prepare lipid vesicles without volatile organic solvents. It is a remarkable development by Prof. Mozafari to formulate microcapsules without the application of toxic solvents, detergents, and harsh treatments such as sonication/microfluidization [38][39]:
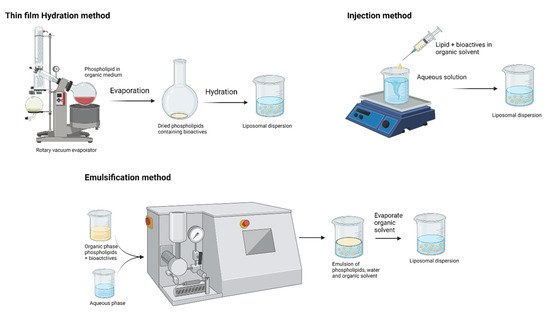
Figure 4. Production techniques of liposomes (created with Biorendor.com).
Table 2. Clinical trials on lipid-based formulations.
| Bioactives | Formulation | Title | Study Aim | Disease/Condition | Reference |
|---|---|---|---|---|---|
| Curcumin | Liposomes | Evaluation of liposomal curcumin in healthy volunteers | Safety and tolerability of increasing doses of intravenous liposomal curcumin | Health volunteers | NCT01403545 (ClinicalTrials.gov) |
| Curcumin | Liposomes | Comparison of curcumin bioavailability | Evaluate the bioavailability of curcumin of eight different formulations | Health volunteers | NCT03530436 (ClinicalTrials.gov) |
| Curcumin | Liposomes | A phase 1 study establishing the safety of intrapleural administration of liposomal curcumin (LipoCurc) as a palliative treatment for malignant pleural effusion | Investigate the safety of administering liposomal curcumin directly to the tumor site | Patient with long-term chest drain | ACTRN12620001216909 (ANZCTR.org.au) |
| Curcumin | LipiSperse® | A randomized double-blind placebo-controlled study to evaluate the effect of curcumin on BDNF levels in otherwise healthy adults | Investigate the bioavailable fraction of curcumin on BDNF levels in healthy adults | Brain-derived neurotrophic factor in healthy adults | ACTRN12621000104853 (ANZCTR.org.au) |
| Coenzyme Q10 | Liposomes | A comparison of the plasma levels and safety of coenzyme Q10 from four different formulations in healthy adult volunteers | Evaluate the bioavailability of coenzyme Q10 in liposomal formulation | Patients with mild to moderate cardiovascular disease | ACTRN12616001527459 (ANZCTR.org.au) |
| Coenzyme Q10 | SEDDS | The impact of micelle size and increased absorption of ubiquinone using a novel delivery system (AquaCelle®) | Evaluate the bioavailability of Coenzyme Q10 in SEDDS formulation | Healthy volunteers | [40] |
| EPA, DHA | SEDDS | A self-emulsifying omega-3 ethyl ester formulation (AquaCelle) significantly improves eicosapentaenoic and docosahexaenoic acid bioavailability in healthy adults | Evaluate the bioavailability of omega-3 fatty acid concentrations of SEDDS formulation | Healthy volunteers under the low-diet condition | [41] |
| Trans-resveratrol | LipiSperse® | Trans-resveratrol oral bioavailability in humans using LipiSperse™ dispersion technology | Pharmacokinetics of resveratrol–LipiSperse® delivery complex | Healthy volunteers | [42] |
-
Add nanoliposomal ingredients to the mixture (preheated to 40–60 °C) of active agent and polyol in a heat-resistant vessel.
-
Heat the mixture at 40–60 °C while stirring in an inert atmosphere (pass nitrogen or argon gas to create an inert atmosphere).
-
Keep the product (nanoliposome) above the phase transition temperature of the phospholipid ingredients under an inert atmosphere for 1 h so that the nanoliposomes will anneal and stabilize. Then bring the nanoliposome to an ambient temperature gradually and store.
3.1.1. Physicochemical Stability
The increasing interest in the liposomal delivery system is due to its functional properties, including efficient encapsulation capacity and biocompatibility with food constituents. However, the main problem associated with the industrial application of liposomes is their insufficient physiochemical stability due to the fragile bilayer phospholipid membranes and oxidation or hydrolysis of the fatty acids. One strategy to improve the physiochemical stability is the application of polysaccharides in liposomal preparation. The addition of polysaccharides (modifying surface charges) and antioxidants to liposomes minimizes the oxidative degradation of liposomes, including high-quality lecithins with low levels of hydroperoxides and transition metals [43]. Lopes, et al. [44] investigated the physicochemical properties and stability of the nisin (antibacterial agent) liposomes covered with polysaccharides (polygalacturonic acid/pectin). Polygalacturonic acid liposomes were stable for over 28 days, with a polydispersity index (PDI) of around 0.2 to 0.3, which indicates that the negatively charged polysaccharides increased the electrostatic repulsion between nanoparticles and thereby increased their stability. Later, the same group coencapsulated nisin and lysozyme in polysaccharide-coated liposomes (polygalacturonic acid/pectin). Polysaccharide-coated liposomes maintained constant particle and zeta potential due to the presence of polysaccharide layers on the surface or induced stabilization through the reduction of charges on the polysaccharide and liposome due to electrostatic screening [45].
Azzi, et al. [46] reported the higher encapsulation capacity and improved physicochemical properties of quercetin encapsulated with cyclodextrin-coated liposomes. Quercetin is a flavonoid known for its beneficial effects, including antiobesity, anticancer, anti-inflammatory, and antioxidant properties. However, poor water solubility and poor chemical stability are the main drawbacks of its application in the food sector. Due to poor aqueous solubility, quercetin possesses low bioavailability and a short biological half-life [47]. The authors revealed that the quercetin-loaded liposomes were stable (evaluated through particle size and quercetin content) even after 12 months of storage (@ 4 °C). Surprisingly, a higher amount of quercetin (98%) was retained after 12 months in the quercetin in cyclodextrin in liposomes. Pu, et al. [48] came up with a different strategy with cationic guar gum (CGG) to increase the stability of curcumin. CGG is the cationic quaternary group of guar gum (GG) and has a good affinity for negatively charged compounds. As a coating material, nonionic GG might coat the negatively charged liposomes with physical absorption, but the cationic CGG is absorbed on the negative surface of the liposomes via electrostatic interaction and remains stable. The negative charge of the liposomal surface was neutralized by the CGG interactions and turned the net charge positive. The authors observed the better protection effect (able to provide higher curcumin retention at 70 °C) of curcumin-loaded liposomes coated with CGG. On the other hand, nanoliposomes have been reported as a promising delivery system for transporting food bioactive compounds.
Plant-based phospholipids (PLs), such as soybean, rapeseed, sunflower PLs, were frequently used for liposomal preparation, and the major PL components include zwitterionic phosphatidylcholine (PC), phosphatidylethanolamine (PE), and anionic phosphatidylinositol (PI). However, those plant-based PLs are more highly susceptible to oxidation and lower physical and storage stability owing to the high degree of unsaturation [49]. Alternatively, animal-based PLs, such as eggs, milk, meat, and marine source, are gaining attention for the fabrication of liposomes. Interestingly, marine PLs (fish roe, krill oil, and fish) are reported for higher PUFAs, which makes them a promising functional ingredient [50]. Milk PLs contain high levels of sphingomyelin (SM) in addition to other PL components and are reported to have a higher degree of saturation. Wu, Mou, Song, Tan, Lai, Shen and Cheong [49] studied the effect of bovine milk and krill PLs for curcumin-loaded liposomes and noticed that the liposome of bovine milk PLs exhibited higher stability with a smaller particle size and greater zeta potential (163 nm and −26.7 mV) than the krill PL liposomes (212 nm and −15.23 mV). Both curcumin liposomes were stable (curcumin retention: 80%) after 40 h exposure in acidic conditions, but the retention rate of curcumin was reduced to 50% at neutral and alkaline conditions. Additionally, curcumin retention is higher in bovine milk PL formulation than the krill PLs attributed to the high amount of phosphatidylcholine in krill PLs, which are susceptible to hydrolysis, resulting in the leakage of curcumin from the phospholipids.
3.1.2. Enhancing the Bioavailability of Polyphenols by Liposomal Technology
There is always a demand for plant-derived bioactive compounds, which are considered natural and safer. Polyphenols are the secondary plant metabolites, which include nearly 10,000 different compounds with one or more aromatic rings and hydroxyl groups. Readers can refer to Brglez Mojzer, et al. [51] for an understanding of the polyphenols and the different extraction methods. Polyphenols are generally classified as flavonoids and phenolic acids. Additionally, flavonoids are further divided into flavones, flavonols, flavanones, and isoflavones, and phenolic acids as hydroxybenzoic and hydroxycinnamic acids [52]. However, the poor absorption and low bioavailability of extracted polyphenols restrict the wide application of polyphenol-based functional foods. Entrapping those polyphenols in the bilayered vesicular systems (liposomes) is one of the ideal ways to improve GI stability and bioavailability.
However, conventional liposomes or nanoparticles do not cross the intestinal mucosal barrier due to their relatively large size [53], are unstable in the gastric environment, and release the entrapped bioactive compounds prior to the target site. From the previous research, it is evident that the bioactive compounds need to be absorbed/transformed into mixed micelles in the GI tract and transported to the epithelial cells. M-cell-mediated transport is reported for the possible absorption route for liposomes and nanoparticles; however, the M-cells may account for 1% of the total epithelial cell population [54]. The following strategies were employed for enhancing the intestinal absorption of encapsulated bioactive compounds.
Curcumin: Curcumin is a polyphenolic compound reported for its antioxidant, anti-inflammatory, antimicrobial, and anticancer properties. However, the wide application of curcumin is limited because of its poor aqueous solubility, rapid metabolism, and low permeability due to its susceptibility to P-glycoprotein efflux [55]. The stability of curcumin in the liposomes can be improved further by converting the liquid formulation into a dry form. Additionally, conventionally either spray drying or freeze drying is used for this purpose. Alternatively, Gopi, et al. [56] prepared liposomal curcuminoids in powder form through a novel nanofiber weaving technology. The release of curcumin from the liposomal curcuminoid powder was evaluated at pH 5.5 (46.2%) and pH 7.4 (40.4%), and the burst release (initial rupture) was observed within 12 h and then followed a slow and sustained release. A slightly acidic pH accelerated the release of curcumin from the formulation, and the strong interaction between phospholipids and curcumin at pH 7.4 showed less desorption.
Recent research on Pluronic-modified liposomes showed promising results in maintaining the stability of bioactives in the GI tract and improving bioaccessibility. Pluronics (also called poloxamer, Kolliphor, and Synperonic) are nonionic triblock copolymers with a central hydrophobic poly (propylene oxide (PPO)) chain with two hydrophilic poly (ethylene oxide (PEO)) on each side [57]. Pluronic-modified liposomes protected the curcumin from degradation, and nearly 50% of curcumin was retained after exposure to 80 °C. Additionally, the bioaccessibility of curcumin was increased to 43.3%, whereas the simple curcumin liposomal formulation was shown to be 26.9% bioaccessible [58]. Further, the same group revealed Fluronic-modified liposomes, and folated Fluronic-modified liposomes are nontoxic to the human KB cell lines [59].
Green tea polyphenols: Green tea comprises more than eight polyphenolic compounds (catechins) and is highly interesting among consumers for its potential health benefits, including antioxidant, antidiabetic, anti-inflammation properties. However, catechins are poorly absorbed in the body. When consuming green tea, only 5% of tea catechins reached the systemic circulation in rats and 1.68% in humans [60]. Liposomal formulation of catechin improves the GI stability and absorption in the body. Ezzat, et al. [61] developed chitosan-coated catechin liposomes and evaluated the pharmacokinetic properties in Wistar rats. Intestinal permeation from the in situ intestinal perfusion study revealed the rapid increase in intestinal absorption of chitosan-coated catechin liposomes (45.80 µg in 30 min); at the same time, conventional liposomal formulation and catechin solution absorbed only 18.26 and 7.36 µg, respectively. Similarly, the bioavailability of chitosan-coated catechin liposome was 1.37-folds higher than that of conventional liposomes and 2.12-folds higher than that of the catechin solution. Therefore, chitosan can directly interact with the tight junctions and facilitates the paracellular transportation and makes the chitosan-entrapped liposomes a promising approach for improving the intestinal absorption of bioactive compounds. Researchers worked with the different strategies to improve the bioavailability of catechins, including nanoencapsulation of catechins and nanoencapsulated catechin-based functional foods, and on different routes of delivering catechin. Recently, Fornasier, et al. [62] formulated catechin hexosomes (lipids aggregated in tubular arrangement) for the topical administration and found increasing penetration of catechins in the pig skin. To enhance the permeation properties, bile salts were used in the formulation and found that hexasomes with bile salts showed deeper transdermal penetration of the catechins than the hexasomes without bile salts.
Resveratrol: Resveratrol (trans-resveratrol; 3,5,4′-trihydroxystilbene) is a nonflavonoid polyphenolic compound found in grapes, red wine, and berries. Resveratrol is known for its health benefits, including anti-inflammatory, antiobesity, antioxidant, anticarcinogenic, and antiaging properties. On the downside, resveratrol is photosensitive and poorly soluble at the low GI phase and has low intestinal absorption and rapid metabolism. To overcome these limitations, nanosized formulations including liposomal entrapment of resveratrol can be employed. The liposomal formulation of resveratrol is gaining attention for its anticancer properties. A combination of anticancer bioactive compounds, resveratrol, and artemisinin in liposomes was evaluated for intestinal cancer cells [63]. The authors found that Eudragit-coated liposomes of both resveratrol and artemisinin reduced the viability of the HT-29 cells to 16% and 37% after 24 h exposure to 10 and 20 µg mL−1, respectively. The increased mortality of tumor cells was attributed to resveratrol’s multiple cellular targets affecting cellular proliferation and growth, including apoptosis, inflammation, invasion, angiogenesis, and metastasis [64].
Proliposomes: Proliposomes are a solidified form of liposomes formulated by removing water content in the liposomal suspension using different techniques: spray drying, freeze drying, vacuum drying, and fluidized bed drying. These dry granular proliposomes do not form a lipid bilayer structure during storing (increases stability), and they can be converted to liposomal formulation upon hydration. In a recent study, Jiao, et al. [65] optimized the supercritical fluid (ScCO2) technique for vitamin C (VC) proliposomes and reported that the optimal conditions were 25 MPa as pressure, 48 °C as temperature, and 0.25 as feeding ratio of vitamin C against phosphatidylcholine. This solid-state formulation ensures stability and is convenient for transportation, storage, and distribution. The solubility of proliposomes loaded with curcumin is estimated to be 98%, indicating the outstanding dispersible characteristics of proliposomes at the hydration step [66]. Further, proliposomes maintained an encapsulation efficiency of 92% after 30 days of storage at room temperature [67]. An in situ intestinal absorption study proved the increase in the absorption of liposomal formulations compared with the free form. Ren, et al. [68] reported a significant increase in absorption rate constant (2.3-fold) and absorptive fraction (1.4-fold) between the proliposome dispersion and its free form (quercetin was the model bioactive compound). Surprisingly, the in vivo pharmacokinetic study revealed a nearly 6.5-fold increase in Cmax (maximum concentration) and a 12-fold increase in AUC (area under the curve) for the quercetin proliposomes. Such drastic increase in oral bioavailability was attributed to the solid formulation of quercetin proliposomes and their stability during GI transit [69]. Recently, Hızır-Kadı, et al. [70] improved the solubility of pollen phenolic extract (PPE) and improved the bioaccessibility by 2-fold compared with the conventional liposomes. However, the authors dissolved PPE in a few drops of ethanol during the proliposome production step, which may restrict the wide application of following this technique in an industrial scale.
Bile salt liposomes: To extend the application of liposomes further, bile salts are incorporated into the encapsulation process of liposomes, and such bile-salt-incorporated liposomes are called ‘nanobilosomes’. Bile salts in the ‘nanobilosomes’ further improve the aqueous solubility and dissolution rate and preserve the liposomes in the GI tract and enhance membrane permeability [71]. This ‘nanobilosome’ can be a promising technique to deliver the Biopharmaceutics Classification System (BCS) class IV bioactive compounds, which exhibit poor solubility and poor intestinal permeability. Mangiferin polyphenol is one of the poorly aqueous soluble (0.162 mg/mL solubility in water) and poorly intestinally permeable compounds (1.96 effective permeability in duodenum) [72]. Nanobilosome formulation improved the aqueous solubility by seven times and had better protection in the simulated gastrointestinal condition due to the electrostatic repulsive force between the bile salts in the formulation and the bile salts in the simulated intestinal fluid [73].
This entry is adapted from the peer-reviewed paper 10.3390/molecules26185510
References
- Arzani, G.; Haeri, A.; Daeihamed, M.; Bakhtiari-Kaboutaraki, H.; Dadashzadeh, S. Niosomal carriers enhance oral bioavailability of carvedilol: Effects of bile salt-enriched vesicles and carrier surface charge. Int. J. Nanomed. 2015, 10, 4797.
- Rein, M.J.; Renouf, M.; Cruz-Hernandez, C.; Actis-Goretta, L.; Thakkar, S.K.; Da Silva Pinto, M. Bioavailability of bioactive food compounds: A challenging journey to bioefficacy. Br. J. Clin. Pharmacol. 2013, 75, 588–602.
- Mishra, D.K.; Shandilya, R.; Mishra, P.K. Lipid based nanocarriers: A translational perspective. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 2023–2050.
- Safari, J.; Zarnegar, Z. Advanced drug delivery systems: Nanotechnology of health design A review. J. Saudi Chem. Soc. 2014, 18, 85–99.
- Ragelle, H.; Danhier, F.; Préat, V.; Langer, R.; Anderson, D.G. Nanoparticle-based drug delivery systems: A commercial and regulatory outlook as the field matures. Expert Opin. Drug Deliv. 2017, 14, 851–864.
- Dabholkar, N.; Waghule, T.; Rapalli, V.K.; Gorantla, S.; Alexander, A.; Saha, R.N.; Singhvi, G. Lipid shell lipid nanocapsules as smart generation lipid nanocarriers. J. Mol. Liq. 2021, 339, 117145.
- Lushchak, O.; Karpenko, R.; Zayahckivska, A.; Koliada, A.; Vaiserman, A. Lipid-Based Nano-delivery of Phytobioactive Compounds in Anti-aging Medicine. In Systemic Delivery Technologies in Anti-Aging Medicine: Methods and Applications; Lai, W.-F., Ed.; Springer: Cham, Switzerland, 2020; pp. 221–245.
- Rezaei, A.; Fathi, M.; Jafari, S.M. Nanoencapsulation of hydrophobic and low-soluble food bioactive compounds within different nanocarriers. Food Hydrocoll. 2019, 88, 146–162.
- González, R.L.; Viuda-Martos, M.; Pérez-Alvarez, J.A.; Fernández-López, J. In vitro digestion models suitable for foods: Opportunities for new fields of application and challenges. Food Res. Int. 2018, 107, 423–436.
- Tamura, M.; Okazaki, Y.; Kumagai, C.; Ogawa, Y. The importance of an oral digestion step in evaluating simulated in vitro digestibility of starch from cooked rice grain. Food Res. Int. 2017, 94, 6–12.
- Ferrua, M.; Singh, R. Modeling the Fluid Dynamics in a Human Stomach to Gain Insight of Food Digestion. J. Food Sci. 2010, 75, R151–R162.
- Sutter, N.; Klinge, M.W.; Mark, E.B.; Nandhra, G.; Haase, A.-M.; Poulsen, J.; Knudsen, K.; Borghammer, P.; Schlageter, V.; Birch, M.; et al. Normative values for gastric motility assessed with the 3D-transit electromagnetic tracking system. Neurogastroenterol. Motil. 2020, 32, e13829.
- Shani-Levi, C.; Alvito, P.; Andrés, A.; Assunção, R.; Barberá, R.; Blanquet-Diot, S.; Bourlieu, C.; Brodkorb, A.; Cilla, A.; Deglaire, A.; et al. Extending in vitro digestion models to specific human populations: Perspectives, practical tools and bio-relevant information. Trends Food Sci. Technol. 2017, 60, 52–63.
- Boland, M.J. Human digestion–A processing perspective. J. Sci. Food Agric. 2016, 96, 2275–2283.
- Dolatabadi, J.E.N.; Omidi, Y. Solid lipid-based nanocarriers as efficient targeted drug and gene delivery systems. TrAC Trends Anal. Chem. 2016, 77, 100–108.
- Porter, C.; Trevaskis, N.; Charman, W. Lipids and lipid-based formulations: Optimizing the oral delivery of lipophilic drugs. Nat. Rev. Drug Discov. 2007, 6, 231–248.
- Edelblum, K.L.; Turner, J.R. Epithelial Cells: Structure, Transport, and Barrier Function. In Mucosal Immunology, 4th ed.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 187–210.
- Borchard, G.; Lueβen, H.L.; de Boer, A.G.; Verhoef, J.; Lehr, C.-M.; Junginger, H.E. The potential of mucoadhesive polymers in enhancing intestinal peptide drug absorption. III: Effects of chitosan-glutamate and carbomer on epithelial tight junctions in vitro. J. Control. Release 1996, 39, 131–138.
- Ji, H.; Tang, J.; Li, M.; Ren, J.; Zheng, N.; Wu, L. Curcumin-loaded solid lipid nanoparticles with Brij78 and TPGS improvedin vivooral bioavailability andin situintestinal absorption of curcumin. Drug Deliv. 2014, 23, 459–470.
- Corr, S.C.; Gahan, C.; Hill, C. M-cells: Origin, morphology and role in mucosal immunity and microbial pathogenesis. FEMS Immunol. Med. Microbiol. 2008, 52, 2–12.
- Ohno, H. Intestinal M cells. J. Biochem. 2016, 159, 151–160.
- Lundquist, P.; Artursson, P. Oral absorption of peptides and nanoparticles across the human intestine: Opportunities, limitations and studies in human tissues. Adv. Drug Deliv. Rev. 2016, 106, 256–276.
- Kou, L.; Sun, J.; Zhai, Y.; He, Z. The endocytosis and intracellular fate of nanomedicines: Implication for rational design. Asian J. Pharm. Sci. 2013, 8, 1–10.
- Swanson, J.A.; Watts, C. Macropinocytosis. Trends Cell Biol. 1995, 5, 424–428.
- Garrett, W.S.; Mellman, I. Studies of endocytosis. In Dendritic Cells: Biology and Clinical Applications, 2nd ed.; Academic Press: San Diego, CA, USA, 2001; pp. 213–230.
- Chai, G.; Xu, Y.; Chen, S.-Q.; Cheng, B.; Hu, F.-Q.; You, J.; Du, Y.-Z.; Yuan, H. Transport Mechanisms of Solid Lipid Nanoparticles across Caco-2 Cell Monolayers and their Related Cytotoxicology. ACS Appl. Mater. Interfaces 2016, 8, 5929–5940.
- Bornhorst, G.M.; Gouseti, O.; Wickham, M.S.; Bakalis, S. Engineering Digestion: Multiscale Processes of Food Digestion. J. Food Sci. 2016, 81, R534–R543.
- Li, C.; Yu, W.; Wu, P.; Chen, X.D. Current in vitro digestion systems for understanding food digestion in human upper gastrointestinal tract. Trends Food Sci. Technol. 2020, 96, 114–126.
- Hur, S.J.; Lim, B.O.; Decker, E.A.; McClements, D.J. In vitro human digestion models for food applications. Food Chem. 2011, 125, 1–12.
- Minekus, M.; Alminger, M.; Alvito, P.; Ballance, S.; Bohn, T.; Bourlieu, C.; Carrière, F.; Boutrou, R.; Corredig, M.; Dupont, D.; et al. A standardised staticin vitrodigestion method suitable for food—An international consensus. Food Funct. 2014, 5, 1113–1124.
- Parthasarathi, S.; Bhushani, J.A.; Anandharamakrishnan, C. Engineered small intestinal system as an alternative to in-situ intestinal permeability model. J. Food Eng. 2018, 222, 110–114.
- Din, F.U.; Aman, W.; Ullah, I.; Qureshi, O.S.; Mustapha, O.; Shafique, S.; Zeb, A. Effective use of nanocarriers as drug delivery systems for the treatment of selected tumors. Int. J. Nanomed. 2017, 12, 7291–7309.
- How, C.W.; Rasedee, A.; Manickam, S.; Rosli, R. Tamoxifen-loaded nanostructured lipid carrier as a drug delivery system: Characterization, stability assessment and cytotoxicity. Colloids Surfaces B. Biointerfaces 2013, 112, 393–399.
- Lim, S.B.; Banerjee, A.; Önyüksel, H. Improvement of drug safety by the use of lipid-based nanocarriers. J. Control. Release 2012, 163, 34–45.
- Verma, S.; Utreja, P. Vesicular nanocarrier based treatment of skin fungal infections: Potential and emerging trends in nanoscale pharmacotherapy. Asian J. Pharm. Sci. 2019, 14, 117–129.
- Mozafari, M.R.; Reed, C.J.; Rostron, C.; Kocum, C.; Piskin, E. Formation and characterisation of non-toxic anionic liposomes for delivery of therapeutic agents to the pulmonary airways. Cell. Mol. Biol. Lett. 2002, 7, 243–244.
- U.S. Food and Drug Administration. Liposome Drug Products: Chemistry, Manufacturing, and Controls; Human Pharmacokinetics and Bioavailability; and Labeling Documentation; FDA-2016-D-2817; Center for Drug Evaluation and Research: Rockville, MD, USA, 2018.
- Khorasani, S.; Danaei, M.; Mozafari, M. Nanoliposome technology for the food and nutraceutical industries. Trends Food Sci. Technol. 2018, 79, 106–115.
- Mozafari, M.R. Nanoliposomes: Preparation and Analysis. Liposomes 2010, 605, 29–50.
- Briskey, D.; Sax, A.T.; Mallard, A.R.; Ng, C.; Rao, A. The impact of micelle size and increased absorption of ubiquinone using a novel delivery system (AquaCelle®). Int. J. Pharmacokinet. 2019, 4, IPK02.
- Bremmell, K.E.; Briskey, D.; Meola, T.; Mallard, A.; Prestidge, C.A.; Rao, A. A self-emulsifying Omega-3 ethyl ester formulation (AquaCelle) significantly improves eicosapentaenoic and docosahexaenoic acid bioavailability in healthy adults. Eur. J. Nutr. 2019, 59, 2729–2737.
- Briskey, D.; Rao, A. Trans-Resveratrol Oral Bioavailability in Humans Using LipiSperse™ Dispersion Technology. Pharmaceutics 2020, 12, 1190.
- Panya, A.; Laguerre, M.; Lecomte, J.; Villeneuve, P.; Weiss, J.; McClements, D.J.; Decker, E.A. Effects of Chitosan and Rosmarinate Esters on the Physical and Oxidative Stability of Liposomes. J. Agric. Food Chem. 2010, 58, 5679–5684.
- Lopes, N.A.; Pinilla, C.M.B.; Brandelli, A. Pectin and polygalacturonic acid-coated liposomes as novel delivery system for nisin: Preparation, characterization and release behavior. Food Hydrocoll. 2017, 70, 1–7.
- Lopes, N.A.; Pinilla, C.M.B.; Brandelli, A. Antimicrobial activity of lysozyme-nisin co-encapsulated in liposomes coated with polysaccharides. Food Hydrocoll. 2019, 93, 1–9.
- Azzi, J.; Jraij, A.; Auezova, L.; Fourmentin, S.; Greige-Gerges, H. Novel findings for quercetin encapsulation and preservation with cyclodextrins, liposomes, and drug-in-cyclodextrin-in-liposomes. Food Hydrocoll. 2018, 81, 328–340.
- Cai, X.; Fang, Z.; Dou, J.; Yu, A.; Zhai, G. Bioavailability of Quercetin: Problems and Promises. Curr. Med. Chem. 2013, 20, 2572–2582.
- Pu, C.; Tang, W.; Li, X.; Li, M.; Sun, Q. Stability enhancement efficiency of surface decoration on curcumin-loaded liposomes: Comparison of guar gum and its cationic counterpart. Food Hydrocoll. 2019, 87, 29–37.
- Wu, Y.; Mou, B.; Song, S.; Tan, C.P.; Lai, O.-M.; Shen, C.; Cheong, L.-Z. Curcumin-loaded liposomes prepared from bovine milk and krill phospholipids: Effects of chemical composition on storage stability, in-vitro digestibility and anti-hyperglycemic properties. Food Res. Int. 2020, 136, 109301.
- Burri, L.; Hoem, N.; Banni, S.; Berge, K. Marine Omega-3 Phospholipids: Metabolism and Biological Activities. Int. J. Mol. Sci. 2012, 13, 15401–15419.
- Brglez Mojzer, E.; Knez Hrnčič, M.; Škerget, M.; Knez, Ž.; Bren, U. Polyphenols: Extraction Methods, Antioxidative Action, Bioavailability and Anticarcinogenic Effects. Molecules 2016, 21, 901.
- Abbas, M.; Saeed, F.; Anjum, F.M.; Afzaal, M.; Tufail, T.; Bashir, M.S.; Ishtiaq, A.; Hussain, S.; Suleria, H.A.R. Natural polyphenols: An overview. Int. J. Food Prop. 2017, 20, 1689–1699.
- Lee, M.-K. Liposomes for enhanced bioavailability of water-insoluble drugs: In vivo evidence and recent approaches. Pharmaceutics 2020, 12, 264.
- Lopes, M.A.; Abrahim, B.A.; Cabral, L.M.; Rodrigues, C.R.; Seiça, R.; Veiga, F.; Ribeiro, A.J. Intestinal absorption of insulin nanoparticles: Contribution of M cells. Nanomed. Nanotechnol. Biol. Med. 2014, 10, 1139–1151.
- Ipar, V.S.; Dsouza, A.; Devarajan, P.V. Enhancing Curcumin Oral Bioavailability Through Nanoformulations. Eur. J. Drug Metab. Pharmacokinet. 2019, 44, 459–480.
- Gopi, S.; Amalraj, A.; Jacob, J.; Kalarikkal, N.; Thomas, S.; Guo, Q. Preparation, characterization and in vitro study of liposomal curcumin powder by cost effective nanofiber weaving technology. New J. Chem. 2018, 42, 5117–5127.
- Hädicke, A.; Blume, A. Interactions of Pluronic block copolymers with lipid vesicles depend on lipid phase and Pluronic aggregation state. Colloid Polym. Sci. 2014, 293, 267–276.
- Li, Z.-L.; Peng, S.; Chen, X.; Zhu, Y.-Q.; Zou, L.-Q.; Liu, W.; Liu, C.-M. Pluronics modified liposomes for curcumin encapsulation: Sustained release, stability and bioaccessibility. Food Res. Int. 2018, 108, 246–253.
- Li, Z.; Xiong, X.; Peng, S.; Chen, X.; Liu, W.; Liu, C. Novel folated pluronic F127 modified liposomes for delivery of curcumin: Preparation, release, and cytotoxicity. J. Microencapsul. 2020, 37, 220–229.
- Cai, Z.-Y.; Li, X.-M.; Liang, J.-P.; Xiang, L.-P.; Wang, K.-R.; Shi, Y.-L.; Yang, R.; Shi, M.; Ye, J.-H.; Lu, J.-L.; et al. Bioavailability of Tea Catechins and Its Improvement. Molecules 2018, 23, 2346.
- Ezzat, H.M.; Elnaggar, Y.S.R.; Abdallah, O.Y. Improved oral bioavailability of the anticancer drug catechin using chitosomes: Design, in-vitro appraisal and in-vivo studies. Int. J. Pharm. 2019, 565, 488–498.
- Fornasier, M.; Pireddu, R.; Del Giudice, A.; Sinico, C.; Nylander, T.; Schillén, K.; Galantini, L.; Murgia, S. Tuning lipid structure by bile salts: Hexosomes for topical administration of catechin. Colloids Surf. B Biointerfaces 2021, 199, 111564.
- Caddeo, C.; Gabriele, M.; Nácher, A.; Fernàndez-Busquets, X.; Valenti, D.; Fadda, A.M.; Pucci, L.; Manconi, M. Resveratrol and artemisinin eudragit-coated liposomes: A strategy to tackle intestinal tumors. Int. J. Pharm. 2021, 592, 120083.
- Jagwani, S.; Jalalpure, S.S.; Dhamecha, D.; Jadhav, K.; A Bohara, R. Pharmacokinetic and Pharmacodynamic Evaluation of Resveratrol Loaded Cationic Liposomes for Targeting Hepatocellular Carcinoma. ACS Biomater. Sci. Eng. 2020, 6, 4969–4984.
- Jiao, Z.; Wang, X.; Han, S.; Zha, X.; Xia, J. Preparation of vitamin C liposomes by rapid expansion of supercritical solution process: Experiments and optimization. J. Drug Deliv. Sci. Technol. 2019, 51, 1–6.
- Silva, G.S.; Jange, C.G.; Rocha, J.S.; Chaves, M.A.; Pinho, S.C. Characterisation of curcumin-loaded proliposomes produced by coating of micronised sucrose and hydration of phospholipid powders to obtain multilamellar liposomes. Int. J. Food Sci. Technol. 2016, 52, 772–780.
- Chaves, M.A.; Pinho, S.C. Curcumin-loaded proliposomes produced by the coating of micronized sucrose: Influence of the type of phospholipid on the physicochemical characteristics of powders and on the liposomes obtained by hydration. Food Chem. 2019, 291, 7–15.
- Ren, J.; Fang, Z.; Jiang, L.; Du, Q. Quercetin-containing self-assemble proliposome preparation and evaluation. J. Liposome Res. 2016, 27, 335–342.
- Desai, M.P.; Labhasetwar, V.; Amidon, G.L.; Levy, R.J. Gastrointestinal Uptake of Biodegradable Microparticles: Effect of Particle Size. Pharm. Res. 1996, 13, 1838–1845.
- Hızır-Kadı, I.; Gültekin-Özgüven, M.; Altin, G.; Demircan, E.; Özçelik, B. Liposomal nanodelivery systems generated from proliposomes for pollen extract with improved solubility and in vitro bioaccessibility. Heliyon 2020, 6, 05030.
- Niu, M.; Tan, Y.; Guan, P.; Hovgaard, L.; Lu, Y.; Qi, J.; Lian, R.; Li, X.; Wu, W. Enhanced oral absorption of insulin-loaded liposomes containing bile salts: A mechanistic study. Int. J. Pharm. 2014, 460, 119–130.
- Ma, H.; Chen, H.; Sun, L.; Tong, L.; Zhang, T. Improving permeability and oral absorption of mangiferin by phospholipid complexation. Fitoterapia 2014, 93, 54–61.
- Thiengkaew, P.; Thanitwatthanasak, S.; Srisala, S.; Jittorntrum, B.; Chunhabundit, R.; Chitprasert, P. Response surface optimization of microfluidic formulations of nanobilosomes for enhancement of aqueous solubility, digestive stability, and cellular antioxidant activity of mangiferin. Food Chem. 2021, 351, 129315.
This entry is offline, you can click here to edit this entry!
