Colitis-complex diarrhea (CCD) in pigs can be defined as a type of diarrhea, which is associated with colonic inflammation and disrupted colonic gut barrier functionality as well as infiltration of neutrophils at the inflamed colon in growing pigs (4–16 weeks post-weaning). It is a challenge for the pig industry as it is associated with the high use of antibiotics, reduced animal welfare, and depressed growth rate. The exact etiology of CCD is still unclear; however, pathogens including Brachyspira (B.) hyodysenteriae, B. pilosicoli, and swine whipworms such as Trichuris (T.) suis have been involved in specific colitis (SC). In the absence of specific pathogens, dietary factors, such as high levels of protein, pelleted feedstuffs, and lack of sufficient antioxidants, can result in non-specific colitis (NSC). On the other hand, supplement of polyunsaturated fatty acids (PUFA) and polyphenols, sufficient supply of essential amino acids (e.g., threonine, cysteine, and proline), short-chain fatty acids (SCFA; especially butyrate), and resistant starch have shown to confer preventing/ameliorating effects on CCD.
- colonic inflammation
- adult pigs
- biomarkers
- dietary strategies
- specific colitis
- non-specific colitis
1. Colonic Epithelium Function
1.1. Colonic Ion Exchange
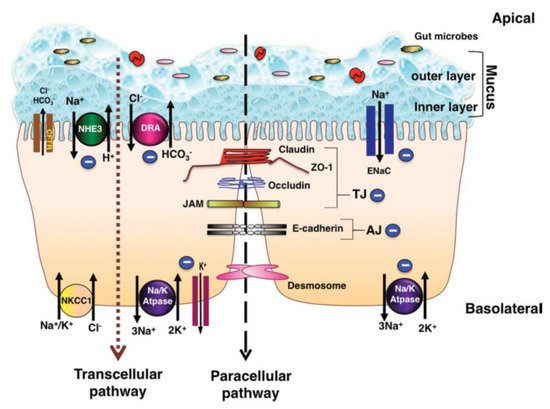
1.2. Cytokines and Luminal Function
1.3. Mucins
2. Colitis
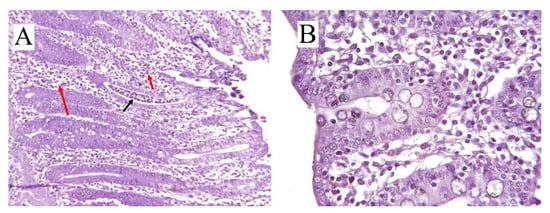
2.1. Specific Colitis
| Type of Colitis | Causative Factor | Affected Site | Pigs Age, Week | Mechanism of Action | Clinical Signs | Gross Lesion | References |
|---|---|---|---|---|---|---|---|
| SC | |||||||
| Swine dysentery | B. hyodysenteriae | Cecum and colon | 6–18 | Absorptive dysfunctionality, hemolysins, and degradative enzymes | Loose stool, mucoid, hemorrhagic diarrhea, dehydration, and retarded growth rate | Inflamed epithelium, mucosal damage, hyperplasia of the crypts, and spirochetal attachment | [31][32][33][34] |
| Spirochetal colitis | B. pilosicoli | Cecum and colon | 4–20 | Absorptive dysfunctionality | Mild non-hemorrhagic, mucoid diarrhea, retarded growth rate | Inflamed epithelium, moderate catarrhal colitis, flaccid and thin luminal wall, appearance of small adherent nodules of digesta | [9][35][33][34][36] |
| Parasitic colitis | T. suis | Cecum and spiral colon | 4–10 | Stimulation of the epithelium and cascading inflammatory responses by hatched eggs and adult worms | Dark loose stool, mucoid to hemorrhagic diarrhea, dehydration, anorexia, and increased feed conversion ratio | Crypt hyperplasia, goblet cell hyperplasia, a general hypertrophy of mucosa, and presence of bipolar eggs | [37][38][39][40][41] |
| NSC | Dietary factors | Cecum and colon | 4–12 | Absorptive dysfunctionality and increased epithelial permeability | Loose and mucoid, non-hemorrhagic diarrhea | Mucosal hyperplasia, mononuclear cell infiltration, multifocal mucosal erosions, increased crypt depth | [33][23][30][42] |
2.1.1. Swine Dysentery
| Biomarkers | Type | Direction | Recovery Site | Causative Factor | Affected Site | Reference |
|---|---|---|---|---|---|---|
| MUC2 and MUC5AC | Mucin | Increased expression | Feces | Colitis, swine dysentry | Large intestine | [17][21][22] |
| LPS 1 | Saccharide | Increased expression | Serum | Gram-negative pathogens, e.g., B. hyodysenteriae | Small and large intestine | [48][49][50][51] |
| Calprotectin and lactoferrin | Protein | Increased expression | Feces and serum | Colitis and inflammatory factors | Large intestine | [52][53][54][55] |
| Na+, Cl−, HCO3-, and K+ | Ion | Reduced absorption and increased luminal accumulation | Feces | B. hyodysenteriae | Large intestine | [9][5][12][44] |
| TNF-α 2, IFN-γ 3, IL-1β 4, IL-6 5, and IL-10 6 | Cytokines | Increased expression | Serum and mucus | Pathogens | Small and large intestine | [1][5][12][23][50][56] |
| NF-κB 7 | Protein | Increased expression in macrophages and in epithelial cells | Serum | IL-1β and TNF-α, LPS, and ROS 8 | Epithelial cells of inflamed colon | [51][56][57][58][59] |
| CRP 8, HP 9, and pig-MAP 10 | Protein | Increased concentration | Serum | LPS, IL-1β, and TNF-α | Epithelial cells of colon, and hepatic cells | [50] |
| FRAP 11, TBARS 12, and ROS 13 | TAC 14 assay | Increased expression | Serum | Oxidative stress | Epithelial cells of colon | [60][61] |
| TEAC 15, CUPRAC 16, AOPP 17, and H2O2 | TAC assay | Increased expression | Saliva | Oxidative stress | Epithelial cells of colon | [61][62] |
2.1.2. Spirochetal Colitis
2.1.3. Parasitic Colitis
2.2. Non-Specific Colitis and Role of Dietary Factors in Inducing Colitis
| Factor | Level | Effect | Reference |
|---|---|---|---|
| Trypsin inhibitor | High | Increased undegraded protein in large intestine and inflammation and causing NSC 1 | [33] |
| Vitamin C and E, glutathione, ubiquinol, polyphenols, and β-carotene | Insufficient | Oxidative distress | [33][59][73][74] |
| Essential amino acids | Insufficient | Oxidative stress by reducing antioxidant enzymes, reduced mucin production | [16][17][42][75] |
| Dietary protein | ≥23% | Increased undegraded protein in large intestine and inflammation due to NH4+, reducing gut barrier function and causing NSC | [23][29][42][76] |
| Soluble NSP 2 and RS 3 | Increased | Ameliorating/preventive effect on large intestinal inflammation, increased SCFA 4, and reduced luminal pH | [77][78][79][80][81][82] |
| Pelleted diet | - | Reduced endogenous enzymes in feedstuffs and causing NSC | [68][83] |
2.2.1. Dietary Crude Protein and NSC
This entry is adapted from the peer-reviewed paper 10.3390/ani11072151
References
- Yang, H.; Jiang, W.; Furth, E.E.; Wen, X.; Katz, J.P.; Sellon, R.K.; Silberg, D.G.; Antalis, T.M.; Schweinfest, C.W.; Wu, G.D. Intestinal inflammation reduces expression of DRA, a transporter responsible for congenital chloride diarrhea. Am. J. Physiol. Gastrointest. Liver Physiol. 1998, 275, G1445–G1453.
- Field, M. Intestinal ion transport and the pathophysiology of diarrhea. J. Clin. Investig. 2003, 111, 931–943.
- Surawicz, C.M. Mechanisms of diarrhea. Curr. Gastroenterol. Rep. 2010, 12, 236–241.
- Binder, H.J. Mechanisms of diarrhea in inflammatory bowel diseases. Ann. N. Y. Acad. Sci. 2009, 1165, 285–293.
- Anbazhagan, A.N.; Priyamvada, S.; Alrefai, W.A.; Dudeja, P.K. Pathophysiology of IBD associated diarrhea. Tissue Barriers 2018, 6, e1463897.
- Musch, M.W.; Arvans, D.L.; Wu, G.D.; Chang, E.B. Functional coupling of the downregulated in adenoma Cl-/base exchanger DRA and the apical Na+/H+ exchangers NHE2 and NHE3. Am. J. Physiol. Gastrointest. Liver Physiol. 2009, 296, G202–G210.
- Khurana, S.; Ganguly, N.K.; Khullar, M.; Panigrahi, D.; Walia, B.N.S. Studies on the mechanism of Salmonella typhimurium enterotoxin-induced diarrhoea. Biochim. Biophys. Acta Mol. Basis Dis. 1991, 1097, 171–176.
- Kaur, T.; Singh, S.; Gorowara, S.; Ganguly, N.K. Role of enteric nervous system in Shigella dysenteriae type 1 toxin-induced fluid secretion in rabbit ileum. J. Diarrhoeal Dis. Res. 1995, 159–165.
- Moeser, A.J.; Blikslager, A.T. Mechanisms of porcine diarrheal disease. J. Am. Vet. Med. Assoc. 2007, 231, 56–67.
- Vernia, P.; Gnaedinger, A.; Hauck, W.; Breuer, R.I. Organic anions and the diarrhea of inflammatory bowel disease. Dig. Dis. Sci. 1988, 33, 1353–1358.
- Madara, J.L.; Stafford, J. Interferon-gamma directly affects barrier function of cultured intestinal epithelial monolayers. J. Clin. Investig. 1989, 83, 724–727.
- Amasheh, S.; Barmeyer, C.; Koch, C.S.; Tavalali, S.; Mankertz, J.; Epple, H.-J.; Gehring, M.M.; Florian, P.; Kroesen, A.-J.; Zeitz, M.; et al. Cytokine-dependent transcriptional down-regulation of epithelial sodium channel in ulcerative colitis. Gastroenterology 2004, 126, 1711–1720.
- Liu, Y. Fatty acids, inflammation and intestinal health in pigs. J. Anim. Sci. Biotechnol. 2015, 6, 41.
- Schulzke, J.D.; Ploeger, S.; Amasheh, M.; Fromm, A.; Zeissig, S.; Troeger, H.; Richter, J.; Bojarski, C.; Schumann, M.; Fromm, M. Epithelial tight junctions in intestinal inflammation. Ann. N. Y. Acad. Sci. 2009, 1165, 294–300.
- Caruso, R.; Lo, B.C.; Núñez, G. Host–microbiota interactions in inflammatory bowel disease. Nat. Rev. Immunol. 2020, 20, 411–426.
- Faure, M.; Mettraux, C.; Moennoz, D.; Godin, J.-P.; Vuichoud, J.; Rochat, F.; Breuillé, D.; Obled, C.; Corthésy-Theulaz, I. Specific amino acids increase mucin synthesis and microbiota in dextran sulfate sodium–treated rats. J. Nutr. 2006, 136, 1558–1564.
- Puiman, P.J.; Jensen, M.; Stoll, B.; Renes, I.B.; de Bruijn, A.C.J.M.; Dorst, K.; Schierbeek, H.; Schmidt, M.; Boehm, G.; Burrin, D.G.; et al. Intestinal Threonine Utilization for Protein and Mucin Synthesis Is Decreased in Formula-Fed Preterm Pigs. J. Nutr. 2011, 141, 1306–1311.
- Van der Sluis, M.; De Koning, B.A.; De Bruijn, A.C.; Velcich, A.; Meijerink, J.P.; Van Goudoever, J.B.; Büller, H.A.; Dekker, J.; Van Seuningen, I.; Renes, I.B. Muc2-deficient mice spontaneously develop colitis, indicating that MUC2 is critical for colonic protection. Gastroenteritis 2006, 131, 117–129.
- Specian, R.D.; Oliver, M.G. Functional biology of intestinal goblet cells. Am. J. Physiol. Cell Physiol. 1991, 260, C183–C193.
- Fuss, I.J.; Strober, W. Chapter 81—Ulcerative Colitis. In Mucosal Immunology, 4th ed.; Mestecky, J., Strober, W., Russell, M.W., Kelsall, B.L., Cheroutre, H., Lambrecht, B.N., Eds.; Academic Press: Boston, MA, USA, 2015; pp. 1573–1612.
- Martin, C.R.; Walker, W.A. Chapter 70—Innate and Mucosal Immunity in the Developing Gastrointestinal Tract: Relationship to Early and Later Disease. In Avery’s Diseases of the Newborn, 9th ed.; Gleason, C.A., Devaskar, S.U., Eds.; W.B. Saunders: Philadelpia, PA, USA, 2012; pp. 994–1006.
- Bengtsson, R.J.; MacIntyre, N.; Guthrie, J.; Wilson, A.D.; Finlayson, H.; Matika, O.; Pong-Wong, R.; Smith, S.H.; Archibald, A.L.; Ait-Ali, T. Lawsonia intracellularis infection of intestinal crypt cells is associated with specific depletion of secreted MUC2 in goblet cells. Vet. Immunol. Immunopathol. 2015, 168, 61–67.
- Wu, Y.; Jiang, Z.; Zheng, C.; Wang, L.; Zhu, C.; Yang, X.; Wen, X.; Ma, X. Effects of protein sources and levels in antibiotic-free diets on diarrhea, intestinal morphology, and expression of tight junctions in weaned piglets. Anim. Nutr. 2015, 1, 170–176.
- Lauridsen, C. From oxidative stress to inflammation: Redox balance and immune system. Poult. Sci. 2019, 98, 4240–4246.
- Stege, H.; Jensen, T.K.; Møller, K.; Baekbo, P.; Jorsal, S. Risk factors for intestinal pathogens in Danish finishing pig herds. Prev. Vet. Med. 2001, 50, 153–164.
- Pedersen, K.S.; Johansen, M.; Angen, O.; Jorsal, S.E.; Nielsen, J.P.; Jensen, T.K.; Guedes, R.; Ståhl, M.; Bækbo, P. Herd diagnosis of low pathogen diarrhoea in growing pigs—A pilot study. Ir. Vet. J. 2014, 67, 24.
- Thomson, J.; Smith, W.J.; Fowler, V.R.; Edwards, S.; Hazzledine, M. Non-specific colitis in pigs: Defining the condition. In Proceedings of the 17th International Pig Veterinary Society Congress, Ames, IA, USA, 2–5 June 2002.
- Constable, P.; Hinchcliff, K.; Done, S.; Grünberg, W. 7—Diseases of the Alimentary Tract: Nonruminant. In Veterinary Medicine, 11st ed.; Constable, P.D., Hinchcliff, K.W., Done, S.H., Grünberg, W., Eds.; W.B. Saunders: Philadelphia, PA, USA, 2017; pp. 175–435.
- Luise, D.; Lauridsen, C.; Bosi, P.; Trevisi, P. Methodology and application of Escherichia coli F4 and F18 encoding infection models in post-weaning pigs. J. Anim. Sci. Biotechnol. 2019, 10, 53.
- Burrough, E.R. Swine Dysentery: Etiopathogenesis and Diagnosis of a Reemerging Disease. Vet. Pathol. 2016, 54, 22–31.
- Zachary, J.F. Chapter 4—Mechanisms of Microbial Infections1. In Pathologic Basis of Veterinary Disease, 6th ed.; Zachary, J.F., Ed.; Mosby: Maryland Heights, MO, USA, 2017; pp. 132–241.e1.
- Hampson, D.J.; Burrough, E.R. Swine Dysentery and Brachyspiral Colitis. In Diseases of Swine; Zimmerman, J.J., Karriker, L.A., Ramirez, A., Schwartz, K.J., Stevenson, G.W., Zhang, J., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2019; pp. 951–970.
- Carr, J.; Chen, S.-P.; Connor, J.F.; Kirkwood, R.; Segalés, J. Pig Health; CRC Press: Boca Raton, FL, USA, 2018.
- Stanton, T.B. The Genus Brachyspira. Prokar 2006, 7, 330–356.
- Jensen, T.; Boye, M.; Møller, K. Extensive intestinal spirochaetosis in pigs challenged with Brachyspira pilosicoli. J. Med. Microbiol. 2004, 53, 309–312.
- Trott, D.J.; Huxtable, C.R.; Hampson, D.J. Experimental infection of newly weaned pigs with human and porcine strains of Serpulina pilosicoli. Infect. Immun. 1996, 64, 4648–4654.
- Lawhorn, D.B. Diarrheal disease in show swine. Tex. FARMER Collect. 2007, 3, 439.
- Kringel, H.; Iburg, T.; Dawson, H.; Aasted, B.; Roepstorff, A. A time course study of immunological responses in Trichuris suis infected pigs demonstrates induction of a local type 2 response associated with worm burden. Int. J. Parasitol. 2006, 36, 915–924.
- Pittman, J.S.; Shepherd, G.; Thacker, B.J.; Myers, G.H. Trichuris suis in finishing pigs: Case report and review. J. Swine Health Prod. 2010, 18, 306–313.
- Roepstorff, A.; Mejer, H.; Nejsum, P.; Thamsborg, S.M. Helminth parasites in pigs: New challenges in pig production and current research highlights. Vet. Parasitol. 2011, 180, 72–81.
- Leroux, L.-P.; Nasr, M.; Valanparambil, R.; Tam, M.; Rosa, B.A.; Siciliani, E.; Hill, D.E.; Zarlenga, D.S.; Jaramillo, M.; Weinstock, J.V.; et al. Analysis of the Trichuris suis excretory/secretory proteins as a function of life cycle stage and their immunomodulatory properties. Sci. Rep. 2018, 8, 15921.
- Thomson, J. Feed-associated colitis of growing pigs and its interaction with enteric infections. Acta Sci. Vet. 2009, 37, s1–s9.
- Gelberg, H.B. Chapter 7—Alimentary System and the Peritoneum, Omentum, Mesentery, and Peritoneal Cavity1. In Pathologic Basis of Veterinary Disease, 6th ed.; Zachary, J.F., Ed.; Mosby: Maryland Heights, MO, USA, 2017; pp. 324–411.e1.
- Argenzio, R.A.; Whipp, S.C.; Glock, R.D. Pathophysiology of Swine Dysentery: Colonic Transport and Permeability Studies. J. Infect. Dis. 1980, 142, 676–684.
- Stege, H.; Jensen, T.K.; Møller, K.; Bækbo, P.; Jorsal, S.E. Prevalence of intestinal pathogens in Danish finishing pig herds. Prev. Vet. Med. 2000, 46, 279–292.
- Quintana-Hayashi, M.P.; Mahu, M.; De Pauw, N.; Boyen, F.; Pasmans, F.; Martel, A.; Premaratne, P.; Fernandez, H.R.; Teymournejad, O.; Maele, L.V. The levels of Brachyspira hyodysenteriae binding to porcine colonic mucins differ between individuals, and binding is increased to mucins from infected pigs with de novo MUC5AC synthesis. Infect. Immun. 2015, 83, 1610–1619.
- Jin, L.; Reynolds, L.P.; Redmer, D.A.; Caton, J.S.; Crenshaw, J.D. Effects of dietary fiber on intestinal growth, cell proliferation, and morphology in growing pigs2. J. Anim. Sci. 1994, 72, 2270–2278.
- Pedersen, K.S.; Kristensen, C.S.; Nielsen, J.P. Demonstration of non-specific colitis and increased crypt depth in colon of weaned pigs with diarrhea. Vet. Q. 2012, 32, 45–49.
- Casas, V.; Vadillo, S.; San Juan, C.; Carrascal, M.; Abian, J. The Exposed Proteomes of Brachyspira hyodysenteriae and B. pilosicoli. Front. Microbiol. 2016, 7, 1103.
- Wyns, H.; Plessers, E.; De Backer, P.; Meyer, E.; Croubels, S. In vivo porcine lipopolysaccharide inflammation models to study immunomodulation of drugs. Vet. Immunol. Immunopathol. 2015, 166, 58–69.
- Gessner, D.K.; Fiesel, A.; Most, E.; Dinges, J.; Wen, G.; Ringseis, R.; Eder, K. Supplementation of a grape seed and grape marc meal extract decreases activities of the oxidative stress-responsive transcription factors NF-κB and Nrf2 in the duodenal mucosa of pigs. Acta Vet. Scand. 2013, 55, 18.
- Lallès, J.-P.; Fagerhol, M.K. Faecal calprotectin: A non invasive marker of inflammation in pigs. ISAH 2005, 1, 405–408.
- Lamb, C.A.; Mansfield, J.C. Measurement of faecal calprotectin and lactoferrin in inflammatory bowel disease. Frontline Gastroenterol. 2011, 2, 13.
- Yamamoto, T.; Shiraki, M.; Bamba, T.; Umegae, S.; Matsumoto, K. Faecal calprotectin and lactoferrin as markers for monitoring disease activity and predicting clinical recurrence in patients with Crohn’s disease after ileocolonic resection: A prospective pilot study. United Eur. Gastroenterol. J. 2013, 1, 368–374.
- Bogere, P.; Choi, Y.J.; Heo, J. Optimization of Fecal Calprotectin Assay for Pig Samples. J. Agric. Life Sci. 2019, 53, 93–104.
- Jobin, C.; Hellerbrand, C.; Licato, L.L.; Brenner, D.A.; Sartor, R.B. Mediation by NF-kappa B of cytokine induced expression of intercellular adhesion molecule 1 (ICAM-1) in an intestinal epithelial cell line, a process blocked by proteasome inhibitors. Gut 1998, 42, 779–787.
- Rogler, G.; Brand, K.; Vogl, D.; Page, S.; Hofmeister, R.; Andus, T.; Knuechel, R.; Baeuerle, P.A.; Schölmerich, J.; Gross, V. Nuclear factor κB is activated in macrophages and epithelial cells of inflamed intestinal mucosa. Gastroenterology 1998, 115, 357–369.
- Ungaro, F.; Rubbino, F.; Danese, S.; D’Alessio, S. Actors and Factors in the Resolution of Intestinal Inflammation: Lipid Mediators as a New Approach to Therapy in Inflammatory Bowel Diseases. Front. Immunol. 2017, 8, 1331.
- Pistol, G.C.; Marin, D.E.; Rotar, M.C.; Ropota, M.; Taranu, I. Bioactive compounds from dietary whole grape seed meal improved colonic inflammation via inhibition of MAPKs and NF-κB signaling in pigs with DSS induced colitis. J. Funct. Foods 2020, 66, 103708.
- Tan, C.; Wei, H.; Sun, H.; Ao, J.; Long, G.; Jiang, S.; Peng, J. Effects of dietary supplementation of oregano essential oil to sows on oxidative stress status, lactation feed intake of sows, and piglet performance. BioMed Res. Int. 2015, 2015, 525218.
- Schuh, S.; Muller, L.K.; Campos, L.P.; Moresco, R.N.; Baldissera, M.D.; de Oliveira, S.C.; Campigotto, G.; Da Silva, A.S.; Paiano, D. Effect of supplementation of newborn piglets with spray dry blood plasma on weight gain and serum biochemical variables. Comp. Clin. Path. 2016, 25, 1029–1033.
- Rubio, C.P.; Mainau, E.; Cerón, J.J.; Contreras-Aguilar, M.D.; Martínez-Subiela, S.; Navarro, E.; Tecles, F.; Manteca, X.; Escribano, D. Biomarkers of oxidative stress in saliva in pigs: Analytical validation and changes in lactation. BMC Vet. Res. 2019, 15, 144.
- Pothoulakis, C.; Castagliuolo, I.; LaMont, J.T. Nerves and intestinal mast cells modulate responses to enterotoxins. Physics 1998, 13, 58–63.
- Argenzio, R.A. Glucose-stimulated fluid absorption in the pig small intestine during the early stage of swine dysentery. Am. J. Vet. Res. 1980, 41, 2000–2006.
- Laber, K.E.; Whary, M.T.; Bingel, S.A.; Goodrich, J.A.; Smith, A.C.; Swindle, M.M. Chapter 15—Biology and Diseases of Swine. In Laboratory Animal Medicine, 2nd ed.; Fox, J.G., Anderson, L.C., Loew, F.M., Quimby, F.W., Eds.; Academic Press: Burlington, NJ, USA, 2002; pp. 615–673.
- Sharma, J.N.; Al-Omran, A.; Parvathy, S.S. Role of nitric oxide in inflammatory diseases. Inflammopharmacology 2007, 15, 252–259.
- Hill, D.E.; Romanowski, R.D.; Urban, J.F. A Trichuris specific diagnostic antigen from culture fluids of Trichuris suis adult worms. Vet. Parasitol. 1997, 68, 91–102.
- Chase-Topping, M.E.; Gunn, G.; Strachan, W.D.; Edwards, S.A.; Smith, W.J.; Hillman, K.; Stefopoulou, S.N.; Thomson, J.R. Epidemiology of porcine non-specific colitis on Scottish farms. Vet. J. 2007, 173, 353–360.
- Taylor-Pickard, J.A.; Stevenson, Z.; Glebocka, K. Formula for the Future: Nutrition or Pathology? Elevating Performance and Health in Pigs and Poultry; Wageningen Academic Pub: Wageningen, The Netherlands, 2008.
- Luecke, R.; McMillen, W.; Thorp, F., Jr.; Tull, C. The relationship of nicotinic acid, tryptophane and protein in the nutrition of the pig. J. Nutr. 1947, 33, 251–261.
- Si, Y.; Zhang, Y.; Zhao, J.; Guo, S.; Zhai, L.; Yao, S.; Sang, H.; Yang, N.; Song, G.; Gu, J.; et al. Niacin Inhibits Vascular Inflammation via Downregulating Nuclear Transcription Factor-B Signaling Pathway. Mediat. Inflamm. 2014, 2014, 263786.
- Salem, H.; Wadie, W. Effect of Niacin on Inflammation and Angiogenesis in a Murine Model of Ulcerative Colitis. Sci. Rep. 2017, 7, 7139.
- Thérond, P.; Bonnefont-Rousselot, D.; Davit-Spraul, A.; Conti, M.; Legrand, A. Biomarkers of oxidative stress: An analytical approach. Curr. Opin. Clin. Nutr. Metab. Care 2000, 3, 373–384.
- Lowe, F. Biomarkers of Oxidative Stress. Syst. Biol. Free Radic. Antioxid. 2014, 22.
- Toledo, J.B.; Furlan, A.C.; Pozza, P.C.; Piano, L.M.; Carvalho, P.L.O.; Peñuela-Sierra, L.M.; Huepa, L.M.D. Effect of the reduction of the crude protein content of diets supplemented with essential amino acids on the performance of piglets weighing 6–15kg. Livest. Sci. 2014, 168, 94–101.
- Nyachoti, C.M.; Omogbenigun, F.O.; Rademacher, M.; Blank, G. Performance responses and indicators of gastrointestinal health in early-weaned pigs fed low-protein amino acid-supplemented diets. J. Anim. Sci. 2006, 84, 125–134.
- Molist, F.; van Oostrum, M.; Pérez, J.F.; Mateos, G.G.; Nyachoti, C.M.; van der Aar, P.J. Relevance of functional properties of dietary fibre in diets for weanling pigs. Anim. Feed Sci. Technol. 2014, 189, 1–10.
- Jha, R.; Fouhse, J.M.; Tiwari, U.P.; Li, L.; Willing, B.P. Dietary Fiber and Intestinal Health of Monogastric Animals. Front. Vet. Sci. 2019, 6, 48.
- Regassa, A.; Nyachoti, C.M. Application of resistant starch in swine and poultry diets with particular reference to gut health and function. Anim. Nutr. 2018, 4, 305–310.
- Souza da Silva, C.; van den Borne, J.J.G.C.; Gerrits, W.J.J.; Kemp, B.; Bolhuis, J.E. Effects of dietary fibers with different physicochemical properties on feeding motivation in adult female pigs. Physiol. Behav. 2012, 107, 218–230.
- Haenen, D.; Zhang, J.; Souza da Silva, C.; Bosch, G.; van der Meer, I.M.; van Arkel, J.; van den Borne, J.J.G.C.; Pérez Gutiérrez, O.; Smidt, H.; Kemp, B.; et al. A Diet High in Resistant Starch Modulates Microbiota Composition, SCFA Concentrations, and Gene Expression in Pig Intestine. J. Nutr. 2013, 143, 274–283.
- Yang, X.; Darko, K.O.; Huang, Y.; He, C.; Yang, H.; He, S.; Li, J.; Li, J.; Hocher, B.; Yin, Y. Resistant Starch Regulates Gut Microbiota: Structure, Biochemistry and Cell Signalling. Cell. Physiol. Biochem. 2017, 42, 306–318.
- Thomson, J.R.; Smith, W.J.; Murray, B.P. Investigations into field cases of porcine colitis with particular reference to infection with Serpulina pilosicoli. Vet. Rec. 1998, 142, 235.
- Al-Sadi, R.; Boivin, M.; Ma, T. Mechanism of cytokine modulation of epithelial tight junction barrier. Front. Biosci. J. Virt. Lib. 2009, 14, 2765.
- Pearce, S.C.; Mani, V.; Boddicker, R.L.; Johnson, J.S.; Weber, T.E.; Ross, J.W.; Rhoads, R.P.; Baumgard, L.H.; Gabler, N.K. Heat stress reduces intestinal barrier integrity and favors intestinal glucose transport in growing pigs. PLoS ONE 2013, 8, e70215.
- Berger, A.L.; Ikuma, M.; Welsh, M.J. Normal gating of CFTR requires ATP binding to both nucleotide-binding domains and hydrolysis at the second nucleotide-binding domain. Proc. Natl. Acad. Sci. USA. 2005, 102, 455–460.
