As an alternative to synthetic pesticides, natural chemistries from living organisms, are not harmful to nontarget organisms and the environment, can be used as biopesticides, nontarget. However, to reduce the reactivity of active ingredients, avoid undesired reactions, protect from physical stress, and control or lower the release rate, encapsulation processes can be applied to biopesticides. The use of supercritical fluid technology (SFT), mainly carbon dioxide (CO2), to encapsulate biopesticides is highlighted, as they reduce the use of organic solvents, have simpler separation processes, and achieve high-purity particles.
- encapsulation techniques
- controlled release
- supercritical fluid technology
1. Introduction
Pesticides are any substance or mixture able to repel, eradicate, or mitigate any pest [3]. Since the intensive application of pesticides, agricultural productivity has increased exponentially. According to The Food and Agriculture Organization of the United Nations (FAO) data, approximately 4 million tonnes of pesticides were used globally or sold to the agricultural sector for crops and seeds in 2018.
The negative effects on soil, air, water quality, agricultural products, and nontarget organisms became noticeable, alongside the evolution of pest resistance due to conventional synthetic pesticide use [2,3]. Less harmful alternatives have been used to control pests. As an example, any pest’s natural enemy can be used for biocontrol. Before this application, different species of organisms are tested against the target plant pathogen, and the most active species is considered for biocontrol.
Another alternative to the use of synthetic pesticides is the application of biopesticides, which are based on naturally occurring living organisms, including animals, plants, and microorganisms [2]. The biopesticide can be retained by physical or chemical interactions within a matrix to provide chemical and physical protection against environmental factors and improve biopesticide stability by decreasing the volatility and reactivity of the active ingredient, a process named encapsulation [7,8]. There are several methods of encapsulation, such as emulsification, coacervation, and spray drying [7]. However, the use of supercritical fluid technology to encapsulate particles has also been explored due to its having several advantages, mainly low environmental impact and high purity [9,10]. Carbon dioxide is the most frequently used supercritical fluid since it has low toxicity, low cost, easy removal, and mild critical properties [11].
2. Biopesticides
Biopesticides are types of natural pesticides consisting of natural products from living organisms, such as animals, plants, microorganisms, or genetically engineered organisms [2]. Biopesticides manage agricultural pests based on their biological effects on these organisms [12,13].
Biopesticides can be classified into three main groups: (1) microbial pesticides, including bacteria, algae, fungi, viruses, or protozoa as active ingredients; (2) Plant-Incorporated-Protectants (PIPs), which are substances produced by plants due to changes in the genetic material; and (3) biochemical pesticides, referring to naturally occurring substances that control pests by non-toxic mechanisms [2,12,13].
Despite the low environmental impact of most biopesticides, since they present non-toxic or less toxic constituents, there are some concerns about the commercialization of natural pesticides, such as regulatory requirements, availability of the active ingredient, and stability of the formulation in storage, transport, and during application. To overcome the instability and lower number of active ingredients used, encapsulation technologies have been developed [32,33].
3. Encapsulation Technologies
Encapsulation is a physical process in which a core material or active substance, such as small solid particles, liquid droplets, or gases are entrapped in an encapsulating agent or wall material to completely or partially isolate the core material, improving its stability, by reducing its volatility and reactivity, and maintaining its viability against adverse environmental conditions, such as pH extremes [11,33,34].
To choose the proper encapsulation technique, the desired application, expected particle size distribution, biocompatibility of the particles, physicochemical properties of the active and the wall material, release mechanism, and process costs are evaluated [11]. There are many encapsulation techniques documented for biopesticides, classified by their chemical or physical processes. Among the chemical processes, the most explored are emulsion polymerization, miniemulsion, melt-dispersion, in situ polymerization, and coacervation. As for the physical processes, spray drying, fluidized bed coating, and ionic gelation encompass the most documented encapsulation techniques for biopesticides and natural substances that have the potential to be biopesticides [7,35,42,43,44,45] (Table 2).
| Method | Description | Drawbacks | Particle Size | Materials | Active Component | Reference | |
|---|---|---|---|---|---|---|---|
| Chemical Processes | Emulsion Polymerization | An organic phase is emulsified in an aqueous phase. Polymerization starts with a water-soluble initiator. Fine oil droplets are spontaneously formed when the surfactant moves from the organic phase to the water phase, resulting in oil-in-water (o/w) emulsion [46,47,48]. When an aqueous phase is emulsified in the organic phase of very low polarity, it results in a water-in-oil (w/o) emulsion. This process is referred to as inverse emulsion polymerization [47]. To achieve greater colloidal stability, emulsifiers are added at concentrations equal to or above their critical micelle concentration [35]. | There is a relatively poor understanding of factors influencing the encapsulation process and there are limitations on the type of organic phases and surfactants used [46]. In addition, particles present low thermal stability [49]. | 55–1300 nm | Citrate buffer and medium-chain triglyceride | Carvacrol | [46,50] |
| 112–594 nm | Polyoxyethylene (20, 40, 60, and 80) and castor oil ether | D-limonene | [51] | ||||
| Miniemulsion Polymerization | Fine monomer droplets are produced by the action of high shear (ultrasonic waves or high-speed homogenizer), so polymer particles are obtained via oil-soluble initiators, through droplet nucleation. However, some monomers have slight solubility in water and a co-stabilizer be also used, in addition to surfactants, to avoid coalescence (Ostwald ripening) [35]. | It is a technique still under improvement without complete knowledge of its mechanism. Additionally, it can present a wide particle size distribution [35]. | 53.25–247.6 nm | Polysorbate 80 (Tween® 80) | Melaleuca alternifolia (tea tree oil), Vitis vinifera (grapes seeds oil), and Punica granatum (pomegranate fruit peel oil) | [52] | |
| Chemical Processes | Melt -Dispersion | The active component is melted in water at a temperature above the melting point and emulsified in a high-pressure homogenizer [53]. | Depending on the temperature set, it can volatilize core components [54]. | 240 nm | Poly-ethylene glycol (PEG) | Garlic essential oil | [54] |
| In situ polymerization | Direct polymerization of a solution of monomers or oligomers is carried out on the core material surface. Deposition and precipitation are controlled by precipitants or changes in pH and temperature [7,55]. | There is little knowledge on precise control of the microencapsulation process, affecting the rational design and efficiency of the microcapsules [55] | 20–110 µm | Phenol and formaldehyde | Neem oil | [56] | |
| 30–600 µm | Phenol, formalin, poly(vinyl alcohol) (PVA), butanol and sodium lauryl sulfate, cardanol, xylene, and resorcinol. | Karanja oil | [57] | ||||
| Complex coacervation |
This technique relies upon a decrease in solubility of the coating polymer when a third component is added to the polymer solution. Two oppositely charged polymers form a wall around the active ingredient, due to the complexation of oppositely charged polyelectrolytes [44,58,59,60]. | This technique usually requires toxic reagents for the coacervate shell [60]. | 35–50 µm | Gelatin and gum Arabic | Metarhizium anisopliae | [61] | |
| Physical Processes | Spray Drying | The core material is homogenized with the carrier, usually an aqueous solution, and then set in a spray dryer Tiny droplets are formed and, by contact with the hot gas, water is evaporated, obtaining a powder or granular product [42,62]. | Highly cost equipment and expensive powder recovery process. High heat consumption and low thermal efficiency [63]. | 1.10–2.09 µm | Chitosan and sodium lignosulfonate | Spinosad | [64] |
| 28.84–52.88 µm | PVA, gum Arabic, and whey protein isolate/maltodextrin | Neem seed oil | [65] | ||||
| 15–20 µm | Maltodextrin, gum Arabic | Trichoderma harzianum | [66] | ||||
| Physical Processes | Fluidized Bed Coating (FBC) | In this method, particles with different diameters are moved around in a fluidized bed and sprayed with a liquid. The solution, either aqueous or organic, evaporates and forms a coating layer around the active ingredient [67]. | FBC can be applied to a limited range of active ingredients since it degrades temperature-sensitive active compounds [49]. It presents difficulties in processing needle or platelet-shaped particles [68]. | - | Biomass | Metarhizium brunneum, Cordyceps fumosorosea and Beauveria bassiana | [69] |
| Ionic Gelation | This technique is based on ionic interactions between charged groups of the polymer and charged groups of the crosslinking agent [70,71]. | It can result in nanoparticles and microparticles with a fragile particulate system, high dispersibility index, and few sites to modify the surface for functional moieties attachment [71]. | - | Alginate, CaCl2, and glycerol | H. bacteriophora | [72] | |
| - | Alginate—multivalent counterions (calcium chloride, zinc sulfate, copper sulfate, cobalt chloride, and ferric chloride) | Bacillus thuringiensis var. israelensis | [73] |
4. Biopesticide Encapsulation Based On Supercritical Fluid Technology
| Supercritical Technology | Active Ingredient | scCO2 role | Material | Solvent | Temperature (°C) | Pressure (MPa) | Particle Size | Reference |
|---|---|---|---|---|---|---|---|---|
| RESS | Atractylodes macrocephala essential oil | Solvent | Phosphatidylcholine and cholesterol | Ethanol | 65 | 30 | 173 nm | [83,84] |
| PGSS | Cydia pomonella granulovirus (CpGV) | Solute | Palm oil-based fat, lecithin-based surfactant, and modified TiO2 as a UV protectant. | - | 65 | 10 | <85 µm | [85] |
| Lavandin oil | Solute | OSA starch and PEG | - | 64–74 | 7.6–8.4 | 21–49 µm | [86] | |
| SSI | 1-octen-3-ol | Solvent | Low-Density Polyethylene (LPDE) films | - | 45 | 7.5–14.5 | - | [87] |
| Thymoquinone and R-(+)-pulegone | Solvent | LDPE films | - | 45 | 10–15 | - | [88] | |
| Pyrethrins | Solvent | Polypropylene films | - | 35–55 | 10–40 | - | [89] | |
| SAA | Rotenone | Cosolute | PEG, alginate, and Polyvinylpyrrolidone (PVP) | Acetone, water, and ethyl acetate | 40–85 | 8–11 | 0.6–1.5 µm | [90] |
| SAF | Ryanodol | Antisolvent | - | Ethanol | 35 | 15 | 5 µm | [91] |
| Supercritical Phase Inversion | Quercetin | Solvent | Cellulose acetate | Acetone | 45–50 | 10–20 | - | [92] |
4.1. Rapid Expansion from Supercritical Solution (RESS)
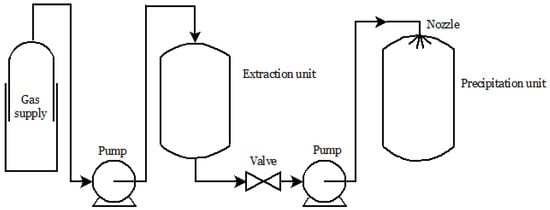
4.2. Particle from Gas Saturated Solutions (PGSS)
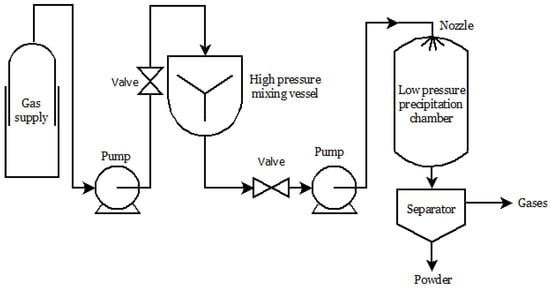
4.3. Supercritical Solvent Impregnation (SSI)
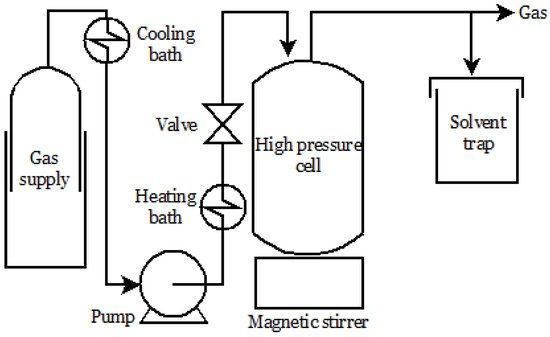
4.4. Supercritical Assisted Atomization (SAA)
4.5. Supercritical Antisolvent Fractionation (SAF)
4.6. Supercritical Phase Inversion and Supercritical Drying
Supercritical Phase Inversion offers the possibility to create porous membranes of polymers that can be loaded with active ingredients. In this technique, a liquid–liquid demixing phenomenon is produced from a polymeric solution by using scCO2 as a non-solvent to steadily remove the organic solvent. The use of scCO2 in this phase inversion avoids the collapse of the pore structure due to its low surface tension and gaslike diffusivity. Moreover, a subsequent membrane drying process is not required. The membrane morphology can be controlled by changing the operating conditions [100] and compounds can be included in the membrane. This process starts with producing polymer solutions by solubilizing the polymer in an organic solvent that should have a proper affinity with scCO2. The final solution is placed in stainless steel caps and processed in a vessel containing scCO2, filled from the bottom up to the desired pressure. Then, this vessel is depressurized, forming dried membranes [101].
4.7. Supercritical Anti-Solvent Precipitation (SAS)
5. Conclusions
This entry is adapted from the peer-reviewed paper 10.3390/molecules26134003
