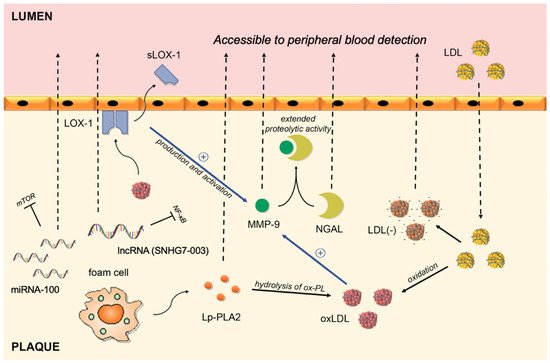
Since atherosclerosis is an everlasting field of research worldwide, there is a multitude of biomarkers whose potential to indicate a vulnerable plaque has been tested. Owing to the pivotal role of lipids and inflammation in the setting of atherosclerosis, most of the biomarkers originated from one of those processes. Conceptually, it is hypothesized that elevated levels of certain lipids will indicate increased susceptibility to the development of unstable atherosclerotic plaque owing to their role in the origin of plaque destabilization. On the other hand, certain molecules can migrate from vulnerable lesion back to circulation, thus creating an opportunity to serve as a biomarker (). In addition, recent development of molecular science brought some novel technologies such as microRNA (miRNA).
2.1. Inflammation-Based Biomarkers
One of the most extensively studied biomarkers in this setting is high sensitivity C-reactive protein (hs-CRP), an established inflammatory biomarker used in everyday practice worldwide [
60]. Aside from the well-known role as the most important acute-phase protein synthetized in the liver, increased CRP levels have been shown to be an independent risk factor for myocardial infarction, predicting CV events better than low-density lipoprotein (LDL) cholesterol [
61,
62,
63]. Studies suggest that apart from the liver, CRP can be synthesized in plaques by macrophages or smooth muscle-like cells [
64]. In line with this, Inoue et al. demonstrated CRP is released both from vulnerable coronary plaques and plaques damaged during percutaneous coronary intervention (PCI), whereas Norja et al. demonstrated that CRP immunoreactivity is associated with the progression of atherosclerotic plaque, especially with the vulnerable coronary plaques [
65,
66]. Apart from representing a biomarker of inflammation, studies suggest that CRP is an effector molecule able to induce a pro-atherothrombotic phenotype in endothelial cells and SMCs and is thus directly implicated in plaque pathophysiology [
67]. Although CRP plasma levels undoubtedly reflect the vulnerability of the plaque, perhaps the biggest setback in its clinical implementation in this setting is its low specificity [
68,
69]. Virtually any inflammatory process in the human body can result in elevated CRP levels. However, CRP could be beneficial as a part of multiple biomarker prognostic score or if used in conjunction with imaging techniques.
Matrix metalloproteinase-9 (MMP-9) belongs to the family of zinc-binding proteolytic enzymes that are capable of degrading most of the extracellular matrix and that take part in all inflammatory processes in humans [
70]. Expression of MMP-9 is markedly upregulated in human macrophages stimulated by oxidized low-density lipoprotein (ox-LDL), suggesting its contribution to matrix degradation in the atherosclerotic plaque and making it susceptible to rupture and/or vascular remodeling [
71,
72]. In line with this, studies on carotid plaques indicate that MMP-9 expression in carotid plaques is higher in vulnerable/symptomatic plaques in comparison to stable ones, whereas serum MMP-9 levels are significantly higher in patients with atheromatous plaques in contrast to patients with fibrous plaques [
73,
74,
75,
76]. Furthermore, multiple studies suggest that serum MMP-9 levels are elevated in patients with unstable plaques and Ferroni et al. suggest that MMP-9 serum levels might even provide an index of plaque activity in the setting of coronary artery disease (CAD) [
77,
78,
79]. Notably, certain polymorphisms of MMP-9 confer a susceptibility risk for CAD. The most consistent evidence with respect to association of MMP-9 polymorphisms and CAD is that regarding the C1562T polymorphism. In a recent meta-analysis, Hassanzadeh-Makoui et al. demonstrated that MMP-9 (C1562T) polymorphism was associated with increased risk of CAD susceptibility in the overall analysis, but markedly in the Asian population [
80]. The pathophysiological background of this association is the fact that this variant of MMP-9 decreases binding potential of the proteins involved in the inhibition of transcription to the DNA sequence, thus playing a role in orchestrating the transcription activity of MMP-9 [
81]. Rather interestingly, this polymorphism exerts a protective role in a wide spectrum of diseases, such as diabetic nephropathy and anterior open bite [
82,
83]. In a large prospective trial (
n = 1127) that included patients with CAD, the association between MMP-9 levels and the risk of fatal CV events showed a hazard ratio of 1.3, even after adjustment for confounders in terms of therapy and other clinical confounders whereas multivariate regression analysis by Ezhov et al. disclosed that MMP-9 is a strong independent predictor of plaque instability in stable CAD patients [
84]. An important line of evidence with regard to MMP-9 implementation was brought by Wang et al. who showed that, in patients presenting with unstable angina, serum MMP-9 levels may discriminate patients who have unstable plaques from patients who do not have plaques, directly implicating a viable clinical usefulness of the MMP-9 [
85]. However, further well-designed studies are needed to establish a putative role of this marker in everyday clinical setting.
A member of the lipocalin superfamily, Neutrophil gelatinase-associated lipocalin (NGAL) is an important regulator of the MMP-9 enzymatic activity and is thus implicated in progression of atherosclerosis [
86]. NGAL creates a complex with MMP-9 which then inhibits degradation of MMP-9, consequently extending its proteolytic activity [
86]. Circulating levels of both NGAL and MMP-9/NGAL complexes are significantly increased in asymptomatic patients with vulnerable carotid plaques, as demonstrated by Eilenberg et al. [
87]. Multiple authors reported higher NGAL levels in patients with ACS in comparison to patients with stable CAD [
88,
89]. In addition, NGAL has been shown in several studies to correlate with poorer prognosis and to predict all-cause mortality and major adverse cardiac events (MACE) in patients with ACS [
90,
91,
92]. However, implication of NGAL in a myriad of processes such as acute kidney injury, heart failure, and stable CAD, as well as inconsistence in data regarding prediction of clinical outcomes reduces its chance for establishment as a biomarker of plaque vulnerability [
89,
93,
94].
Another molecule that requires attention is soluble part of the Lectin-like oxidized low-density lipoprotein receptor-1 (sLOX-1), produced by shedding of the LOX-1 [
95]. Interaction between ox-LDL and its principal receptor, the LOX-1, appears to play a role in vascular dysfunction, including cells apoptosis and MMP production and activation, evoking the plaque rupture or erosion [
96,
97,
98,
99]. LOX-1 is abundantly present advanced in human atherosclerotic lesions. However, the main advantage of LOX-1 in terms of biomarker value is that its soluble part, the sLOX-1, is significantly elevated during the acute stage of ACS whilst not in general acute inflammatory diseases or stable CAD [
100,
101]. In fact, elevated sLOX-1 levels were detectable at an earlier stage after the onset of ACS in comparison to those of troponin-T (TnT), indicating that sLOX-1 reflects the atherosclerotic plaque vulnerability/rupture even before ischemic cardiac damage becomes clinically evident [
100]. Furthermore, sLOX, but neither hsCRP nor hsTnT, can differentiate ACS with plaque rupture from those without, and ACS with thin-cap fibroatheroma from those without, as shown by OCT studies [
102]. Additionally, the accuracy of the ACS diagnosis improved when sLOX-1 and hs-TnT were measured in combination [
103].
2.2. Lipid-Based Biomarkers
It has so far been well established by large epidemiological studies that higher lipid levels correlate with the occurrence of major CV events [
104,
105,
106]. Hence, since major CV events arise from vulnerable plaques, it is reasonable to infer that lipid status could indicate plaque vulnerability. Unlike in plasma, where LDL can be scarcely modified, in the atherosclerotic plaque, LDL is easily modified under the effect of free radicals and enzymatic activity [
107]. More importantly, these modified LDLs gain inflammatory properties and become aggregated, impeding their return to the circulation [
103,
108]. However, a small portion of these reach plasma and it is therefore hypothesized that these could serve as biomarkers.
Ox-LDL is one of the key pathophysiologic contributors to the atherosclerotic plaque development and progression [
109]. It has been demonstrated by multiple authors that amount of plaque ox-LDL correlates with plaque instability, especially in symptomatic carotid artery disease [
110,
111,
112]. However, the translation of experimental evidence in humans with aimed at the demonstration of the association between ox-LDL plasma levels with CV events proved to be difficult as it resulted in contrasting findings [
113,
114,
115,
116]. This example highlights the challenging nature of plaque biomarker implementation in clinical practice, as although ox-LDL is a major participant in the in proinflammatory process associated with plaque rupture, its plasma levels do not seem to reliably reflect the vulnerability of the plaque.
Electronegative LDL (LDL(−)) is a modified fraction of the LDL that holds physical and chemical characteristics that differ from those of native LDL, such as increased lipoprotein-associated phospholipase A2 activity (Lp-PLA2), ceramide, clusterine, non-esterified fatty acid content, as well as increased aggregation level [
117]. LDL(−) normally comprises around 3−5% of the total LDL plasma quantity, yet in certain CV pathologies this fraction rises [
118]. A moderate rise of LDL(−) is commonly observed in patients with classical CV risk factors such as hypercholesterolemia, active smoking, diabetes, and metabolic syndrome [
119,
120,
121,
122]. Moreover, LDL(−) is an important mediator of atherogenesis which triggers the detrimental cascade by binding to LOX-1, the aforementioned inflammatory biomarker [
123]. It has been recently shown that the most electronegative and the most atherogenic LDL fraction, the L5 fraction, is elevated in plasma of patients in STEMI [
124]. In addition, L5 from STEMI patients can enhance platelet aggregation in vitro [
125]. L5 levels have been also elevated in patients with ischemic stroke, and Shen et al. argue that L5 could thus serve as a marker of plaque vulnerability in those patients [
125]. Even though this biomarker could be promising, a much larger body of well-design studies is needed to confirm it.
Lp-PLA2, originally named platelet-activating factor acetylhydrolase (PAF-AH), is an enzyme formed by macrophages and foam cells in atherosclerotic plaque where it is responsible for hydrolysis of oxidized phospholipids (ox-PL) on LDL particles and subsequent release of proinflammatory lipids [
126]. It has been shown that stable atherosclerotic plaques contain only small amount of Lp-PLA2, unlike vulnerable plaques which are abundant in this molecule, markedly inside necrotic core [
127]. Importantly, unlike CRP, which is elevated in most inflammatory processes, Lp-PLA2 represents a vascular-specific inflammatory marker and its plasma concentration is stable in terms of time, which largely increases the putative role of this molecule as a biomarker [
128]. In addition, Lp-PLA2 level is independent of insulin resistance, unlike most of the other biomarkers [
129]. In a study by Sarlon-Bartoli et al., authors demonstrated that patients with confirmed unstable carotid plaque, even if asymptomatic, have higher Lp-PLA2 plasma levels [
130]. Similarly, Dong-Ling et al. demonstrated the same in patients with coronary artery plaques, highlighting that the specificity of serum Lp-PLA2 was stronger than that of hs-CRP in this setting [
131]. Furthermore, multiple epidemiological studies and meta-analysis demonstrated that Lp-PLA2 is independently associated with the risk of CAD, but rather interestingly, not with atherosclerosis in abdominal aorta [
132,
133,
134]. This discrepancy was further investigated by Fenning et al., which confirmed the differential role of Lp-PLA2 in the inflammatory cascade between different plaques [
135]. The authors hypothesize that the observed discrepancy arises from differences in flow hemodynamics and embryologic origin of the vasculature. This discrepancy is very important from our point of view as it highlights the difficulty of using non-cardiac vasculature as a surrogate marker for detection and management of atherosclerotic plaques in coronary arteries. In summary, LpPLA2 seems to be a potentially beneficial adjunctive biomarker as it is both specific for vascular inflammation and, relatively speaking, for CAD.