Alzheimer’s disease (AD) is the most common cause of dementia and the sixth cause of death in the world, constituting a major health problem for aging societies. This disease is a neurodegenerative continuum with well-established pathology hallmarks, namely the deposition of amyloid-β (Aβ) peptides in extracellular plaques and intracellular hyperphosphorylated forms of the microtubule associated protein tau forming neurofibrillary tangles (NFTs), accompanied by neuronal and synaptic loss. Interestingly, patients who will eventually develop AD manifest brain pathology decades before clinical symptoms appear. Among all the proposed pathogenic mechanisms to understand the etiology of Alzheimer’s disease (AD), increased oxidative stress seems to be a robust and early disease feature where many of those hypotheses converge.
- oxidative stress
- Alzheimer’s disease
1. Introduction
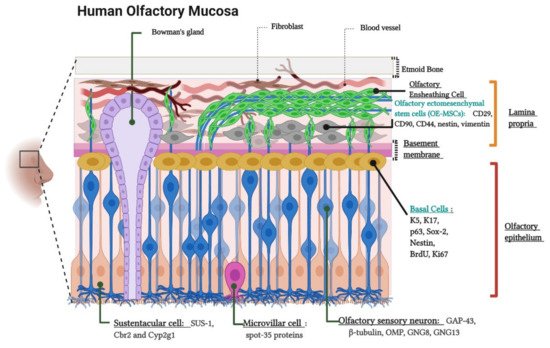
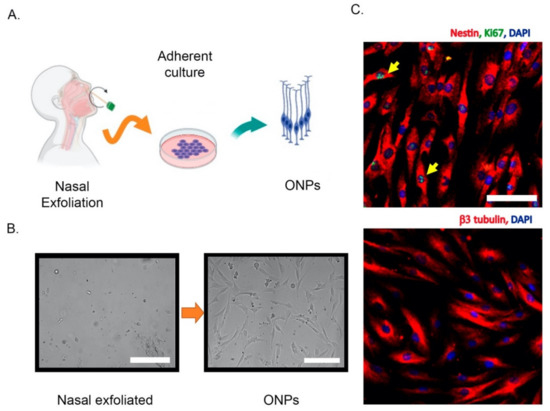
2. Olfactory Neuroepithelium and the Non-Invasive Isolation of ONPs
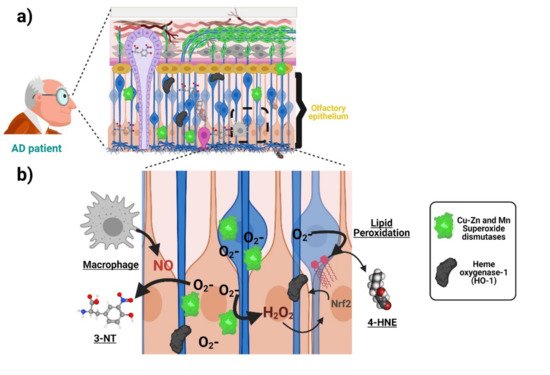
3. Alzheimer’s Disease-Related Oxidative Stress in the Olfactory Epithelium and ONPs
| Pathogenic Mechanism | Main Finding | Cellular Type | Lineage | References |
|---|---|---|---|---|
| Amyloid/Tau | Platelets from AD patients reproduce the increased amyloidogenic processing of AβPP | Platelets | Non-neuronal | [61] |
| Amyloid/Tau | AD platelets harbor increased levels of a higher molecular weight tau isoform | Platelets | Non-neuronal | [62] |
| Amyloid/Tau | Alteration of AβPP, BACE, and ADAM 10 levels in early stages of the disease | Platelets | Non-neuronal | [63][64][65] |
| Amyloid/Tau | It is suggested a decreased non-amyloidogenic processing of AβPP by a lack of nicastrin mRNA expression in samples obtained from AD patients | Lymphocytes | Non-neuronal | [66] |
| Amyloid/Tau | Altered balance between Aβ-oligomers and PKCε levels in AD. Loss of PKCε-mediated inhibition of Aβ |
Fibroblasts | Non-neuronal | [67] |
| Amyloid/Tau | Higher Aβ42/Aβ40 ratio compared to control cells | PSEN1 iPSC-derived neural progenitors |
Neuronal | [68] |
| Amyloid/Tau | Mutation alters the initial cleavage site of γ-secretase, resulting in an increased generation of Aβ42, in addition to an increase in the levels of total and phosphorylated tau | Neuron-derived iPSCs from patients harboring the London FAD AβPP mutation V717I |
Neuronal | [69] |
| Amyloid/Tau | Oligomeric forms of canonical Aβ impairs synaptic plasticity |
Cortical neurons from three genetic forms of AD —PSEN1 L113_I114insT, AβPP duplication (AβPPDp), and Ts21— generated from iPSCs | Neuronal | [70] |
| Amyloid/Tau | Increase in the content and changes in the subcellular distribution of t-tau and p-tau in cells from AD patients compared to controls | Non-invasively isolated ONPs | Neuronal | [9] |
| Mitochondria | Compromise of mitochondrial COX from AD patients |
Platelets | Non-neuronal | [71] |
| Mitochondria | Platelets isolated from AD patients show decreased ATP levels | Platelets | Non-neuronal | [72] |
| Mitochondria | AD lymphocytes exhibit impairment of total OXPHOS capacity | Lymphocytes | Non-neuronal | [73] |
| Mitochondria | AD skin fibroblasts show increased production of CO2 and reduced oxygen uptake suggesting that mitochondrial electron transport chain might be compromised |
Fibroblasts | Non-neuronal | [74] |
| Mitochondria | AD fibroblasts present reduction in mitochondrial length and a dysfunctional mitochondrial bioenergetics profile | Fibroblasts | Non-neuronal | [75] |
| Mitochondria | SAD fibroblasts exhibit aged mitochondria, and their recycling process is impaired | Fibroblasts | Non-neuronal | [76] |
| Mitochondria | Patient-derived cells show increased levels of oxidative phosphorylation chain complexes | Human induced pluripotent stem cell-derived neuronal cells (iN cells) from SAD patients |
Neuronal | [77] |
| Mitochondria | Mitophagy failure as a consequence of lysosomal dysfunction |
iPSC-derived neurons from FAD1 patients harboring PSEN1 A246E mutation | Neuronal | [78] |
| Mitochondria | Neurons exhibit defective mitochondrial axonal transport |
iPSC-derived neurons from an AD patient carrying AβPP -V715M mutation | Neuronal | [79] |
| Oxidative Stress | Increased activity of the antioxidant enzyme catalase in probable AD patients | Erythrocytes | Non-neuronal | [80] |
| Oxidative Stress | Increased production and content of thiobarbituric acid-reactive substances (TBARS), superoxide dismutase (SOD), and nitric oxide synthase (NOS) |
Erythrocytes and Platelets | Non-neuronal | [81] |
| Oxidative Stress | Increase in the content of the unfolded version of p53 as well as reduced SOD activity | Peripheral blood mononuclear cells (PBMCs) | Non-neuronal | [82] |
| Oxidative Stress | Exacerbated response to NFKB pathway | PBMCs | Non-neuronal | [83] |
| Oxidative Stress | Increased ROS production in response to H2O2 | PBMCs | Non-neuronal | [59] |
| Oxidative Stress | AD lymphocytes were more prone to cell death after a H2O2 challenge | Lymphocytes | Non-neuronal | [84] |
| Oxidative Stress | Reduced antioxidant capacity of FAD lymphocytes and fibroblasts together with increased lipid peroxidation on their plasma membrane | Lymphocytes and Fibroblasts | Non-neuronal | [85] |
| Oxidative Stress | Aβ peptides were better internalized and generated greater oxidative damage in FAD fibroblasts | Fibroblasts | Non-neuronal | [86] |
| Oxidative Stress | Aβ peptide caused a higher increase in the oxidation of HSP60 | Fibroblasts | Non-neuronal | [87] |
| Oxidative Stress | Reduction in the levels of Vimentin in samples from AD patients | iPSCs-derived neurons from AD patient | Neuronal | [58] |
| Oxidative Stress | Increased levels of hydroxynonenal, Nɛ-(carboxymethyl)lysine), and heme oxygenase-1 in samples from AD patients | Biopsy-derived ONPs | Neuronal | [24] |
| Oxidative Stress | Increased susceptibility to oxidative-stress-induced cell death | Biopsy-derived ONPs | Neuronal | [25] |
| ER-Stress | Impaired ER Ca2+ and ER stress in PBMCs from MCIs and mild AD patients | PBMCs | Non-neuronal | [88] |
| ER-Stress | Accumulation of Aβ oligomers induced ER and oxidative stress | iPSC-derived neural cells from a patient carrying APP-E693Δ mutation and a sporadic AD patient | Neuronal | [89] |
| ER-Stress | Aβ-S8C dimer triggers an ER stress response more prominent in AD neuronal cultures where several genes from the UPR were upregulated | iPSC-derived neuronal cultures carrying the AD-associated TREM2 R47H variant | Neuronal | [90] |
| ER-Stress | Accumulation of Aβ oligomers in iPSC-derived neurons from AD patients leads to increased ER stress |
iPSC-derived neurons from patients with an AβPP-E693Δ mutation | Neuronal | [91] |
This entry is adapted from the peer-reviewed paper 10.3390/ijms22126311
References
- Haque, R.U.; Levey, A.I.; Alzheimer’s disease: A clinical perspective and future nonhuman primate research opportunities. Proceedings of the National Academy of Sciences of the United States of America 2019, 116 (52), 26224-26229, 10.1073/pnas.1912954116.
- Serrano-Pozo, A.; Frosch, M.P.; Masliah, E.; Hyman, B.T.; Neuropathological alterations in Alzheimer disease. Cold Spring Harbor perspectives in medicine 2011, 1, a006189, 10.1101/cshperspect.a006189.
- Perrin, R.J.; Fagan, A.M.; Holtzman, D.M.; Multimodal techniques for diagnosis and prognosis of Alzheimer’s disease. Nature 2009, 461, 916–922, 10.1038/nature08538.
- Jack, C.R., Jr.; Knopman, D.S.; Jagust, W.J.; Shaw, L.M.; Aisen, P.S.; Weiner, M.W.; Petersen, R.C.; Trojanowski, J.Q.; Hypothetical model of dynamic biomarkers of the Alzheimer’s pathological cascade. The Lancet. Neurology 2010, 9, 119–128, 10.1016/S1474-4422(09)70299-6.
- Trushina, E.; Alzheimer’s disease mechanisms in peripheral cells: Promises and challenges. Alzheimer’s & dementia 2019, 5, 652–660, 10.1016/j.trci.2019.06.008..
- Penney, J.; Ralvenius, W.T.; Tsai, L.H.; Modeling Alzheimer’s disease with iPSC-derived brain cells. Molecular psychiatry 2020, 25, 148–167, 10.1038/s41380-019-0468-3.
- Mackay-Sim, A; Concise review: Patient-derived olfactory stem cells: New models for brain diseases. Stem cells 2012, 30, 2361–2365, 10.1002/stem.1220.
- Jimenez-Vaca, A.L.; Benitez-King, G.; Ruiz, V.; Ramirez-Rodriguez, G.B.; Hernandez-de la Cruz, B.; Salamanca-Gomez, F.A.; Gonzalez-Marquez, H.; Ramirez-Sanchez, I.; Ortiz-Lopez, L.; Velez-Del Valle, C.; et al. Exfoliated Human Olfactory Neuroepithelium: A Source of Neural Progenitor Cells. Molecular neurobiology 2018, 55, 2516–2523, 10.1007/s12035-017-0500-z.
- Riquelme, A.; Valdes-Tovar, M.; Ugalde, O.; Maya-Ampudia, V.; Fernandez, M.; Mendoza-Duran, L.; Rodriguez-Cardenas, L.; Benitez-King, G.; Potential Use of Exfoliated and Cultured Olfactory Neuronal Precursors for In Vivo Alzheimer’s Disease Diagnosis: A Pilot Study. Cellular and molecular neurobiology 2020, 40, 87–98, 10.1007/s10571-019-00718-z.
- Wolozin, B.; Zheng, B.; Loren, D.; Lesch, K.P.; Lebovics, R.S.; Lieberburg, I.; Sunderland, T.; Beta/A4 domain of APP: Antigenic differences between cell lines. Journal of neuroscience research 1992, 33, 189–195, 10.1002/jnr.490330202.
- Wolozin, B.; Bacic, M.; Merrill, M.J.; Lesch, K.P.; Chen, C.; Lebovics, R.S.; Sunderland, T.; Differential expression of carboxyl terminal derivatives of amyloid precursor protein among cell lines. Journal of neuroscience research 1992, 33, 163–169, 10.1002/jnr.490330121.
- Butterfield, D.A.; Halliwell, B.; Oxidative stress, dysfunctional glucose metabolism and Alzheimer disease. Nature reviews. Neuroscience 2019, 20, 148–160, 10.1038/s41583-019-0132-6.
- Nunomura, A.; Perry, G.; Aliev, G.; Hirai, K.; Takeda, A.; Balraj, E.K.; Jones, P.K.; Ghanbari, H.; Wataya, T.; Shimohama, S.; et al. Oxidative damage is the earliest event in Alzheimer disease. Journal of neuropathology and experimental neurology 2001, 60, 759–767, 10.1093/jnen/60.8.759.
- Smith, M.A.; Nunomura, A.; Lee, H.G.; Zhu, X.; Moreira, P.I.; Avila, J.; Perry, G.; Chronological primacy of oxidative stress in Alzheimer disease. Neurobiology of aging 2005, 26, 579–580, 10.1016/j.neurobiolaging.2004.09.021.
- Gonzalez-Reyes, R.E.; Nava-Mesa, M.O.; Vargas-Sanchez, K.; Ariza-Salamanca, D.; Mora-Munoz, L.; Involvement of Astrocytes in Alzheimer’s Disease from a Neuroinflammatory and Oxidative Stress Perspective. Frontiers in molecular neuroscience 2017, 10, 427, 10.3389/fnmol.2017.00427.
- Habas, A.; Hahn, J.; Wang, X.; Margeta, M.; Neuronal activity regulates astrocytic Nrf2 signaling. Proceedings of the National Academy of Sciences of the United States of America 2013, 110, 18291–18296, 10.1073/pnas.1208764110.
- Qiu, J.; Dando, O.; Febery, J.A.; Fowler, J.H.; Chandran, S.; Hardingham, G.E.; Neuronal Activity and Its Role in Controlling Antioxidant Genes. International journal of molecular sciences 2020, 21, 1933, 10.3390/ijms21061933.
- Baxter, P.S.; Hardingham, G.E.; Adaptive regulation of the brain’s antioxidant defences by neurons and astrocytes. Free radical biology & medicine 2016, 100, 147–152, 10.1016/j.freeradbiomed.2016.06.027.
- Araque, A.; Parpura, V.; Sanzgiri, R.P.; Haydon, P.G.; Tripartite synapses: Glia, the unacknowledged partner. Trends in neurosciences 1999, 22, 208–215, 10.1016/s0166-2236(98)01349-6.
- Perea, G.; Navarrete, M.; Araque, A.; Tripartite synapses: Astrocytes process and control synaptic information. Trends in neurosciences 2009, 32, 421–431, 10.1016/j.tins.2009.05.001.
- Wang, Q.; Wang, Y.; Shimony, J.S.; Owen, C.J.; Liu, J.; Fagan, A.M.; Cairns, N.J.; Ances, B.; Morris, J.C.; Benzinger, T.L.S.; et al. IC-P-172: Simultaneous Quantification of White Matter Abnormalities and Vasogenic Edema in Early Alzheimer Disease.. Alzheimer’s & dementia 2016, 12, P125-P126, 10.1016/j.jalz.2016.06.203.
- Kitchen, P.; Salman, M.M.; Halsey, A.M.; Clarke-Bland, C.; MacDonald, J.A.; Ishida, H.; Vogel, H.J.; Almutiri, S.; Logan, A.; Kreida, S.; et al. Targeting Aquaporin-4 Subcellular Localization to Treat Central Nervous System Edema. Cell 2020, 181, 784–799 e719, 10.1016/j.cell.2020.03.037.
- Getchell, M.L.; Shah, D.S.; Buch, S.K.; Davis, D.G.; Getchell, T.V; 3-Nitrotyrosine immunoreactivity in olfactory receptor neurons of patients with Alzheimer’s disease: Implications for impaired odor sensitivity. . Neurobiology of aging 2003, 24, 663–673, 10.1016/s0197-4580(02)00195-1.
- Ghanbari, H.A.; Ghanbari, K.; Harris, P.L.; Jones, P.K.; Kubat, Z.; Castellani, R.J.; Wolozin, B.L.; Smith, M.A.; Perry, G.; Oxidative damage in cultured human olfactory neurons from Alzheimer’s disease patients. Aging cell 2004, 3, 41–44, 10.1111/j.1474-9728.2004.00083.x.
- Nelson, V.M.; Dancik, C.M.; Pan, W.; Jiang, Z.G.; Lebowitz, M.S.; Ghanbari, H.A.; PAN-811 inhibits oxidative stress-induced cell death of human Alzheimer’s disease-derived and age-matched olfactory neuroep-ithelial cells via suppression of intracellular reactive oxygen species. Journal of Alzheimer’s disease : JAD 2009, 17, 611–619, 10.3233/JAD-2009-1078.
- Jafek, B.W.; Ultrastructure of human nasal mucosa. The Laryngoscope 1983, 93, 1576–1599, 10.1288/00005537-198312000-00011.
- Moran, D.T.; Rowley, J.C., 3rd; Jafek, B.W.; Lovell, M.A.; The fine structure of the olfactory mucosa in man. Journal of neurocytology 1982, 11, 721–746, 10.1007/bf01153516.
- Morrison, E.E.; Costanzo, R.M.; Morphology of the human olfactory epithelium. The Journal of comparative neurology 1990, 297, 1–13, 10.1002/cne.902970102.
- Verhaagen, J.; Oestreicher, A.B.; Gispen, W.H.; Margolis, F.L.; The expression of the growth associated protein B50/GAP43 in the olfactory system of neonatal and adult rats. The Journal of Neuroscience : The official journal of the Society for Neuroscience 1989, 9, 683–691, 10.1523/JNEUROSCI.09-02-00683.1989.
- Holbrook, E.H.; Wu, E.; Curry, W.T.; Lin, D.T.; Schwob, J.E.; Immunohistochemical characterization of human olfactory tissue.. Laryngoscope 2011, 121, 1687–1701, 10.1002/lary.21856..
- Fletcher, R.B.; Das, D.; Gadye, L.; Street, K.N.; Baudhuin, A.; Wagner, A.; Cole, M.B.; Flores, Q.; Choi, Y.G.; Yosef, N.; et al. Deconstructing Olfactory Stem Cell Trajectories at Single-Cell Resolution. Cell stem cell 2017, 20, 817–830 e818, 10.1016/j.stem.2017.04.003.
- Hahn, C.G.; Han, L.Y.; Rawson, N.E.; Mirza, N.; Borgmann-Winter, K.; Lenox, R.H.; Arnold, S.E.; In vivo and in vitro neurogenesis in human olfactory epithelium. The Journal of comparative neurology 2005, 483, 154–163, 10.1002/cne.20424.
- Chen, X.; Fang, H.; Schwob, J.E.; Multipotency of purified, transplanted globose basal cells in olfactory epithelium. The Journal of comparative neurology 2004, 469, 457–474, 10.1002/cne.11031.
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E.; et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317, 10.1080/14653240600855905.
- Delorme, B.; Nivet, E.; Gaillard, J.; Haupl, T.; Ringe, J.; Deveze, A.; Magnan, J.; Sohier, J.; Khrestchatisky, M.; Roman, F.S.; et al. The human nose harbors a niche of olfactory ectomesenchymal stem cells displaying neurogenic and osteogenic properties. Stem cells and development 2010, 19, 853–866, 10.1089/scd.2009.0267.
- Murrell, W.; Feron, F.; Wetzig, A.; Cameron, N.; Splatt, K.; Bellette, B.; Bianco, J.; Perry, C.; Lee, G.; Mackay-Sim, A.; et al. Multipotent stem cells from adult olfactory mucosa. Developmental dynamics: An official publication of the American Association of Anatomists 2005, 233, 496–515, 10.1002/dvdy.20360.
- Tanos, T.; Saibene, A.M.; Pipolo, C.; Battaglia, P.; Felisati, G.; Rubio, A.; Isolation of putative stem cells present in human adult olfactory mucosa. PLoS ONE 2017, 12, e0181151, 10.1371/journal.pone.0181151.
- Matigian, N.; Abrahamsen, G.; Sutharsan, R.; Cook, A.L.; Vitale, A.M.; Nouwens, A.; Bellette, B.; An, J.; Anderson, M.; Beckhouse, A.G.; et al. Disease-specific, neurosphere-derived cells as models for brain disorders. Disease models & mechanisms 2010, 3, 785–798, 10.1242/dmm.005447.
- Feron, F.; Gepner, B.; Lacassagne, E.; Stephan, D.; Mesnage, B.; Blanchard, M.P.; Boulanger, N.; Tardif, C.; Deveze, A.; Rousseau, S.; et al. Olfactory stem cells reveal MOCOS as a new player in autism spectrum disorders. Molecular psychiatry 2016, 21, 1215–1224, 10.1038/mp.2015.106.
- Stewart, R.; Wali, G.; Perry, C.; Lavin, M.F.; Feron, F.; Mackay-Sim, A.; Sutharsan, R.; A Patient-Specific Stem Cell Model to Investigate the Neurological Phenotype Observed in Ataxia-Telangiectasia. Methods in molecular biology 2017, 1599, 391–400, 10.1007/978-1-4939-6955-5_28.
- Fan, Y.; Wali, G.; Sutharsan, R.; Bellette, B.; Crane, D.I.; Sue, C.M.; Mackay-Sim, A.; Low dose tubulin-binding drugs rescue peroxisome trafficking deficit in patient-derived stem cells in Hereditary Spastic Paraplegia. Biology open 2014, 3, 494–502, 10.1242/bio.20147641.
- Ayala-Grosso, C.A.; Pieruzzini, R.; Diaz-Solano, D.; Wittig, O.; Abrante, L.; Vargas, L.; Cardier, J.; Amyloid-abeta Peptide in olfactory mucosa and mesenchymal stromal cells of mild cognitive impairment and Alzheimer’s disease patients. Brain pathology 2015, 25, 136–145, 10.1111/bpa.12169.
- Solis-Chagoyan, H.; Flores-Soto, E.; Valdes-Tovar, M.; Cercos, M.G.; Calixto, E.; Montano, L.M.; Barajas-Lopez, C.; Sommer, B.; Aquino-Galvez, A.; Trueta, C.; et al. Purinergic Signaling Pathway in Human Olfactory Neuronal Precursor Cells. Stem cells international 2019, 2019, 2728786, 10.1155/2019/2728786.
- Benitez-King, G.; Riquelme, A.; Ortiz-Lopez, L.; Berlanga, C.; Rodriguez-Verdugo, M.S.; Romo, F.; Calixto, E.; Solis-Chagoyan, H.; Jimenez, M.; Montano, L.M.; et al. A non-invasive method to isolate the neuronal linage from the nasal epithelium from schizophrenic and bipolar diseases.. Journal of neuroscience methods 2011, 201, 35–45, 10.1016/j.jneumeth.2011.07.009.
- Wolozin, B.; Sunderland, T.; Zheng, B.B.; Resau, J.; Dufy, B.; Barker, J.; Swerdlow, R.; Coon, H.; Continuous culture of neuronal cells from adult human olfactory epithelium. Journal of molecular neuroscience: MN 1992, 3, 137–146, 10.1007/bf02919405.
- Gomez, G.; Rawson, N.E.; Hahn, C.G.; Michaels, R.; Restrepo, D.; Characteristics of odorant elicited calcium changes in cultured human olfactory neurons. Journal of neuroscience research 2000, 62, 737–749, 10.1002/1097-4547(20001201)62:5<737::AID-JNR14>3.0.CO;2-A.
- Yazinski, S.; Gomez, G.; Time course of structural and functional maturation of human olfactory epithelial cells in vitro. Journal of neuroscience research 2014, 92, 64–73, 10.1002/jnr.23296.
- Kagan, V.E.; Tyurina, Y.Y.; Sun, W.Y.; Vlasova, II.; Dar, H.; Tyurin, V.A.; Amoscato, A.A.; Mallampalli, R.; van der Wel, P.C.A.; He, R.R.; et al. Redox phospholipidomics of enzymatically generated oxygenated phospholipids as specific signals of programmed cell death. Free radical biology & medicine 2020, 147, 231–241, 10.1016/j.freeradbiomed.2019.12.028.
- Andersen, J.K.; Oxidative stress in neurodegeneration: Cause or consequence? . Nature medicine 2004, 10 Suppl, S18-25, 10.1038/nrn1434.
- Sutherland, G.T.; Chami, B.; Youssef, P.; Witting, P.K.; Oxidative stress in Alzheimer’s disease: Primary villain or physiological by-product?. Redox report: Communications in free radical research 2013, 18, 134–141, 10.1179/1351000213Y.0000000052.
- Butterfield, D.A.; Boyd-Kimball, D; Oxidative Stress, Amyloid-beta Peptide, and Altered Key Molecular Pathways in the Pathogenesis and Progression of Alzheimer’s Disease. Journal of Alzheimer’s disease: JAD 2018, 62, 1345–1367, 10.3233/JAD-170543.
- Tonnies, E.; Trushina, E.; Oxidative Stress, Synaptic Dysfunction, and Alzheimer’s Disease. Journal of Alzheimer’s disease: JAD 2017, 57, 1105–1121, 10.3233/JAD-161088.
- Kulkarni-Narla, A.; Getchell, T.V.; Schmitt, F.A.; Getchell, M.L.; Manganese and copper-zinc superoxide dismutases in the human olfactory mucosa: Increased immunoreactivity in Alzheimer’s disease. Experimental neurology 1996, 140, 115–125, 10.1006/exnr.1996.0122.
- Chuah, M.I.; Getchell, M.L.; Metallothionein in olfactory mucosa of Alzheimer’s disease patients and apoE-deficient mice. Neuroreport 1999, 10, 1919–1924, 10.1097/00001756-199906230-00023.
- Calhoun-Haney, R.; Murphy, C.; Apolipoprotein epsilon4 is associated with more rapid decline in odor identification than in odor threshold or Dementia Rating Scale scores. Brain and cognition 2005, 58, 178–182, 10.1016/j.bandc.2004.10.004.
- Gilbert, P.E.; Murphy, C.; The effect of the ApoE epsilon4 allele on recognition memory for olfactory and visual stimuli in patients with pathologically confirmed Alzheimer’s disease, probable Alzheimer’s disease, and healthy elderly controls. Journal of clinical and experimental neuropsychology 2004, 26, 779–794, 10.1080/13803390490509439.
- Wang, Q.S.; Tian, L.; Huang, Y.L.; Qin, S.; He, L.Q.; Zhou, J.N.; Olfactory identification and apolipoprotein E epsilon 4 allele in mild cognitive impairment. Brain research 2002, 951, 77–81, 10.1016/s0006-8993(02)03137-2.
- Chen, M.; Lee, H.K.; Moo, L.; Hanlon, E.; Stein, T.; Xia, W.; Common proteomic profiles of induced pluripotent stem cell-derived three-dimensional neurons and brain tissue from Alzheimer patients. Journal of proteomics 2018, 182, 21–33, 10.1016/j.jprot.2018.04.032.
- Ponce, D.P.; Salech, F.; SanMartin, C.D.; Silva, M.; Xiong, C.; Roe, C.M.; Henriquez, M.; Quest, A.F.; Behrens, M.I.; Increased susceptibility to oxidative death of lymphocytes from Alzheimer patients correlates with dementia severity. Current Alzheimer research 2014, 11, 892–898, 10.2174/1567205011666141001113135.
- Salech, F.; Ponce, D.P.; SanMartin, C.D.; Rogers, N.K.; Chacon, C.; Henriquez, M.; Behrens, M.I.; PARP-1 and p53 Regulate the Increased Susceptibility to Oxidative Death of Lymphocytes from MCI and AD Patients. Frontiers in aging neuroscience 2017, 9, 310, 10.3389/fnagi.2017.00310.
- Tang, K.; Hynan, L.S.; Baskin, F.; Rosenberg, R.N.; Platelet amyloid precursor protein processing: A bio-marker for Alzheimer’s disease. Journal of the neurological sciences 2006, 240, 53–58, 10.1016/j.jns.2005.09.002.
- Neumann, K.; Farias, G.; Slachevsky, A.; Perez, P.; Maccioni, R.B.; Human platelets tau: A potential peripheral marker for Alzheimer’s disease. Journal of Alzheimer’s disease: JAD 2011, 25, 103–109, 10.3233/JAD-2011-101641.
- Colciaghi, F.; Marcello, E.; Borroni, B.; Zimmermann, M.; Caltagirone, C.; Cattabeni, F.; Padovani, A.; Di Luca, M.; Platelet APP, ADAM 10 and BACE alterations in the early stages of Alzheimer disease. Neurology 2004, 62, 498–501, 10.1212/01.wnl.0000106953.49802.9c.
- Colciaghi, F.; Borroni, B.; Pastorino, L.; Marcello, E.; Zimmermann, M.; Cattabeni, F.; Padovani, A.; Di Luca, M.; [alpha]-Secretase ADAM10 as well as [alpha]APPs is reduced in platelets and CSF of Alzheimer disease patients. Molecular medicine 2002, 8, 67–74, 10.1007/BF03402076.
- Vignini, A.; Sartini, D.; Morganti, S.; Nanetti, L.; Luzzi, S.; Provinciali, L.; Mazzanti, L.; Emanuelli, M.; Platelet amyloid precursor protein isoform expression in Alzheimer’s disease: Evidence for peripheral marker. International journal of immunopathology and pharmacology 2011, 24, 529–534, 10.1177/039463201102400229.
- Herrera-Rivero, M.; Soto-Cid, A.; Hernandez, M.E.; Aranda-Abreu, G.E; Tau, APP, NCT and BACE1 in lymphocytes through cognitively normal ageing and neuropathology. Anais da Academia Brasileira de Ciencias 2013, 85, 1489–1496, 10.1590/0001-376520130013.
- Khan, T.K.; Sen, A.; Hongpaisan, J.; Lim, C.S.; Nelson, T.J.; Alkon, D.L.; PKCepsilon deficits in Alzheimer’s disease brains and skin fibroblasts. Journal of Alzheimer’s disease: JAD 2015, 43, 491–509, 10.3233/JAD-141221.
- Sproul, A.A.; Jacob, S.; Pre, D.; Kim, S.H.; Nestor, M.W.; Navarro-Sobrino, M.; Santa-Maria, I.; Zimmer, M.; Aubry, S.; Steele, J.W.; et al. Characterization and molecular profiling of PSEN1 familial Alzheimer’s disease iPSC-derived neural progenitors. PLoS ONE 2014, 9, e84547, 10.1371/journal.pone.0084547.
- Muratore, C.R.; Rice, H.C.; Srikanth, P.; Callahan, D.G.; Shin, T.; Benjamin, L.N.; Walsh, D.M.; Selkoe, D.J.; Young-Pearse, T.L.; The familial Alzheimer’s disease APPV717I mutation alters APP processing and Tau expression in iPSC-derived neurons. Human molecular genetics 2014, 23, 3523–3536, 10.1093/hmg/ddu064.
- Hu, N.W.; Corbett, G.T.; Moore, S.; Klyubin, I.; O’Malley, T.T.; Walsh, D.M.; Livesey, F.J.; Rowan, M.J.; Extracellular Forms of Abeta and Tau from iPSC Models of Alzheimer’s Disease Disrupt Synaptic Plasticity.. Cell reports 2018, 23, 1932–1938, 10.1016/j.celrep.2018.04.040.
- Bosetti, F.; Brizzi, F.; Barogi, S.; Mancuso, M.; Siciliano, G.; Tendi, E.A.; Murri, L.; Rapoport, S.I.; Solaini, G.; Cytochrome c oxidase and mitochondrial F1F0-ATPase (ATP synthase) activities in platelets and brain from patients with Alz-heimer’s disease. Neurobiology of aging 2002, 23, 371–376, 10.1016/s0197-4580(01)00314-1.
- Cardoso, S.M.; Proenca, M.T.; Santos, S.; Santana, I.; Oliveira, C.R.; Cytochrome c oxidase is decreased in Alzheimer’s disease platelets. Neurobiology of aging 2004, 25, 105–110, 10.1016/s0197-4580(03)00033-2.
- Leuner, K.; Schulz, K.; Schutt, T.; Pantel, J.; Prvulovic, D.; Rhein, V.; Savaskan, E.; Czech, C.; Eckert, A.; Muller, W.E.; et al. Peripheral mitochondrial dysfunction in Alzheimer’s disease: Focus on lymphocytes. Molecular neurobiology 2012, 46, 194–204, 10.1007/s12035-012-8300-y.
- Sims, N.R.; Finegan, J.M.; Blass, J.P.; Altered metabolic properties of cultured skin fibroblasts in Alzheimer’s disease. Annals of neurology 1987, 21, 451–457, 10.1002/ana.410210507.
- Perez, M.J.; Ponce, D.P.; Osorio-Fuentealba, C.; Behrens, M.I.; Quintanilla, R.A.; Mitochondrial Bioenergetics Is Altered in Fibroblasts from Patients with Sporadic Alzheimer’s Disease. Frontiers in neuroscience 2017, 11, 553, 10.3389/fnins.2017.00553.
- Martin-Maestro, P.; Gargini, R.; Garcia, E.; Perry, G.; Avila, J.; Garcia-Escudero, V.; Slower Dynamics and Aged Mitochondria in Sporadic Alzheimer’s Disease. Oxidative medicine and cellular longevity 2017, 2017, 9302761, 10.1155/2017/9302761.
- Birnbaum, J.H.; Wanner, D.; Gietl, A.F.; Saake, A.; Kundig, T.M.; Hock, C.; Nitsch, R.M.; Tackenberg, C.; Oxidative stress and altered mitochondrial protein expression in the absence of amyloid-beta and tau pathology in iPSC-derived neurons from sporadic Alzheimer’s disease patients. Stem cell research 2018, 27, 121–130, 10.1016/j.scr.2018.01.019.
- Martin-Maestro, P.; Gargini, R.; A., A.S.; Garcia, E.; Anton, L.C.; Noggle, S.; Arancio, O.; Avila, J.; Garcia-Escudero, V.; Mitophagy Failure in Fibroblasts and iPSC-Derived Neurons of Alzheimer’s Disease-Associated Presenilin 1 Mutation. Frontiers in molecular neuroscience 2017, 10, 291, 10.3389/fnmol.2017.00291.
- Li, L.; Roh, J.H.; Kim, H.J.; Park, H.J.; Kim, M.; Koh, W.; Heo, H.; Chang, J.W.; Nakanishi, M.; Yoon, T.; et al. The First Generation of iPSC Line from a Korean Alzheimer’s Disease Patient Carrying APP-V715M Mutation Exhibits a Distinct Mitochondrial Dysfunction. Experimental neurobiology 2019, 28, 329–336, 10.5607/en.2019.28.3.329.
- Repetto, M.G.; Reides, C.G.; Evelson, P.; Kohan, S.; de Lustig, E.S.; Llesuy, S.F.; Peripheral markers of oxidative stress in probable Alzheimer patients. European journal of clinical investigation 1999, 29, 643–649, 10.1046/j.1365-2362.1999.00506.x.
- Kawamoto, E.M.; Munhoz, C.D.; Glezer, I.; Bahia, V.S.; Caramelli, P.; Nitrini, R.; Gorjao, R.; Curi, R.; Scavone, C.; Marcourakis, T.; et al. Oxidative state in platelets and erythrocytes in aging and Alzheimer’s disease. Neurobiology of aging 2005, 26, 857–864, 10.1016/j.neurobiolaging.2004.08.011.
- Ihara, Y.; Hayabara, T.; Sasaki, K.; Kawada, R.; Nakashima, Y.; Kuroda, S.; Relationship between oxidative stress and apoE phenotype in Alzheimer’s disease. Acta neurologica Scandinavica 2000, 102, 346–349, 10.1034/j.1600-0404.2000.102006346.x.
- Ascolani, A.; Balestrieri, E.; Minutolo, A.; Mosti, S.; Spalletta, G.; Bramanti, P.; Mastino, A.; Caltagirone, C.; Macchi, B.; Dysregulated NF-kappaB pathway in peripheral mononuclear cells of Alzheimer’s disease patients. Current Alzheimer research 2012, 9, 128–137, 10.2174/156720512799015091.
- Behrens, M.I.; Silva, M.; Salech, F.; Ponce, D.P.; Merino, D.; Sinning, M.; Xiong, C.; Roe, C.M.; Quest, A.F.; Inverse susceptibility to oxidative death of lymphocytes obtained from Alzheimer’s patients and skin cancer survivors: Increased apoptosis in Alzheimer’s and reduced necrosis in cancer. The journals of gerontology. Series A, Biological Sciences and Medical Sciences 2012, 67, 1036–1040, 10.1093/gerona/glr258.
- Cecchi, C.; Fiorillo, C.; Sorbi, S.; Latorraca, S.; Nacmias, B.; Bagnoli, S.; Nassi, P.; Liguri, G.; Oxidative stress and reduced antioxidant defenses in peripheral cells from familial Alzheimer’s patients. Free radical biology & medicine 2002, 33, 1372–1379, 10.1016/s0891-5849(02)01049-3.
- Cecchi, C.; Fiorillo, C.; Baglioni, S.; Pensalfini, A.; Bagnoli, S.; Nacmias, B.; Sorbi, S.; Nosi, D.; Relini, A.; Liguri, G.; et al. Increased susceptibility to amyloid toxicity in familial Alzheimer’s fibroblasts. Neurobiology of aging 2007, 28, 863–876, 10.1016/j.neurobiolaging.2006.05.014.
- Choi, J.; Malakowsky, C.A.; Talent, J.M.; Conrad, C.C.; Carroll, C.A.; Weintraub, S.T.; Gracy, R.W.; Anti-apoptotic proteins are oxidized by Abeta25-35 in Alzheimer’s fibroblasts. Biochimica et biophysica acta 2003, 1637, 135–141, 10.1016/s0925-4439(02)00227-2.
- Mota, S.I.; Costa, R.O.; Ferreira, I.L.; Santana, I.; Caldeira, G.L.; Padovano, C.; Fonseca, A.C.; Baldeiras, I.; Cunha, C.; Letra, L.; et al. Oxidative stress involving changes in Nrf2 and ER stress in early stages of Alzheimer’s disease. Biochimica et biophysica acta 2015, 1852, 1428–1441, 10.1016/j.bbadis.2015.03.015.
- Piccini, A.; Fassio, A.; Pasqualetto, E.; Vitali, A.; Borghi, R.; Palmieri, D.; Nacmias, B.; Sorbi, S.; Sitia, R.; Tabaton, M.; et al. Fibroblasts from FAD-linked presenilin 1 mutations display a normal unfolded protein response but overproduce Abeta42 in response to tunicamycin. Neurobiology of disease 2004, 15, 380–386, 10.1016/j.nbd.2003.11.013.
- Martins, S.; Muller-Schiffmann, A.; Erichsen, L.; Bohndorf, M.; Wruck, W.; Sleegers, K.; Van Broeckhoven, C.; Korth, C.; Adjaye, J.; IPSC-Derived Neuronal Cultures Carrying the Alzheimer’s Disease Associated TREM2 R47H Variant Enables the Construction of an Abeta-Induced Gene Regulatory Network. International journal of molecular sciences 2020, 21, 4516, 10.3390/ijms21124516.
- Kondo, T.; Asai, M.; Tsukita, K.; Kutoku, Y.; Ohsawa, Y.; Sunada, Y.; Imamura, K.; Egawa, N.; Yahata, N.; Okita, K.; et al. Modeling Alzheimer’s disease with iPSCs reveals stress phenotypes associated with intracellular Abeta and differential drug responsiveness. Cell stem cell 2013, 12, 487–496, 10.1016/j.stem.2013.01.009.
