Metallic nanostructures (MNs) and metal-organic frameworks (MOFs) play a pivotal role by articulating their significance in high-performance supercapacitors along with conducting polymers (CPs). The interaction and synergistic pseudocapacitive effect of MNs with CPs have contributed to enhance the specific capacitance and cyclic stability. Among various conjugated heterocyclic CPs, polypyrrole (PPy) (prevalently knows as “synthetic metal”) is exclusively studied because of its excellent physicochemical properties, ease of preparation, flexibility in surface modifications, and unique molecular structure–property relationships. Numerous researchers attempted to improve the low electronic conductivity of MNs and MOFs, by incorporating conducting PPy and/or used decoration strategy. This was succeeded by fine-tuning this objective, which managed to get outstanding supercapacitive performances.
- conducting polymers
- cyclic voltammetry
- electrode materials
- metal oxides
- polypyrrole
- supercapacitors
- synthetic metal
1. Introduction
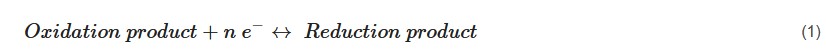 Comparatively, pseudocapacitors can accumulate greater electrochemical storage electricity and demonstrate higher energy density than EDLCs (for an illustration, see Figure 1). Accordingly, the synergistic and tunable complimenting properties of diverse nanoarchitecture metal-organic frameworks (metal oxides/phosphides/sulfides) with conjugated organic polymers, especially polypyrrole (PPy) derivatives, have found widespread application in fabricating electrochemical sensors and energy storage technologies [6][7][8][9].
Comparatively, pseudocapacitors can accumulate greater electrochemical storage electricity and demonstrate higher energy density than EDLCs (for an illustration, see Figure 1). Accordingly, the synergistic and tunable complimenting properties of diverse nanoarchitecture metal-organic frameworks (metal oxides/phosphides/sulfides) with conjugated organic polymers, especially polypyrrole (PPy) derivatives, have found widespread application in fabricating electrochemical sensors and energy storage technologies [6][7][8][9].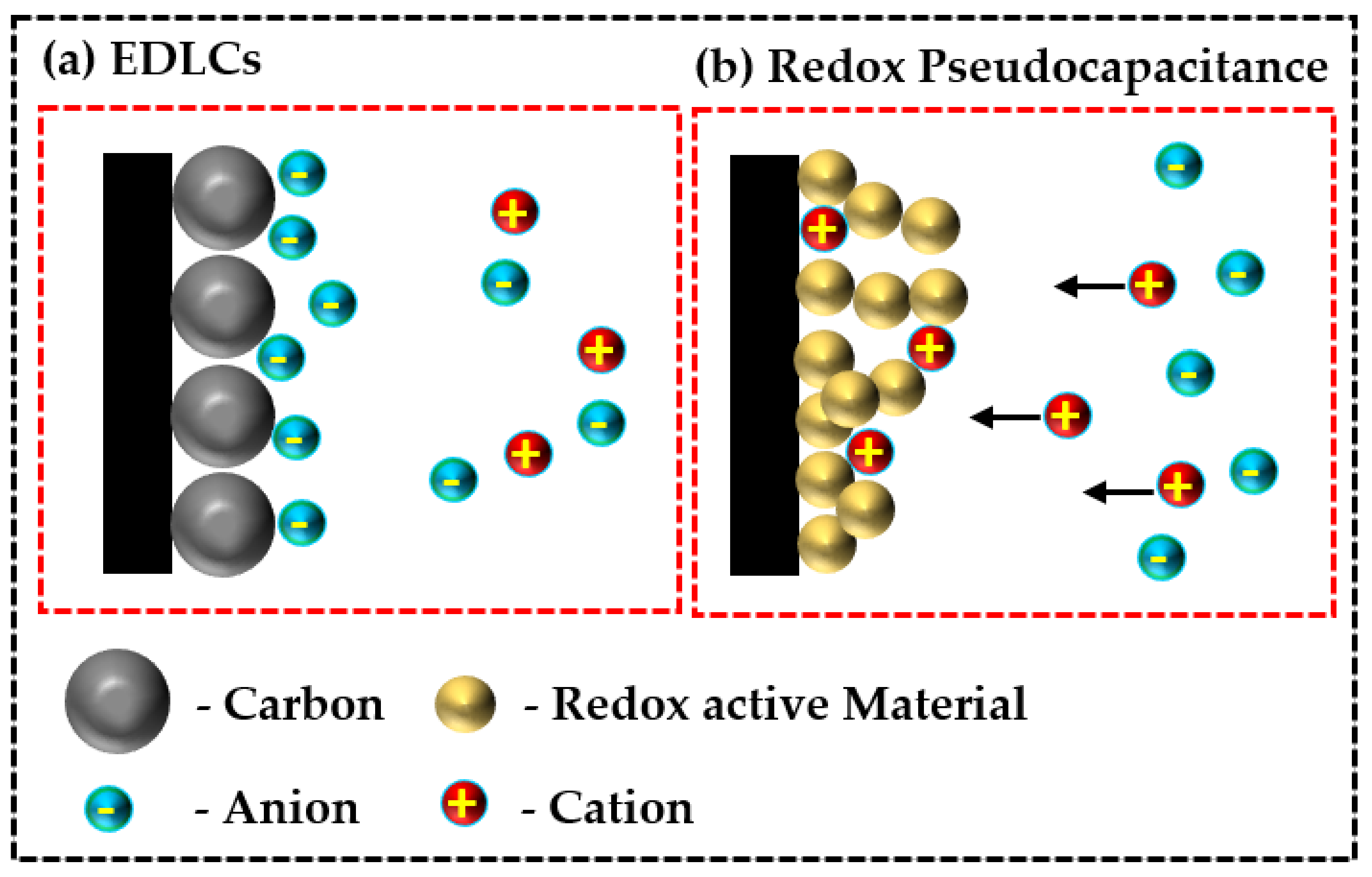 Figure 1. Schematic illustration of (a) electric double layer supercapacitors (EDLCs) and (b) redox pseudocapacitors.
Figure 1. Schematic illustration of (a) electric double layer supercapacitors (EDLCs) and (b) redox pseudocapacitors.2. Polypyrrole-Based Hybrid Metallic Nanostructures as Electrode Materials for High Performance Supercapacitors
3. Concluding Remarks
This entry is adapted from the peer-reviewed paper 10.3390/met11060905
References
- Ocheje, M.U.; Charron, B.P.; Nyayachavadi, A.; Rondeau-Gagné, S. Stretchable electronics: Recent progress in the preparation of stretchable and self-healing semiconducting conjugated polymers. Flex. Print. Electron. 2017, 2, 043002.
- Wang, M.; Baek, P.; Akbarinejad, A.; Barker, D.; Travas-Sejdic, J. Conjugated polymers and composites for stretchable organic electronics. J. Mater. Chem. C 2019, 7, 5534–5552.
- The Nobel Prize. The Nobel Prize in Chemistry 2000. Available online: (accessed on 16 December 2020).
- Naresh, U.; Kumar, R.J.; Ramesh, S.; Chandra Babu Naidu, C.B.K.; Basha, D.B.; Banerjee, P.; Srinivas, K. Conducting Polymer Derived Materials for Batteries. In Conducting Polymers-Based Energy Storage Materials; Inamuddin, Boddula, R., Ahmer, M.F., Asiri, A.M., Eds.; CRC Press; Taylor & Francis Group: Abingdon, UK, 2019; Chapter 3; pp. 65–78.
- Kumar, A.B.V.K.; Chaudhary, S.; Ramana, C.H.V.V. Conducting Polymer-Metal-Based Binary Composites for Supercapacitor Applications. In Conducting Polymers-Based Energy Storage Materials; Inamuddin, Boddula, R., Ahmer, M.F., Asiri, A.M., Eds.; CRC Press; Taylor & Francis Group: Abingdon, UK, 2019; Chapter 14; pp. 209–234.
- Pal, B.; Yang, S.; Ramesh, S.; Thangadurai, V.; Jose, R. Electrolyte selection for supercapacitive devices: A critical review. Nanoscale Adv. 2019, 1, 3807–3835.
- Kim, B.K.; Sy, S.; Yu, A.; Zhang, J. Electrochemical Supercapacitors for Energy Storage and Conversion. In Handbook of Clean Energy Systems; John Whiley and Sons: Hoboken, NJ, USA, 2015; pp. 1–25.
- Huang, S.; Zhu, X.; Sarkar, S.; Zhao, Y. Challenges and opportunities for supercapacitors. APL Mater. 2019, 7, 100901.
- Patterson, N.; Xiao, B.; Ignaszak, A. Polypyrrole decorated metal–organic frameworks for supercapacitor devices. RSC Adv. 2020, 10, 20162–20172.
- Dall’Olio, A.; Dascola, G.; Varraca, V.; Bocchi, V. Electron paramagnetic resonance and conductivity of an electrolytic oxy-purrole black. Gazz. Chim. Ital. 1961, 46, 279, OCLC Number: 28804047.
- Gardini, G. The Oxidation of Monocyclic Pyrroles. Adv. Heterocycl. Chem. 1973, 15, 67–98.
- Kızılcan, N.; Öz, N.K.; Ustamehmetoglu, B.; Akar, A. High conductive copolymers of polypyrrole-α,ω-diamine polydimethylsiloxane. Eur. Polym. J. 2006, 42, 2361–2368.
- Jones, R.A.; Bean, G.P. Oxidation and Reduction of the Pyrrole Ring. In Organic Chemistry: A Series of Monographs; Elsevier BV: Amsterdam, The Netherlands, 1977; Volume 34, Chapter 5; pp. 209–247.
- Kanazawa, K.K.; Diaz, A.F.; Geiss, R.H.; Gill, W.D.; Kwak, J.F.; Logan, J.A.; Rabolt, J.F.; Street, G.B. ‘Organic metals’: Polypyrrole, a stable synthetic ‘metallic’ polymer. J. Chem. Soc. Chem. Commun. 1979, 19, 854–855.
- Lv, H.; Pan, Q.; Song, Y.; Liu, X.-X.; Liu, T. A Review on Nano-/Microstructured Materials Constructed by Electrochemical Technologies for Supercapacitors. Nano-Micro Lett. 2020, 12, 1–56.
- Abdah, M.A.A.M.; Azman, N.H.N.; Kulandaivalu, S.; Sulaiman, Y. Review of the use of transition-metal-oxide and conducting polymer-based fibres for high-performance supercapacitors. Mater. Des. 2020, 186, 108199.
- Wang, J.; Li, X.; Du, X.; Wang, J.; Ma, H.; Jing, X. Polypyrrole composites with carbon materials for supercapacitors. Chem. Pap. 2017, 71, 293–316.
- Moyseowicz, A.; Pająk, K.; Gajewska, K.; Gryglewicz, G. Synthesis of Polypyrrole/Reduced Graphene Oxide Hybrids via Hydrothermal Treatment for Energy Storage Applications. Materials 2020, 13, 2273.
- Borenstein, A.; Hanna, O.; Attias, R.; Luski, S.; Brousse, T.; Aurbach, D. Carbon-based composite materials for supercapacitor electrodes: A review. J. Mater. Chem. A 2017, 5, 12653–12672.
- Feng, M.; Lu, W.; Zhou, Y.; Zhen, R.; He, H.; Wang, Y.; Li, C. Synthesis of polypyrrole/nitrogen-doped porous carbon matrix composite as the electrode material for supercapacitors. Sci. Rep. 2020, 10, 1–12.
- Wang, L.; Zhang, C.; Jiao, X.; Yuan, Z. Polypyrrole-based hybrid nanostructures grown on textile for wearable supercapacitors. Nano Res. 2019, 12, 1129–1137.
- Li, Z.; Cai, J.; Cizek, P.; Niu, H.; Du, Y.; Lin, T. A self-supported, flexible, binder-free pseudo-supercapacitor electrode material with high capacitance and cycling stability from hollow, capsular polypyrrole fibers. J. Mater. Chem. A 2015, 3, 16162–16167.
- Dubal, D.; Patil, S.; Kim, W.B.; Lokhande, C. Supercapacitors based on electrochemically deposited polypyrrole nanobricks. Mater. Lett. 2011, 65, 2628–2631.
- Shinde, S.S.; Gund, G.S.; Kumbhar, V.S.; Patil, B.H.; Lokhande, C.D. Novel chemical synthesis of polypyrrole thin film electrodes for supercapacitor application. Eur. Polym. J. 2013, 49, 3734–3739.
- Huang, Y.; Tao, J.; Meng, W.; Zhu, M.; Fu, Y.; Gao, Y.; Zhi, C. Super-high rate stretchable polypyrrole-based supercapacitors with excellent cycling stability. Nano Energy 2015, 11, 518–525.
- Gan, J.K.; Lim, Y.S.; Huang, N.M.; Lim, H.N. Hybrid silver nanoparticle/nanocluster-decorated polypyrrole for high-performance supercapacitors. RSC Adv. 2015, 5, 75442–75450.
- Iqbal, J.; Numan, A.; Ansari, M.O.; Jagadish, P.R.; Jafer, R.; Bashir, S.; Mohamad, S.; Ramesh, K.; Ramesh, S. Facile synthesis of ternary nanocomposite of polypyrrole incorporated with cobalt oxide and silver nanoparticles for high performance supercapattery. Electrochim. Acta 2020, 348, 136313.
- Elahi, H.; Munir, K.; Eugeni, M.; Atek, S.; Gaudenzi, P. Energy Harvesting towards Self-Powered IoT Devices. Energies 2020, 13, 5528.
