Rhes is one of the most interesting genes regulated by thyroid hormones that, through the inhibition of the striatal cAMP/PKA pathway, acts as a modulator of dopamine neurotransmission. Rhes mRNA is expressed at high levels in the dorsal striatum, with a medial-to-lateral expression gradient reflecting that of both dopamine D2 and adenosine A2A receptors. Rhes transcript is also present in the hippocampus, cerebral cortex, olfactory tubercle and bulb, substantia nigra pars compacta (SNc) and ventral tegmental area of the rodent brain. In line with Rhes-dependent regulation of dopaminergic transmission, data showed that lack of Rhes enhanced cocaine- and amphetamine-induced motor stimulation in mice.
- substantia nigra
- mTOR
- SUMO E3 ligase
- Huntington’s disease
- 3
- 4-methylenedioxymethamphetamine (MDMA)
- autophagy
- L-Dopa-induced dyskinesia (LID)
- mitophagy
1. Discovery of Rhes
1.1. Protein Structure
1.2. Anatomical Brain Localization
2. Ontogeny of Rhes and Its Striatal Regulation
2.1. Rhes Is Modulated by Thyroid Hormones
2.2. Rhes Expression Is Regulated by Dopamine Innervation
3. Rhes Intracellular Signaling
3.1. In Vitro Modulation of Rhes-Dependent cAMP/PKA Signaling
3.2. Rhes Affects Striatal cAMP/PKA Signaling in Mice
3.3. Rhes Affects the PI3K/Akt Signaling Pathway
4. Rhes Involvement in Huntington Disease and L-DOPA-Induced Dyskinesia
4.1. Rhes Acts as SUMO E3 Ligase for the Mutant Huntingtin
4.2. Role of Rhes in Modulating HD-Dependent Phenotypes in Animal Models
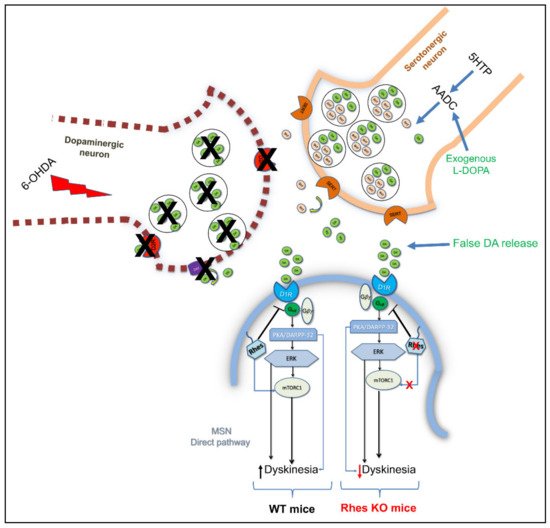
4.3. Rhes Affects L-DOPA-Induced Dyskinesia (LID) in PD Mouse Model
5. Involvement of Rhes in Parkinson’s Disease: Focus on Rhes Regulation of Nigrostriatal Neurons’ Survival
5.1. Rhes Counteracts Nigrostriatal Degeneration during Ageing in a Gender-Dependent Manner
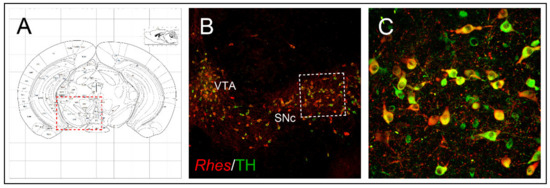
5.2. Rhes Reduces the MDMA-Induced Dopaminergic Degeneration and Neuroinflammation Affecting the Nigrostriatal System
This entry is adapted from the peer-reviewed paper 10.3390/ijms22105326
