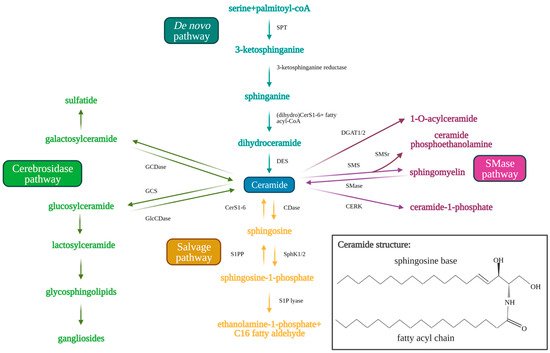
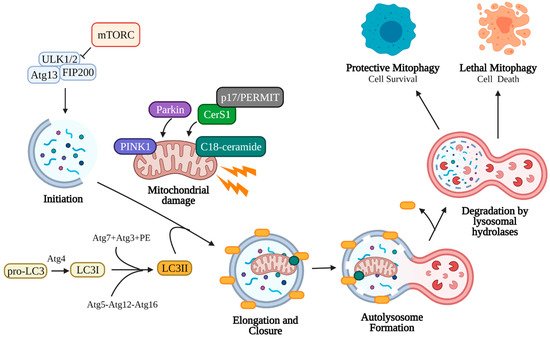
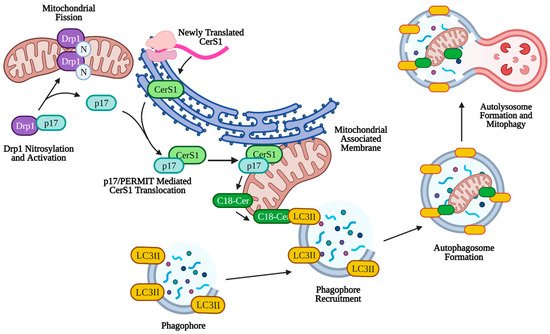
CerS1-6 function in the production of variable length ceramides that have distinct effects on cancer cell growth and survival. CerS1 is notable for its synthesis of C18-ceramide, a pro-apoptotic and anti-tumorigenic ceramide that has been shown to be downregulated in head and neck squamous cell carcinoma (HNSCC) compared to healthy control tissue [
60,
61,
62,
63,
64]. C18-ceramide production by CerS1 is inhibited in HNSCC through transcriptional repression via histone deacetylase 1 (HDAC1)-dependent inhibition of Sp1 at the promoter, and post-transcriptionally by miR574-5p that induces translation of a CerS1 isoform 2 splice variant common in HNSCC tumor tissues [
65]. Reduced C18-ceramide levels in HNSCC has been associated with tumor metastasis, advanced stage cancer, and increased lymphovascular invasion [
61,
62,
66]. C18-ceramide has also been shown to interact with inhibitor 2 of protein phosphatase 2A (I2PP2A), which indirectly activates PP2A and induces degradation of c-Myc in lung adenocarcinoma cell lines [
47,
67,
68,
69,
70,
71]. CerS1 depletion has been associated with poorer prognosis and patient outcomes in breast cancer and neuroblastoma [
64,
72]. Reduced levels of C18-ceramide were also found in colorectal cancer and glioma tissue, where exogenous C18-ceramide was found to induce ER-stress and lethal autophagy in the latter [
73,
74]. Like CerS1, CerS4 also contributes to C18-C20 ceramide synthesis. CerS4 mRNA was found to be higher in early stage, non-metastatic HNSCC, melanoma, and renal cell carcinoma tumors compared to later stage, aggressive, metastatic tumors. Knocking down CerS4 in lung adenocarcinoma and HNSCC cell lines reduced cell migration in vitro and liver metastasis from murine mammary cancer cells in vivo [
75].
CerS2 generates longer C22–C24 ceramides and was similarly found to be downregulated in many cancers. Mice that expressed a catalytically inactive mutant CerS2 developed hepatocellular carcinoma (HCC) at a young age (8 weeks), while mice that were deficient for CerS2 developed liver adenoma and HCC later in adulthood (7–10 months) [
76,
77,
78]. Similarly, CerS2 knockout mice were susceptible to azoxymethane induction of colon carcinoma and dextran sodium sulfate induction of colitis [
36]. CerS2 overexpression in MDA-MB-231 breast cancer cells was shown to decrease cell migration and invasion, and CerS2 expression was associated with improved patient survival in breast, ovarian, lung, and liver cancer [
65,
79]. CerS5 has been noted to be upregulated in colorectal cancer and was associated with poor patient survival and 5-year cancer recurrence [
73,
80,
81]. CerS5 has also been noted to be a biomarker for colorectal cancer [
81]. Interestingly, CerS5 and CerS6 knockdown was shown to sensitize mice to azoxymethane/dextran sodium sulfate-induced colitis and increased cases of colitis-associated colon cancer [
82,
83]. CerS6 is notable for its generation of C16-ceramide, which is involved in apoptosis induction and the protection of ER and Golgi membrane integrity in cancer. CerS6 derived C16-ceramide was found to induce apoptosis in lung adenocarcinoma cells following non-genotoxic folate stress through p53 transcriptional targeting [
84]. C16-ceramide induced apoptosis through BAX in HeLa cells following irradiation, and C16-ceramide also activated caspase 3 translocation to the nucleus resulting in increased tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) sensitivity in colon cancer cells [
85,
86]. Paradoxically, C16-ceramide was found elevated in oral and gastric cancer [
64,
87,
88]. CerS6 overexpression has been described as a biomarker in gastric cancer, and CerS6 overexpression was associated with poor patient survival, invasion, and metastasis in gastric cancer [
88]. It was found that HNSCC exhibited upregulated CerS6 derived C16-ceramide, which resulted in a protective effect seen towards the ER and Golgi membrane integrity. When CerS6 was inhibited, activating transcription factor 6 (ATF6) induced ER-stress resulting in cell death [
10,
87,
89,
90]. Overall, these studies suggest that distinct/paradoxic biological roles of ceramides in cancer are context dependent regulated by their subcellular localization and down-stream targets.