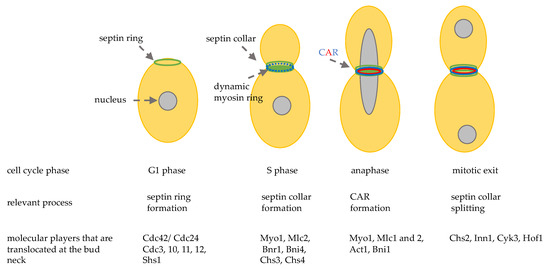
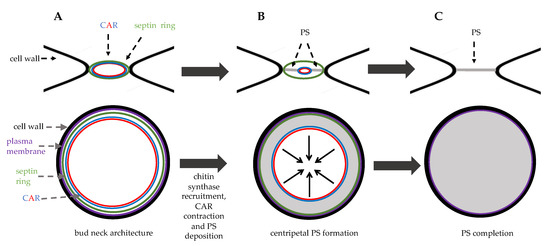
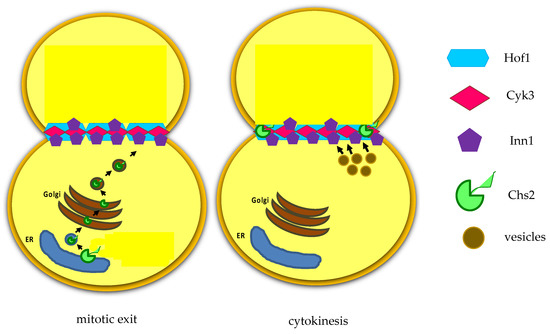
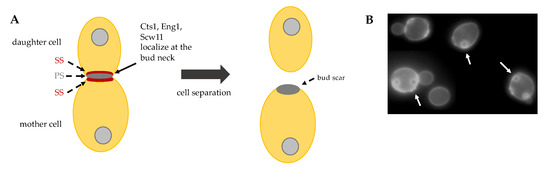
The activity of these hydrolytic enzymes is tightly regulated: they are specifically transcribed in the daughter cell and are correctly delivered at the daughter bud neck by the Golgi pathway. The regulatory proteins Cbk1 and Mob2 localize to the neck and to the daughter nuclei at the end of mitosis [
47], in particular they form a complex with the transcriptional factor Ace2 and prevent its export from the nucleus, thus ensuring daughter-specific expression of cell separation factors [
48]. In addition, Cts1 is post-translationally modified and delivered to the daughter bud neck through the Golgi pathway [
49]. Recently, a new level of Cts1 regulation has been described: upon incomplete septation, Fir1 inhibits Cbk1 activity, thus blocking production and secretion of septum degrading enzymes [
50]. This mechanism inhibits cell separation until septation is properly executed and, therefore, enforces cytokinesis order of events.
To sum up, the successful completion of cell separation depends upon the precise construction of the trilaminar septum in coordination with the division cycle, and then on the temporal and spatial regulation of chitinase and glucanase action.
4. Checkpoint Pathways that Control Cytokinesis
Cytokinesis must be tightly coordinated with the nuclear cycle in order to maintain genetic stability. Eukaryotic cells have evolved several checkpoints and checkpoint-like mechanisms that preserve the integrity of cell division.
The mitotic checkpoint is an evolutionarily conserved signaling pathway that blocks mitosis progression in case of problems in the mitotic spindle, in chromosome attachment to MTs, or in presence of mistakes in chromosome segregation. In budding yeast, the division site is determined before mitotic spindle formation, which implies that the bipolar spindle must be properly positioned at the bud neck and aligned perpendicularly with respect to the division axis before anaphase onset. Two pathways direct spindle positioning, the dynein pathway and Kar9 pathway, and the spindle orientation checkpoint (SPOC) blocks mitotic exit and cytokinesis in the case of defects [
52]. If the checkpoint fails, cytokinesis occurs, even if the nucleus divides into the mother cell, thus causing the formation of aneuploid cells. The target of the SPOC is Tem1, the G protein at the top of MEN pathway.
Mitotic Exit Network (MEN) in
S. cerevisiae and Septation Initiation Network (SIN) in S.
pombe are two signaling pathways that coordinate mitosis progression from anaphase onset to cell separation. MEN is a signaling cascade with the GTPase Tem1 at the top, several protein kinases, among which the Hippo-like kinase Cdc15 and the LATS-like kinase Dbf2, and the final target is the phosphatase Cdc14. MEN pathway leads to complete Cdc14 activation that causes a decrease in mitotic kinase activity (Cdk1), triggering mitotic exit [
53]. Before anaphase onset, MEN components localize at the spindle pole bodies (the yeast centrosomes), but later they are found at the bud neck, where they ensure that mitotic exit only occurs after acto-myosin ring contraction and septum deposition [
54]. In addition, in telophase, Cdc14 dephosphorylates cytoplasmic and bud-neck associated targets of Cdk1 and contributes to reorganize actin cytoskeleton and target secretion vesicles to the bud neck [
55,
56]. In
S. pombe the SIN controls CAR contraction and septum deposition, rather than mitotic exit and blocks septation in the case of problems [
57]. Homologs of Cdc14, MEN, and SIN components are also found in other yeasts:
Candida albicans, Aspergillus nidulans, and
Ashbya gossypii. Budding yeast Cdc14 is conserved in higher eukaryotes, however these homologs do not seem to control mitotic exit, rather they are involved in other processes, such as DNA replication, DNA damage repair, nuclear organization, mitotic entry, mitotic spindle assembly, and cytokinesis [
58]. In
Drosophila and human cells, MEN homologs are part of the Hippo pathway that controls centrosome duplication, cell proliferation, and apoptosis [
59,
60]. The conserved Hippo pathway inhibits cell proliferation by the activation of LATS1 and LATS2 kinases and by p53 stabilization (see below).
Mammalian cells have a checkpoint that arrest cells in mitosis in response to several problems in order to preserve genetic stability and cell survival, rarely cells can escape this arrest and enter mitosis without chromosome segregation, thus becoming tetraploid [
61,
62]. Importantly, another checkpoint control is active in human cells to recognize cytokinesis failure and induce proliferation arrest in G1. Indeed, in tetraploid cells, arising either by failure of mitotic spindle or of cytokinesis, the checkpoint induces a p53-dependent cell cycle arrest, thus preventing the proliferation of aneuploid cells and carcinogenesis [
63]. However it is not clear how, at which stage and by which signal (from extra chromosomes or extra centrosomes) p53 is activated by tetraploidy. Interestingly, cytokinesis failure activates the Hippo pathway: LATS2, which is an important kinase of Hippo pathway, translocates from centrosomes to the nucleus and stabilizes p53 in the presence of additional centrosomes [
64,
65]. In addition, extra centrosomes can activate RAC1, which is known to antagonize RhoA, which leads to LATS2 activation [
66]. If p53 function is lost, cells can override the cell cycle block that is induced by tetraploidization, accumulate chromosome aberrations, and start to proliferate without control.
Cytokinesis can be blocked even when the furrow started to ingress: the abscission checkpoint delays cytokinesis completion and can even drive the regression of the cleavage furrow in the presence of chromatin bridges or lagging chromosomes [
67]. The abscission delay is dependent upon the localization of the Aurora B kinase at the midbody. Aurora B translocates from kinetochores to the division site after anaphase onset. At the midbody, activated Aurora B phosphorylates the ESCRT-III subunit Chmp4c, but the molecular mechanism that delays abscission is not completely understood.
Human cells are well equipped with several pathways that block tetraploid cells proliferation since cytokinesis failure can be tumorigenic. However tetraploidization is used by evolution and it can be important for certain tissues to gain new advantageous traits, such as resistance to drugs or stimuli (see also paragraph 6).
Interesting data suggest new connections between cytokinesis and DNA damage, in particular cytokinesis might be regulated in response to DNA damage. Damaged DNA should not be segregated, so, in this case, cytokinesis must be blocked to prevent the cleavage furrow from cutting damaged DNA. Proteins that are involved in DNA repair, such as BRCA2 and BCCIP, may be directly involved in cytokinesis, as their deficiency induces cytokinetic abnormalities [
68]. The polo kinase Plk1 is an important regulator of mitosis and cytokinesis and it is also a modulator of the DNA damage checkpoint [
69]. The budding yeast DNA damage checkpoint kinase Rad53 localizes at the division site and associates with septins [
70]. Other experiments suggest that DNA damage pathways may regulate cytokinesis proteins by modulating their the expression or their post-translational modifications, as p53 and Rb pathways inhibit the expression of cytokinetic proteins, such as Plk1, ECT2, anillin, and survivin [
71,
72], and BRCT inhibits Aurora B kinase activity by Poly(ADP-ribosyl)ation in response to DNA strand breaks [
73]. It is reasonable that DNA damage, and perhaps incompletely replicated DNA, activates pathways that promote DNA repair, arrests cell cycle progression, and blocks cytokinesis completion.