The dogma of mitochondria as the major source of energy in supporting sperm motility should be critically reconsidered in the light of several experimental data pointing to a major role of glycolysis in mammalian spermatozoa. In this light, the reported positive correlation between the mitochondrial membrane potential (ΔΨm) and motility of ejaculated spermatozoa cannot be explained convincingly by an impaired mitochondrial ATP generation only. Evidence has been produced suggesting that, in human sperm, dysfunctional mitochondria represent the main site of generation of reactive oxygen species (ROS). Furthermore, in these organelles, a complex bidirectional relationship could exist between ROS generation and apoptosis-like events that synergize with oxidative stress in impairing sperm biological integrity and functions. Despite the activity of enzymatic and non-enzymatic antioxidant factors, human spermatozoa are particularly vulnerable to oxidative stress, which plays a major role in male factor infertility.
- adenosine trisphosphate (ATP)
- apoptosis
- glycolysis
- mitochondria
- oxidative phosphorylation
- spermatozoa
- energetic metabolism
- sperm motility
- reactive oxygen species (ROS)
1. Introduction
metabolism and oxidative/apoptotic events.
2. Are Mitochondria Really the Energetic Motor of Mammalian Sperm?
The role of mitochondria in the energetic support of sperm motility is a matter of debate [11,12,13]. Two pathways can account for the generation of ATP in mammalian spermatozoa, glycolysis and mitochondrial respiration. As mitochondrial OXPHOS is much more efficient than glycolysis in generating ATP, it has been widely accepted that the ATP needed for sperm motility is synthesized by mitochondrial respiration.
In mammalian spermatozoa, mitochondria rearrange in tubular structures that are helically distributed around the anterior portion of the axoneme, constituting the midpiece [14,15]. As the sperm flagellum is long and thin and mitochondria are confined in its proximal end, the question has been raised as to whether OXPHOS-derived ATP can passively diffuse through the entire flagellum to efficiently support axoneme activity. In sea urchin sperm, a shuttle mechanism to facilitate the ATP diffusion along the flagellum is provided by the creatine phosphate (CrP) that buffers the ATP/adenosine diphosphate (ADP) ratio at the expense of CrP/creatine [16]. However, mammalian spermatozoa lack or contain only low amounts of CrP or other phosphagens [17,18], making it unlikely that the CrP shuttle plays a major role in providing ATP from mitochondria to the axoneme. Indeed, spermatozoa from knockout mouse models where the gene for the mitochondrial isotype of creatine kinase had been inactivated exhibited similar motility patterns to the wild-type controls [19]. These legitimate considerations shifted the focus from OXPHOS to glycolysis.
Although mitochondrial respiration is more efficient than glycolysis in generating ATP molecules, key enzymes of glycolysis are tethered to the fibrous sheath of the principal piece [20,21,22,23], and hence they might assure an efficient production of ATP for dynein ATPase locally in the entire length of the flagellum. Consistent with this view, in mouse [3], bovine [24] and human spermatozoa [5,6,7], motility was not affected by mitochondrial inhibition when glucose was available in the extracell ular medium. We previously demonstrated that in a medium lacking glycolysable sugars, the presence of substrates for OXPHOS such as pyruvate and lactate fully supported the motility of human spermatozoa [7].
Interestingly, under such experimental conditions, the addition of 2-Deoxy-D-glucose (DOG), which inhibits glycolysis by competing with glucose for key enzymes, significantly decreased sperm motility [7]. This evidence was incompatible with the hypothesis that ATP is synthesized in mitochondria and then provided to the entire axoneme by diffusion.
On the contrary, these findings supported the notion that ATP produced by OXPHOS is used to drive gluconeogenesis and thus to supply glucose to glycolytic enzymes for ATP production in the principal piece.
In this light, glycolysis would compensate for any lack of ATP production by mitochondria in maintaining sperm motility, and mitochondrial OXPHOS inhibition could depress motility only under experimental conditions of concomitant glycolysis blockage. However, differences among the species exist, as stallion spermatozoa rely primarily on mitochondrial respiration to generate energy required for motility [25]. Overall, it is conceivable that both glycolysis and OXPHOS contribute to ATP production, depending on each other in controlling sperm functions according to the different availability of energetic substrates in the environment [4]. Of note, in female genital tract fluids, the concentrations of lactate are higher than those of glycolysable substrates [26,27,28,29], suggesting a possible major role of mitochondrial respiration in supporting sperm motility. This hypothesis could explain why spermatozoa retain a high number of mitochondria during their differentiation, despite the dramatical decrease in the cellular volume resulting from the removal of any unnecessary structure. Anyway, an obligatory role for glycolysis seems to be confirmed by the loss of progressive motility in spermatozoa of mouse models where the gene for sperm-specific glyceraldehyde-3-phosphate dehydrogenases had been knocked out [4]. In this view, the reported correlation of the mitochondrial membrane potential (∆Ψm) [30] or mitochondrial morphologic integrity [31] with the motility of ejaculated spermatozoa cannot be explained convincingly by an impaired mitochondrial ATP generation only.
Noteworthy, in human spermatozoa, a mitochondrial dysfunction could affect motility when it is accompanied by an intrinsic generation of ROS. Oxidative stress, indeed, is responsible for membrane lipid peroxidation [5,32] and promotes the activation of mitochondrial pathways resulting in apoptosis-like changes.
3. Biochemistry of Reactive Oxygen Species: An Overview
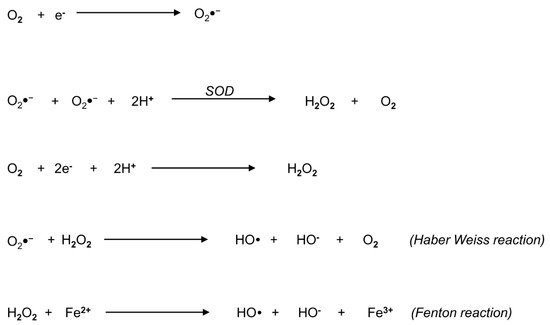
4. Origin of ROS in Semen
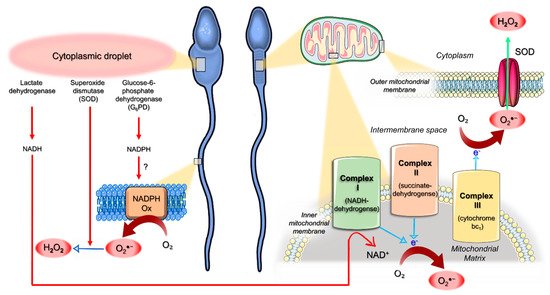
5. Mitochondria as an Interplay Center between Oxidative Stress and Apoptotic Events
6. Pathophysiology of Oxidative Stress in Human Spermatozoa
7. Conclusions
References
- Cummins, J. Mitochondrial DNA in mammalian reproduction. Rev. Reprod. 1998, 3, 172–182. [Google Scholar] [CrossRef] [PubMed]
- Saraste, M. Oxidative phosphorylation at the fin de siècle. Science 1999, 283, 1488–1493. [Google Scholar] [CrossRef] [PubMed]
- Mukai, C.; Okuno, M. Glycolysis plays a major role for adenosine triphosphate supplementation in mouse sperm flagellar movement. Biol. Reprod. 2004, 71, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Miki, K.; Qu, W.; Goulding, E.H.; Willis, W.D.; Bunch, D.O.; Strader, L.F.; Perreault, S.D.; Eddy, E.M.; O’Brien, D.A. Glyceraldehyde 3-phosphate dehydrogenase-S, a sperm-specific glycolytic enzyme, is required for sperm motility and male fertility. Proc. Natl. Acad. Sci. USA 2004, 101, 16501–16506. [Google Scholar] [CrossRef]
- Losano, J.D.A.; Padín, J.F.; Méndez-López, I.; Angrimani, D.S.R.; García, A.G.; Barnabe, V.H.; Nichi, M. The Stimulated Glycolytic Pathway Is Able to Maintain ATP Levels and Kinetic Patterns of Bovine Epididymal Sperm Subjected to Mitochondrial Uncoupling. Oxid. Med. Cell Longev. 2017, 1682393. [Google Scholar] [CrossRef]
- Koppers, A.J.; De Iuliis, G.N.; Finnie, J.M.; McLaughlin, E.A.; Aitken, R.J. Significance of mitochondrial reactive oxygen species in the generation of oxidative stress in spermatozoa. J. Clin. Endocrinol. Metab. 2008, 93, 3199–3207. [Google Scholar] [CrossRef]
- Nascimento, J.M.; Shi, L.Z.; Tam, J.; Chandsawangbhuwana, C.; Durrant, B.; Botvinick, E.L.; Berns, M.W. Comparison of glycolysis and oxidative phosphorylation as energy sources for mammalian sperm motility, using the combination of fluorescence imaging, laser tweezers, and real-time automated tracking and trapping. J. Cell Physiol. 2008, 217, 745–751. [Google Scholar] [CrossRef]
- Barbonetti, A.; Vassallo, M.R.; Fortunato, D.; Francavilla, S.; Maccarrone, M.; Francavilla, F. Energetic metabolism and human sperm motility: Impact of CB1 receptor activation. Endocrinology 2010, 151, 5882–5892. [Google Scholar] [CrossRef]
- Ortega-Ferrusola, C.; García, B.M.; Gallardo-Bolaños, J.M.; González-Fernández, L.; Rodríguez-Martinez, H.; Tapia, J.A.; Peña, F.J. Apoptotic markers can be used to forecast the freezeability of stallion spermatozoa. Anim. Reprod. Sci. 2009, 114, 393–403. [Google Scholar] [CrossRef]
- Amaral, A.; Paiva, C.; Attardo Parrinello, C.; Estanyol, J.M.; Ballescà, J.L.; Ramalho-Santos, J.; Oliva, R. Identification of proteins involved in human sperm motility using high-throughput differential proteomics. J. Proteome Res. 2014, 13, 5670–5684. [Google Scholar] [CrossRef]
- Ford, W.C. Glycolysis and sperm motility: Does a spoonful of sugar help the flagellum go round? Hum. Reprod. Update 2006, 12, 269–274. [Google Scholar] [CrossRef]
- Miki, K. Energy metabolism and sperm function. Soc. Reprod. Fertil. Suppl. 2007, 65, 309–325. [Google Scholar]
- Ruiz-Pesini, E.; Díez-Sánchez, C.; López-Pérez, M.J.; Enríquez, J.A. The role of the mitochondrion in sperm function: Is there a place for oxidative phosphorylation or is this a purely glycolytic process? Curr. Top. Dev. Biol. 2007, 77, 3–19. [Google Scholar] [PubMed]
- Tombes, R.M.; Shapiro, B.M. Metabolite channeling: A phosphorylcreatine shuttle to mediate high energy phosphate transport between sperm mitochondrion and tail. Cell 1985, 41, 325–334. [Google Scholar] [CrossRef]
- Smith, M.B.; Babcock, D.F.; Lardy, H.A. A 31P NMR study of the epididymis and epididymal sperm of the bull and hamster. Biol. Reprod. 1985, 33, 1029–1040. [Google Scholar] [CrossRef] [PubMed]
- Robitaille, P.M.; Robitaille, P.A.; Martin, P.A.; Brown, G.G. Phosphorus-31 nuclear magnetic resonance studies of spermatozoa from the boar, ram, goat and bull. Comp. Biochem. Physiol. B 1987, 87, 285–296. [Google Scholar] [CrossRef]
- Steeghs, K.; Oerlemans, F.; Wieringa, B. Mice deficient in ubiquitous mitochondrial creatine kinase are viable and fertile. Biochim. Biophys. Acta 1995, 1230, 130–138. [Google Scholar] [CrossRef]
- Westhoff, D.; Kamp, G. Glyceraldehyde 3-phosphate dehydrogenase is bound to the fibrous sheath of mammalian spermatozoa. J. Cell Sci. 1997, 110 Pt 15, 1821–1829. [Google Scholar] [CrossRef]
- Travis, A.J.; Foster, J.A.; Rosenbaum, N.A.; Visconti, P.E.; Gerton, G.L.; Kopf, G.S.; Moss, S.B. Targeting of a germ cell-specific type 1 hexokinase lacking a porin-binding domain to the mitochondria as well as to the head and fibrous sheath of murine spermatozoa. Mol. Biol. Cell. 1998, 9, 263–276. [Google Scholar] [CrossRef]
- Mori, C.; Nakamura, N.; Welch, J.E.; Gotoh, H.; Goulding, E.H.; Fujioka, M.; Eddy, E.M. Mouse spermatogenic cell-specific type 1 hexokinase (mHk1-s) transcripts are expressed by alternative splicing from the mHk1 gene and the HK1-S protein is localized mainly in the sperm tail. Mol. Reprod. Dev. 1998, 49, 374–385. [Google Scholar] [CrossRef]
- Angulo, C.; Rauch, M.C.; Droppelmann, A.; Reyes, A.M.; Slebe, J.C.; Delgado-López, F.; Guaiquil, V.H.; Vera, J.C.; Concha, I.I. Hexose transporter expression and function in mammalian spermatozoa: Cellular localization and transport of hexoses and vitamin C. J. Cell Biochem. 1998, 71, 189–203. [Google Scholar] [CrossRef]
- Davila, M.P.; Muñoz, P.M.; Bolaños, J.M.; Stout, T.A.; Gadella, B.M.; Tapia, J.A.; da Silva, C.B.; Ferrusola, C.O.; Peña, F.J. Mitochondrial ATP is required for the maintenance of membrane integrity in stallion spermatozoa, whereas motility requires both glycolysis and oxidative phosphorylation. Reproduction 2016, 152, 683–694. [Google Scholar] [CrossRef]
- Windsor, D.P. Mitochondrial function and ram sperm fertility. Reprod. Fertil. Dev. 1997, 9, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.R. Chemical interference with sperm metabolic pathways. J. Reprod. Fertil. Suppl. 1998, 53, 227–234. [Google Scholar]
- Galantino-Homer, H.L.; Florman, H.M.; Storey, B.T.; Dobrinski, I.; Kopf, G.S. Bovine sperm capacitation: Assessment of phosphodiesterase activity and intracellular alkalinization on capacitation-associated protein tyrosine phosphorylation. Mol. Reprod. Dev. 2004, 67, 487–500. [Google Scholar] [CrossRef]
- Harris, S.E.; Gopichandran, N.; Picton, H.M.; Leese, H.J.; Orsi, N.M. Nutrient concentrations in murine follicular fluid and the female reproductive tract. Theriogenology 2005, 64, 992–1006. [Google Scholar] [CrossRef]
- Paoli, D.; Gallo, M.; Rizzo, F.; Baldi, E.; Francavilla, S.; Lenzi, A.; Lombardo, F.; Gandini, L. Mitochondrial membrane potential profile and its correlation with increasing sperm motility. Fertil. Steril. 2011, 95, 2315–2319. [Google Scholar] [CrossRef] [PubMed]
- Pelliccione, F.; Micillo, A.; Cordeschi, G.; D’Angeli, A.; Necozione, S.; Gandini, L.; Lenzi, A.; Francavilla, F.; Francavilla, S. Altered ultrastructure of mitochondrial membranes is strongly associated with unexplained asthenozoospermia. Fertil. Steril. 2011, 95, 641–646. [Google Scholar] [CrossRef]
- Barbonetti, A.; Vassallo, M.R.; Di Rosa, A.; Leombruni, Y.; Felzani, G.; Gandini, L.; Lenzi, A.; Necozione, S.; Francavilla, S.; Francavilla, F. Involvement of mitochondrial dysfunction in the adverse effect exerted by seminal plasma from men with spinal cord injury on sperm motility. Andrology 2013, 1, 456–463. [Google Scholar] [CrossRef]
- Aitken, R.J.; Fisher, H. Reactive oxygen species generation and human spermatozoa: The balance of benefit and risk. BioEssays 1994, 16, 259–267. [Google Scholar] [CrossRef]
- Aitken, R.J. Reactive oxygen species as mediators of sperm capacitation and pathological damage. Mol. Reprod. Dev. 2017, 84, 1039–1052. [Google Scholar] [CrossRef] [PubMed]
- Kehrer, J.P.; Robertson, J.D.; Smith, C.V. Free Radicals and Reactive Oxygen Species. Compr. Toxicol. 2010, 1, 277–307. [Google Scholar]
- Tremellen, K. Oxidative stress and male infertility—A clinical perspective. Hum. Reprod. Update 2008, 14, 243–258. [Google Scholar] [CrossRef]
- Aitken, R.J.; Koppers, A.J. Apoptosis and DNA damage in human spermatozoa. Asian J. Androl. 2011, 13, 36–42. [Google Scholar] [CrossRef]
- Plante, M.; de Lamirande, E.; Gagnon, C. Reactive oxygen species released by activated neutrophils, but not by deficient spermatozoa, are sufficient to affect normal sperm motility. Fertil. Steril. 1994, 62, 387–393. [Google Scholar] [CrossRef]
- Micillo, A.; Vassallo, M.R.; Cordeschi, G.; D’Andrea, S.; Necozione, S.; Francavilla, F.; Francavilla, S.; Barbonetti, A. Semen leukocytes and oxidative-dependent DNA damage of spermatozoa in male partners of subfertile couples with no symptoms of genital tract infection. Andrology 2016, 4, 808–815. [Google Scholar] [CrossRef]
- Castellini, C.; D’Andrea, S.; Martorella, A.; Minaldi, E.; Necozione, S.; Francavilla, F.; Francavilla, S.; Barbonetti, A. Relationship between leukocytospermia, reproductive potential after assisted reproductive technology, and sperm parameters: A systematic review and meta-analysis of case-control studies. Andrology 2020, 8, 125–135. [Google Scholar] [CrossRef]
- Agarwal, A.; Salch, R.H.; Bedaiwy, M.A. Role of reactive oxygen species in the pathophysiology of human reproduction. Fertil. Steril. 2003, 79, 829–843. [Google Scholar] [CrossRef]
- Aitken, R.J.; Paterson, M.; Fisher, H.; Buckingham, D.W.; van Duin, M. Redox regulation of tyrosine phosphorylation in human spermatozoa and its role in the control of human sperm function. J. Cell Sci. 1995, 108, 2017–2025. [Google Scholar] [CrossRef]
- Orlando, C.; Krausz, C.; Forti, G.; Casano, R. Simultaneous measurement of sperm LDH, LDH-X, CPK activities and ATP content in normospermic and oligozoospermic men. Int. J. Androl. 1994, 17, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Gomez, E.; Buckingham, D.W.; Brindle, J.; Lanzafame, F.; Irvine, D.S.; Aitken, R.J. Development of an image analysis system to monitor the retention of residual cytoplasm by human spermatozoa: Correlation with biochemical markers of the cytoplasmic space, oxidative stress, and sperm function. J. Androl. 1996, 17, 276–287. [Google Scholar]
- Han, D.; Williams, E.; Cadenas, E. Mitochondrial respiratory chain-dependent generation of superoxide anion and its release into the intermembrane space. Biochem. J. 2001, 353 Pt 2, 411–416. [Google Scholar] [CrossRef]
- Chen, Y.R.; Zweier, J.L. Cardiac mitochondria and reactive oxygen species generation. Circ. Res. 2014, 114, 524–537. [Google Scholar] [CrossRef]
- Han, D.; Antunes, F.; Canali, R.; Rettori, D.; Cadenas, E. Voltage-dependent anion channels control the release of the superoxide anion from mitochondria to cytosol. J. Biol. Chem. 2003, 278, 5557–5563. [Google Scholar] [CrossRef]
- Balaban, R.S.; Nemoto, S.; Finkel, T. Mitochondria, oxidants, and aging. Cell 2005, 120, 483–495. [Google Scholar] [CrossRef]
- Aitken, R.J.; Whiting, S.; De Iuliis, G.N.; McClymont, S.; Mitchell, L.A.; Baker, M.A. Electrophilic aldehydes generated by sperm metabolism activate mitochondrial reactive oxygen species generation and apoptosis by targeting succinate dehydrogenase. J. Biol. Chem. 2012, 287, 33048–33060. [Google Scholar] [CrossRef] [PubMed]
- Hotchkiss, R.S.; Strasser, A.; McDunn, J.E.; Swanson, P.E. Cell death. N. Engl. J. Med. 2009, 361, 1570–1583. [Google Scholar] [CrossRef]
- Li, P.; Nijhawan, D.; Budihardjo, I.; Srinivasula, S.M.; Ahmad, M.; Alnemri, E.S.; Wang, X. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 1997, 91, 479–489. [Google Scholar] [CrossRef]
- Du, C.; Fang, M.; Li, Y.; Li, L.; Wang, X. Smac, a mitochondrial protein that promotes cytochrome c-dependent caspase activation by eliminating IAP inhibition. Cell 2000, 102, 33–42. [Google Scholar] [CrossRef]
- Goldstein, J.C.; Muñoz-Pinedo, C.; Ricci, J.E.; Adams, S.R.; Kelekar, A.; Schuler, M.; Tsien, R.Y.; Green, D.R. Cytochrome c is released in a single step during apoptosis. Cell Death Differ. 2005, 12, 453–462. [Google Scholar] [CrossRef]
- Wu, H.; Tschopp, J.; Lin, S.C. Smac mimetics and TNFalpha: A dangerous liaison? Cell 2007, 131, 655–658. [Google Scholar] [CrossRef] [PubMed]
- Strasser, A. The role of BH3-only proteins in the immune system. Nat. Rev. Immunol. 2005, 5, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Jones, D.P. Superoxide in apoptosis. Mitochondrial generation triggered by cytochrome c loss. J. Biol. Chem. 1998, 273, 11401–11404. [Google Scholar] [CrossRef] [PubMed]
- Barbonetti, A.; Castellini, C.; Di Giammarco, N.; Santilli, G.; Francavilla, S.; Francavilla, F. In vitro exposure of human spermatozoa to bisphenol A induces pro-oxidative/apoptotic mitochondrial dysfunction. Reprod. Toxicol. 2016, 66, 61–67. [Google Scholar] [CrossRef]
- De Lamirande, E.; Gagnon, C. Human sperm hyperactivation and capacitation as parts of an oxidative process. Free Radic. Biol. Med. 1993, 14, 157–166. [Google Scholar] [CrossRef]
- Aitken, R.J.; Ryan, A.L.; Baker, M.A.; McLaughlin, E.A. Redox activity associated with the maturation and capacitation of mammalian spermatozoa. Free Radic. Biol. Med. 2004, 36, 994–1010. [Google Scholar] [CrossRef]
- De Lamirande, E.; Tsai, C.; Harakat, A.; Gagnon, C. Involvement of reactive oxygen species in human sperm arcosome reaction induced by A23187, lysophosphatidylcholine, and biological fluid ultrafiltrates. J. Androl. 1998, 19, 585–594. [Google Scholar]
- Aitken, R.J.; Buckingham, D.W.; Brindle, J.; Gomez, E.; Baker, H.W.; Irvine, D.S. Analysis of sperm movement in relation to the oxidative stress created by leukocytes in washed sperm preparations and seminal plasma. Hum. Reprod. 1995, 10, 2061–2071. [Google Scholar] [CrossRef]
- Aitken, R.J. Free radicals, lipid peroxidation and sperm function. Reprod. Fertil. Dev. 1995, 7, 659–668. [Google Scholar] [CrossRef]
- Kodama, H.; Kuribayashi, Y.; Gagnon, C. Effect of sperm lipid peroxidation on fertilization. J. Androl. 1996, 17, 151–157. [Google Scholar]
- Griveau, J.F.; Le Lannou, D. Reactive oxygen species and human spermatozoa: Physiology and pathology. Int. J. Androl. 1997, 20, 61–69. [Google Scholar] [CrossRef]
- Armstrong, J.S.; Rajasekaran, M.; Chamulitrat, W.; Gatti, P.; Hellstrom, W.J.; Sikka, S.C. Characterization of reactive oxygen species induced e_ects on human spermatozoa movement and energy metabolism. Free Radic. Biol. Med. 1999, 26, 869–880. [Google Scholar] [CrossRef]
- Aitken, R.J.; Gibb, Z.; Baker, M.A.; Drevet, J.; Gharagozloo, P. Causes and consequences of oxidative stress in spermatozoa. Reprod. Fertil. Dev. 2016, 28, 1–10. [Google Scholar] [CrossRef]
- Amaral, A.; Lourenço, B.; Marques, M.; Ramalho-Santos, J. Mitochondria functionality and sperm quality. Reproduction 2013, 146, 163–174. [Google Scholar] [CrossRef]
- Aitken, R.J.; Drevet, J.R. The importance of oxidative stress in determining the functionality of mammalian spermatozoa: A two-edged sword. Antioxidants 2020, 9, 111. [Google Scholar] [CrossRef]
- Weir, C.P.; Robaire, B. Spermatozoa have decreased antioxidant enzymatic capacity and increased reactive oxygen species production during aging in the Brown Norway rat. J. Androl. 2007, 28, 229–240. [Google Scholar] [CrossRef] [PubMed]
- Bourgeron, T. Mitochondrial function and male infertility. Results Probl. Cell Differ. 2000, 28, 187–210. [Google Scholar] [PubMed]
- Twigg, J.; Fulton, N.; Gomez, E.; Irvine, D.S.; Aitken, R.J. Analysis of the impact of intracellular reactive oxygen species generation on the structural and functional integrity of human spermatozoa: Lipid peroxidation, DNA fragmentation and effectiveness of antioxidants. Hum. Reprod. 1998, 13, 1429–1436. [Google Scholar] [CrossRef] [PubMed]
- Sanocka, D.; Kurpisz, M. Reactive oxygen species and sperm cells. Reprod. Biol. Endocrinol. 2004, 2, 12. [Google Scholar] [CrossRef]
- Mennella, M.R.; Jones, R. Properties of spermatozoal superoxide dismutase and lack of involvement of superoxides in metal-ion-catalysed lipid-peroxidation and reactions in semen. Biochem. J. 1980, 191, 289–297. [Google Scholar] [CrossRef]
- Zini, A.; de Lamirande, E.; Gagnon, C. Reactive oxygen species in semen of infertile patients: Levels of superoxide dismutase- and catalase-like activities in seminal plasma and spermatozoa. Int. J. Androl. 1993, 16, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Vernet, P.; Aitken, R.J.; Drevet, J.R. Antioxidant strategies in the epididymis. Mol. Cell Endocrinol. 2004, 216, 31–39. [Google Scholar] [CrossRef] [PubMed]
This entry is adapted from the peer-reviewed paper 10.3390/antiox10050695
