Ischemia with non-obstructive coronary arteries (INOCA) is an increasingly recognized disease, with a prevalence of 3 to 4 million individuals, and is associated with a higher risk of morbidity, mortality, and a worse quality of life. Persistent angina in many patients with INOCA is due to coronary microvascular dysfunction (CMD), which can be difficult to diagnose and treat. A coronary flow reserve <2.5 is used to diagnose endothelial-independent CMD. Antianginal treatments are often ineffective in endothelial-independent CMD and thus novel treatment modalities are currently being studied for safety and efficacy. CD34+ cell therapy is a promising treatment option for these patients, as it has been shown to promote vascular repair and enhance angiogenesis in the microvasculature.
- CD34 stem cell therapy
- coronary microvascular dysfunction
- refractory angina
- ischemia with non-obstructive coronary arteries
1. Introduction
Ischemia with non-obstructive coronary arteries (INOCA) is an increasingly recognized disease with an estimated prevalence of 3 to 4 million individuals [1]. INOCA is characterized by signs and symptoms of ischemia in the absence of obstructive disease [2,3,4,5,6,7]. Although the pathophysiology is not completely understood, coronary microvascular dysfunction (CMD) has been shown to play a critical role and is reported in 47–64% of INOCA patients [1,5,8,9,10,11]. CMD encompasses endothelial-dependent and endothelial-independent microvascular dysfunction. Endothelium-dependent CMD stems from the inability of endothelial cells to produce vasodilatory substrates, thus blunting adequate myocardial perfusion during stress. Conversely, endothelial-independent CMD is detected as abnormal microvascular dilation resulting in a reduced coronary flow reserve (CFR) in response to adenosine. Endothelium-independent CMD results from the inability of smooth muscles to dilate despite the presence of vasodilatory substrates. CMD is associated with increased rates of major adverse cardiac events (MACE), including heart failure (HF), myocardial infarction (MI), and non-fatal stroke during long-term follow-up [2,3,4,11]. However, the current treatment is limited to risk factor management and antianginal medications, with no specific therapy for ischemia, which can significantly limit quality of life.
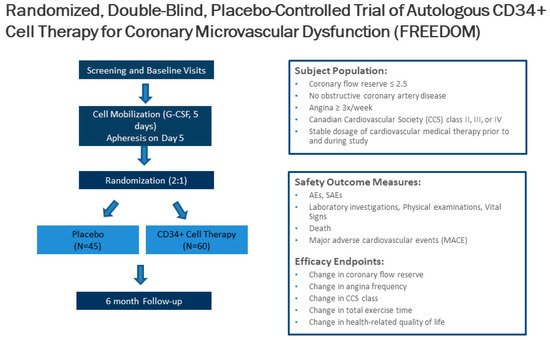
Cell therapy using autologous stem cells expressing CD34 (CD34+) is a novel therapeutic option for INOCA patients with CMD and refractory angina given the ability of CD34+ cells to repair the microcirculation [12,13,14]. Data from animal models indicate that CD34+ stem cells differentiate into endothelial cells, which incorporate into new vasculature and aid in the release of angiogenic cytokines, thus promoting vascular repair in the microcirculation, leading to improved myocardial perfusion in tissues damaged by acute and chronic ischemia [12,13,14,15,16,17,18]. Three consecutive, randomized, double-blinded, placebo-controlled trials in the United States, in patients with obstructive coronary artery disease (CAD) and Canadian Cardiovascular Society (CCS) class 3–4 refractory angina, established the feasibility of the intra-myocardial delivery of auto-CD34+ cells, showing significant improvements in short- and long-term anginal frequency (AF) and total exercise time (TET) [15,16,18]. Meta-analysis of these three trials demonstrated significant improvement in AF and TET, and a reduction in mortality [19]. PreSERVE-AMI, a large clinical trial using CD34+ cell therapy for left ventricular dysfunction post-ST-segment elevation MI (STEMI), demonstrated the safety and potential efficacy of the intracoronary infusion of CD34+ cell therapy in post-MI patients [20]. A recent two-center, phase 1 feasibility and safety trial (ESCaPE-CMD, NCT03508609) using autologous CD34+ stem cells in INOCA patients with endothelial-independent CMD demonstrated a significant improvement in CFR, AF, CCS class and quality of life [21]. These results led to an ongoing, double-blinded, placebo-controlled trial (FREEDOM, NCT04614467) to demonstrate the safety and efficacy of the intracoronary delivery of CD34+ stem cells. We will review the preclinical and clinical data that led to the development and application of cell-based therapy for ischemic repair, with a focus on microvascular repair in INOCA patients with CMD.
2. Clinical Studies on CD34 Therapy for Ischemic Disease
Although the majority of the clinical trials have been in patients with no-option refractory angina [15,16,17,18,19], CD34+ stem cells have also been investigated in clinical studies for peripheral ischemia [31,32], nonischemic cardiomyopathy [33], myocardial infarction [20], and ischemic stroke [34,35].
2.1. CD34 Therapy for Peripheral Ischemia
2.2. CD34 Therapy for Nonischemic Cardiomyopathy
2.3. CD34 Therapy for Myocardial Infarction
2.4. CD34 Therapy for Ischemic Stroke
2.5. CD34 Therapy for Refractory Angina
Successful phase 1 and phase 2 trials have shown the safety and efficacy of intramyocardial CD34+ stem cell therapy in treating no-option refractory angina. The phase 1, double-blinded, placebo-controlled trial enrolled 24 patients with CCS class 3 or 4 angina on optimal medical treatment, and demonstrated initial safety as well as clinical improvement in the CD34+ treated patients [15]. Under physiological conditions, the circulating CD34+ cell concentration is too low in the peripheral circulation. Thus, stimulation with granulocyte colony-stimulating factor (G-CSF) at 5 μg/kg per day for 4–5 days, followed by leukapheresis at day 5 with subsequent CD34+ enrichment, was used to harvest autologous mobilized CD34+ cells. The phase 2 ACT 34 trial enrolled 167 patients who were randomized to receive CD34+ stem cells in low doses (1 × 105 cells/kg, n = 55), high doses (5 × 105 cells/kg, n = 56), and an equal volume placebo diluent (n = 56) [16]. Treatment was distributed between 10 distinct ischemic sites with viable myocardium using a NOGA Myostar® mapping injection catheter. Both CD34+ cell therapy-treated patient groups showed significant improvements in the primary endpoint of AF at 6 months and 12 months. Compared to the placebo group, the low-dose treatment group showed a reduction in AF (6 months: 6.81 vs. 10.91 episodes per week, p = 0.02; 12 months: 6.3 vs. 11.0 episodes per week, p = 0.035) and improvement in TET at 6 and 12 months (6 months: 139 ± 151 vs. 69 ± 122 s, p = 0.014; 12 months: 140 ± 171 vs. 58 ± 146 s, p = 0.017) [16]. A two-year follow-up of 130 of those patients showed that autologous CD34+ cell therapy was associated with persistent improvements in AF in both the low-dose and high-dose groups (p = 0.03) [17]. Additionally, there was a decrease in mortality (p = 0.08) and MACE (p = 0.08).
The phase 3 RENEW, randomized, double-blinded, placebo-controlled trial was originally designed to obtain FDA approval using improvements in TET as the primary endpoint. Unfortunately, the trial was stopped early due to financial issues with the sponsor. Among the 112 enrolled patients, the improvement in TET was 61.0 s at 3 months (95% confidence interval (CI): −2.9 to 124.8, p = 0.06), 46.2 s at 6 months (95% CI: −28.0 to 120.4, p = 0.22), and 36.6 s at 12 months (95% CI: –56.1 to 129.2, p = 0.43) [18]. Furthermore, AF was significantly improved at 6 months (relative risk (RR): 0.58 by intention to treat, p = 0.02). As the study was incomplete, researchers were unable to conclusively determine the efficacy of CD34+ cell therapy for refractory angina patients. Further retrospective data analyses have shown that CD34+ treatment decreased long-term mortality (24% vs. 47%, p = 0.02), costs related to cardiac care (62% reduction translating to an average of USD 5500, p = 0.03), and interventional coronary procedures at 12 months (1.2 ± 0.91 vs. 0.32 ± 0.75 events, p < 0.0001) [36]. In all three trials, placebo patients also underwent treatment with G-CSF, leukapheresis, and intramyocardial injections. Therefore, the trials were completely blinded.
A 2018 meta-analysis of these three randomized, double-blinded trials (n = 304)—phase 1 and 2 ACT-34, ACT-34 extension and phase 3 RENEW—showed an improvement in TET, AF, and MACE in patients with obstructive coronary artery disease and refractory angina receiving intramyocardial autologous CD34+ cell therapy [19]. TET improved by 46.6 s at 3 months (p = 0.007), 49.5 s at 6 months (p = 0.016), and 44.7 s at 12 months (p = 0.065). Additionally, the relative AF decreased–0.78 at 3 months (p = 0.032), –0.66 at 6 months (p = 0.012), and –0.58 at 12 months (p = 0.011). Lastly, there was a significant decrease in mortality (12.1% vs. 2.5%, p = 0.0025) and MACE (38.9% vs. 30.0%, p = 0.14) in patients receiving intramyocardial autologous CD34+ cell therapy compared to placebo at 24 months (Figure 1).
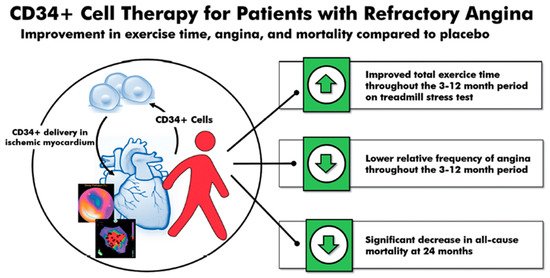
Although the three previous refractory angina trials used intramyocardial injection, the intracoronary delivery of CD34+ stem cells has also been shown to be safe and effective. This potentially allows for a safer and more established delivery route without losing the effectiveness of therapy. A 2010 study from China of 112 patients showed the safety and feasibility of intracoronary CD34+ stem cell therapy in refractory angina patients [37]. In this single-center study, there were no differences in the frequency of adverse events between the placebo and treatment groups, showing that the intracoronary delivery of bone-marrow-derived CD34+ cells is safely tolerated. Additionally, the study showed that patients receiving intracoronary CD34+ stem cell therapy had a greater reduction in AF at 3 months (–14.6 ± 4.8 vs. –4.5 ± 0.3 episodes, p < 0.01) and 6 months (–15.6 ± 4.0 vs. –3.0 ± 1.2 events, p < 0.01) compared to placebo. Once again, the placebo group also showed a significant decrease in AF from baseline, indicating a strong placebo effect. This study also found a statistically significant decrease in use of nitroglycerin and CCS class, as well as an increase in TET in patients receiving intracoronary CD34+ stem cells [37]. In summary, extensive clinical trial data have demonstrated the outstanding safety of CD34+ stem cell therapy, and consistently showed clinical improvements in conditions characterized by perfusion abnormalities. No studies to date have reported an increased risk of uncontrolled cell growth of CD34+ inoculated tissues, angiomas or cancer.
3. CD34 Therapy as a Novel Treatment for Coronary Microvascular Dysfunction
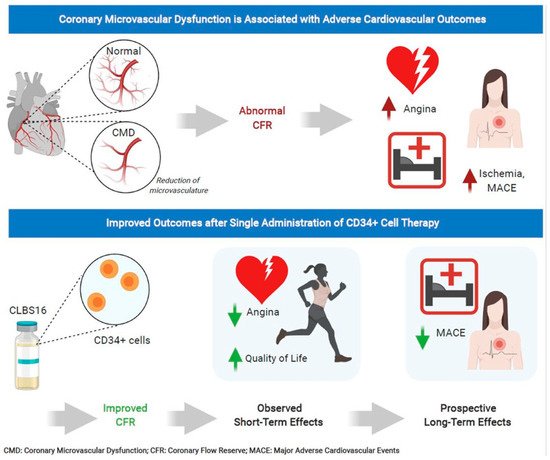
4. Conclusions
Endothelial-independent CMD, associated with a reduced CFR in response to intracoronary adenosine, increases the risk of MACE, MI, heart failure hospitalizations, and mortality in INOCA patients. However, no effective specific therapy to date exists for CMD. Cell therapy using autologous CD34+ stem cells is a promising new therapeutic option for INOCA patients with CMD. Through the induction of capillary growth, direct incorporation into damaged vasculature, and the upregulation of proliferative cytokines, CD34+ stem cell therapy can enhance angiogenesis and restore the microcirculation in acute and chronic ischemia. Three consecutive randomized and double-blinded trials have demonstrated the safety and efficacy of the reduction in CCS class 3–4 refractory angina from obstructive CAD via the delivery of intramyocardial CD34+ stem cells, and a pooled analysis of these studies showed significant improvements in AF and TET and a reduction of mortality. Feasibility and safety studies on the use of autologous CD34+ cell therapy in INOCA patients with endothelial-independent CMD and persistent angina showed significant improvement in CFR in 6 months, with no cell-related adverse events. The use of CD34+ cell therapy in CMD and persistent angina will be determined by the results of the ongoing phase 2 FREEDOM trial (NCT04614467). Given the high prevalence and the lack of specific treatment options in patient with INOCA and underlying CMD, additional research is needed to identify novel therapies.
This entry is adapted from the peer-reviewed paper 10.3390/cells10051137
