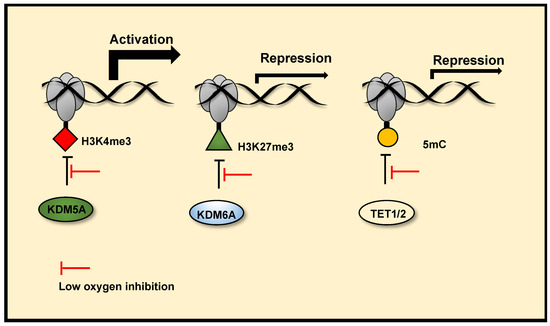
Histone methylation is a recognised mechanism controlling chromatin structure and is associated with regulation of gene transcription, with some marks clearly leading to open chromatin, while others are firmly associated with closed conformation [
228]. The family of enzymes predominantly responsible for histone demethylation are JmjC demethylases, which are part of the JmjC-domain containing group of 2-OGDs that includes demethylases and hydroxylases (JmjC 2-ODGs)). In vitro studies demonstrate the varied oxygen sensitivities of these enzymes, some are potentially direct molecular oxygen sensors ( and
Supplementary Table S1). Oxygen affinities, oxygen availability and protein expression levels will likely dictate JmjC demethylase activities in hypoxia. Several groups have reported increases in total levels of histone methylation modifications in response to hypoxia across a range of human and mouse cell types and human tumours using immunoblotting, immunohistochemistry, immunofluorescence and quantitative proteomics ([
229], reviewed in [
16]). ChIP-sequencing approaches have revealed site-specific, hypoxia-induced changes in Histone (H)3 Lysine (K)4 trimethylation (me3) [
230,
231], H3K36me3 [
230] and H3K27me3 [
25,
231], which correlate with changes in gene expression. There is now evidence, through the use of in vitro and in cell histone demethylation assays, in coordination with mutagenesis analysis, gene expression analysis and histone methylation analysis, that Lysine Demethylase (KDM) 6A (KDM6A) [
25] and potentially KDM5A [
230], which demethylate H3K27me3 and H3K4me3 respectively, are inhibited by reduced oxygen levels in hypoxia. These result in increased histone methylation modifications, which coordinate hypoxia inducible gene transcriptional changes () and hypoxia induced cellular responses. Specifically, KDM6A inhibition in hypoxia triggers hypermethylation of H3K27me3 at a subset of hypoxia repressed gene promoters, reducing their expression. Conversely, potential KDM5A inhibition in hypoxia, triggers H3K4me3 hypermethylation at a subset of hypoxia inducible gene promoters, this precedes increases in their expression in hypoxia and is required for their full transcriptional activation in hypoxia. In cell and in vitro H3K9me3 demethylation assays have also revealed that the demethylase activity of KDM4A is highly sensitive to oxygen concentrations over physiologically relevant ranges [
24]. Although there are a range of reported in vitro oxygen affinities, (
Supplementary Table S1) the data from cells show that KDM4A activity is sensitive to oxygen levels. There is evidence that reduced histone demethylase activity of KDM4A in severe hypoxia (<0.5% oxygen), along with KDM4C, triggers increased expression of a subset of androgen receptor target genes [
232]. Thus, KDM4A can also be classed as an oxygen sensor. Interestingly, KDM4A has been shown to positively regulate HIF-1α levels via H3K9me3 demethylation at the
HIF1A gene locus, this effect is observed in mild hypoxia (2% oxygen), but impaired at severe hypoxia (<0.1% oxygen) [
233]. This may provide a mechanism of maintaining HIF-1α levels in conditions of mild hypoxia specifically, where there is still residual PHD activity. Future work should investigate if the oxygen sensitive H3K9me3 demethylase activity of KDM4A is linked to control of gene expression and chromatin regulation in hypoxia. Importantly, some JmjC demethylases remain active at low oxygen concentrations and function in hypoxia through their histone demethylase activity. KDM4C [
234] and KDM3A [
235,
236] display HIF coactivator activity in hypoxia via demethylation of H3K9 at HIF target gene promoters, facilitating transcriptional activation at these genes. The co-activator role of KDM3A in hypoxia, via retained histone demethylation activity appears to be context dependent, as other groups have reported reduced activity KDM3A in hypoxia [
237]. Furthermore, many JmjC histone demethylases are HIF target genes that are upregulated in hypoxia (reviewed in [
215]), this is thought in part to be a compensatory mechanism to counteract reduced demethylase activity, acting as a hypoxia feedback loop similar to what is seen with transcriptional upregulation of PHD2/3 by HIF. Thus, there is complex crosstalk between histone methylation, gene expression and hypoxia, mediated in part by JmjC demethylases. However, further characterisation of the oxygen sensitives of JmjC demethylases is needed.