Chemotherapy-induced peripheral neuropathy (CIPN), one of major dose-limiting side effects of first-line chemotherapeutic agents such as paclitaxel, oxaliplatin, vincristine, and bortezomib is resistant to most of existing medicines.
- high mobility group box 1 (HMGB1)
- chemotherapy-induced peripheral neuropathy (CIPN)
1. Introduction
Chemotherapy-induced peripheral neuropathy (CIPN) is one of the major dose-limiting side effects of first-line chemotherapeutic agents such as paclitaxel, oxaliplatin, vincristine, and bortezomib. In terms of evidence-based medicine, no drugs are recommended for the prevention of CIPN, and duloxetine is the only agent that has limited benefit in treating established CIPN [1]. The mechanisms for development and maintenance of CIPN have not been fully understood, although preclinical studies have provided evidence for some possible mechanisms for CIPN [2], such as neuroimmune interactions [3], mitochondrial dysfunction [4], reactive oxygen species (ROS) accumulation [5], and transcriptional or functional upregulation of cation channels [6][7] in the spinal cord, dorsal root ganglion (DRG), and peripheral sensory neurons. Our preclinical studies have demonstrated the involvement of high mobility group box 1 (HMGB1) in the development and maintenance of CIPN [8][9][10][11]. HMGB1 (also known as amphoterin) is a non-histone nuclear protein that has highly conserved amino acid sequences during evolution and is ubiquitously expressed in most mammalian cell types [12]. HMGB1 is essential for life, because HMGB1-deficient mice die within 24 h of birth due to hypoglycemia [13]. In the nucleus, HMGB1 contributes to nucleosome stability and sliding, DNA replication and repair, gene transcription, etc. On the other hand, HMGB1 is actively secreted by activated immune cells and passively released from necrotic cells in pathological conditions including inflammation. Extracellular HMGB1 acts as a damage-associated molecular pattern (DAMP) protein via activation of several pattern recognition receptors (PRRs) including Toll-like receptors (TLRs), receptors for advance glycosylation end products (RAGE), C-X-C motif chemokine receptor 4 (CXCR4), etc., leading to acceleration of inflammation and pain [11][13][14][15][16][17][18][19][20]. There are plenty of papers showing that an anti-HMGB1-neutralizing antibody (HMGB1-nAb) [21][22] strongly suppresses somatic or visceral pathological pain with inflammatory and/or neuropathic components [23][24][25][26][27][28][29][30][31][32][33][34]. We have shown that the HMGB1-nAb strongly prevents the development of CIPN in rodents [8][9][10]. Similarly, recombinant human soluble thrombomodulin (TM) [thrombomodulin alfa (TMα), ART-123, recomodulin®], capable of promoting thrombin-dependent HMGB1 degradation [11][35][36], also prevents the development of CIPN in rodent models [8][9][10]. Intriguingly, the efficacy of TMα in preventing CIPN in humans has been confirmed by a placebo-controlled, randomized, double-blind phase IIa study [37]. Thus, targeting extracellular HMGB1 is considered a promising strategy to prevent CIPN.
2. Role of HMGB1 in CIPN
2.1. Involvement of Endogenous HMGB1 in the Development of Pathological Pain Including CIPN
In 2001, it was reported for the first time that injection of HMGB1 around the sciatic nerve induced mechanical allodynia in rats [38]. Recently, increasing evidence has unveiled the pro-nociceptive role of HMGB1 in the peripheral tissue and spinal cord [16][18][29][39][40], and demonstrated that endogenous HMGB1 is involved in the pathogenesis of various types of intractable pain [11][15], including inflammatory pain [28][29][39], visceral pain [19][20][30][32], neuropathic pain [23][31][41][42][43], cancer pain [33], and post-stroke pain [34]. Endogenous HMGB1 also appears to play a key role in the development of CIPN in rats or mice treated with cancer chemotherapeutics, such as paclitaxel, oxaliplatin, and vincristine, considering the complete prevention of CIPN by inactivation of HMGB1 with HMGB1-nAb or TMα (Figure 1) [8][9][10][11].
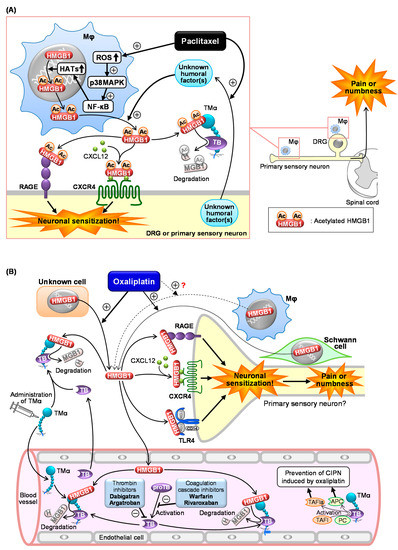
Figure 1. Scheme of the mechanisms underlying chemotherapy-induced peripheral neuropathy (CIPN) in mice treated with paclitaxel (A) or oxaliplatin (B). (A) Paclitaxel causes macrophage (Mφ) accumulation in the dorsal root ganglion (DRG) and sciatic nerves (primary sensory neurons) and upregulates histone acetyltransferases (HATs) via the ROS/p38MAPK/NF-κB pathway, resulting in acetylation and cytoplasmic translocation of nuclear HMGB1 followed by its secretion. Unknown humoral factors released from neurons in response to paclitaxel promote the HMGB1 release from Mφ. The secreted HMGB1 causes neuronal excitation through activation of RAGE, TLR4, and the CXCL12/CXCR4 axis, leading to CIPN characterized by pain or numbness. TMα prevents CIPN following paclitaxel treatment by degrading extracellular HMGB1 in a thrombin (TB)-dependent manner. (B) Oxaliplatin induces HMGB1 release possibly from multiple cells including Schwann cells, leading to CIPN through neuronal sensitization via activation of RAGE, TLR4, and CXCL12/CXCR4 pathways. Exogenously applied TMα and endothelial TM accelerate thrombin-induced degradation of HMGB1, thereby reducing the CIPN induced by oxaliplatin. Anticoagulants such as dabigatran, argatroban, warfarin, and rivaroxaban reduce the thrombin-dependent degradation of HMGB1 by TM and TMα, thereby aggravating CIPN and cancelling the anti-CIPN effect of TMα.
2.2. Roles of Macrophage-Derived HMGB1 and Its Upstream/Downstream Molecules in CIPN Caused by Paclitaxel
Actually, macrophages accumulate in the sciatic nerve and dorsal root ganglion (DRG) from the CIPN mice treated with paclitaxel (Figure 1A) [9][44]. In macrophage-like RAW264.7 cells, paclitaxel causes cytoplasmic translocation and extracellular secretion of nuclear HMGB1, which involves the activation of the ROS/p38MAPK/NF-κB pathway and subsequent upregulation of histone acetyltransferases (HATs), possibly essential for acetylation of HMGB1 (Figure 1A) [9]. Deletion of ROS by N-acetylcysteine (NAC), an antioxidant, prevents the development of CIPN in mice treated with paclitaxel in vivo (Table 1), in agreement with the in vitro experiments using macrophages [9]. Co-culture experiments using macrophage-like RAW264.7 cells and neuron-like NG108-15 cells suggest that “unknown humoral factors” derived from neurons accelerate HMGB1 secretion by macrophages in response to paclitaxel (Figure 1A) [9]. Thus, a crosstalk between macrophages and neurons mediated by HMGB1 and unknown humoral factors may play a key role in CIPN development after paclitaxel treatment.
Antagonists of RAGE or CXCR4, membrane receptors targeted by at-HMGB1, prevent the development of CIPN caused by paclitaxel (Figure 1A) [9]. On the other hand, the role of TLR4 in the paclitaxel-induced CIPN appears to be different among species or strains. TAK-242, a TLR4 antagonist capable of penetrating into the CNS, prevents CIPN after paclitaxel treatment in C57BL/6 mice and Sprague–Dawley rats, but not in ddY mice [9][45]. It is to note that intraperitoneal (i.p.) administration of LPS-RS, a peripherally preferential TLR4 antagonist, does not prevent the CIPN in C57BL/6 mice (Table 1) [9], while intrathecal administration of LPS-RS blocks rat CIPN caused by paclitaxel [46]. Therefore, the CIPN caused by paclitaxel appears to involve central, but not peripheral, TLR4 in rats and C57BL/6 mice. Interestingly, there is a report showing that macrophage TLR9, an intracellular compartment receptor for HMGB1, participates in the CIPN caused by paclitaxel in male, but not female, mice, suggesting the existence of sex dimorphism in the role of TLR9 in the development of CIPN [47]. The same study has shown the involvement of TLR9 in the paclitaxel-induced release of TNF and CXCL1 from male, but not female, macrophages [47]. Similar sex dimorphisms have also been described in the role of TLR4 in pain processing [39][48]. Collectively, activation of RAGE and CXCR4 possibly by macrophage-derived at-HMGB1 may play a major part in CIPN due to paclitaxel.
2.3. Role of HMGB1 and Its Upstream/Downstream Molecules in CIPN Caused by Oxaliplatin
We have shown that the inactivation of HMGB1 with HMGB1-nAb or TMα and pharmacological blockade of RAGE, CXCR4, or TLR4 prevent CIPN in rodents treated with oxaliplatin (Figure 2B) [10]. Peripheral TLR4 appears to be involved in the CIPN caused by oxaliplatin, because systemic administration of LPS-RS as well as TAK-242 exhibited a preventive effect in this CIPN model (Figure 1B). Most surprisingly, macrophages do not play a major role in the CIPN caused by oxaliplatin, since depletion of macrophages by liposomal clodronate as well as inhibition of macrophage-derived HMGB1 by minocycline or ethyl pyruvate had no effect on the CIPN in mice treated with oxaliplatin [10]. An independent group [49] has suggested the involvement of macrophages in the same CIPN model, being apparently inconsistent with our results [10]. However, their study showed that liposomal clodronate only slightly (by 30%) reduced the oxaliplatin-induced allodynia, suggesting that the participation of macrophage-derived HMGB1 in the CIPN caused by oxaliplatin should be very minor, if any [50]. This notion is further supported by our findings that the number of macrophages in the sciatic nerve did not increase in mice treated with oxaliplatin. Thus, the origin of HMGB1 involved in CIPN caused by oxaliplatin is still open to question (Figure 1B). Alternatively, it is likely that HMGB1 derived from multiple cells including macrophages contributes to the development of CIPN following oxaliplatin treatment (Figure 1B), because oxaliplatin at relatively high concentrations (3–10 µM) causes HMGB1 release from Schwann cells and macrophage-like RAW264.7 cells in vitro [10]. Our study to explore other origins that secrete HMGB1 in response to oxaliplatin is now in progress.
This entry is adapted from the peer-reviewed paper 10.3390/ijms22010367
References
- Loprinzi, C.L.; Lacchetti, C.; Bleeker, J.; Cavaletti, G.; Chauhan, C.; Hertz, D.L.; Kelley, M.R.; Lavino, A.; Lustberg, M.B.; Paice, J.A.; et al. Prevention and Management of Chemotherapy-Induced Peripheral Neuropathy in Survivors of Adult Cancers: ASCO Guideline Update. J. Clin. Oncol. 2020, 38, 3325–3348.
- Flatters, S.J.L.; Dougherty, P.M.; Colvin, L.A. Clinical and preclinical perspectives on Chemotherapy-Induced Peripheral Neuropathy (CIPN): A narrative review. Br. J. Anaesth. 2017, 119, 737–749.
- Lees, J.G.; Makker, P.G.; Tonkin, R.S.; Abdulla, M.; Park, S.B.; Goldstein, D.; Moalem-Taylor, G. Immune-mediated processes implicated in chemotherapy-induced peripheral neuropathy. Eur. J. Cancer 2017, 73, 22–29.
- Trecarichi, A.; Flatters, S.J.L. Mitochondrial dysfunction in the pathogenesis of chemotherapy-induced peripheral neuropathy. Int. Rev. Neurobiol. 2019, 145, 83–126.
- Shim, H.S.; Bae, C.; Wang, J.; Lee, K.H.; Hankerd, K.M.; Kim, H.K.; Chung, J.M.; La, J.H. Peripheral and central oxidative stress in chemotherapy-induced neuropathic pain. Mol. Pain 2019, 15, 1744806919840098.
- Nakagawa, T.; Kaneko, S. Roles of Transient Receptor Potential Ankyrin 1 in Oxaliplatin-Induced Peripheral Neuropathy. Biol. Pharm. Bull. 2017, 40, 947–953.
- Tomita, S.; Sekiguchi, F.; Deguchi, T.; Miyazaki, T.; Ikeda, Y.; Tsubota, M.; Yoshida, S.; Nguyen, H.D.; Okada, T.; Toyooka, N.; et al. Critical role of Cav3.2 T-type calcium channels in the peripheral neuropathy induced by bortezomib, a proteasome-inhibiting chemotherapeutic agent, in mice. Toxicology 2019, 413, 33–39.
- Nishida, T.; Tsubota, M.; Kawaishi, Y.; Yamanishi, H.; Kamitani, N.; Sekiguchi, F.; Ishikura, H.; Liu, K.; Nishibori, M.; Kawabata, A. Involvement of high mobility group box 1 in the development and maintenance of chemotherapy-induced peripheral neuropathy in rats. Toxicology 2016, 365, 48–58.
- Sekiguchi, F.; Domoto, R.; Nakashima, K.; Yamasoba, D.; Yamanishi, H.; Tsubota, M.; Wake, H.; Nishibori, M.; Kawabata, A. Paclitaxel-induced HMGB1 release from macrophages and its implication for peripheral neuropathy in mice: Evidence for a neuroimmune crosstalk. Neuropharmacology 2018, 141, 201–213.
- Tsubota, M.; Fukuda, R.; Hayashi, Y.; Miyazaki, T.; Ueda, S.; Yamashita, R.; Koike, N.; Sekiguchi, F.; Wake, H.; Wakatsuki, S.; et al. Role of non-macrophage cell-derived HMGB1 in oxaliplatin-induced peripheral neuropathy and its prevention by the thrombin/thrombomodulin system in rodents: Negative impact of anticoagulants. J. Neuroinflamm. 2019, 16, 199.
- Tsujita, R.; Tsubota, M.; Sekiguchi, F.; Kawabata, A. Role of high-mobility group box 1 and its modulation by thrombomodulin/thrombin axis in neuropathic and inflammatory pain. Br. J. Pharmacol. 2020.
- Kang, R.; Chen, R.; Zhang, Q.; Hou, W.; Wu, S.; Cao, L.; Huang, J.; Yu, Y.; Fan, X.G.; Yan, Z.; et al. HMGB1 in health and disease. Mol. Aspects Med. 2014, 40, 1–116.
- Calogero, S.; Grassi, F.; Aguzzi, A.; Voigtlander, T.; Ferrier, P.; Ferrari, S.; Bianchi, M.E. The lack of chromosomal protein Hmg1 does not disrupt cell growth but causes lethal hypoglycaemia in newborn mice. Nat. Genet. 1999, 22, 276–280.
- Yanai, H.; Ban, T.; Taniguchi, T. High-mobility group box family of proteins: Ligand and sensor for innate immunity. Trends Immunol. 2012, 33, 633–640.
- Wan, W.; Cao, L.; Khanabdali, R.; Kalionis, B.; Tai, X.; Xia, S. The Emerging Role of HMGB1 in Neuropathic Pain: A Potential Therapeutic Target for Neuroinflammation. J. Immunol. Res. 2016, 2016, 6430423.
- Yamasoba, D.; Tsubota, M.; Domoto, R.; Sekiguchi, F.; Nishikawa, H.; Liu, K.; Nishibori, M.; Ishikura, H.; Yamamoto, T.; Taga, A.; et al. Peripheral HMGB1-induced hyperalgesia in mice: Redox state-dependent distinct roles of RAGE and TLR4. J. Pharmacol. Sci. 2016, 130, 139–142.
- VanPatten, S.; Al-Abed, Y. High Mobility Group Box-1 (HMGb1): Current Wisdom and Advancement as a Potential Drug Target. J. Med. Chem. 2018, 61, 5093–5107.
- Tsujita, R.; Tsubota, M.; Hayashi, Y.; Saeki, H.; Sekiguchi, F.; Kawabata, A. Role of Thrombin in Soluble Thrombomodulin-Induced Suppression of Peripheral HMGB1-Mediated Allodynia in Mice. J. Neuroimmune. Pharmacol. 2018, 13, 179–188.
- Hiramoto, S.; Tsubota, M.; Yamaguchi, K.; Okazaki, K.; Sakaegi, A.; Toriyama, Y.; Tanaka, J.; Sekiguchi, F.; Ishikura, H.; Wake, H.; et al. Cystitis-Related Bladder Pain Involves ATP-Dependent HMGB1 Release from Macrophages and Its Downstream H2S/Cav3.2 Signaling in Mice. Cells 2020, 9, 1748.
- Irie, Y.; Tsubota, M.; Maeda, M.; Hiramoto, S.; Sekiguchi, F.; Ishikura, H.; Wake, H.; Nishibori, M.; Kawabata, A. HMGB1 and its membrane receptors as therapeutic targets in an intravesical substance P-induced bladder pain syndrome mouse model. J. Pharmacol. Sci. 2020, 143, 112–116.
- Liu, K.; Mori, S.; Takahashi, H.K.; Tomono, Y.; Wake, H.; Kanke, T.; Sato, Y.; Hiraga, N.; Adachi, N.; Yoshino, T.; et al. Anti-high mobility group box 1 monoclonal antibody ameliorates brain infarction induced by transient ischemia in rats. FASEB J. 2007, 21, 3904–3916.
- Nishibori, M.; Mori, S.; Takahashi, H.K. Anti-HMGB1 monoclonal antibody therapy for a wide range of CNS and PNS diseases. J. Pharmacol. Sci. 2019, 140, 94–101.
- Shibasaki, M.; Sasaki, M.; Miura, M.; Mizukoshi, K.; Ueno, H.; Hashimoto, S.; Tanaka, Y.; Amaya, F. Induction of high mobility group box-1 in dorsal root ganglion contributes to pain hypersensitivity after peripheral nerve injury. Pain 2010, 149, 514–521.
- Tong, W.; Wang, W.; Huang, J.; Ren, N.; Wu, S.X.; Li, Y.Q. Spinal high-mobility group box 1 contributes to mechanical allodynia in a rat model of bone cancer pain. Biochem. Biophys. Res. Commun. 2010, 395, 572–576.
- Otoshi, K.; Kikuchi, S.; Kato, K.; Sekiguchi, M.; Konno, S. Anti-HMGB1 neutralization antibody improves pain-related behavior induced by application of autologous nucleus pulposus onto nerve roots in rats. Spine 2011, 36, E692–E698.
- Ren, P.C.; Zhang, Y.; Zhang, X.D.; An, L.J.; Lv, H.G.; He, J.; Gao, C.J.; Sun, X.D. High-mobility group box 1 contributes to mechanical allodynia and spinal astrocytic activation in a mouse model of type 2 diabetes. Brain Res. Bull. 2012, 88, 332–337.
- Nakamura, Y.; Morioka, N.; Abe, H.; Zhang, F.F.; Hisaoka-Nakashima, K.; Liu, K.; Nishibori, M.; Nakata, Y. Neuropathic pain in rats with a partial sciatic nerve ligation is alleviated by intravenous injection of monoclonal antibody to high mobility group box-1. PLoS ONE 2013, 8, e73640.
- Tanaka, J.; Seki, Y.; Ishikura, H.; Tsubota, M.; Sekiguchi, F.; Yamaguchi, K.; Murai, A.; Umemura, T.; Kawabata, A. Recombinant human soluble thrombomodulin prevents peripheral HMGB1-dependent hyperalgesia in rats. Br. J. Pharmacol. 2013, 170, 1233–1241.
- Agalave, N.M.; Larsson, M.; Abdelmoaty, S.; Su, J.; Baharpoor, A.; Lundback, P.; Palmblad, K.; Andersson, U.; Harris, H.; Svensson, C.I. Spinal HMGB1 induces TLR4-mediated long-lasting hypersensitivity and glial activation and regulates pain-like behavior in experimental arthritis. Pain 2014, 155, 1802–1813.
- Tanaka, J.; Yamaguchi, K.; Ishikura, H.; Tsubota, M.; Sekiguchi, F.; Seki, Y.; Tsujiuchi, T.; Murai, A.; Umemura, T.; Kawabata, A. Bladder pain relief by HMGB1 neutralization and soluble thrombomodulin in mice with cyclophosphamide-induced cystitis. Neuropharmacology 2014, 79, 112–118.
- Zhang, F.F.; Morioka, N.; Harano, S.; Nakamura, Y.; Liu, K.; Nishibori, M.; Hisaoka-Nakashima, K.; Nakata, Y. Perineural expression of high-mobility group box-1 contributes to long-lasting mechanical hypersensitivity via matrix metalloprotease-9 up-regulation in mice with painful peripheral neuropathy. J. Neurochem. 2016, 136, 837–850.
- Irie, Y.; Tsubota, M.; Ishikura, H.; Sekiguchi, F.; Terada, Y.; Tsujiuchi, T.; Liu, K.; Nishibori, M.; Kawabata, A. Macrophage-derived HMGB1 as a Pain Mediator in the Early Stage of Acute Pancreatitis in Mice: Targeting RAGE and CXCL12/CXCR4 Axis. J. Neuroimmune. Pharmacol. 2017, 12, 693–707.
- An, K.; Rong, H.; Ni, H.; Zhu, C.; Xu, L.; Liu, Q.; Chen, Y.; Zheng, Y.; Huang, B.; Yao, M. Spinal PKC activation-Induced neuronal HMGB1 translocation contributes to hyperalgesia in a bone cancer pain model in rats. Exp. Neurol. 2018, 303, 80–94.
- Matsuura, W.; Harada, S.; Liu, K.; Nishibori, M.; Tokuyama, S. Evidence of a role for spinal HMGB1 in ischemic stress-induced mechanical allodynia in mice. Brain Res. 2018, 1687, 1–10.
- Abeyama, K.; Stern, D.M.; Ito, Y.; Kawahara, K.; Yoshimoto, Y.; Tanaka, M.; Uchimura, T.; Ida, N.; Yamazaki, Y.; Yamada, S.; et al. The N-terminal domain of thrombomodulin sequesters high-mobility group-B1 protein, a novel antiinflammatory mechanism. J. Clin. Investig. 2005, 115, 1267–1274.
- Ito, T.; Kawahara, K.; Okamoto, K.; Yamada, S.; Yasuda, M.; Imaizumi, H.; Nawa, Y.; Meng, X.; Shrestha, B.; Hashiguchi, T.; et al. Proteolytic cleavage of high mobility group box 1 protein by thrombin-thrombomodulin complexes. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1825–1830.
- Kotaka, M.; Saito, Y.; Kato, T.; Satake, H.; Makiyama, A.; Tsuji, Y.; Shinozaki, K.; Fujiwara, T.; Mizushima, T.; Harihara, Y.; et al. A placebo-controlled, double-blind, randomized study of recombinant thrombomodulin (ART-123) to prevent oxaliplatin-induced peripheral neuropathy. Cancer Chemother. Pharmacol. 2020, 86, 607–618.
- Chacur, M.; Milligan, E.D.; Gazda, L.S.; Armstrong, C.; Wang, H.; Tracey, K.J.; Maier, S.F.; Watkins, L.R. A new model of sciatic inflammatory neuritis (SIN): Induction of unilateral and bilateral mechanical allodynia following acute unilateral peri-sciatic immune activation in rats. Pain 2001, 94, 231–244.
- Rudjito, R.; Agalave, N.M.; Farinotti, A.B.; Lundback, P.; Szabo-Pardi, T.; Price, T.J.; Harris, H.E.; Burton, M.D.; Svensson, C.I. Sex- and cell-dependent contribution of peripheral HMGB1 and TLR4 in arthritis-induced pain. Pain 2020.
- Hayashi, Y.; Tsujita, R.; Tsubota, M.; Saeki, H.; Sekiguchi, F.; Honda, G.; Kawabata, A. Human soluble thrombomodulin-induced blockade of peripheral HMGB1-dependent allodynia in mice requires both the lectin-like and EGF-like domains. Biochem. Biophys. Res. Commun. 2018, 495, 634–638.
- Tomita, S.; Sekiguchi, F.; Kasanami, Y.; Naoe, K.; Tsubota, M.; Wake, H.; Nishibori, M.; Kawabata, A. Cav3.2 overexpression in L4 dorsal root ganglion neurons after L5 spinal nerve cutting involves Egr-1, USP5 and HMGB1 in rats: An emerging signaling pathway for neuropathic pain. Eur. J. Pharmacol. 2020, 888, 173587.
- Morioka, N.; Miyauchi, K.; Miyashita, K.; Kochi, T.; Zhang, F.F.; Nakamura, Y.; Liu, K.; Wake, H.; Hisaoka-Nakashima, K.; Nishibori, M.; et al. Spinal high-mobility group box-1 induces long-lasting mechanical hypersensitivity through the toll-like receptor 4 and upregulation of interleukin-1beta in activated astrocytes. J. Neurochem. 2019, 150, 738–758.
- Thakur, V.; Sadanandan, J.; Chattopadhyay, M. High-Mobility Group Box 1 Protein Signaling in Painful Diabetic Neuropathy. Int. J. Mol. Sci. 2020, 21, 881.
- Zhang, H.; Li, Y.; de Carvalho-Barbosa, M.; Kavelaars, A.; Heijnen, C.J.; Albrecht, P.J.; Dougherty, P.M. Dorsal Root Ganglion Infiltration by Macrophages Contributes to Paclitaxel Chemotherapy-Induced Peripheral Neuropathy. J. Pain 2016, 17, 775–786.
- Li, Y.; Tatsui, C.E.; Rhines, L.D.; North, R.Y.; Harrison, D.S.; Cassidy, R.M.; Johansson, C.A.; Kosturakis, A.K.; Edwards, D.D.; Zhang, H.; et al. Dorsal root ganglion neurons become hyperexcitable and increase expression of voltage-gated T-type calcium channels (Cav3.2) in paclitaxel-induced peripheral neuropathy. Pain 2017, 158, 417–429.
- Li, Y.; Zhang, H.; Zhang, H.; Kosturakis, A.K.; Jawad, A.B.; Dougherty, P.M. Toll-like receptor 4 signaling contributes to Paclitaxel-induced peripheral neuropathy. J. Pain 2014, 15, 712–725.
- Luo, X.; Huh, Y.; Bang, S.; He, Q.; Zhang, L.; Matsuda, M.; Ji, R.R. Macrophage Toll-like Receptor 9 Contributes to Chemotherapy-Induced Neuropathic Pain in Male Mice. J. Neurosci. 2019, 39, 6848–6864.
- Agalave, N.M.; Rudjito, R.; Farinotti, A.B.; Khoonsari, P.E.; Sandor, K.; Nomura, Y.; Szabo-Pardi, T.A.; Urbina, C.M.; Palada, V.; Price, T.J.; et al. Sex-dependent role of microglia in disulfide HMGB1-mediated mechanical hypersensitivity. Pain 2020.
- Gu, H.; Wang, C.; Li, J.; Yang, Y.; Sun, W.; Jiang, C.; Li, Y.; Ni, M.; Liu, W.T.; Cheng, Z.; et al. High mobility group box-1-toll-like receptor 4-phosphatidylinositol 3-kinase/protein kinase B-mediated generation of matrix metalloproteinase-9 in the dorsal root ganglion promotes chemotherapy-induced peripheral neuropathy. Int. J. Cancer 2020, 146, 2810–2821.
- Gao, Y.; Lv, X.; Yang, H.; Peng, L.; Ci, X. Isoliquiritigenin exerts antioxidative and anti-inflammatory effects via activating the KEAP-1/Nrf2 pathway and inhibiting the NF-kappaB and NLRP3 pathways in carrageenan-induced pleurisy. Food Funct. 2020, 11, 2522–2534.
