Cistus albidus is one of the approximately 20 species of the Cistus genus. The genus’ name is derived from the ancient Greek term kistos. It is supposed that the name alludes to the woody capsule fruits. Evergreen in its Mediterranean homeland and between 50 and 250 centimeters tall, this shrub is called albidus, not because of the colour of its flowers, but because its leaves are finely covered with white hair (trichomes).
- phytochemistry
- pharmacology
- polyphenols
- terpenes
- traditional uses
1. Botanical Characteristics

Vegetative Development
2. Phytochemical Constituents
2.1. Terpenes
2.1.1. Mono- and Sesquiterpenes from the Essential Oils
| No | Compound | Structure/Class | Presence in | Analytical Reference | Pharmacology |
|---|---|---|---|---|---|
| 1 | cis-α-Bergamotene | polycyclic monoterpene hydrocarbon | 🌱✿ | [30][31][35] | n/a |
| 2 | trans-α-Bergamotene | 🌱 | [29][30] | ||
| 3 | Borneol | polycyclic monoterpene alcohol |
🌱 | [34] | blood brain barrier (BBB) permeability improvement, intercellular tight junction (TJ) loosening [36] |
| 4 | Camphor | oxygenated polycyclic monoterpene | 🌱 | [32] | analgesic, antinociceptive [37]; antimicrobial, antiviral [38]; anticancer [39]; antitussive [40]; skin penetration enhancer [41]; |
| 5 | Carene | polycyclic monoterpene hydrocarbon |
🌱✿ | [34] | antiviral [42]; enhances bone mineralization [43]; anti-inflammatory [44]; |
| 6 | Carvacrol | monocyclic monoterpene alcohol | 🌱✿ | [31] | antibacterial (64); antifungal [45]; antioxidant [46]; anticancer [47]; anti-inflammatory, analgesic [48]; antiobesity [49]; hepatoprotective [50]; spasmolytic [51]; vasorelaxant [52]; |
| 7 | β-Cyclocitral | oxygenated monocyclic monoterpene | 🌱 | [35] | n/a |
| 8 | p-Cymene | monocyclic monoterpene hydrocarbon |
🌱 | [31][32] | anti-inflamatory, antinociceptive, antioxidant [53]; antidiabetic [54]; |
| 9 | p-Cymenene | monocyclic monoterpene hydrocarbon |
🌱✿ | [32] | n/a |
| 10 | Isobornyl formate | oxygenated polycyclic monoterpene | 🌱 | [32] | n/a |
| 11 | (D-)Limonene | monocyclic monoterpene hydrocarbon | 🌱✿ | [29][30][31][32] | anticancer, anticholesterol [55]; antidepressant [56]; |
| 12 | Linalool | acyclic monoterpene alcohol |
🌱✿ | [33][34] | antibacterial, antifungal [57]; anxiolytic [58]; anticancer, antioxidant [59]; analgesic [60]; anti-inflammatory [61]; |
| 13 | cis-linalool oxide | oxygenated heteromonocyclic monoterpene |
[33] | n/a | |
| 14 | Myrcene | acyclic monoterpene hydrocarbon | 🌱 | [31][33] | analgesic, antinociceptive [62]; |
| 15 | Neryl acetate | acyclic monoterpene hydrocarbon | 🌱✿ | [35] | n/a |
| 16 | (E)-Ocimene | acyclic monoterpene hydrocarbon | 🌱✿ | [31] | anticancer [63]; anticonvulsant [64]; |
| 17 | (Z)-β-Ocimene | [31] | anticancer [63]; antibacterial [65]; |
||
| 18 | α-Phellandrene | monocyclic monoterpene hydrocarbon |
🌱 | [31] | antifungal [66]; antidepressant [67]; anti-inflammatory, antihyperalgesic [67][68], analgesic, antinociceptive [68]; anticancer [69]; |
| 19 | β-Phellandrene | ✿ | [31] | n/a | |
| 20 | α-Pinene | polycyclic monoterpene hydrocarbon |
🌱 | [29][31][33] | antifungal, anti-inflammatory, antioxidant [70]; anticancer [71]; anti-Leishmania [72]; gastroprotective [73][74]; antibacterial [75]; antiviral [76]; neuroprotective [77]; |
| 21 | β-Pinene | 🌱✿ | [29] | anticancer [78]; antimicrobial [79]; gastroprotective [74]; neuroprotective [80]; |
|
| 22 | Piperitone | oxygenated monocyclic monoterpene | 𐩕 | [31] | n/a |
| 23 | Sabinene | polycyclic monoterpene hydrocarbon | 🌱 | [31] | n/a |
| 24 | cis-Sabinene hydrate | oxygenated polycyclic monoterpene | [31] | ||
| 25 | Safranal | oxygenated monocyclic monoterpene | 🌱 | [35] | antioxidant [81]; antimicrobial [82]; anticonvulsant [83]; antidepressant, anxiolytic [84]; gastroprotective [85]; |
| 26 | α-Terpinene | monocyclic monoterpene hydrocarbon |
🌱 | [31] | antioxidant [86]; antimicrobial [87]; |
| 27 | Δ-Terpinene | 🌱✿ | [34] | n/a | |
| 28 | γ-Terpinene | ✿ | [31] | antimicrobial [87]; | |
| 29 | α-Terpineol | monocyclic monoterpene alcohol |
✿𐩕 | [31][32][34][35][88] | antioxidant, anticancer, antinociceptive, anticonvulsant, sedative, antibronchitis, antihypertensive, vasorelaxant, cardioprotective [89]; |
| 30 | 4-Terpineol | 🌱 | [31][35] | antimicrobial [90]; gastroprotective [91]; |
|
| 31 | Thymol | monocyclic monoterpene hydrocarbon |
🌱✿ | [31][32] | anti-inflammatory, antioxidant, antimicrobial, immunostimulatory, anticancer [92]; cardioprotective [93]; antihypertensive [52]; antihyperglycemic [94]; antinociceptive [95]; gastroprotective [96]; anxiolytic [97]; |
| 32 | Abscisic acid | oxygenated monocyclic sesquiterpene |
🌱 | [98][99] | antidiabetic [100]; antinociceptive [101]; |
| 33 | α-Amorphene | polycyclic sesquiterpene hydrocarbon |
🌱 | [32][35] | n/a |
| 34 | Aromadendrene | polycyclic sesquiterpene hydrocarbon |
🌱✿ | [29][30][35] | antimicrobial [102]; |
| 35 | allo-Aromadendrene | polycyclic sesquiterpene hydrocarbon |
🌱✿ | [22][23][28][30][31][32][33][34][35] | |
| 36 | allo-Aromadendrene epoxide | oxygenated polycyclic sesquiterpene |
🌱 | [35] | |
| 37 | Bisabola-2,10-diene(1-9)oxide | oxygenated polycyclic sesquiterpene |
🌱 | [35] | n/a |
| 38 | β-Bisabolene | monocyclic sesquiterpene hydrocarbon |
[30] | anticancer [103] | |
| 39 | epi-α-Bisabolol | monocyclic sesquiterpene alcohol |
🌱✿ | [28][30][31][35] | anti-inflammatory [104]; antimicrobial [105]; anticancer [106]; |
| 40 | α-Bisabolol | 🌱 | [23][35] | n/a | |
| 41 | β-Bisabolol | 🌱✿ | [30] | n/a | |
| 42 | β-Bourbonene | polycyclic sesquiterpene hydrocarbon |
🌱✿ | [22][23][28][29][31][32][33][34][35] | n/a |
| 43 | 1,5-di-epi-Bourbonene (α or β) | 🌱 | [35] | ||
| 44 | Bulnesol | polycyclic sesquiterpene alcohol |
🌱✿ | [31] | n/a |
| 45 | Cadalene | polycyclic sesquiterpene hydrocarbon |
🌱 | [35] | n/a |
| 46 | Cadina-1,4-diene | polycyclic sesquiterpene hydrocarbon |
🌱✿ | [30][35] | n/a |
| 47 | α-Cadinene | polycyclic sesquiterpene hydrocarbon |
🌱 | [32][35] | n/a |
| 48 | cis-γ-Cadinene | 🌱✿𐩕 | [31] | ||
| 49 | trans-γ-Cadinene | 🌱 | [28] | ||
| 50 | γ-Cadinene | 🌱 | [33][34][35] | ||
| 51 | δ-Cadinene | 🌱✿𐩕 | [23][28][30][31][32][34][35] | ||
| 52 | α-Cadinol | polycycylic sesquiterpene alcohol |
🌱✿ | [23][30][31][35] | antifungal [107]; |
| 53 | T-Cadinol | [28][31][35][108] | anticancer [109] | ||
| 54 | α-Calacorene | polycyclic sesquiterpene hydrocarbon |
🌱 | [32][35] | n/a |
| 55 | β-Calacorene | [35] | |||
| 56 | Calamenene | polycyclic sesquiterpene hydrocarbon |
🌱 | [32][35] | anticancer [109]; |
| 57 | Caryophylladienol I | polycyclic sesquiterpene alcohol |
🌱 | [35] | n/a |
| 58 | Caryophylladienol II | [35] | |||
| 59 | β-Caryophyllene | polycyclic sesquiterpene hydrocarbon |
🌱✿𐩕 | [22][23][28][29][30][31][32][33][34][35] | antioxidant, antimicrobial, antitumor, anticancer [110]; anti-inflammatory, neuroprotective [111]; anxiolytic, antidepressant [112]; anticonvulsant [113]; analgesic [114]; |
| 60 | β-Caryophyllene epoxide |
oxygenated polycyclic sesquiterpene |
🌱 | [23][28][30][31][35] | anticancer, analgesic [114]; |
| 61 | Caryophyllenol II | polycyclic sesquiterpene alcohol |
🌱 | [35] | n/a |
| 62 | 8,14-Cedranoxide | oxygenated sesquiterpene |
🌱 | [28] | n/a |
| 63 | α-Cedrene | polycyclic sesquiterpene hydrocarbon |
🌱 | [29] | n/a |
| 64 | α-Copaene | polycyclic sesquiterpene hydrocarbon |
🌱 | [22][28][29][31][32][34][35] | antioxidant, anticancer [115]; neuroprotective [116]; |
| 65 | β-Copaene | 🌱✿𐩕 | [28][32][35] | ||
| 66 | α-Corocalene | polycyclic sesquiterpene hydrocarbon |
🌱 | [35] | n/a |
| 67 | α-Cubebene | polycyclic sesquiterpene hydrocarbon |
🌱✿ | [22][31][35] | antioxidant, neuroprotective [117]; antimicrobial [118]; anti-inflammatory [119]; |
| 68 | β-Cubebene | 🌱 | [22][28][31] | ||
| 69 | Cubebol | polycyclic sesquiterpene alcohol |
🌱 | [35] | n/a |
| 70 | 4-epi-Cubebol | [35] | |||
| 71 | 1,10-di-epiCubenol | polycyclic sesquiterpene alchohol |
🌱✿ | [28][31][35] | n/a |
| 72 | Cubenol | 🌱 | [35] | ||
| 73 | 1-epi-Cubenol | 🌱✿ | [28][31][35] | ||
| 74 | ar-Curcumen-15-al | oxygenated monocyclic sesquiterpene | 🌱 | [28][35] | n/a |
| 75 | ar-Curcumene | monocyclic sesquiterpene hydrocarbon |
🌱✿ | [22][23][28][29][30][31][32][33][34][35][108] | n/a |
| 76 | β-Curcumene | ✿ | [30] | ||
| 77 | γ-Curcumene | 🌱✿ | [30][31] | ||
| 78 | Curcuphenol | monocyclic sesquiterpene alcohol |
🌱✿ | [30][35] | anticancer [120]; |
| 79 | Cyclosativene | polycyclic sesquiterpene hydrocarbon |
🌱✿ | [31] | n/a |
| 80 | Dehydrosesquicineole | oxygenated polycyclic sesquiterpene |
🌱 | [35] | n/a |
| 81 | Bicyclo-Elemene | polycyclic sesquiterpene hydrocarbon |
🌱 | [32] | n/a |
| 82 | β-Elemene | monocyclic sesquiterpene hydrocarbon |
🌱 | [30][35] | anticancer, anti-inflammatory [121]; |
| 83 | γ-Elemene | 🌱 | [30] | ||
| 84 | δ-Elemene | 🌱✿ | [30][31][32] | ||
| 85 | Elemol | monocyclic sesquiterpene alcohol |
🌱 | [23][30][31] | n/a |
| 86 | β-Eudesma 4(15), 7 dien-1β-ol | polycyclic sesquiterpene alcohol |
🌱 | [35] | n/a |
| 87 | α-Eudesmol | 🌱 | [28] | neuroprotective [122]; | |
| 88 | β-Eudesmol | 🌱✿ | [28][30] | anti-allergic, anti-inflammatory [123]; anticancer [124]; |
|
| 89 | γ-Eudesmol | 🌱✿ | [30][31] | n/a | |
| 90 | 10-epi-γ-Eudesmol | [30][31] | n/a | ||
| 91 | Kunseaol | monocyclic sesquiterpene alcohol |
🌱 | [35] | n/a |
| 92 | Bicyclo-Germacrene | polycyclic sesquiterpene hydrocarbon |
🌱 | [35] | n/a |
| 93 | Germacrene B | monocyclic sesquiterpene hydrocarbon |
🌱 | [29][30] | |
| 94 | Germacrene D | 🌱✿𐩕 | [22][23][29][30][31][33][34][35] | anticancer [125]; anti-inflammatory, analgesic [126]; antioxidant [127]; |
|
| 95 | Iso-Germacrene D | 🌱 | [35] | ||
| 96 | β-Germacrenol | monocyclic sesquiterpene alcohol |
🌱 | [35] | n/a |
| 97 | Globulol | polycyclic sesquiterpene alcohol |
🌱✿ | [28][30] | n/a |
| 98 | α-Guaia-6,10(14)-diene-4β-ol | polycyclic sesquiterpene alcohol |
🌱 | [35] | n/a |
| 99 | Guaiene | polycyclic sesquiterpene hydrocarbon |
🌱 | [28] | n/a |
| 100 | Guaiol | polycyclic sesquiterpene alcohol |
🌱✿ | [30][31] | n/a |
| 101 | α-Gurjunene | polycyclic sesquiterpene hydrocarbon |
🌱✿ | [29][31][33][34] | n/a |
| 102 | β-Gurjunene | [31] | |||
| 103 | β-Himachalene | polycyclic sesquiterpene hydrocarbon |
✿𐩕 | [31] | n/a |
| 104 | α-Humulene | monocyclic sesquiterpene hydrocarbon |
🌱✿ | [22][23][28][29][30][31][33][34][35] | antitumor, anti- inflammatory, antimicrobial [128]; |
| 105 | Iso-Calamendiol | polycyclic sesquiterpene alcohol |
🌱 | [35] | n/a |
| 106 | Iso-Italicene | polycyclic sesquiterpene hydrocarbon |
🌱 | [28] | n/a |
| 107 | Juniper camphor | polycyclic sesquiterpene alcohol |
🌱✿ | [31] | n/a |
| 108 | Ledol | polycyclic sesquiterpene alcohol |
🌱 | [35][88] | n/a |
| 109 | α-Longipinene | polycyclic sesquiterpene hydrocarbon |
🌱 | [28] | n/a |
| 110 | cis-Muurola-4(14),5-diene | polycyclic sesquiterpene hydrocarbon |
🌱✿ | [31][35] | n/a |
| 111 | α-Muurolene | [22][30][31][32][34] | |||
| 112 | γ-Muurolene | [28][31][35] | |||
| 113 | 14-hydroxi-α-Muurolene | polycyclic sesquiterpene alcohol |
🌱 | [28] | n/a |
| 114 | α-Muurolol | 🌱✿ | [31] | ||
| 115 | epi-α-Muurolol | [30] | |||
| 116 | T-Muurolol | [23][28][31][31][35] | |||
| 117 | E-Nerolidol | acyclic sesquiterpene alcohol |
🌱✿ | [30] | antihyperlipidemic, anti-inflammatory, anti-uterine fibroids [129]; anticancer [130]; |
| 118 | (E)-Nuciferol | moncyclic sesquiterpene alcohol |
🌱 | [35] | n/a |
| 119 | β-Oplopenone | oxygenated polycyclic sesquiterpene |
🌱 | [35] | n/a |
| 120 | Salvial-4(14)-en-1-one | oxygenated polycyclic sesquiterpene |
🌱 | [35] | n/a |
| 121 | α-Santalene | polycyclic sesquiterpene hydrocarbon |
🌱 | [32] | n/a |
| 122 | cis-α-Santalol | polycyclic sesquiterpene alcohol |
🌱 | [28] | antihyperglycemic, antioxidant [131]; |
| 123 | Selin-11-en-4-α-ol | polycyclic sesquiterpene alcohol |
🌱 | [88] | anxiolytic, sedative [132]; |
| 124 | Selina-3,7(11)-diene | polycyclic sesquiterpene hydrocarbon |
🌱 | [32] | n/a |
| 125 | α-Selinene | polycyclic sesquiterpene hydrocarbon |
🌱 | [32] | n/a |
| 126 | β-Sesquiphellandrene | monocyclic sesquiterpene hydrocarbon |
🌱✿ | [23][29][31][35] | anticancer [133]; antioxidant [134]; |
| 127 | trans-Sesquisabinene hydrate |
polycyclic sesquiterpene alcohol |
🌱 | [30] | n/a |
| 128 | Shyobunone | oxygenated monocyclic sesquiterpene |
🌱 | [22][35] | neuroprotective, acetyl-cholinesterase inhibition [135]; |
| 129 | 6-epi-Shyobunone | [35] | |||
| 130 | iso-Shyobunone | [35] | |||
| 131 | Spathulenol | polycyclic sesquiterpene alcohol |
🌱✿ | [28][30][31][35] | neuroprotective [136]; antibacterial, antioxidant, anti-inflammatory, anticancer [137]; |
| 132 | Spathulenol isomer | 🌱 | [30] | ||
| 133 | ar-Turmerol | monocyclic sesquiterpene alcohol |
🌱✿ | [28][30] | n/a |
| 134 | Valerianol | polycyclic sesquiterpene alcohol |
🌱✿ | [31] | n/a |
| 135 | Viridiflorol | polycyclic sesquiterpene alcohol |
🌱✿ | [30][35] | anti-arthritic, analgesic, antinociceptive [138]; anticancaer [139]; antioxidant, antibacterial, anti-inflammatory [140]; |
| 136 | Xanthorrhizol | monocyclic sesquiterpene alcohol |
🌱 | [28][35] | anticancer [141]; antimicrobial, antibacterial [142]; antihypolipidemic [143]; anti-inflammatory [144]; |
| 137 | α-Ylangene | polycyclic sesquiterpene hydrocarbon |
🌱 | [32] | n/a |
| 138 | β-Ylangene | 🌱 | [35] | ||
| 139 | α-Zingiberene | monocyclic sesquiterpene hydrocarbon |
🌱✿𐩕 | [22][23][28][29][30][31][32][33][34][35] | analgesic, neuroprotective [145]; anticancer [146]; anti-inflammatory [147]; |
| 140 | Zingiberenol | monocyclic sesquiterpene alcohol |
🌱 | [35] | n/a |
| 141 | 15,16-Dinorlabd-8(20)-en-13-one | oxygenated diterpene |
🌱 | [88] | n/a |
| 142 | Geranyl-p-cymene | monocyclic diterpene hydrocarbon |
🌱 | [35] | n/a |
| 143 | Geranyl α-terpinene | monocyclic diterpene hydrocarbon |
🌱 | [35] | n/a |
| 144 | Geranyl linalool | acyclic diterpene alcohol |
🌱 | [35] | n/a |
| 145 | Gibberellin | oxygenated polycyclic diterpene |
🌱 | [4] | n/a |
| 146 | Manool | polycyclic diterpene alcohol |
🌱 | [88] | antioxidant, anti-inflammatory [148]; anticancer [149]; antihypertensive [150]; cardioprotective [151]; |
| 147 | Manoyl oxide | polycyclic oxygenated diterpene |
🌱 | [108] | anticancer [152]; |
| 148 | 13-epi-Manoyl oxide | [29][35][108] | |||
| 149 | Methyl Neoabietate | oxygenated polycyclic diterpene |
🌱 | [29] | n/a |
| 150 | lutein | oxygenated polycyclic tetraterpene |
🌱 | [4] | antioxidant, anti-inflammatory, neuroprotective [153]; anticancer [154]; hepatoprotective [155]; cardiopreotective [156]; |
| 151 | neoxanthin | oxygenated polycyclic tetraterpene |
🌱 | [4] | antihyperlipidemic [157]; |
| 152 | zeaxanthin | oxygenated polycyclic tetraterpene |
🌱 | [4] | n/a |
2.1.2. Phenylpropanoids from the Essential Oils
2.1.3. Diterpenes
2.1.4. Tetraterpenes
2.2. Phenolic Compounds
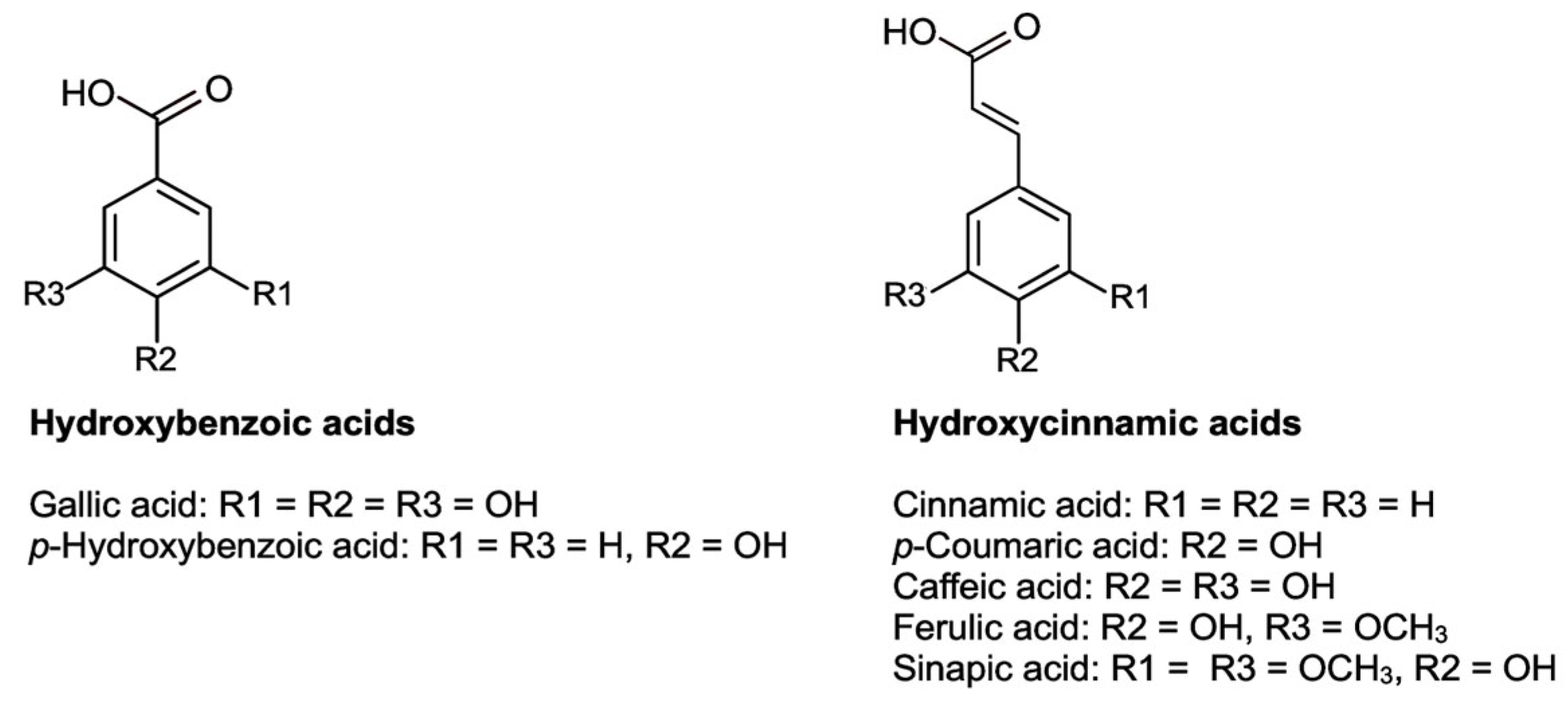
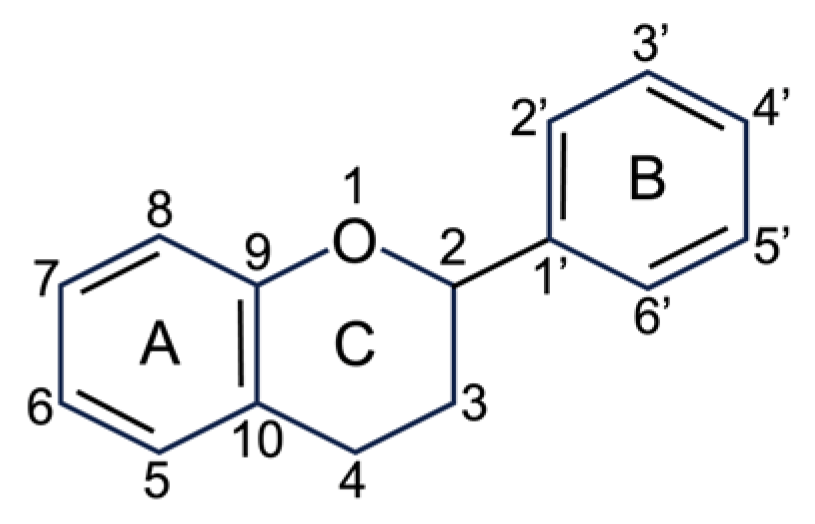
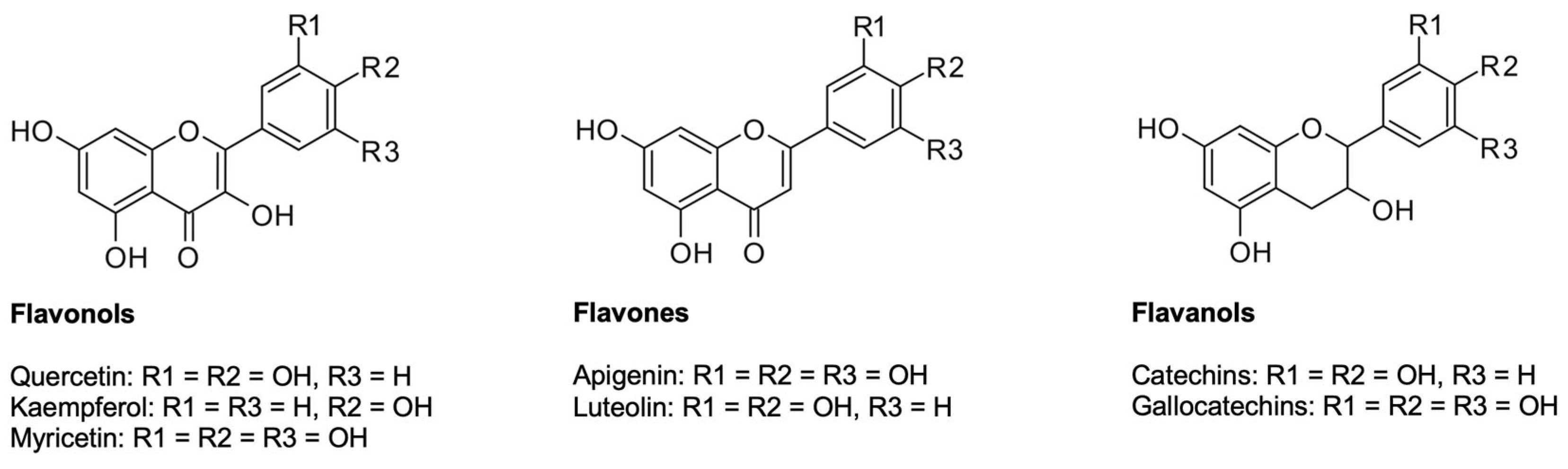
2.2.1. Flavonoids
2.2.2. Phenolic Acids
2.3. Carbonylic Compounds
2.4. Phytohormones and Vitamin E
2.5. Alkanes
2.6. Other Compounds: Fatty and Carboxylic Acids
3. Preparation Methods of the C. albidus Extracts
Studies of the pharmacological properties of traditional medicines based on C. albidus preparations are related to the presence of terpenoids and polyphenols. In order to understand the use that has been given to C. albidus traditionally, it is necessary to first review the preparation methods used in popular medicine of this plant due to their influence on the pharmacological effect. 6.1. Traditional Preparations For the different applications, traditionally, only the aerial parts of C. albidus were harvested, mainly the leaves, but also flowers, flower buds, and to a lesser extent stems. The traditional preparation of C. albidus varies from an infusion to a prolonged decoction, while the dose usually used is around 3 g per 100 mL of water, taking a cup (150 mL) two or three times a day [171]. Within traditional preparations, decoction is the most used technique. It consists of boiling the plant material for a certain period of time and letting it rest afterwards. This method is primarily suitable for thermostable and water-soluble phytochemicals. During decoction, several compounds undergo chemical modifications. For example, catechins undergo epimerization, which is a change in their configuration relative to one of their stereogenic centres. Epimers, specifically epicatechins and epigallocatechins, have been shown to have important health benefits. It has been found that this epimerization occurs more readily in water with alkaline pH values than in purified water [172]. In addition, it has been shown that at temperatures greater than 98ºC, epimerization occurs faster than its degradation [173], so it can be deduced that the traditional preparation of C. albidus is the most effective way to extract catechins and their epimers. However, for green tea, the levels of epicatechin, epicatechin gallate, epigallocatechin, and epigallocatechin gallate were reported to increase only during the first 3 to 5 min of preparation (infusion at 85ºC), and the proportion of these flavonoids decreased as time increased. In contrast, another study found that levels of catechin, gallocatechin, and gallocatechin gallate increased continuously with the length of preparation time [174]. Taking these results into account, the pharmacological activities referred to in traditional use could be optimized by limiting the decoction time. Nonetheless, thermolabile compounds are lost in the decoction process. As a result, monoterpenes should not be contained in the resulting extract. Sesquiterpenes, however, would not be affected by extracting temperatures around 100 degrees but by low solubility in water due to their lipophilic character. It can therefore be assumed that terpenes play a minor role in the traditional decoction of plant material. On the other side, probably in order to use the entire compound spectrum of the plant, based on both terpenes and polyphenols, the dried and crushed leaves sometimes were used directly (orally) [175][176][177][178][179]. This usage ensures that the resulting medicine is rich in polyphenols terpenes and other volatiles. However, a loss of several terpenes and oxidative reactions could be induced by the drying process, as reported for other species as Cannabis sativa [180][181].
4. Therapeutical Uses
4.1. Traditional Uses
Plant resources have always been an integral part of human society throughout history. Until the middle of the last century, traditional medicines provided an alternative and inexpensive source of primary health care for the rural population. However, with access to synthetic drugs, a large number of medicinal plants became obsolete, the memory of which in the population, after only two generations, is being lost. One of these medicinal plants is C. albidus, which has been used in traditional folk medicine for a variety of illnesses [171][182][183][184], especially for the treatment of fever, diarrhea and other gastrointestinal illnesses [185], skin diseases, rheumatism, and various inflammatory diseases [165]. For the sake of completeness, it is mentioned here that C. albidus has also been used as a tanning agent [164], as an insect repellent, and as a substitute for tobacco, highly appreciated, moreover, for its hypotensive effect [31][184][186]. The decoction of leaves was traditionally used in the Spanish Levant as a tranquilizer, in the Baixa Plana as a sedative [171], and as a remedy against Parkinson’s symptoms in Mallorca [187][188]. To relieve toothache, mouthwashes were made with a decoction of its leaves and flowers. A sip of the resulting liquid, once cold, was kept in the mouth for some time [179][189][190][191]. In addition, the decoction of the aerial parts was used as an external antiseptic, for wounds and skin infections [190][191][192][193]. In the Spanish Basque Country, several uses were reported. For example, decoction was applied for the treatment of ulcers and for the treatment of gangrene, and fresh leaves were used directly on the wound for disinfection [194]. In the Mediterranean region, the decoction of the aerial parts (leaves, stems, and flowers) has been used to regulate blood pressure [195][196]. It has also been a frequent remedy for hemorrhoids and to treat bruises and varicose veins [187]. The decoction of flowers and leaves has also been popularly used as an analgesic for oral infections [293 and for hepatoprotection in Granada and Mallorca [187][195]. The decoction of the fresh aerial parts, including the flowers, was used as a remedy against colds and flu infections, and against bronchitis [175][187][196] and whooping cough [171]. In the Spanish peninsula, C. albidus decoction has also been used as a remedy for osteoarthritis in the province of Jaen [179] and for rheumatism in the Valencian community and the Province of Jaen [189][197]. In addition, it was used as an external antiseptic for wound healing and skin infections in the provinces of Castellon, Mallorca, and Almería [171][187][190], and in Morocco [176][178]. In Sardinia (Italy), a traditional use is reported in poultices and ointments, which were applied directly to the wound [198]. In cases of gastrointestinal infections, in Almería (Spain), an infusion of dried leaves was prepared to reduce abdominal pain [190][195]. Against colic, in Castilla-La Mancha and Murcia (Spain) an infusion of young and tender shoots was administered, but it was also supplied by oral ingestion of the powder of dry leaves for treatment [175]. The dried leaf powder also served as an antidiarrheal in Jaen [179]. Infusions of fresh flowers and leaves have been used as an antiseptic for the urinary tract in Murcia [199] and also as an anti-inflammatory for orchitis in Valencia [200].
This entry is adapted from the peer-reviewed paper 10.3390/plants12162988
References
- Guzmán, B.; Vargas, P. Long-Distance Colonization of the Western Mediterranean by Cistus ladanifer (Cistaceae) despite the Absence of Special Dispersal Mechanisms. J. Biogeogr. 2009, 36, 954–968.
- Müller, M.; Siles, L.; Cela, J.; Munné-Bosch, S. Perennially Young: Seed Production and Quality in Controlled and Natural Populations of Cistus albidus Reveal Compensatory Mechanisms That Prevent Senescence in Terms of Seed Yield and Viability. J. Exp. Bot. 2014, 65, 287–297.
- Pérez-Llorca, M.; Casadesús, A.; Müller, M.; Munné-Bosch, S. Leaf Orientation as Part of the Leaf Developmental Program in the Semi-Deciduous Shrub, Cistus albidus L.: Diurnal, Positional, and Photoprotective Effects During Winter. Front. Plant Sci. 2019, 10, 767.
- Pérez-Llorca, M.; Casadesús, A.; Munné-Bosch, S.; Müller, M. Contrasting Patterns of Hormonal and Photoprotective Isoprenoids in Response to Stress in Cistus albidus during a Mediterranean Winter. Planta 2019, 250, 1409–1422.
- Munné-Bosch, S.; Jubany-Marí, T.; Alegre, L. Enhanced Photo- and Antioxidative Protection, and Hydrogen Peroxide Accumulation in Drought-Stressed Cistus clusii and Cistus albidus Plants. Tree Physiol. 2003, 23, 1–12.
- Casadesús, A.; Bouchikh, R.; Pérez-Llorca, M.; Munné-Bosch, S. Linking Jasmonates with Vitamin E Accumulation in Plants: A Case Study in the Mediterranean Shrub Cistus albidus L. Planta 2021, 253, 36.
- Brossa, R.; Pintó-Marijuan, M.; Francisco, R.; López-Carbonell, M.; Chaves, M.M.; Alegre, L. Redox Proteomics and Physiological Responses in Cistus albidus Shrubs Subjected to Long-Term Summer Drought Followed by Recovery. Planta 2015, 241, 803–822.
- Roy, J.; Sonie, L. Germination and Population Dynamics of Cistus Species in Relation to Fire. J. Appl. Ecol. 1992, 29, 647.
- Casadesús, A.; Bouchikh, R.; Munné-Bosch, S. Contrasting Seasonal Abiotic Stress and Herbivory Incidence in Cistus albidus L. Plants Growing in Their Natural Habitat on a Mediterranean Mountain. J. Arid Environ. 2022, 206, 104842.
- Cabezudo, B.; Pérez Latorre, A.V.; Navarro, T.; Nieto Caldera, J.M. Estudios Fenomorfológicos En La Vegetación Del Sur de España. II. Alcornocales Mesomediterráneos. (Montes de Málaga, Málaga). Acta Bot. Malacit. 1993, 18, 179–188.
- Blasco, S.; Mateu, I. Flowering and Fruiting Phenology and Breeding System of Cistus albidus L. Acta Bot. Gallica 1995, 142, 245–251.
- Hernández, I.; Miret, J.A.; Van Der Kelen, K.; Rombaut, D.; Van Breusegem, F.; Munné-Bosch, S. Zeatin Modulates Flower Bud Development and Tocopherol Levels in Cistus albidus (L.) Plants as They Age. Plant Biol. 2015, 17, 90–96.
- Siles, L.; Müller, M.; Cela, J.; Hernández, I.; Alegre, L.; Munné-Bosch, S. Marked Differences in Seed Dormancy in Two Populations of the Mediterranean Shrub, Cistus albidus L. Plant Ecol. Divers. 2017, 10, 231–240.
- Rizzotto, M. Ricerche tassonomiche e corologiche sulle Cistaceae. 1: Il genere Cistus L. in Italia. Webbia 1979, 33, 343–378.
- Robles, C.; Dutoit, T.; Bonin, G. Inhibition Mechanisms and Successional Processes: A Case Study of Cistus albidus L. in Provence. Ecosyst. Sustain. Dev. 1998, 1, 437–446.
- Thanos, A.C.; Geroghiou, K.; Kadis, C.; Pantazi, C. Cistaceae: A plant family with hard seeds. Isr. J. Bot. 1993, 41, 251–263.
- Trabaud, L.; Oustric, J. Heat Requirements for Seed Germination of Three Cistus Species in the Garrigue of Southern France. Flora 1989, 183, 321–325.
- Trabaud, L.; Renard, P. Do light and litter influence the recruitment of cistus spp. Stands? Isr. J. Plant Sci. 1999, 47, 1–9.
- Baskin, J.M.; Baskin, C.C. A Classification System for Seed Dormancy. Seed Sci. Res. 2004, 14, 1–16.
- Barrajón-Catalán, E.; Fernández-Arroyo, S.; Roldán, C.; Guillén, E.; Saura, D.; Segura-Carretero, A.; Micol, V. A Systematic Study of the Polyphenolic Composition of Aqueous Extracts Deriving from Several Cistus Genus Species: Evolutionary Relationship: Polyphenolic Characterization of Cistus Aqueous Extracts. Phytochem. Anal. 2011, 22, 303–312.
- Polunin, O.; Schauer, T.; Everard, B. Pflanzen Europas; BLV-Bestimmungsbuch; BLV(-Verl. Ges.): München, Germany, 1971; ISBN 978-3-405-10929-5.
- Ormeño, E.; Baldy, V.; Ballini, C.; Fernandez, C. Production and Diversity of Volatile Terpenes from Plants on Calcareous and Siliceous Soils: Effect of Soil Nutrients. J. Chem. Ecol. 2008, 34, 1219–1229.
- Robles, C.; Garzino, S. Essential Oil Composition of Cistus albidus Leaves. Phytochemistry 1998, 48, 1341–1345.
- Castells, E.; Peñuelas, J. Is There a Feedback between N Availability in Siliceous and Calcareous Soils and Cistus albidus Leaf Chemical Composition? Oecologia 2003, 136, 183–192.
- El Mamoun, I.; Mouna, F.; Mohammed, A.; Najib, B.; Zine-El Abidine, T.; Abdelkarim, G.; Didier, B.; Laurent, L.; Abdelaziz, S. Zinc, Lead, and Cadmium Tolerance and Accumulation in Cistus libanotis, Cistus albidus and Cistus salviifolius: Perspectives on Phytoremediation. Remediat. J. 2020, 30, 73–80.
- Lukas, B.; Jovanovic, D.; Schmiderer, C.; Kostas, S.; Kanellis, A.; Gómez Navarro, J.; Aytaç, Z.; Koç, A.; Sözen, E.; Novak, J. Intraspecific Genetic Diversity of Cistus creticus L. and Evolutionary Relationships to Cistus albidus L. (Cistaceae): Meeting of the Generations? Plants 2021, 10, 1619.
- ISO Standard No. 9235:2013; International Organization for Standardization Aromatic Natural Raw Materials. ISO: Geneva, Switzerland, 2013.
- Bechlaghem, K.; Allali, H.; Benmehdi, H.; Aissaoui, N.; Flamini, G. Chemical Analysis of the Essential Oils of Three Cistus Species Growing in North- West of Algeria. Agric. Conspec. Sci. 2019, 84, 283–293.
- Llusià, J.; Peñuelas, J.; Ogaya, R.; Alessio, G. Annual and Seasonal Changes in Foliar Terpene Content and Emission Rates in Cistus albidus L. Submitted to Soil Drought in Prades Forest (Catalonia, NE Spain). Acta Physiol. Plant. 2010, 32, 387–394.
- Palá-Paúl, J.; Velasco-Negueruela, A.; Pérez-Alonso, M.J.; Sanz, J. Seasonal Variation in Chemical Composition of Cistus albidus L. from Spain. J. Essent. Oil Res. 2005, 17, 19–22.
- Maccioni, S.; Baldini, R.; Cioni, P.L.; Tebano, M.; Flamini, G. In Vivo Volatiles Emission and Essential Oils from Different Organs and Pollen Of Cistus albidus from Caprione (Eastern Liguria, Italy). Flavour Fragr. J. 2007, 22, 61–65.
- Morales-Soto, A.; Oruna-Concha, M.J.; Elmore, J.S.; Barrajón-Catalán, E.; Micol, V.; Roldán, C.; Segura-Carretero, A. Volatile Profile of Spanish Cistus Plants as Sources of Antimicrobials for Industrial Applications. Ind. Crops Prod. 2015, 74, 425–433.
- Ormeño, E.; Mévy, J.P.; Vila, B.; Bousquet-Mélou, A.; Greff, S.; Bonin, G.; Fernandez, C. Water Deficit Stress Induces Different Monoterpene and Sesquiterpene Emission Changes in Mediterranean Species. Relationship between Terpene Emissions and Plant Water Potential. Chemosphere 2007, 67, 276–284.
- Ormeño, E.; Bousquet-Mélou, A.; Mévy, J.-P.; Greff, S.; Robles, C.; Bonin, G.; Fernandez, C. Effect of Intraspecific Competition and Substrate Type on Terpene Emissions from Some Mediterranean Plant Species. J. Chem. Ecol. 2007, 33, 277–286.
- Paolini, J.; Tomi, P.; Bernardini, A.-F.; Bradesi, P.; Casanova, J.; Kaloustian, J. Detailed Analysis of the Essential Oil from Cistus albidus L. by Combination of GC/RI, GC/MS and 13C-NMR Spectroscopy. Nat. Prod. Res. 2008, 22, 1270–1278.
- Gao, H.; Gao, X. Brain Targeted Drug Delivery Systems: A Focus on Nanotechnology and and Nanoparticulates; Academic Press: London, UK, 2019; ISBN 978-0-12-814002-4.
- Xu, H.; Blair, N.T.; Clapham, D.E. Camphor Activates and Strongly Desensitizes the Transient Receptor Potential Vanilloid Subtype 1 Channel in a Vanilloid-Independent Mechanism. J. Neurosci. Off. J. Soc. Neurosci. 2005, 25, 8924–8937.
- Chen, W.; Vermaak, I.; Viljoen, A. Camphor—A Fumigant during the Black Death and a Coveted Fragrant Wood in Ancient Egypt and Babylon—A Review. Molecules 2013, 18, 5434–5454.
- Banerjee, S.; Welsch, C.W.; Rao, A.R. Modulatory Influence of Camphor on the Activities of Hepatic Carcinogen Metabolizing Enzymes and the Levels of Hepatic and Extrahepatic Reduced Glutathione in Mice. Cancer Lett. 1995, 88, 163–169.
- Laude, E.A.; Morice, A.H.; Grattan, T.J. The Antitussive Effects of Menthol, Camphor and Cineole in Conscious Guinea-Pigs. Pulm. Pharmacol. 1994, 7, 179–184.
- Liu, H.; Zhou, Y.; Sun, Y.; Sheng, X.; Zhang, J.; Zhang, Z.; Ding, J. Effect of Menthol and Camphor on Permeation of Compound Diphenhydramine Cream in Vitro. Cent South Pharm. 2011, 9, 97–101.
- Patra, J.K.; Das, G.; Bose, S.; Banerjee, S.; Vishnuprasad, C.N.; Pilar Rodriguez-Torres, M.; Shin, H. Star Anise (Illicium verum): Chemical Compounds, Antiviral Properties, and Clinical Relevance. Phytother. Res. 2020, 34, 1248–1267.
- Jeong, J.-G.; Kim, Y.S.; Min, Y.K.; Kim, S.H. Low Concentration of 3-Carene Stimulates the Differentiation of Mouse Osteoblastic MC3T3-E1 Subclone 4 Cells. Phytother. Res. 2008, 22, 18–22.
- Gil, M.L.; Jimenez, J.; Ocete, M.A.; Zarzuelo, A.; Cabo, M.M. Comparative Study of Different Essential Oils of Bupleurum gibraltaricum Lamarck. Pharmazie 1989, 44, 284–287.
- Pina-Vaz, C.; Gonçalves Rodrigues, A.; Pinto, E.; Costa-de-Oliveira, S.; Tavares, C.; Salgueiro, L.; Cavaleiro, C.; Gonçalves, M.J.; Martinez-de-Oliveira, J. Antifungal Activity of Thymus Oils and Their Major Compounds. J. Eur. Acad. Dermatol. Venereol. 2004, 18, 73–78.
- Aeschbach, R.; Löliger, J.; Scott, B.C.; Murcia, A.; Butler, J.; Halliwell, B.; Aruoma, O.I. Antioxidant Actions of Thymol, Carvacrol, 6-Gingerol, Zingerone and Hydroxytyrosol. Food Chem. Toxicol. 1994, 32, 31–36.
- Jayakumar, S.; Madankumar, A.; Asokkumar, S.; Raghunandhakumar, S.; Gokula dhas, K.; Kamaraj, S.; Divya, M.G.J.; Devaki, T. Potential Preventive Effect of Carvacrol against Diethylnitrosamine-Induced Hepatocellular Carcinoma in Rats. Mol. Cell. Biochem. 2012, 360, 51–60.
- Hotta, M.; Nakata, R.; Katsukawa, M.; Hori, K.; Takahashi, S.; Inoue, H. Carvacrol, a Component of Thyme Oil, Activates PPAR alpha and Gamma and Suppresses COX-2 Expression. J. Lipid Res. 2010, 51, 132–139.
- Cho, S.; Choi, Y.; Park, S.; Park, T. Carvacrol Prevents Diet-Induced Obesity by Modulating Gene Expressions Involved in Adipogenesis and Inflammation in Mice Fed with High-Fat Diet. J. Nutr. Biochem. 2012, 23, 192–201.
- Decker, K.; Keppler, D. Galactosamine Induced Liver Injury. Prog. Liver Dis. 1972, 4, 183–199.
- Boskabady, M.H.; Jandaghi, P. Relaxant Effects of Carvacrol on Guinea Pig Tracheal Chains and Its Possible Mechanisms. Pharmazie 2003, 58, 661–663.
- Peixoto-Neves, D.; Silva-Alves, K.S.; Gomes, M.D.M.; Lima, F.C.; Lahlou, S.; Magalhães, P.J.C.; Ceccatto, V.M.; Coelho-de-Souza, A.N.; Leal-Cardoso, J.H. Vasorelaxant Effects of the Monoterpenic Phenol Isomers, Carvacrol and Thymol, on Rat Isolated Aorta. Fundam. Clin. Pharmacol. 2010, 24, 341–350.
- Quintans-Júnior, L.; Moreira, J.C.F.; Pasquali, M.A.B.; Rabie, S.M.S.; Pires, A.S.; Schröder, R.; Rabelo, T.K.; Santos, J.P.A.; Lima, P.S.S.; Cavalcanti, S.C.H.; et al. Antinociceptive Activity and Redox Profile of the Monoterpenes (+)-Camphene, p -Cymene, and Geranyl Acetate in Experimental Models. ISRN Toxicol. 2013, 2013, 459530.
- Arabloei Sani, M.; Yaghmaei, P.; Hajebrahimi, Z.; Hayati Roodbari, N. Therapeutic Effect of P-Cymene on Lipid Profile, Liver Enzyme, and Akt/Mtor Pathway in Streptozotocin-Induced Diabetes Mellitus in Wistar Rats. J. Obes. 2022, 1–12.
- Sun, J. D-Limonene: Safety and Clinical Applications. Altern. Med. Rev. J. Clin. Ther. 2007, 12, 259–264.
- Lorigooini, Z.; Boroujeni, S.N.; Sayyadi-Shahraki, M.; Rahimi-Madiseh, M.; Bijad, E.; Amini-Khoei, H. Limonene through Attenuation of Neuroinflammation and Nitrite Level Exerts Antidepressant-Like Effect on Mouse Model of Maternal Separation Stress. Behav. Neurol. 2021, 2021, 8817309.
- Pattnaik, S.; Subramanyam, V.R.; Bapaji, M.; Kole, C.R. Antibacterial and Antifungal Activity of Aromatic Constituents of Essential Oils. Microbios 1997, 89, 39–46.
- Linck, V.M.; da Silva, A.L.; Figueiró, M.; Caramão, E.B.; Moreno, P.R.H.; Elisabetsky, E. Effects of Inhaled Linalool in Anxiety, Social Interaction and Aggressive Behavior in Mice. Phytomedicine 2010, 17, 679–683.
- Jana, S.; Patra, K.; Sarkar, S.; Jana, J.; Mukherjee, G.; Bhattacharjee, S.; Mandal, D.P. Antitumorigenic Potential of Linalool Is Accompanied by Modulation of Oxidative Stress: An in Vivo Study in Sarcoma-180 Solid Tumor Model. Nutr. Cancer 2014, 66, 835–848.
- Sugawara, Y.; Hara, C.; Aoki, T.; Sugimoto, N.; Masujima, T. Odor Distinctiveness between Enantiomers of Linalool: Difference in Perception and Responses Elicited by Sensory Test and Forehead Surface Potential Wave Measurement. Chem. Senses 2000, 25, 77–84.
- Peana, A.T.; D’Aquila, P.S.; Panin, F.; Serra, G.; Pippia, P.; Moretti, M.D.L. Anti-Inflammatory Activity of Linalool and Linalyl Acetate Constituents of Essential Oils. Phytomedicine 2002, 9, 721–726.
- Jansen, C.; Shimoda, L.M.N.; Kawakami, J.K.; Ang, L.; Bacani, A.J.; Baker, J.D.; Badowski, C.; Speck, M.; Stokes, A.J.; Small-Howard, A.L.; et al. Myrcene and Terpene Regulation of TRPV1. Channels 2019, 13, 344–366.
- Bomfim, L.M.; Menezes, L.R.A.; Rodrigues, A.C.B.C.; Dias, R.B.; Gurgel Rocha, C.A.; Soares, M.B.P.; Neto, A.F.S.; Nascimento, M.P.; Campos, A.F.; Silva, L.C.R.C.e.; et al. Antitumour Activity of the Microencapsulation of Annona vepretorum Essential Oil. Basic Clin. Pharmacol. Toxicol. 2016, 118, 208–213.
- Sayyah, M.; Nadjafnia, L.; Kamalinejad, M. Anticonvulsant Activity and Chemical Composition of Artemisia dracunculus L. Essential Oil. J. Ethnopharmacol. 2004, 94, 283–287.
- Pulaj, B.; Mustafa, B.; Nelson, K.; Quave, C.L.; Hajdari, A. Chemical Composition and in Vitro Antibacterial Activity of Pistacia terebinthus Essential Oils Derived from Wild Populations in Kosovo. BMC Complement. Altern. Med. 2016, 16, 147.
- Zhang, J.; Sun, H.; Chen, S.; Zeng, L.; Wang, T. Anti-Fungal Activity, Mechanism Studies on α-Phellandrene and Nonanal against Penicillium cyclopium. Bot. Stud. 2017, 58, 13.
- Piccinelli, A.C.; Santos, J.A.; Konkiewitz, E.C.; Oesterreich, S.A.; Formagio, A.S.N.; Croda, J.; Ziff, E.B.; Kassuya, C.A.L. Antihyperalgesic and Antidepressive Actions of (R)-(+)-Limonene, α-Phellandrene, and Essential Oil from Schinus terebinthifolius Fruits in a Neuropathic Pain Model. Nutr. Neurosci. 2015, 18, 217–224.
- Lima, D.F.; Brandão, M.S.; Moura, J.B.; Leitão, J.M.R.S.; Carvalho, F.A.A.; Miúra, L.M.C.V.; Leite, J.R.S.A.; Sousa, D.P.; Almeida, F.R.C. Antinociceptive Activity of the Monoterpene α-Phellandrene in Rodents: Possible Mechanisms of Action. J. Pharm. Pharmacol. 2012, 64, 283–292.
- Lin, J.-J.; Lu, K.-W.; Ma, Y.-S.; Tang, N.-Y.; Wu, P.-P.; Wu, C.-C.; Lu, H.-F.; Lin, J.-G.; Chung, J.-G. Alpha-Phellandrene, a Natural Active Monoterpene, Influences a Murine WEHI-3 Leukemia Model in Vivo by Enhancing Macrophague Phagocytosis and Natural Killer Cell Activity. In Vivo 2014, 28, 583–588.
- Karthikeyan, R.; Kanimozhi, G.; Prasad, N.R.; Agilan, B.; Ganesan, M.; Srithar, G. Alpha Pinene Modulates UVA-Induced Oxidative Stress, DNA Damage and Apoptosis in Human Skin Epidermal Keratinocytes. Life Sci. 2018, 212, 150–158.
- Hou, J.; Zhang, Y.; Zhu, Y.; Zhou, B.; Ren, C.; Liang, S.; Guo, Y. α-Pinene Induces Apoptotic Cell Death via Caspase Activation in Human Ovarian Cancer Cells. Med. Sci. Monit. 2019, 25, 6631–6638.
- Rodrigues, K.A.d.F.; Amorim, L.V.; Dias, C.N.; Moraes, D.F.C.; Carneiro, S.M.P.; Carvalho, F.A.d.A. Syzygium Cumini (L.) Skeels Essential Oil and Its Major Constituent α-Pinene Exhibit Anti-Leishmania Activity through Immunomodulation in Vitro. J. Ethnopharmacol. 2015, 160, 32–40.
- Pinheiro, M.A.; Magalhães, R.; Torres, D.; Cavalcante, R.; Mota, F.X.; Oliveira Coelho, E.A.; Moreira, H.; Lima, G.; da Costa Araújo, P.; Cardoso, J.L.; et al. Gastroprotective Effect of Alpha-Pinene and Its Correlation with Antiulcerogenic Activity of Essential Oils Obtained from Hyptis Species. Pharmacogn. Mag. 2015, 11, 123.
- Matthews Jucá, D.; da Silva, M.; Palheta Junior, R.; de Lima, F.; Okoba, W.; Lahlou, S.; de Oliveira, R.; dos Santos, A.; Magalhães, P. The Essential Oil of Eucalyptus tereticornis and Its Constituents, α- and β-Pinene, Show Accelerative Properties on Rat Gastrointestinal Transit. Planta Med. 2011, 77, 57–59.
- Utegenova, G.; Pallister, K.; Kushnarenko, S.; Özek, G.; Özek, T.; Abidkulova, K.; Kirpotina, L.; Schepetkin, I.; Quinn, M.; Voyich, J. Chemical Composition and Antibacterial Activity of Essential Oils from Ferula L. Species against Methicillin-Resistant Staphylococcus aureus. Molecules 2018, 23, 1679.
- Yang, Z.; Wu, N.; Zu, Y.; Fu, Y. Comparative Anti-Infectious Bronchitis Virus (IBV) Activity of (-)-Pinene: Effect on Nucleocapsid (N) Protein. Molecules 2011, 16, 1044–1054.
- Zamyad, M.; Abbasnejad, M.; Esmaeili-Mahani, S.; Mostafavi, A.; Sheibani, V. The Anticonvulsant Effects of Ducrosia anethifolia (Boiss) Essential Oil Are Produced by Its Main Component Alpha-Pinene in Rats. Arq. Neuropsiquiatr. 2019, 77, 106–114.
- Wang, Y.; Wu, C.; Zhang, Q.; Shan, Y.; Gu, W.; Wang, S. Design, Synthesis and Biological Evaluation of Novel β-Pinene-Based Thiazole Derivatives as Potential Anticancer Agents via Mitochondrial-Mediated Apoptosis Pathway. Bioorg. Chem. 2019, 84, 468–477.
- Liao, S.; Shang, S.; Shen, M.; Rao, X.; Si, H.; Song, J.; Song, Z. One-Pot Synthesis and Antimicrobial Evaluation of Novel 3-Cyanopyridine Derivatives of (−)-β-Pinene. Bioorg. Med. Chem. Lett. 2016, 26, 1512–1515.
- Felipe, C.F.B.; Albuquerque, A.M.S.; de Pontes, J.L.X.; de Melo, J.Í.V.; Rodrigues, T.C.M.L.; de Sousa, A.M.P.; Monteiro, Á.B.; Ribeiro, A.E.d.S.; Lopes, J.P.; de Menezes, I.R.A.; et al. Comparative Study of Alpha- and Beta-Pinene Effect on PTZ-Induced Convulsions in Mice. Fundam. Clin. Pharmacol. 2019, 33, 181–190.
- Assimopoulou, A.N.; Sinakos, Z.; Papageorgiou, V.P. Radical Scavenging Activity of Crocus sativus L. Extract and Its Bioactive Constituents. Phytother. Res. 2005, 19, 997–1000.
- Pintado, C.; de Miguel, A.; Acevedo, O.; Nozal, L.; Novella, J.L.; Rotger, R. Bactericidal Effect of Saffron (Crocus sativus L.) on Salmonella enterica during Storage. Food Control 2011, 22, 638–642.
- Hosseinzadeh, H.; Talebzadeh, F. Anticonvulsant Evaluation of Safranal and Crocin from Crocus sativus in Mice. Fitoterapia 2005, 76, 722–724.
- Hosseinzadeh, H.; Noraei, N.B. Anxiolytic and Hypnotic Effect of Crocus sativus Aqueous Extract and Its Constituents, Crocin and Safranal, in Mice. Phytother. Res. 2009, 23, 768–774.
- Kianbakht, S.; Mozaffari, K. Effects of Saffron and Its Active Constituents, Crocin and Safranal, on Prevention of Indomethacin Induced Gastric Ulcers in Diabetic and Nondiabetic Rats. JMPIR 2009, 8, 30–38.
- Rudbäck, J.; Bergström, M.A.; Börje, A.; Nilsson, U.; Karlberg, A.-T. α-Terpinene, an Antioxidant in Tea Tree Oil, Autoxidizes Rapidly to Skin Allergens on Air Exposure. Chem. Res. Toxicol. 2012, 25, 713–721.
- Baldissera, M.D.; Grando, T.H.; Souza, C.F.; Gressler, L.T.; Stefani, L.M.; da Silva, A.S.; Monteiro, S.G. In Vitro and in Vivo Action of Terpinen-4-Ol, γ-Terpinene, and α-Terpinene against Trypanosoma evansi. Exp. Parasitol. 2016, 162, 43–48.
- Fadel, H.; Kebbi, S.; Chalchat, J.-C.; Figueredo, G.; Chalard, P.; Benayache, F.; Ghedadba, N.; Benayache, S. Identification of Volatile Components and Antioxidant Assessment of the Aerial Part Extracts from an Algerian Cistus albidus L. of the Aures Region. J. New Technol. Mater. 2020, 10, 38–46.
- Khaleel, C.; Tabanca, N.; Buchbauer, G. α-Terpineol, a Natural Monoterpene: A Review of Its Biological Properties. Open Chem. 2018, 16, 349–361.
- Couladis, M.; Chinou, I.B.; Tzakou, O.; Petrakis, P.V. Composition and Antimicrobial Activity of the Essential Oil Of Hypericum rumeliacum Subsp.Apollinis (Boiss. & Heldr.). Phytother. Res. 2003, 17, 152–154.
- Souza, R.; Cardoso, M.; Menezes, C.; Silva, J.; De Sousa, D.; Batista, J. Gastroprotective Activity of α-Terpineol in Two Experimental Models of Gastric Ulcer in Rats. Daru J. Fac. Pharm. Tehran Univ. Med. Sci. 2011, 19, 277–281.
- Nagoor Meeran, M.F.; Javed, H.; Al Taee, H.; Azimullah, S.; Ojha, S.K. Pharmacological Properties and Molecular Mechanisms of Thymol: Prospects for Its Therapeutic Potential and Pharmaceutical Development. Front. Pharmacol. 2017, 8, 380.
- Nagoor Meeran, M.F.; Jagadeesh, G.S.; Selvaraj, P. Thymol, a Dietary Monoterpene Phenol Abrogates Mitochondrial Dysfunction in β-Adrenergic Agonist Induced Myocardial Infarcted Rats by Inhibiting Oxidative Stress. Chem. Biol. Interact. 2016, 244, 159–168.
- Saravanan, S.; Pari, L. Role of Thymol on Hyperglycemia and Hyperlipidemia in High Fat Diet-Induced Type 2 Diabetic C57BL/6J Mice. Eur. J. Pharmacol. 2015, 761, 279–287.
- Mendes, S.S.; Bomfim, R.R.; Jesus, H.C.R.; Alves, P.B.; Blank, A.F.; Estevam, C.S.; Antoniolli, A.R.; Thomazzi, S.M. Evaluation of the Analgesic and Anti-Inflammatory Effects of the Essential Oil of Lippia gracilis Leaves. J. Ethnopharmacol. 2010, 129, 391–397.
- Bukovská, A.; Cikoš, Š.; Juhás, Š.; Il’ková, G.; Rehák, P.; Koppel, J. Effects of a Combination of Thyme and Oregano Essential Oils on TNBS-Induced Colitis in Mice. Mediators Inflamm. 2007, 1–9.
- Bhandari, S.S.; Kabra, M.P. To Evaluate Anti-Anxiety Activity of Thymol. J. Acute Dis. 2014, 3, 136–140.
- López-Carbonell, M.; Gabasa, M.; Jáuregui, O. Enhanced Determination of Abscisic Acid (ABA) and Abscisic Acid Glucose Ester (ABA-GE) in Cistus albidus Plants by Liquid Chromatography-Mass Spectrometry in Tandem Mode. Plant Physiol. Biochem. 2009, 47, 256–261.
- Pérez-Llorca, M.; Caselles, V.; Müller, M.; Munné-Bosch, S. The Threshold between Life and Death in Cistus albidus L. Seedlings: Mechanisms Underlying Drought Tolerance and Resilience. Tree Physiol. 2021, 41, 1861–1876.
- Bassaganya-Riera, J.; Skoneczka, J.; Kingston, D.G.J.; Krishnan, A.; Misyak, S.A.; Guri, A.J.; Pereira, A.; Carter, A.B.; Minorsky, P.; Tumarkin, R.; et al. Mechanisms of Action and Medicinal Applications of Abscisic Acid. Curr. Med. Chem. 2010, 17, 467–478.
- Mollashahi, M.; Abbasnejad, M.; Esmaeili-Mahani, S. Phytohormone Abscisic Acid Elicits Antinociceptive Effects in Rats through the Activation of Opioid and Peroxisome Proliferator-Activated Receptors β/δ. Eur. J. Pharmacol. 2018, 832, 75–80.
- Mulyaningsih, S.; Sporer, F.; Zimmermann, S.; Reichling, J.; Wink, M. Synergistic Properties of the Terpenoids Aromadendrene and 1,8-Cineole from the Essential Oil of Eucalyptus globulus against Antibiotic-Susceptible and Antibiotic-Resistant Pathogens. Phytomedicine 2010, 17, 1061–1066.
- Yeo, S.K.; Ali, A.Y.; Hayward, O.A.; Turnham, D.; Jackson, T.; Bowen, I.D.; Clarkson, R. β-Bisabolene, a Sesquiterpene from the Essential Oil Extract of Opoponax (Commiphora guidottii), Exhibits Cytotoxicity in Breast Cancer Cell Lines: Anti-tumour properties of β-bisabolene. Phytother. Res. 2016, 30, 418–425.
- Maurya, A.K.; Singh, M.; Dubey, V.; Srivastava, S.; Luqman, S.; Bawankule, D.U. α-(-)-Bisabolol Reduces pro-Inflammatory Cytokine Production and Ameliorates Skin Inflammation. Curr. Pharm. Biotechnol. 2014, 15, 173–181.
- van Zyl, R.L.; Seatlholo, S.T.; van Vuuren, S.F.; Viljoen, A.M. The Biological Activities of 20 Nature Identical Essential Oil Constituents. J. Essent. Oil Res. 2006, 18, 129–133.
- Seki, T.; Kokuryo, T.; Yokoyama, Y.; Suzuki, H.; Itatsu, K.; Nakagawa, A.; Mizutani, T.; Miyake, T.; Uno, M.; Yamauchi, K.; et al. Antitumor Effects of α-Bisabolol against Pancreatic Cancer. Cancer Sci. 2011, 102, 2199–2205.
- Ho, C.-L.; Liao, P.-C.; Wang, E.I.-C.; Su, Y.-C. Composition and Antifungal Activities of the Leaf Essential Oil of Neolitsea parvigemma from Taiwan. Nat. Prod. Commun. 2011, 6, 1357–1360.
- Mastino, P.M.; Marchetti, M.; Costa, J.; Usai, M. Comparison of Essential Oils from Cistus Species Growing in Sardinia. Nat. Prod. Res. 2017, 31, 299–307.
- Takei, M.; Umeyama, A.; Arihara, S. T-Cadinol and Calamenene Induce Dendritic Cells from Human Monocytes and Drive Th1 Polarization. Eur. J. Pharmacol. 2006, 537, 190–199.
- Dahham, S.; Tabana, Y.; Iqbal, M.; Ahamed, M.; Ezzat, M.; Majid, A.; Majid, A. The Anticancer, Antioxidant and Antimicrobial Properties of the Sesquiterpene β-Caryophyllene from the Essential Oil of Aquilaria crassna. Molecules 2015, 20, 11808–11829.
- Ojha, S.; Javed, H.; Azimullah, S.; Haque, M.E. β-Caryophyllene, a Phytocannabinoid Attenuates Oxidative Stress, Neuroinflammation, Glial Activation, and Salvages Dopaminergic Neurons in a Rat Model of Parkinson Disease. Mol. Cell. Biochem. 2016, 418, 59–70.
- Bahi, A.; Al Mansouri, S.; Al Memari, E.; Al Ameri, M.; Nurulain, S.M.; Ojha, S. β-Caryophyllene, a CB2 Receptor Agonist Produces Multiple Behavioral Changes Relevant to Anxiety and Depression in Mice. Physiol. Behav. 2014, 135, 119–124.
- de Oliveira, C.C.; de Oliveira, C.V.; Grigoletto, J.; Ribeiro, L.R.; Funck, V.R.; Grauncke, A.C.B.; de Souza, T.L.; Souto, N.S.; Furian, A.F.; Menezes, I.R.A.; et al. Anticonvulsant Activity of β-Caryophyllene against Pentylenetetrazol-Induced Seizures. Epilepsy Behav. 2016, 56, 26–31.
- Fidyt, K.; Fiedorowicz, A.; Strządała, L.; Szumny, A. β-Caryophyllene and β-Caryophyllene Oxide-Natural Compounds of Anticancer and Analgesic Properties. Cancer Med. 2016, 5, 3007–3017.
- Türkez, H.; Çelik, K.; Toğar, B. Effects of Copaene, a Tricyclic Sesquiterpene, on Human Lymphocytes Cells in Vitro. Cytotechnology 2014, 66, 597–603.
- Turkez, H.; Togar, B.; Tatar, A. Tricyclic Sesquiterpene Copaene Prevents H2O2-Induced Neurotoxicity. J. Intercult. Ethnopharmacol. 2014, 3, 21.
- Park, S.Y.; Park, S.J.; Park, N.J.; Joo, W.H.; Lee, S.-J.; Choi, Y.-W. α-Iso-Cubebene Exerts Neuroprotective Effects in Amyloid Beta Stimulated Microglia Activation. Neurosci. Lett. 2013, 555, 143–148.
- Lee, S.K.; Kim, S.D.; Lee, H.Y.; Baek, S.-H.; Ko, M.J.; Son, B.G.; Park, S.; Choi, Y.W.; Bae, Y.-S. α-Iso-Cubebene, a Natural Compound Isolated from Schisandra chinensis Fruit, Has Therapeutic Benefit against Polymicrobial Sepsis. Biochem. Biophys. Res. Commun. 2012, 426, 226–231.
- Baek, S.E.; Jang, E.J.; Choi, J.M.; Choi, Y.W.; Kim, C.D. α-Iso-Cubebene Attenuates Neointima Formation by Inhibiting HMGB1-Induced Monocyte to Macrophage Differentiation via Suppressing ROS Production. Int. Immunopharmacol. 2022, 111, 109121.
- Rodrigo, G.; Almanza, G.R.; Cheng, Y.; Peng, J.; Hamann, M.; Duan, R.-D.; Åkesson, B. Antiproliferative Effects of Curcuphenol, a Sesquiterpene Phenol. Fitoterapia 2010, 81, 762–766.
- Bai, Z.; Yao, C.; Zhu, J.; Xie, Y.; Ye, X.-Y.; Bai, R.; Xie, T. Anti-Tumor Drug Discovery Based on Natural Product β-Elemene: Anti-Tumor Mechanisms and Structural Modification. Molecules 2021, 26, 1499.
- Asakura, K.; Matsuo, Y.; Oshima, T.; Kihara, T.; Minagawa, K.; Araki, Y.; Kagawa, K.; Kanemasa, T.; Ninomiya, M. ω-Agatoxin IVA-Sensitive Ca2+ Channel Blocker, α-Eudesmol, Protects against Brain Injury after Focal Ischemia in Rats. Eur. J. Pharmacol. 2000, 394, 57–65.
- Moon, P.-D.; Han, N.-R.; Lee, J.S.; Kim, H.-Y.; Hong, S.; Kim, H.-J.; Yoo, M.-S.; Kim, H.-M.; Jeong, H.-J. β-Eudesmol Inhibits Thymic Stromal Lymphopoietin through Blockade of Caspase-1/NF-ΚB Signal Cascade in Allergic Rhinitis Murine Model. Chem. Biol. Interact. 2018, 294, 101–106.
- Narahara, C.; Saeheng, T.; Chaijaroenkul, W.; Dumre, S.P.; Na-Bangchang, K.; Karbwang, J. β-Eudesmol Induces the Expression of Apoptosis Pathway Proteins in Cholangiocarcinoma Cell Lines. J. Res. Med. Sci. Off. J. Isfahan Univ. Med. Sci. 2020, 25, 7.
- Dhyani, P.; Sati, P.; Sharma, E.; Attri, D.C.; Bahukhandi, A.; Tynybekov, B.; Szopa, A.; Sharifi-Rad, J.; Calina, D.; Suleria, H.A.R.; et al. Sesquiterpenoid Lactones as Potential Anti-Cancer Agents: An Update on Molecular Mechanisms and Recent Studies. Cancer Cell Int. 2022, 22, 305.
- Guedes, J.B.; do Nascimento, A.L.; Costa, W.K.; de Veras, B.O.; de Aguiar, J.C.R.d.O.F.; Navarro, D.M.d.A.F.; Napoleão, T.H.; da Silva, M.V.; de Oliveira, A.M.; Correia, M.T.d.S. Eugenia gracillima Essential Oil Has Pharmaceutical Applications in Pain and Inflammation without Toxic Effects in Mice. J. Ethnopharmacol. 2023, 303, 115941.
- Casiglia, S.; Bruno, M.; Bramucci, M.; Quassinti, L.; Lupidi, G.; Fiorini, D.; Maggi, F. Kundmannia sicula (L.) DC: A Rich Source of Germacrene D. J. Essent. Oil Res. 2017, 29, 437–442.
- Mendes De Lacerda Leite, G.; De Oliveira Barbosa, M.; Pereira Lopes, M.J.; De Araújo Delmondes, G.; Bezerra, D.S.; Araújo, I.M.; Carvalho De Alencar, C.D.; Melo Coutinho, H.D.; Peixoto, L.R.; Barbosa-Filho, J.M.; et al. Pharmacological and Toxicological Activities of α-Humulene and Its Isomers: A Systematic Review. Trends Food Sci. Technol. 2021, 115, 255–274.
- Sabir, U.; Irfan, H.M.; Alamgeer Ullah, A.; Althobaiti, Y.S.; Alshehri, F.S.; Niazi, Z.R. Downregulation of Hepatic Fat Accumulation, Inflammation and Fibrosis by Nerolidol in Purpose Built Western-Diet-Induced Multiple-Hit Pathogenesis of NASH Animal Model. Biomed. Pharmacother. 2022, 150, 112956.
- Dong, J.-R.; Chang, W.-W.; Chen, S.-M. Nerolidol Inhibits Proliferation of Leiomyoma Cells via Reactive Oxygen Species-Induced DNA Damage and Downregulation of the ATM/Akt Pathway. Phytochemistry 2021, 191, 112901.
- Misra, B.B.; Dey, S. Evaluation of in Vivo Anti-Hyperglycemic and Antioxidant Potentials of α-Santalol and Sandalwood Oil. Phytomedicine 2013, 20, 409–416.
- Garlet, Q.I.; Souza, C.F.; Rodrigues, P.; Descovi, S.N.; Martinez-Rodríguez, G.; Baldisserotto, B.; Heinzmann, B.M. GABAa Receptor Subunits Expression in Silver Catfish (Rhamdia Quelen) Brain and Its Modulation by Nectandra grandiflora Nees Essential Oil and Isolated Compounds. Behav. Brain Res. 2019, 376, 112178.
- Tyagi, A.K.; Prasad, S.; Yuan, W.; Li, S.; Aggarwal, B.B. Identification of a Novel Compound (β-Sesquiphellandrene) from Turmeric (Curcuma longa) with Anticancer Potential: Comparison with Curcumin. Investig. New Drugs 2015, 33, 1175–1186.
- Zhao, J.; Zhang, J.; Yang, B.; Lv, G.-P.; Li, S.-P. Free Radical Scavenging Activity and Characterization of Sesquiterpenoids in Four Species of Curcuma Using a TLC Bioautography Assay and GC-MS Analysis. Molecules 2010, 15, 7547–7557.
- Martins, R.M.G.; Xavier-Júnior, F.H.; Barros, M.R.; Menezes, T.M.; de Assis, C.R.D.; de Melo, A.C.G.R.; Veras, B.O.; Ferraz, V.P.; Filho, A.A.M.; Yogui, G.T.; et al. Impact on Cholinesterase-Inhibition and in Silico Investigations of Sesquiterpenoids from Amazonian Siparuna guianensis Aubl. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2021, 252, 119511.
- Manjima, R.B.; Ramya, S.; Kavithaa, K.; Paulpandi, M.; Saranya, T.; Harysh Winster, S.B.; Balachandar, V.; Arul, N. Spathulenol Attenuates 6-Hydroxydopamine Induced Neurotoxicity in SH-SY5Y Neuroblastoma Cells. Gene Rep. 2021, 25, 101396.
- do Nascimento, K.F.; Moreira, F.M.F.; Alencar Santos, J.; Kassuya, C.A.L.; Croda, J.H.R.; Cardoso, C.A.L.; Vieira, M.d.C.; Góis Ruiz, A.L.T.; Ann Foglio, M.; de Carvalho, J.E.; et al. Antioxidant, Anti-Inflammatory, Antiproliferative and Antimycobacterial Activities of the Essential Oil of Psidium guineense Sw. and Spathulenol. J. Ethnopharmacol. 2018, 210, 351–358.
- de Matos Balsalobre, N.; dos Santos, E.; Mariano dos Santos, S.; Arena, A.C.; Konkiewitz, E.C.; Ziff, E.B.; Nazari Formagio, A.S.; Leite Kassuya, C.A. Potential Anti-Arthritic and Analgesic Properties of Essential Oil and Viridiflorol Obtained from Allophylus edulis Leaves in Mice. J. Ethnopharmacol. 2023, 301, 115785.
- Akiel, M.A.; Alshehri, O.Y.; Aljihani, S.A.; Almuaysib, A.; Bader, A.; Al-Asmari, A.I.; Alamri, H.S.; Alrfaei, B.M.; Halwani, M.A. Viridiflorol Induces Anti-Neoplastic Effects on Breast, Lung, and Brain Cancer Cells through Apoptosis. Saudi J. Biol. Sci. 2022, 29, 816–821.
- Trevizan, L.N.F.; Nascimento, K.F.d.; Santos, J.A.; Kassuya, C.A.L.; Cardoso, C.A.L.; Vieira, M.d.C.; Moreira, F.M.F.; Croda, J.; Formagio, A.S.N. Anti-Inflammatory, Antioxidant and Anti-Mycobacterium tuberculosis Activity of Viridiflorol: The Major Constituent of Allophylus edulis (A. St.-Hil., A. Juss. & Cambess.) Radlk. J. Ethnopharmacol. 2016, 192, 510–515.
- Oon, S.F.; Nallappan, M.; Tee, T.T.; Shohaimi, S.; Kassim, N.K.; Sa’ariwijaya, M.S.F.; Cheah, Y.H. Xanthorrhizol: A Review of Its Pharmacological Activities and Anticancer Properties. Cancer Cell Int. 2015, 15, 100.
- Mordukhova, E.A.; Kim, D.; Kim, W.-G.; Hwang, J.-K.; Pan, J.-G. The Food-Grade Antimicrobial Xanthorrhizol Targets the Enoyl-ACP Reductase (FabI) in Escherichia coli. Bioorg. Med. Chem. Lett. 2020, 30, 127651.
- Oon, S.F.; Nallappan, M.; Kassim, N.K.; Shohaimi, S.; Sa’ariwijaya, M.S.F.; Tee, T.T.; Cheah, Y.H. Hypolipidemic Activities of Xanthorrhizol Purified from Centrifugal TLC. Biochem. Biophys. Res. Commun. 2016, 478, 1403–1408.
- Cho, J.Y.; Hwang, J.-K.; Chun, H.S. Xanthorrhizol Attenuates Dextran Sulfate Sodium-Induced Colitis via the Modulation of the Expression of Inflammatory Genes in Mice. Life Sci. 2011, 88, 864–870.
- Borgonetti, V.; Governa, P.; Manetti, F.; Galeotti, N. Zingiberene, a Non-Zinc-Binding Class I HDAC Inhibitor: A Novel Strategy for the Management of Neuropathic Pain. Phytomedicine 2023, 111, 154670.
- Bou, D.; Lago, J.; Figueiredo, C.; Matsuo, A.; Guadagnin, R.; Soares, M.; Sartorelli, P. Chemical Composition and Cytotoxicity Evaluation of Essential Oil from Leaves of Casearia sylvestris, Its Main Compound α-Zingiberene and Derivatives. Molecules 2013, 18, 9477–9487.
- Ferreira, B.A.; Silva, R.F.; de Moura, F.B.R.; Narduchi, C.T.; Deconte, S.R.; Sartorelli, P.; Tomiosso, T.C.; Lago, J.H.G.; Araújo, F. de A. α-Zingiberene, a Sesquiterpene from Essential Oil from Leaves of Casearia sylvestris, Suppresses Inflammatory Angiogenesis and Stimulates Collagen Deposition in Subcutaneous Implants in Mice. Nat. Prod. Res. 2022, 36, 5858–5862.
- Ravera, S.; Esposito, A.; Degan, P.; Caicci, F.; Manni, L.; Liguori, A.; Bisio, A.; Iobbi, V.; Schito, A.; Traverso, C.E.; et al. The diterpene manool extracted from Salvia tingitana lowers free radical production in retinal rod outer segments by inhibiting the extramitochondrial F1 F0 ATP synthase. Cell Biochem. Funct. 2021, 39, 528–535.
- Nicolella, H.D.; de Oliveira, P.F.; Munari, C.C.; Costa, G.F.D.; Moreira, M.R.; Veneziani, R.C.S.; Tavares, D.C. Differential Effect of Manool—A Diterpene from Salvia officinalis, on Genotoxicity Induced by Methyl Methanesulfonate in V79 and HepG2 Cells. Food Chem. Toxicol. 2014, 72, 8–12.
- Monteiro, A.S.e.N.; Campos, D.R.; Albuquerque, A.A.S.; Evora, P.R.B.; Ferreira, L.G.; Celotto, A.C. Efeito Do Diterpeno Manool Sobre a Pressão Arterial e Reatividade Vascular Em Ratos Normotensos e Hipertensos. Arq. Bras. Cardiol. 2020, 115, 669–677.
- Castro, C.H.; Pontes, C.N.R. Efeitos Cardiovasculares Do Diterpeno Manool Em Ratos Normotensos e Hipertensos. Arq. Bras. Cardiol. 2020, 115, 678–679.
- Angelopoulou, D.; Demetzos, C.; Dimas, C.; Perdetzoglou, D.; Loukis, A. Essential Oils and Hexane Extracts from Leaves and Fruits of Cistus monspeliensis. Cytotoxic Activity of Ent-13-Epi-Manoyl Oxide and Its Isomers. Planta Med. 2001, 67, 168–171.
- Kim, J.-H.; Na, H.-J.; Kim, C.-K.; Kim, J.-Y.; Ha, K.-S.; Lee, H.; Chung, H.-T.; Kwon, H.J.; Kwon, Y.-G.; Kim, Y.-M. The Non-Provitamin A Carotenoid, Lutein, Inhibits NF-KappaB-Dependent Gene Expression through Redox-Based Regulation of the Phosphatidylinositol 3-Kinase/PTEN/Akt and NF-KappaB-Inducing Kinase Pathways: Role of H(2)O(2) in NF-KappaB Activation. Free Radic. Biol. Med. 2008, 45, 885–896.
- Zhang, W.-L.; Zhao, Y.-N.; Shi, Z.-Z.; Cong, D.; Bai, Y.-S. Lutein Inhibits Cell Growth and Activates Apoptosis via the PI3K/AKT/MTOR Signaling Pathway in A549 Human Non-Small-Cell Lung Cancer Cells. J. Environ. Pathol. Toxicol. Oncol. Off. 2018, 37, 341–350.
- Li, S.; Ding, Y.; Niu, Q.; Xu, S.; Pang, L.; Ma, R.; Jing, M.; Feng, G.; Tang, J.X.; Zhang, Q.; et al. Lutein Has a Protective Effect on Hepatotoxicity Induced by Arsenic via Nrf2 Signaling. BioMed Res. Int. 2015, 2015, 315205.
- Ouyang, B.; Li, Z.; Ji, X.; Huang, J.; Zhang, H.; Jiang, C. The Protective Role of Lutein on Isoproterenol-Induced Cardiac Failure Rat Model through Improving Cardiac Morphology, Antioxidant Status via Positively Regulating Nrf2/HO-1 Signalling Pathway. Pharm. Biol. 2019, 57, 529–535.
- Okada, T.; Nakai, M.; Maeda, H.; Hosokawa, M.; Sashima, T.; Miyashita, K. Suppressive Effect of Neoxanthin on the Differentiation of 3T3-L1 Adipose Cells. J. Oleo Sci. 2008, 57, 345–351.
- Teuscher, E. Biogene Arzneimittel: Lehrbuch der Pharmazeutischen Biologie; 2020; ISBN 978-3-8047-3607-8. Available online: https://www.amazon.de/Biogene-Arzneimittel-Lehrbuch-Pharmazeutischen-Biologie/dp/3804736076 (accessed on 10 August 2023).
- Tomás-Menor, L.; Morales-Soto, A.; Barrajón-Catalán, E.; Roldán-Segura, C.; Segura-Carretero, A.; Micol, V. Correlation between the Antibacterial Activity and the Composition of Extracts Derived from Various Spanish Cistus Species. Food Chem. Toxicol. 2013, 55, 313–322.
- Sticher, O.; Heilmann, J.; Zündorf, I.; Hänsel, R.; Steinegger, E. Pharmakognosie—Phytopharmazie; 10., völlig neu Bearbeitete Auflage.; Wissenschaftliche Verlagsgesellschaft: Stuttgart, Germany, 2015; ISBN 978-3-8047-3144-8.
- Belitz, H.-D.; Grosch, W.; Schieberle, P. Lehrbuch der Lebensmittelchemie; Springer-Lehrbuch; Sechste, Vollständig Überarbeitete Auflage; Springer: Berlin/Heidelberg, Germany, 2008; ISBN 978-3-540-73201-3.
- Mastino, P.; Marchetti, M.A.; Costa, J.; Juliano, C.; Usai, M. Analytical Profiling of Phenolic Compounds in Extracts of Three Cistus Species from Sardinia and Their Potential Antimicrobial and Antioxidant Activity. Chem. Biodivers. 2021, 18, e2100053.
- Qa’dan, F.; Petereit, F.; Nahrstedt, A. Prodelphinidin Trimers and Characterization of a Proanthocyanidin Oligomer from Cistus albidus. Pharmazie 2003, 58, 416–419.
- Wiesner, J.V. Die Rohstoffe des Pflanzenreiches; Verlag W. Engelmann: Leipzig, Berlin, Germany, 1921.
- Gonçalves, S.; Gomes, D.; Costa, P.; Romano, A. The Phenolic Content and Antioxidant Activity of Infusions from Mediterranean Medicinal Plants. Ind. Crops Prod. 2013, 43, 465–471.
- Legrum, W. Riechstoffe, Zwischen Gestank und Duft: Vorkommen, Eigenschaften und Anwendung von Riechstoffen und deren Gemischen; Studienbücher Chemie; 1. Aufl.; Vieweg + Teubner: Wiesbaden, Germany, 2011; ISBN 978-3-8348-1245-2.
- Fahlbusch, K.-G.; Hammerschmidt, F.-J.; Panten, J.; Pickenhagen, W.; Schatkowski, D.; Bauer, K.; Garbe, D.; Surburg, H. Flavors and Fragrances. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH Verlag GmbH & Co. KGaA, Ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2003; p. a11_141. ISBN 978-3-527-30673-2.
- Fenaroli, G.; Burdock, G.A. Fenaroli’s Handbook of Flavor Ingredients, 4th ed.; CRC Press: Boca Raton, FL, USA, 2002; ISBN 978-0-8493-0946-5.
- Oñate, M.; Munné-Bosch, S. Loss of Flower Bud Vigour in the Mediterranean Shrub, Cistus albidus L. at Advanced Developmental Stages. Plant Biol. 2010, 12, 475–483.
- Jubany-Mari, T.; Munne-Bosch, S.; Lopez-Carbonell, M.; Alegre, L. Hydrogen Peroxide Is Involved in the Acclimation of the Mediterranean Shrub, Cistus albidus L., to Summer Drought. J. Exp. Bot. 2008, 60, 107–120.
- Mulet Pascual, L. Estudio Etnobotánico de La Provincia de Castellón; Diputación de Castellón: Castellón, Spain, 1991; ISBN 978-84-86895-24-2.
- Wang, H. Epimerisation of Catechins in Green Tea Infusions. Food Chem. 2000, 70, 337–344.
- Wang, R.; Zhou, W.; Jiang, X. Reaction Kinetics of Degradation and Epimerization of Epigallocatechin Gallate (EGCG) in Aqueous System over a Wide Temperature Range. J. Agric. Food Chem. 2008, 56, 2694–2701.
- Saklar, S.; Ertas, E.; Ozdemir, I.S.; Karadeniz, B. Effects of Different Brewing Conditions on Catechin Content and Sensory Acceptance in Turkish Green Tea Infusions. J. Food Sci. Technol. 2015, 52, 6639–6646.
- Verde, A.; Fajardo, J.; Obón, C.; Cebrían, F.; Rivera, D. Guía de las Plantas Medicinales de Castilla-La Mancha; Altabán Ediciones: Albacete, Spain, 2008; ISBN 987-84-96465-53-4.
- Alami Merrouni, I.; Kharchoufa, L.; Bencheikh, N.; Elachouri, M. Ethnobotanical Profile of Medicinal Plants Used by People of North-Eastern Morocco: Cross-Cultural and Historical Approach (Part I). Ethnobot. Res. Appl. 2021, 21, 1–45.
- Las Plantas En La Cultura Popular de La Provincia de Albacete; Fajardo, J.; Verde, A.; Rivera, D.; Obón, C. (Eds.) Serie I—Estudios; Instituto de Estudios Albacetenses “Don Juan Manuel” de la Excma; Diputacioń de Albacete: Albacete, Spain, 2000; ISBN 978-84-95394-08-8.
- Ajjoun, M.; Kharchoufa, L.; Alami Merrouni, I.; Elachouri, M. Moroccan Medicinal Plants Traditionally Used for the Treatment of Skin Diseases: From Ethnobotany to Clinical Trials. J. Ethnopharmacol. 2022, 297, 115532.
- Fernández Ocaña, A.M. Estudio Etnobotánico En El Parque Natural de Las Sierras de Cazorla, Segura y Las Villas; Investigación Química de Un Grupo de Especies Interesantes, Departamento de Biología Animal, Vegetal y Ecología, Facultad de Ciencias Experimentales, Universidad de Jaén: Jáen, Spain, 2000.
- Fiorini, D.; Molle, A.; Nabissi, M.; Santini, G.; Benelli, G.; Maggi, F. Valorizing Industrial Hemp (Cannabis sativa L.) by-Products: Cannabidiol Enrichment in the Inflorescence Essential Oil Optimizing Sample Pre-Treatment Prior to Distillation. Ind. Crops Prod. 2019, 128, 581–589.
- Ross, S.A.; ElSohly, M.A. The Volatile Oil Composition of Fresh and Air-Dried Buds of Cannabis sativa. J. Nat. Prod. 1996, 59, 49–51.
- Alcaraz Ariza, F.J.; Le Houérou, H.-N. Flora Básica de la Región de Murcia; Sociedad Cooperativa de Enseñanza Severo Ochoa: Murcia, Spain, 2002; ISBN 978-84-600-9378-7.
- Stübing, G.; Peris, J.B. Plantas Medicinales de la Comunidad Valenciana; Generalitat Valenciana, Conselleria de Medio Ambiete: Valencia, Spain, 1998; ISBN 978-84-482-1805-8.
- Segarra i Durà, E. Etnobotànica Farmacèutica de Gàtova; Universitat de València: València, Spain, 2008; ISBN 978-84-370-6976-0.
- Pardo de Santayana, M. Inventario Español de los Conocimientos Tradicionales Relativos de la Biodiversidad: Primera fase: Introducción, Metodología y Fichas; Ministerio para la Transición Ecológica: Madrid, Spain, 2014; ISBN 978-84-491-1401-4.
- Ledesma, J. Estudio Del Uso Tradicional de Las Plantas Silvestres En La Sierra de Montsant; Trabajo Fin de Carrera, Escuela de Ingeniería Forestal de Lleida: Lleida, Spain, 2004.
- Carrió Cabrer, M.E. Contribució a l’etnobotànica de Mallorca; La Biodiversitat Vegetal i La Seva Gestió En Una Illa Mediterrània, Universidad de Barcelona: Barcelona, Spain, 2013.
- Carrió, E.; Vallès, J. Ethnobotany of Medicinal Plants Used in Eastern Mallorca (Balearic Islands, Mediterranean Sea). J. Ethnopharmacol. 2012, 141, 1021–1040.
- Belda, A.; Zaragozí, B.; Belda, I.; Martínez, J.; Seva, E. Traditional Knowledge of Medicinal Plants in the Serra de Mariola Natural Park, South-Eastern Spain. Afr. J. Tradit. Complement. Altern. Med. 2013, 10, 299–309.
- Martínez-Lirola, M.J.; González-Tejero, M.R.; Molero-Mesa, J. Ethnobotanical Resources in the Province of Almeria, Spain: Campos de Nijar. Econ. Bot. 1996, 50, 40–56.
- Torres-Montes, F. Nombres y Usos Tradicionales de Las Plantas Silvestres En Almería (Estudio Linguistico y Etnografico); Diputación de Almería e Instituto de Estudios Almerienses: Almería, Spain, 2004; ISBN 978-84-8108-313-2.
- Alarcόn, R.; Pardo-de-Santayana, M.; Priestley, C.; Morales, R.; Heinrich, M. Medicinal and Local Food Plants in the South of Alava (Basque Country, Spain). J. Ethnopharmacol. 2015, 176, 207–224.
- Ortuño Moya, I.M. Etnobotánica de Los Villares y Valdepeñas de Jaén (Sur de La Península Ibérica); Universidad de Jaén—Departamento de Biología Animal, Vegetal y Ecología, Facultad de Ciencias Experimentales: Jaén, Spain, 2004.
- Goicoetxea Marcaida, A. Etnobotánica y Medicina Popular en el País Vasco; Pastor: Madrid, Spain, 2017; ISBN 978-84-95461-61-2.
- González-Tejero García, M.R. Investigaciones Etnobotánicas En La Provincia de Granada; Departamento de Biología Vegetal, Facultad de Farmacia, Universidad de Granada: Granada, Spain, 1989.
- Rivera, D.; Alcaraz, F.; Verde, A.; Fajardo, J.; Obón, C. Las Plantas en la Cultura Popular; SOMEHN (Sociedad Mediterránea de Historia Natural): Murcia, Spain, 2008; ISBN 978-84-931786-0-4.
- Guzmán, A. Aproximación a La Etnobotánica de La Provincia de Jaén; Departamento de Biología Vegetal, Universidad de Granada: Granada, Spain, 1997.
- Atzei, A.D. Le Piante Nella Tradizione Popolare Della Sardegna: Documentazione Sugli Usi Alimentari, Aromatizzanti, Profumieri, Artigianali, Cosmetici, Medicinali, Veterinari, Magici, Ornamentali, Rituali, Religiosi, Tintori, Antiparassitari e Vari, Delle Piante; C. Delfino: Sassari, Italy, 2003; ISBN 978-88-7138-298-2.
- Rivera, D.; Robleda, A.; Obón, C.; Cano, F. Introducción al Mundo de Las Plantas Medicinales En Murcia, 1st ed.; Ayuntamiento de Murcia: Murcia, Spain, 1994; ISBN 978-84-606-1922-2.
- Pellicer, J. Costumari Botànic: Recerques Etnobotàniques a Les Comarques Centrals Valencianes; Farga monogràfica, 2nd ed.; Edicions del Bullent: Picanya, Spain, 2000; ISBN 978-84-96187-13-9.
