On 30 January 2020, WHO declared COVID-19 a public health emergency of global concern. COVID-19 became pandemic on 11 March 2020, and spread unprecedently. No country was prepared to face its impact. Major fears started to be expressed for Africa, where dramatic consequences were expected, due to the weakness of health systems. The limited resources, in terms of qualified physicians, medical equipment and hospital infrastructures marked an undeniable inequality and highlighted the need to empower local capacity, raising the level of preparedness of African nations against infection outbreaks.
- SARS-CoV-2
- COVID-19
- infections
- Africa
1. COVID-19 in Africa
2. Genetic Variants of SARS-CoV-2 Reported in Africa
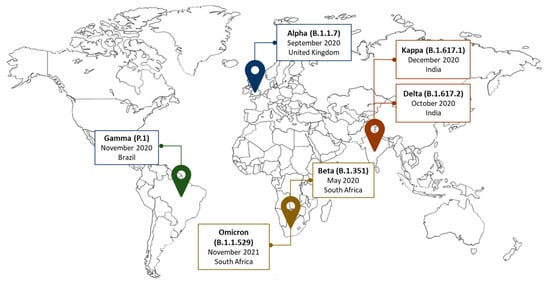
3. The Transmissibility of SARS-CoV-2 and the Difficulty of Applying Standardized Protection Measures in Africa
4. The Comorbidities Underlying COVID-19 Severity in Africa
5. Vaccine Inequity Distribution in Africa
This entry is adapted from the peer-reviewed paper 10.3390/covid4020014
References
- United Nations World Population Prospects 2019: Highlights. Available online: https://population.un.org/wpp/Publications/Files/wpp2019_10KeyFindings.pdf (accessed on 9 April 2023).
- The Fund for Peace Global Data. Available online: https://fragilestatesindex.org/global-data/ (accessed on 19 March 2023).
- Bwire, G.; Ario, A.R.; Eyu, P.; Ocom, F.; Wamala, J.F.; Kusi, K.A.; Ndeketa, L.; Jambo, K.C.; Wanyenze, R.K.; Talisuna, A.O. The COVID-19 Pandemic in the African Continent. BMC Med. 2022, 20, 167.
- Udoakang, A.; Oboh, M.; Henry-Ajala, A.; Anyigba, C.; Omoleke, S.; Amambua-Ngwa, A.; Paemka, L.; Awandare, G.; Quashie, P. Low COVID-19 Impact in Africa: The Multifactorial Nexus. Open Res. Afr. 2021, 4, 47.
- Cilliers, J.; Kwasi, S.; Yeboua, K.; Oosthuizen, M.; Alexander, K.; Pooe, T.; Moyer, J. Impact of COVID-19 in Africa: A Scenario Analysis to 2030. SSRN Electron. J. 2020.
- Dong, E.; Du, H.; Gardner, L. An Interactive Web-Based Dashboard to Track COVID-19 in Real Time. Lancet Infect. Dis. 2020, 20, 533–534.
- El-Sadr, W.M.; Justman, J. Africa in the Path of COVID-19. N. Engl. J. Med. 2020, 383, e11.
- Mutombo, P.N.; Fallah, M.P.; Munodawafa, D.; Kabel, A.; Houeto, D.; Goronga, T.; Mweemba, O.; Balance, G.; Onya, H.; Kamba, R.S.; et al. COVID-19 Vaccine Hesitancy in Africa: A Call to Action. Lancet Glob. Health 2022, 10, e320–e321.
- Hussein, M.I.H.; Albashir, A.A.D.; Elawad, O.A.M.A.; Homeida, A. Malaria and COVID-19: Unmasking Their Ties. Malar. J. 2020, 19, 457.
- Yazdanbakhsh, M.; Kremsner, P.G.; Van Ree, R. Immunology: Allergy, Parasites, and the Hygiene Hypothesis. Science 2002, 296, 490–494.
- Arts, R.J.W.; Moorlag, S.J.C.F.M.; Novakovic, B.; Li, Y.; Wang, S.Y.; Oosting, M.; Kumar, V.; Xavier, R.J.; Wijmenga, C.; Joosten, L.A.B.; et al. BCG Vaccination Protects against Experimental Viral Infection in Humans through the Induction of Cytokines Associated with Trained Immunity. Cell Host Microbe 2018, 23, 89–100.e5.
- Rolot, M.; Dougall, A.M.; Chetty, A.; Javaux, J.; Chen, T.; Xiao, X.; Machiels, B.; Selkirk, M.E.; Maizels, R.M.; Hokke, C.; et al. Helminth-Induced IL-4 Expands Bystander Memory CD8+ T Cells for Early Control of Viral Infection. Nat. Commun. 2018, 9, 4516.
- Boum, Y.; Bebell, L.M.; Bisseck, A.-C.Z.-K. Africa Needs Local Solutions to Face the COVID-19 Pandemic. Lancet 2021, 397, 1238–1240.
- Nguimkeu, P.; Tadadjeu, S. Why Is the Number of COVID-19 Cases Lower than Expected in Sub-Saharan Africa? A Cross-Sectional Analysis of the Role of Demographic and Geographic Factors. World Dev. 2021, 138, 105251.
- Mwiinde, A.M.; Siankwilimba, E.; Sakala, M.; Banda, F.; Michelo, C. Climatic and Environmental Factors Influencing COVID-19 Transmission—An African Perspective. Trop. Med. Infect. Dis. 2022, 7, 433.
- Chan, K.H.; Peiris, J.S.M.; Lam, S.Y.; Poon, L.L.M.; Yuen, K.Y.; Seto, W.H. The Effects of Temperature and Relative Humidity on the Viability of the SARS Coronavirus. Adv. Virol. 2011, 2011, 734690.
- Sharif, N.; Sarkar, M.K.; Ahmed, S.N.; Ferdous, R.N.; Nobel, N.U.; Parvez, A.K.; Talukder, A.A.; Dey, S.K. Environmental Correlation and Epidemiologic Analysis of COVID-19 Pandemic in Ten Regions in Five Continents. Heliyon 2021, 7, e06576.
- Whittemore, P.B. COVID-19 Fatalities, Latitude, Sunlight, and Vitamin D. Am. J. Infect. Control 2020, 48, 1042–1044.
- Shao, L.; Ge, S.; Jones, T.; Santosh, M.; Silva, L.F.O.; Cao, Y.; Oliveira, M.L.S.; Zhang, M.; BéruBé, K. The Role of Airborne Particles and Environmental Considerations in the Transmission of SARS-CoV-2. Geosci. Front. 2021, 12, 101189.
- Adekunle, I.A.; Tella, S.A.; Oyesiku, K.O.; Oseni, I.O. Spatio-Temporal Analysis of Meteorological Factors in Abating the Spread of COVID-19 in Africa. Heliyon 2020, 6, e04749.
- Salyer, S.J.; Maeda, J.; Sembuche, S.; Kebede, Y.; Tshangela, A.; Moussif, M.; Ihekweazu, C.; Mayet, N.; Abate, E.; Ouma, A.O.; et al. The First and Second Waves of the COVID-19 Pandemic in Africa: A Cross-Sectional Study. Lancet 2021, 397, 1265–1275.
- World Health Organization (WHO). WHO Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int/ (accessed on 3 November 2021).
- Diop, B.Z.; Ngom, M.; Pougué Biyong, C.; Pougué Biyong, J.N. The Relatively Young and Rural Population May Limit the Spread and Severity of COVID-19 in Africa: A Modelling Study. BMJ Glob. Health 2020, 5, e002699.
- World Data International Tourism: The Most Popular Countries. Available online: https://www.worlddata.info/tourism.php (accessed on 9 April 2023).
- Bradshaw, D.; Dorrington, R.; Moultrie, T.; Groenewald, P.; Moultrie, H. Underestimated COVID-19 Mortality in WHO African Region. Lancet Glob. Health 2022, 10, e1559.
- Chitungo, I.; Dzobo, M.; Hlongwa, M.; Dzinamarira, T. COVID-19: Unpacking the Low Number of Cases in Africa. Public Health Pract. 2020, 1, 100038.
- Lucero-Prisno, D.E.; Adebisi, Y.A.; Lin, X. Current Efforts and Challenges Facing Responses to 2019-NCoV in Africa. Glob. Health Res. Policy 2020, 5, 21.
- Jensen, N.; Kelly, A.H.; Avendano, M. The COVID-19 Pandemic Underscores the Need for an Equity-Focused Global Health Agenda. Humanit. Soc. Sci. Commun. 2021, 8, 15.
- Smolen, K.K.; Cai, B.; Gelinas, L.; Edgardo, S.; Iii, F.; Larsen, M.; Speert, D.P.; Cooper, P.J.; Esser, M.; Marchant, A.; et al. Single-Cell Analysis of Innate Cytokine Responses to Pattern Recognition Receptor Stimulation in Children across Four Continents. J. Immunol. 2014, 193, 3003–3012.
- Mascola, J.R.; Graham, B.S.; Fauci, A.S. SARS-CoV-2 Viral Variants—Tackling a Moving Target. JAMA 2021, 325, 1261–1262.
- Prévost, J.; Finzi, A. The Great Escape? SARS-CoV-2 Variants Evading Neutralizing Responses. Cell Host Microbe 2021, 29, 322–324.
- Tao, K.; Tzou, P.L.; Nouhin, J.; Gupta, R.K.; de Oliveira, T.; Kosakovsky Pond, S.L.; Fera, D.; Shafer, R.W. The Biological and Clinical Significance of Emerging SARS-CoV-2 Variants. Nat. Rev. Genet. 2021, 22, 757–773.
- World Health Organization (WHO) Tracking SARS-CoV-2 Variants. Available online: https://www.who.int/activities/tracking-SARS-CoV-2-variants (accessed on 24 January 2024).
- Tessema, S.K.; Nkengasong, J.N. Understanding COVID-19 in Africa. Nat. Rev. Immunol. 2021, 21, 469–470.
- Viana, R.; Moyo, S.; Amoako, D.G.; Tegally, H.; Scheepers, C.; Althaus, C.L.; Anyaneji, U.J.; Bester, P.A.; Boni, M.F.; Chand, M.; et al. Rapid Epidemic Expansion of the SARS-CoV-2 Omicron Variant in Southern Africa. Nature 2022, 603, 679–686.
- Gao, S.-J.; Guo, H.; Luo, G. Omicron Variant (B.1.1.529) of SARS-CoV-2, a Global Urgent Public Health Alert! J. Med. Virol. 2022, 94, 1255–1256.
- Chang, D.; Chang, X.; He, Y.; Tan, K.J.K. The Determinants of COVID-19 Morbidity and Mortality across Countries. Sci. Rep. 2022, 12, 5888.
- Ma, Q.; Liu, J.; Liu, Q.; Kang, L.; Liu, R.; Jing, W.; Wu, Y.; Liu, M. Global Percentage of Asymptomatic SARS-CoV-2 Infections Among the Tested Population and Individuals with Confirmed COVID-19 Diagnosis: A Systematic Review and Meta-Analysis. JAMA Netw. Open 2021, 4, e2137257.
- Christie, B. COVID-19: Early Studies Give Hope Omicron Is Milder than Other Variants. BMJ 2021, 375, n3144.
- World Health Organization (WHO). TAG-VE Statement on the Meeting of 3 January on the COVID-19 Situation in China. Available online: https://www.who.int/news/item/04-01-2023-tag-ve-statement-on-the-3rd-january-meeting-on-the-covid-19-situation-in-china (accessed on 9 April 2023).
- Cao, Y.; Jian, F.; Wang, J.; Yu, Y.; Song, W.; Yisimayi, A.; Wang, J.; An, R.; Chen, X.; Zhang, N.; et al. Imprinted SARS-CoV-2 Humoral Immunity Induces Convergent Omicron RBD Evolution. Nature 2023, 614, 521–529.
- Sun, Y.; Wang, M.; Lin, W.; Dong, W.; Xu, J. Evolutionary Analysis of Omicron Variant BF.7 and BA.5.2 Pandemic in China. J. Biosaf. Biosecur. 2023, 5, 14–20.
- Bai, Y.; Yao, L.; Wei, T.; Tian, F.; Jin, D.-Y.; Chen, L.; Wang, M. Presumed Asymptomatic Carrier Transmission of COVID-19. JAMA 2020, 323, 1406–1407.
- Lauer, S.A.; Grantz, K.H.; Bi, Q.; Jones, F.K.; Zheng, Q.; Meredith, H.R.; Azman, A.S.; Reich, N.G.; Lessler, J. The Incubation Period of Coronavirus Disease 2019 (COVID-19) From Publicly Reported Confirmed Cases: Estimation and Application. Ann. Intern. Med. 2020, 172, 577–582.
- Azkur, A.K.; Akdis, M.; Azkur, D.; Sokolowska, M.; van de Veen, W.; Brüggen, M.-C.; O’Mahony, L.; Gao, Y.; Nadeau, K.; Akdis, C.A. Immune Response to SARS-CoV-2 and Mechanisms of Immunopathological Changes in COVID-19. Allergy 2020, 75, 1564–1581.
- Gandhi, R.T.; Lynch, J.B.; del Rio, C. Mild or Moderate COVID-19. N. Engl. J. Med. 2020, 383, 1757–1766.
- Wang, D.; Hu, B.; Hu, C.; Zhu, F.; Liu, X.; Zhang, J.; Wang, B.; Xiang, H.; Cheng, Z.; Xiong, Y.; et al. Clinical Characteristics of 138 Hospitalized Patients with 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA-J. Am. Med. Assoc. 2020, 323, 1061–1069.
- Liu, J.; Liao, X.; Qian, S.; Yuan, J.; Wang, F.; Liu, Y.; Wang, Z.; Wang, F.-S.; Liu, L.; Zhang, Z. Community Transmission of Severe Acute Respiratory Syndrome Coronavirus 2, Shenzhen, China, 2020. Emerg. Infect. Dis. 2020, 26, 1320–1323.
- Chan, J.F.-W.; Yuan, S.; Kok, K.-H.; To, K.K.-W.; Chu, H.; Yang, J.; Xing, F.; Liu, J.; Yip, C.C.-Y.; Poon, R.W.-S.; et al. A Familial Cluster of Pneumonia Associated with the 2019 Novel Coronavirus Indicating Person-to-Person Transmission: A Study of a Family Cluster. Lancet 2020, 395, 514–523.
- Burke, R.M.; Midgley, C.M.; Dratch, A.; Fenstersheib, M.; Haupt, T.; Holshue, M.; Ghinai, I.; Jarashow, M.C.; Lo, J.; McPherson, T.D.; et al. Active Monitoring of Persons Exposed to Patients with Confirmed COVID-19-United States, January-February 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 245–246.
- van Doremalen, N.; Bushmaker, T.; Morris, D.H.; Holbrook, M.G.; Gamble, A.; Williamson, B.N.; Tamin, A.; Harcourt, J.L.; Thornburg, N.J.; Gerber, S.I.; et al. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. N. Engl. J. Med. 2020, 382, 1564–1567.
- Van Damme, W.; Dahake, R.; van de Pas, R.; Vanham, G.; Assefa, Y. COVID-19: Does the Infectious Inoculum Dose-Response Relationship Contribute to Understanding Heterogeneity in Disease Severity and Transmission Dynamics? Med. Hypotheses 2021, 146, 110431.
- Gralton, J.; Tovey, E.R.; McLaws, M.-L.; Rawlinson, W.D. Respiratory Virus RNA Is Detectable in Airborne and Droplet Particles. J. Med. Virol. 2013, 85, 2151–2159.
- Somsen, G.A.; van Rijn, C.; Kooij, S.; Bem, R.A.; Bonn, D. Small Droplet Aerosols in Poorly Ventilated Spaces and SARS-CoV-2 Transmission. Lancet Respir. Med. 2020, 8, 658–659.
- Stadnytskyi, V.; Bax, C.E.; Bax, A.; Anfinrud, P. The Airborne Lifetime of Small Speech Droplets and Their Potential Importance in SARS-CoV-2 Transmission. Proc. Natl. Acad. Sci. USA 2020, 117, 11875.
- UNICEF. Water Security for All; UNICEF: New York, NY, USA, 2021.
- Guan, W.; Ni, Z.; Hu, Y.; Liang, W.; Ou, C.; He, J.; Liu, L.; Shan, H.; Lei, C.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720.
- Peng, L.; Liu, J.; Xu, W.; Luo, Q.; Chen, D.; Lei, Z.; Huang, Z.; Li, X.; Deng, K.; Lin, B.; et al. SARS-CoV-2 Can Be Detected in Urine, Blood, Anal Swabs, and Oropharyngeal Swabs Specimens. J. Med. Virol. 2020, 92, 1676–1680.
- Wu, Y.; Guo, C.; Tang, L.; Hong, Z.; Zhou, J.; Dong, X.; Yin, H.; Xiao, Q.; Tang, Y.; Qu, X.; et al. Prolonged Presence of SARS-CoV-2 Viral RNA in Faecal Samples. Lancet Gastroenterol. Hepatol. 2020, 5, 434–435.
- Wang, W.; Xu, Y.; Gao, R.; Lu, R.; Han, K.; Wu, G.; Tan, W. Detection of SARS-CoV-2 in Different Types of Clinical Specimens. JAMA 2020, 323, 1843–1844.
- Xiao, F.; Tang, M.; Zheng, X.; Liu, Y.; Li, X.; Shan, H. Evidence for Gastrointestinal Infection of SARS-CoV-2. Gastroenterology 2020, 158, 1831–1833.e3.
- Zheng, S.; Fan, J.; Yu, F.; Feng, B.; Lou, B.; Zou, Q.; Xie, G.; Lin, S.; Wang, R.; Yang, X.; et al. Viral Load Dynamics and Disease Severity in Patients Infected with SARS-CoV-2 in Zhejiang Province, China, January-March 2020: Retrospective Cohort Study. BMJ 2020, 369, m1443.
- UNICEF; World Health Organization (WHO). Progress on Drinking Water, Sanitation and Hygiene 2000–2017; UNICEF: New York, NY, USA, 2019.
- Sun, S.; Han, J. Open Defecation and Squat Toilets, an Overlooked Risk of Fecal Transmission of COVID-19 and Other Pathogens in Developing Communities. Environ. Chem. Lett. 2021, 19, 787–795.
- World Health Organization. Breastfeeding and COVID-19: WHO Scientific Brief; World Health Organization: Geneva, Switzerland, 2020; pp. 1–3.
- Bhattacharjee, N.V.; Schaeffer, L.E.; Marczak, L.B.; Ross, J.M.; Swartz, S.J.; Albright, J.; Gardner, W.M.; Shields, C.; Sligar, A.; Schipp, M.F.; et al. Mapping Exclusive Breastfeeding in Africa between 2000 and 2017. Nat. Med. 2019, 25, 1205–1212.
- Bhattacharjee, N.V.; Schaeffer, L.E.; Hay, S.I.; Lu, D.; Schipp, M.F.; Lazzar-Atwood, A.; Donkers, K.M.; Abady, G.G.; Abd-Allah, F.; Abdelalim, A.; et al. Mapping Inequalities in Exclusive Breastfeeding in Low- and Middle-Income Countries, 2000–2018. Nat. Hum. Behav. 2021, 5, 1027–1045.
- World Health Organization (WHO); UNICEF. Global Nutrition Targets 2025: Breastfeeding Policy Brief; World Health Organization: Geneva, Switzerland, 2014.
- Dong, L.; Tian, J.; He, S.; Zhu, C.; Wang, J.; Liu, C.; Yang, J. Possible Vertical Transmission of SARS-CoV-2 From an Infected Mother to Her Newborn. JAMA 2020, 323, 1846–1848.
- Alabi, Q.K.; Oyedeji, A.S.; Kayode, O.O.; Kajewole-Alabi, D.I. Impact of COVID-19 Pandemic on Mother and Child Health in Sub-Saharan Africa—A Review. Pediatr. Res. 2023, 94, 1278–1283.
- Lawal, Y. Africa’s Low COVID-19 Mortality Rate: A Paradox? Int. J. Infect. Dis. 2021, 102, 118–122.
- Dalal, S.; Beunza, J.J.; Volmink, J.; Adebamowo, C.; Bajunirwe, F.; Njelekela, M.; Mozaffarian, D.; Fawzi, W.; Willett, W.; Adami, H.-O.; et al. Non-Communicable Diseases in Sub-Saharan Africa: What We Know Now. Int. J. Epidemiol. 2011, 40, 885–901.
- World Health Organization (WHO). Deaths from Noncommunicable Diseases on the Rise in Africa. Available online: https://www.afro.who.int/news/deaths-noncommunicable-diseases-rise-africa (accessed on 9 April 2023).
- Ajaero, C.K.; De Wet-Billings, N.; Atama, C.; Agwu, P.; Eze, E.J. The Prevalence and Contextual Correlates of Non-Communicable Diseases among Inter-Provincial Migrants and Non-Migrants in South Africa. BMC Public Health 2021, 21, 999.
- Vos, T.; Lim, S.S.; Abbafati, C.; Abbas, K.M.; Abbasi, M.; Abbasifard, M.; Abbasi-Kangevari, M.; Abbastabar, H.; Abd-Allah, F.; Abdelalim, A.; et al. Global Burden of 369 Diseases and Injuries in 204 Countries and Territories, 1990-2019: A Systematic Analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222.
- Cole, J.B.; Florez, J.C. Genetics of Diabetes Mellitus and Diabetes Complications. Nat. Rev. Nephrol. 2020, 16, 377–390.
- World Health Organization (WHO). African Region Tops World in Undiagnosed Diabetes: WHO Analysis. Available online: https://www.afro.who.int/news/african-region-tops-world-undiagnosed-diabetes-who-analysis (accessed on 28 March 2023).
- Anjorin, A.A.; Abioye, A.I.; Asowata, O.E.; Soipe, A.; Kazeem, M.I.; Adesanya, I.O.; Raji, M.A.; Adesanya, M.; Oke, F.A.; Lawal, F.J.; et al. Comorbidities and the COVID-19 Pandemic Dynamics in Africa. Trop. Med. Int. Health 2021, 26, 2–13.
- Schoen, K.; Horvat, N.; Guerreiro, N.F.C.; de Castro, I.; de Giassi, K.S. Spectrum of Clinical and Radiographic Findings in Patients with Diagnosis of H1N1 and Correlation with Clinical Severity. BMC Infect. Dis. 2019, 19, 964.
- Banik, G.R.; Alqahtani, A.S.; Booy, R.; Rashid, H. Risk Factors for Severity and Mortality in Patients with MERS-CoV: Analysis of Publicly Available Data from Saudi Arabia. Virol. Sin. 2016, 31, 81–84.
- Yang, J.K.; Feng, Y.; Yuan, M.Y.; Yuan, S.Y.; Fu, H.J.; Wu, B.Y.; Sun, G.Z.; Yang, G.R.; Zhang, X.L.; Wang, L.; et al. Plasma Glucose Levels and Diabetes Are Independent Predictors for Mortality and Morbidity in Patients with SARS. Diabet. Med. 2006, 23, 623–628.
- Hussain, A.; Bhowmik, B.; do Vale Moreira, N.C. COVID-19 and Diabetes: Knowledge in Progress. Diabetes Res. Clin. Pract. 2020, 162, 108142.
- Burki, T. COVID-19 and Diabetes in Africa: A Lethal Combination. Lancet Diabetes Endocrinol. 2022, 10, 23.
- Kerolos, M.M.; Ruge, M.; Gill, A.; Planek, M.I.; Volgman, A.S.; Du-Fay-De-Lavallaz, J.M.; Gomez, J.M.D.; Suboc, T.M.; Williams, K.A.; Abusin, S. Clinical Outcomes of COVID-19 Infection in Patients with Pre-Existing Cardiovascular Disease. Am. Heart J. Plus Cardiol. Res. Pract. 2022, 20, 100189.
- Nkomo, V.T. Epidemiology and Prevention of Valvular Heart Diseases and Infective Endocarditis in Africa. Heart 2007, 93, 1510.
- Anabwani, G.M.; Amoa, A.B.; Muita, A.K. Epidemiology of Rheumatic Heart Disease among Primary School Children in Western Kenya. Int. J. Cardiol. 1989, 23, 249–252.
- McLaren, M.J.; Hawkins, D.M.; Koornhof, H.J.; Bloom, K.R.; Bramwell-Jones, D.M.; Cohen, E.; Gale, G.E.; Kanarek, K.; Lachman, A.S.; Lakier, J.B.; et al. Epidemiology of Rheumatic Heart Disease in Black Shcoolchildren of Soweto, Johannesburg. Br. Med. J. 1975, 3, 474.
- Marijon, E.; Ou, P.; Celermajer, D.S.; Ferreira, B.; Mocumbi, A.O.; Jani, D.; Paquet, C.; Jacob, S.; Sidi, D.; Jouven, X. Prevalence of Rheumatic Heart Disease Detected by Echocardiographic Screening. N. Engl. J. Med. 2007, 357, 470–476.
- Ibrahim-Khalil, S.; Elhag, M.; Ali, E.; Mahgoub, F.; Hakiem, S.; Omer, N.; Shafie, S.; Mahgoub, E. An Epidemiological Survey of Rheumatic Fever and Rheumatic Heart Disease in Sahafa Town, Sudan. J. Epidemiol. Community Health 1992, 46, 477.
- Oli, K.; Tekle-Haimanot, R.; Forsgren, L.; Ekstedt, J. Rheumatic Heart Disease Prevalence among Schoolchildren of an Ethiopian Rural Town. Cardiology 1992, 80, 152–155.
- Nyasulu, P.S.; Tamuzi, J.L.; Erasmus, R.T. Burden, Causation, and Particularities of Long-COVID in African Populations: A Rapid Systematic Review. medRxiv 2023.
- Margolin, E.; Burgers, W.A.; Sturrock, E.D.; Mendelson, M.; Chapman, R.; Douglass, N.; Williamson, A.-L.; Rybicki, E.P. Prospects for SARS-CoV-2 Diagnostics, Therapeutics and Vaccines in Africa. Nat. Rev. Microbiol. 2020, 18, 690–704.
- Western Cape Department of Health in Collaboration with the National Institute for Communicable; Diseases, S.A. Risk Factors for Coronavirus Disease 2019 (COVID-19) Death in a Population Cohort Study from the Western Cape Province, South Africa. Clin. Infect. Dis. 2021, 73, e2005.
- Motta, I.; Centis, R.; D’Ambrosio, L.; García-García, J.-M.; Goletti, D.; Gualano, G.; Lipani, F.; Palmieri, F.; Sánchez-Montalvá, A.; Pontali, E.; et al. Tuberculosis, COVID-19 and Migrants: Preliminary Analysis of Deaths Occurring in 69 Patients from Two Cohorts. Pulmonology 2020, 26, 233–240.
- Nägele, F.; Graber, M.; Hirsch, J.; Pölzl, L.; Sahanic, S.; Fiegl, M.; Hau, D.; Engler, C.; Lechner, S.; Stalder, A.K.; et al. Correlation between Structural Heart Disease and Cardiac SARS-CoV-2 Manifestations. Commun. Med. 2022, 2, 142.
- Tomasoni, D.; Italia, L.; Adamo, M.; Inciardi, R.M.; Lombardi, C.M.; Solomon, S.D.; Metra, M. COVID-19 and Heart Failure: From Infection to Inflammation and Angiotensin II Stimulation. Searching for Evidence from a New Disease. Eur. J. Heart Fail. 2020, 22, 957–966.
- Omoush, S.A.; Alzyoud, J.A.M. The Prevalence and Impact of Coinfection and Superinfection on the Severity and Outcome of COVID-19 Infection: An Updated Literature Review. Pathogens 2022, 11, 445.
- Blanco, J.-R.; Cobos-Ceballos, M.-J.; Navarro, F.; Sanjoaquin, I.; Arnaiz de las Revillas, F.; Bernal, E.; Buzon-Martin, L.; Viribay, M.; Romero, L.; Espejo-Perez, S.; et al. Pulmonary Long-Term Consequences of COVID-19 Infections after Hospital Discharge. Clin. Microbiol. Infect. 2021, 27, 892–896.
- Raisi-Estabragh, Z.; Cooper, J.; Salih, A.; Raman, B.; Lee, A.M.; Neubauer, S.; Harvey, N.C.; Petersen, S.E. Cardiovascular Disease and Mortality Sequelae of COVID-19 in the UK Biobank. Heart 2023, 109, 119.
- World Health Organization (WHO). Obesity Rising in Africa, WHO Analysis Finds. Available online: https://www.afro.who.int/news/obesity-rising-africa-who-analysis-finds (accessed on 12 March 2023).
- Arentz, M.; Yim, E.; Klaff, L.; Lokhandwala, S.; Riedo, F.X.; Chong, M.; Lee, M. Characteristics and Outcomes of 21 Critically Ill Patients With COVID-19 in Washington State. JAMA 2020, 323, 1612–1614.
- Bhatraju, P.K.; Ghassemieh, B.J.; Nichols, M.; Kim, R.; Jerome, K.R.; Nalla, A.K.; Greninger, A.L.; Pipavath, S.; Wurfel, M.M.; Evans, L.; et al. COVID-19 in Critically Ill Patients in the Seattle Region—Case Series. N. Engl. J. Med. 2020, 382, 2012–2022.
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W.; Barnaby, D.P.; Becker, L.B.; Chelico, J.D.; Cohen, S.L.; et al. Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA 2020, 323, 2052–2059.
- Nakeshbandi, M.; Maini, R.; Daniel, P.; Rosengarten, S.; Parmar, P.; Wilson, C.; Kim, J.M.; Oommen, A.; Mecklenburg, M.; Salvani, J.; et al. The Impact of Obesity on COVID-19 Complications: A Retrospective Cohort Study. Int. J. Obes. 2020, 44, 1832–1837.
- Nagy, É.; Cseh, V.; Barcs, I.; Ludwig, E. The Impact of Comorbidities and Obesity on the Severity and Outcome of COVID-19 in Hospitalized Patients—A Retrospective Study in a Hungarian Hospital. Int. J. Environ. Res. Public Health 2023, 20, 1372.
- Katona, P.; Katona-Apte, J. The Interaction between Nutrition and Infection. Clin. Infect. Dis. 2008, 46, 1582–1588.
- Mertens, E.; Peñalvo, J.L. The Burden of Malnutrition and Fatal COVID-19: A Global Burden of Disease Analysis. Front. Nutr. 2021, 7, 351.
- Menshawey, R.; Menshawey, E.; Alserr, A.H.K.; Abdelmassih, A.F. Low Iron Mitigates Viral Survival: Insights from Evolution, Genetics, and Pandemics—A Review of Current Hypothesis. Egypt. J. Med. Hum. Genet. 2020, 21, 75.
- Williams, A.M.; Ladva, C.N.; Leon, J.S.; Lopman, B.A.; Tangpricha, V.; Whitehead, R.D.; Armitage, A.E.; Wray, K.; Morovat, A.; Pasricha, S.R.; et al. Changes in Micronutrient and Inflammation Serum Biomarker Concentrations after a Norovirus Human Challenge. Am. J. Clin. Nutr. 2019, 110, 1456–1464.
- Cavezzi, A.; Troiani, E.; Corrao, S. COVID-19: Hemoglobin, Iron, and Hypoxia beyond Inflammation. A Narrative Review. Clin. Pract. 2020, 10, 1271.
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and Clinical Characteristics of 99 Cases of 2019 Novel Coronavirus Pneumonia in Wuhan, China: A Descriptive Study. Lancet 2020, 395, 507–513.
- Chen, X.; Zhao, B.; Qu, Y.; Chen, Y.; Xiong, J.; Feng, Y.; Men, D.; Huang, Q.; Liu, Y.; Yang, B.; et al. Detectable Serum Severe Acute Respiratory Syndrome Coronavirus 2 Viral Load (RNAemia) Is Closely Correlated with Drastically Elevated Interleukin 6 Level in Critically Ill Patients with Coronavirus Disease 2019. Clin. Infect. Dis. 2020, 71, 1937–1942.
- Liu, B.; Li, M.; Zhou, Z.; Guan, X.; Xiang, Y. Can We Use Interleukin-6 (IL-6) Blockade for Coronavirus Disease 2019 (COVID-19)-Induced Cytokine Release Syndrome (CRS)? J. Autoimmun. 2020, 111, 102452.
- Martins, A.C.; Almeida, J.I.; Lima, I.S.; Kapitão, A.S.; Gozzelino, R. Iron Metabolism and the Inflammatory Response. IUBMB Life 2017, 69, 442–450.
- Alipour, R.; Hashemi, S.H.; Mikaeili, F. Serum Iron Level in Patients with COVID-19: A Case Report Study. Int. J. Res. Med. Sci. 2020, 8, 2658.
- Nai, A.; Lorè, N.I.; Pagani, A.; De Lorenzo, R.; Di Modica, S.; Saliu, F.; Cirillo, D.M.; Rovere-Querini, P.; Manfredi, A.A.; Silvestri, L. Hepcidin Levels Predict COVID-19 Severity and Mortality in a Cohort of Hospitalized Italian Patients. Am. J. Hematol. 2021, 96, E32–E35.
- Ganz, T. Anemia of Inflammation. N. Engl. J. Med. 2019, 381, 1148–1157.
- Yağcı, S.; Serin, E.; Acicbe, Ö.; Zeren, M.İ.; Odabaşı, M.S. The Relationship between Serum Erythropoietin, Hepcidin, and Haptoglobin Levels with Disease Severity and Other Biochemical Values in Patients with COVID-19. Int. J. Lab. Hematol. 2021, 43, 142–151.
- Litton, E.; Lim, J. Iron Metabolism: An Emerging Therapeutic Target in Critical Illness. Crit. Care 2019, 2019, 573–584.
- Shah, A.; Frost, J.N.; Aaron, L.; Donovan, K.; Drakesmith, H.; McKechnie, S.R.; Stanworth, S.J. Systemic Hypoferremia and Severity of Hypoxemic Respiratory Failure in COVID-19. Crit. Care 2020, 24, 320.
- Bu, X.; Yao, Y.; Li, X. Immune Checkpoint Blockade in Breast Cancer Therapy. In Advances in Experimental Medicine and Biology; Springer: Berlin/Heidelberg, Germany, 2017.
- The Lancet. COVID-19 in Africa: A Lesson in Solidarity. Lancet 2021, 398, 185.
- Herzog, L.M.; Norheim, O.F.; Emanuel, E.J.; McCoy, M.S. Covax Must Go beyond Proportional Allocation of COVID Vaccines to Ensure Fair and Equitable Access. BMJ 2021, 372, m4853.
- World Health Organization (WHO). Fair Allocation Mechanism for COVID-19 Vaccines through the COVAX Facility. Available online: https://www.who.int/publications/m/item/fair-allocation-mechanism-for-covid-19-vaccines-through-the-covax-facility (accessed on 9 April 2023).
- World Health Organization (WHO). WHO Lists Two Additional COVID-19 Vaccines for Emergency Use and COVAX Roll-Out. Available online: https://www.who.int/news/item/15-02-2021-who-lists-two-additional-covid-19-vaccines-for-emergency-use-and-covax-roll-out (accessed on 1 February 2023).
- Mwai, P. COVID-19 Africa: Who Is Getting the Vaccine? Available online: https://www.bbc.com/news/56100076 (accessed on 2 April 2021).
- Our World in Data Coronavirus (COVID-19) Vaccinations. Available online: https://ourworldindata.org/covid-vaccinations#what-share-of-the-population-has-received-at-least-one-dose-of-vaccine (accessed on 19 March 2023).
- World Health Organization (WHO). Africa COVID-19 Vaccination Dashboard. Available online: https://app.powerbi.com/view?r=eyJrIjoiOTI0ZDlhZWEtMjUxMC00ZDhhLWFjOTYtYjZlMGYzOWI4NGIwIiwidCI6ImY2MTBjMGI3LWJkMjQtNGIzOS04MTBiLTNkYzI4MGFmYjU5MCIsImMiOjh9 (accessed on 9 April 2023).
- Kabakama, S.; Konje, E.T.; Dinga, J.N.; Kishamawe, C.; Morhason-Bello, I.; Hayombe, P.; Adeyemi, O.; Chimuka, E.; Lumu, I.; Amuasi, J.; et al. Commentary on COVID-19 Vaccine Hesitancy in Sub-Saharan Africa. Trop. Med. Infect. Dis. 2022, 7, 130.
- World Health Organization (WHO). Joint Statement on Dose Donations of COVID-19 Vaccines to African Countries. Available online: https://www.who.int/news/item/29-11-2021-joint-statement-on-dose-donations-of-covid-19-vaccines-to-african-countries (accessed on 9 April 2023).
- World Health Organization (WHO). Strategy to Achieve Global COVID-19 Vaccination by Mid-2022. Available online: https://www.who.int/publications/m/item/strategy-to-achieve-global-covid-19-vaccination-by-mid-2022 (accessed on 1 February 2023).
- Nature. Africa Is Bringing Vaccine Manufacturing Home. Nature 2022, 602, 184.
- Paton, J. BioNTech to Send COVID Vaccine Production Units to Africa. Available online: https://www.bloomberg.com/news/articles/2022-02-16/biontech-to-send-covid-vaccine-production-units-to-africa#xj4y7vzkg (accessed on 12 March 2023).
- Wellcome. Scaling Up African Vaccine Manufacturing Capacity. Available online: https://cms.wellcome.org/sites/default/files/2023-01/Wellcome-Biovac-BCG-Scaling-up-African-vaccine-manufacturing-capacity-report-2023_0.pdf (accessed on 24 January 2024).
- World Health Organization (WHO). Africa Continued Delivery of Immunization Services in the Context of COVID-19 Pandemic. Available online: https://www.afro.who.int/news/continued-delivery-immunization-services-context-covid-19-pandemic (accessed on 28 March 2023).
- Hakizimana, D.; Ntizimira, C.; Mbituyumuremyi, A.; Hakizimana, E.; Mahmoud, H.; Birindabagabo, P.; Musanabaganwa, C.; Gashumba, D. The Impact of COVID-19 on Malaria Services in Three High Endemic Districts in Rwanda: A Mixed-Method Study. Malar. J. 2022, 21, 48.
- Desta, A.A.; Woldearegay, T.W.; Gebremeskel, E.; Alemayehu, M.; Getachew, T.; Gebregzabiher, G.; Ghebremedhin, K.D.; Zgita, D.N.; Aregawi, A.B.; Redae, G. Impacts of COVID-19 on Essential Health Services in Tigray, Northern Ethiopia: A Pre-Post Study. PLoS ONE 2021, 16, e0256330.
- Jensen, C.; McKerrow, N.H. Child Health Services during a COVID-19 Outbreak in KwaZulu-Natal Province, South Africa. S. Afr. Med. J. 2020, 111, 114.
- Harvey, R. The Ignored Pandemic: The Dual Crises of Gender-Based Violence and COVID-19; Oxfam: Oxford, UK, 2021.
- Mednick, S. COVID-19 Brings Abuse and Other Fears to Displaced Women in South Sudan. Available online: https://www.thenewhumanitarian.org/feature/2020/05/06/coronavirus-south-sudan-women-abuse-gender-violence (accessed on 24 January 2024).
- International Labour Organization. COVID-19 Impact on Child Labour and Forced Labour: The Response of the IPEC+ Flagship Programme; International Labour Organization: Geneva, Switzerland, 2020.
