Ciliary beat frequency (CBF) within the ciliated epithelium of the nasal tract can be stimulated to a higher frequency and provide increased protection against transient exposure to airway irritants. Smokers as well as non-smokers exposed to secondhand tobacco smoke were found to have higher CBFs. However, with extended exposure to irritants, persistent upregulated CBF can damage and remodel the epithelial layer with fewer protective cilia. Additionally, mucociliary clearance (MCC), the innate defense mechanism of the respiratory system, traps particles and pathogens within the mucous layer of the epithelium and propels them out of the airways through ciliary activity. However, this mechanism becomes defective as disease progresses, increasing susceptibility to viral respiratory infections.
- COVID-19
- SARS-CoV-2
- smoker’s paradox
- ciliary beat frequency
- mucociliary clearance
- tobacco exposure
- ACE2
- ciliated epithelium
1. Introduction
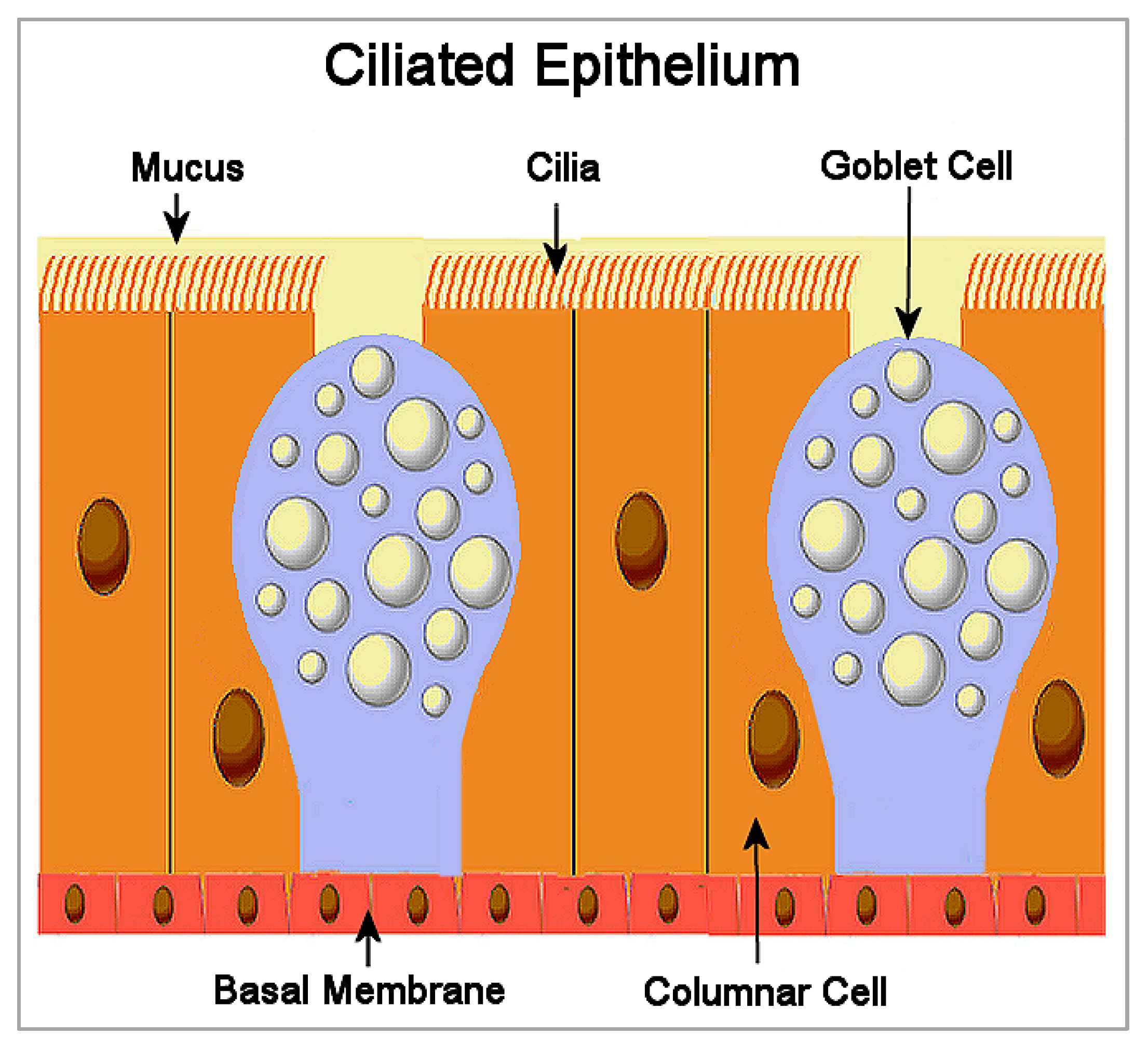
2. Ciliated Epithelium
3. Mucociliary Clearance and SARS-CoV-2 Infection
This entry is adapted from the peer-reviewed paper 10.3390/biomed2010009
References
- Jiang, C.; Chen, Q.; Xie, M. Smoking increases the risk of infectious diseases: A narrative review. Tob. Induc. Dis. 2020, 18, 60.
- Xu, X.; Chen, P.; Wang, J.; Feng, J.; Zhou, H.; Li, X.; Zhong, W.; Hao, P. Evolution of the novel coronavirus from the ongoing Wuhan outbreak and modeling of its spike protein for risk of human transmission. Sci. China Life Sci. 2020, 63, 457–460.
- Zhang, H.; Rostami, M.R.; Leopold, P.L.; Mezey, J.G.; O’Beirne, S.L.; Strulovici-Barel, Y.; Crystal, R.G. Expression of the SARS-CoV-2 ACE2 Receptor in the Human Airway Epithelium. Am. J. Respir. Crit. Care Med. 2020, 202, 219–229.
- Usman, M.S.; Siddiqi, T.J.; Khan, M.S.; Patel, U.K.; Shahid, I.; Ahmed, J.; Kalra, A.; Michos, E.D. Is there a smoker’s paradox in COVID-19? BMJ Evid.-Based Med. 2021, 26, 279–284.
- Vardavas, C.I.; Nikitara, K. COVID-19 and smoking: A systematic review of the evidence. Tob. Induc. Dis. 2020, 18, 20.
- Lippi, G.; Henry, B.M. Active smoking is not associated with severity of coronavirus disease 2019 (COVID-19). Eur. J. Intern. Med. 2020, 75, 107–108.
- Yanover, C.; Mizrahi, B.; Kalkstein, N.; Marcus, K.; Akiva, P.; Barer, Y.; Shalev, V.; Chodick, G. What Factors Increase the Risk of Complications in SARS-CoV-2-Infected Patients? A Cohort Study in a Nationwide Israeli Health Organization. JMIR Public Health Surveill. 2020, 6, e20872.
- Hippisley-Cox, J.; Young, D.; Coupland, C.; Channon, K.M.; Tan, P.S.; Harrison, D.A.; Rowan, K.; Aveyard, P.; Pavord, I.D.; Watkinson, P.J. Risk of severe COVID-19 disease with ACE inhibitors and angiotensin receptor blockers: Cohort study including 8.3 million people. Heart 2020, 106, 1503–1511.
- Meini, S.; Fortini, A.; Andreini, R.; Sechi, L.A.; Tascini, C. The Paradox of the Low Prevalence of Current Smokers Among COVID-19 Patients Hospitalized in Nonintensive Care Wards: Results From an Italian Multicenter Case–Control Study. Nicotine Tob. Res. 2020, 23, 1436–1440.
- Rossato, M.; Russo, L.; Mazzocut, S.; Di Vincenzo, A.; Fioretto, P.; Vettor, R. Current smoking is not associated with COVID-19. Eur. Respir. J. 2020, 55, 2001290.
- Van Westen-Lagerweij, N.A.; Meijer, E.; Meeuwsen, E.G.; Chavannes, N.H.; Willemsen, M.C.; Croes, E.A. Are smokers protected against SARS-CoV-2 infection (COVID-19)? The origins of the myth. NPJ Prim. Care Respir. Med. 2021, 31, 10.
- Le Bras, A. Light smoking and CVD risk. Nat. Rev. Cardiol. 2018, 15, 136.
- Widdicombe, J.H.; Wine, J.J. Airway Gland Structure and Function. Physiol. Rev. 2015, 95, 1241–1319.
- Kuek, L.E.; Lee, R.J. First contact: The role of respiratory cilia in host-pathogen interactions in the airways. Am. J. Physiol. Lung Cell. Mol. Physiol. 2020, 319, L603–L619.
- Cicuta, P. The use of biophysical approaches to understand ciliary beating. Biochem. Soc. Trans. 2020, 48, 221–229.
- Ueno, H.; Yasunaga, T.; Shingyoji, C.; Hirose, K. Dynein pulls microtubules without rotating its stalk. Proc. Natl. Acad. Sci. USA 2008, 105, 19702–19707.
- Zhou, H.; Wang, X.; Brighton, L.; Hazucha, M.; Jaspers, I.; Carson, J.L. Increased nasal epithelial ciliary beat frequency associated with lifestyle tobacco smoke exposure. Inhal. Toxicol. 2009, 21, 875–881.
- Fu, Y.; Tong, J.; Meng, F.; Hoeltig, D.; Liu, G.; Yin, X.; Herrler, G. Ciliostasis of airway epithelial cells facilitates influenza A virus infection. Vet. Res. 2018, 49, 65.
- Brown, R.B. Sodium Toxicity in the Nutritional Epidemiology and Nutritional Immunology of COVID-19. Medicina 2021, 57, 739.
- Aghapour, M.; Raee, P.; Moghaddam, S.J.; Hiemstra, P.S.; Heijink, I.H. Airway Epithelial Barrier Dysfunction in Chronic Obstructive Pulmonary Disease: Role of Cigarette Smoke Exposure. Am. J. Respir. Cell. Mol. Biol. 2018, 58, 157–169.
- Hadar, T.; Yaniv, E.; Shvili, Y.; Koren, R.; Shvero, J. Histopathological changes of the nasal mucosa induced by smoking. Inhal. Toxicol. 2009, 21, 1119–1122.
- Bustamante-Marin, X.M.; Ostrowski, L.E. Cilia and Mucociliary Clearance. Cold Spring Harb. Perspect. Biol. 2017, 9, a028241.
- Koparal, M.; Kurt, E.; Altuntas, E.E.; Dogan, F. Assessment of mucociliary clearance as an indicator of nasal function in patients with COVID-19: A cross-sectional study. Eur. Arch. Otorhinolaryngol. 2021, 278, 1863–1868.
- Hou, Y.J.; Okuda, K.; Edwards, C.E.; Martinez, D.R.; Asakura, T.; Dinnon, K.H., 3rd; Kato, T.; Lee, R.E.; Yount, B.L.; Mascenik, T.M.; et al. SARS-CoV-2 Reverse Genetics Reveals a Variable Infection Gradient in the Respiratory Tract. Cell 2020, 182, 429–446.e14.
- Oran, D.P.; Topol, E.J. Prevalence of asymptomatic SARS-CoV-2 infection: A narrative review. Ann. Intern. Med. 2020, 173, 362–367.
- Nalbandian, A.; Sehgal, K.; Gupta, A.; Madhavan, M.V.; McGroder, C.; Stevens, J.S.; Cook, J.R.; Nordvig, A.S.; Shalev, D.; Sehrawat, T.S. Post-acute COVID-19 syndrome. Nat. Med. 2021, 27, 601–615.
- CDC. Health Effects of Secondhand Smoke. Centers for Disease Control and Prevention—Smoking & Tobacco Use. Available online: https://www.cdc.gov/tobacco/data_statistics/fact_sheets/secondhand_smoke/health_effects/index.htm (accessed on 29 January 2022).
- Nishiga, M.; Wang, D.W.; Han, Y.; Lewis, D.B.; Wu, J.C. COVID-19 and cardiovascular disease: From basic mechanisms to clinical perspectives. Nat. Rev. Cardiol. 2020, 17, 543–558.
