Oral cancer is becoming more and more frequent worldwide. Despite the widely available prevention, it is one of the most common cancers in the world, with 476,125 new cases and 225,900 deaths in 2020. Among the causes of carcinogenesis in the oral cavity, tobacco smoking or chewing, alcohol consumption, occupational exposure, risky sexual behaviour, genetic factors, and environmental pollution are widely mentioned. Smoking is the most prominent risk factor for oral cancer due to the carcinogenic chemicals in cigarette smoke, including nitrosamines, benzopyrenes, and aromatic amines. The risk of oral cancer is three times higher in smokers compared to non-smokers. In addition, the combination of cigarette smoking and frequent heavy alcohol consumption increases the risk of developing cancer by several times.
- oral dysplasia
- oral epithelial dysplasia
- immunohistochemistry
- histological grading
1. Biomarkers Related to Cell Division and Proliferation
2. Biomarkers Related to Epithelial–Mesenchymal Transition (EMT)
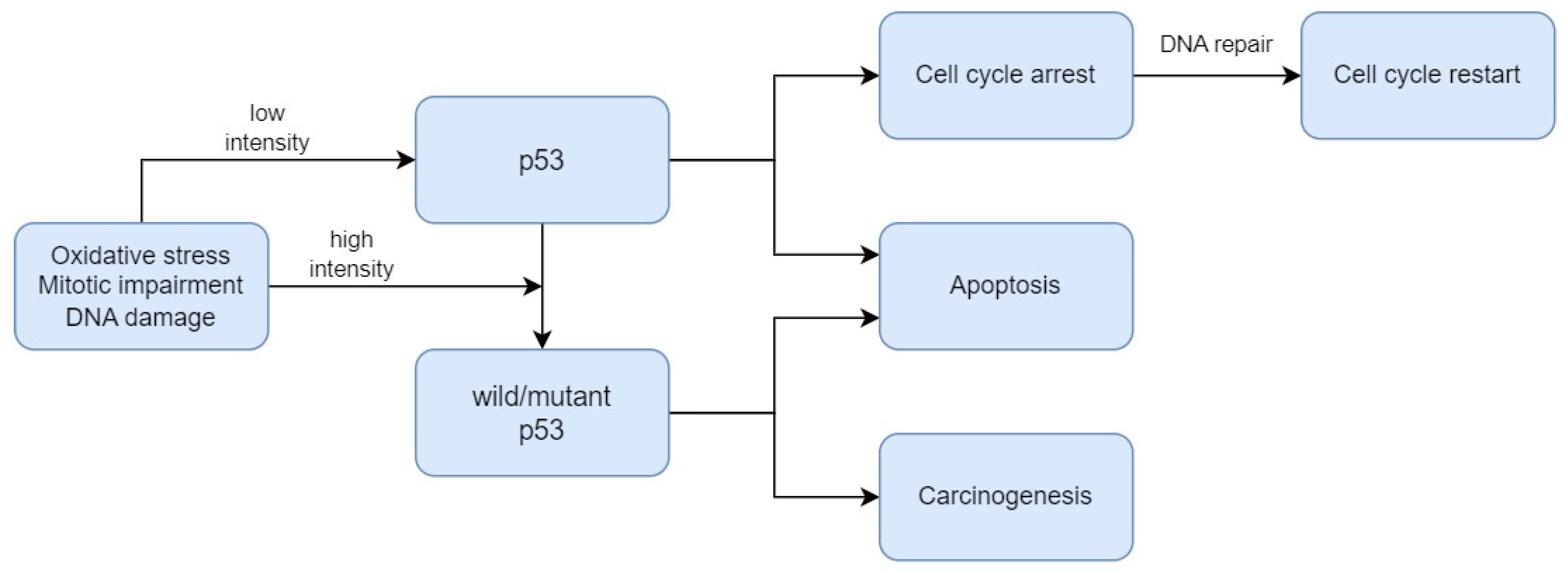
3. Biomarkers Related to Cell Death Regulation
4. Biomarkers Related to Cellular Metabolism
5. Biomarkers Related to Extracellular Signalling Pathways
This entry is adapted from the peer-reviewed paper 10.3390/biomedicines12030577
References
- Wolgemuth, D.J. Function of Cyclins in Regulating the Mitotic and Meiotic Cell Cycles in Male Germ Cells. Cell Cycle Georget. 2008, 7, 3509–3513.
- Wang, Z. Cell Cycle Progression and Synchronization: An Overview. Methods Mol. Biol. 2022, 2579, 3–23.
- Loyer, P.; Trembley, J.H. Roles of CDK/Cyclin Complexes in Transcription and Pre-mRNA Splicing: Cyclins L and CDK11 at the Cross-Roads of Cell Cycle and Regulation of Gene Expression. Semin. Cell Dev. Biol. 2020, 107, 36–45.
- Zhang, W.; Liu, Y.; Jang, H.; Nussinov, R. Cell Cycle Progression Mechanisms: Slower Cyclin-D/CDK4 Activation and Faster Cyclin-E/CDK2. BioRxiv Prepr. Serv. Biol. 2023.
- Steurer, S.; Riemann, C.; Büscheck, F.; Luebke, A.M.; Kluth, M.; Hube-Magg, C.; Hinsch, A.; Höflmayer, D.; Weidemann, S.; Fraune, C.; et al. P63 Expression in Human Tumors and Normal Tissues: A Tissue Microarray Study on 10,200 Tumors. Biomark. Res. 2021, 9, 7.
- Bavle, R.M.; Paremala, K.; Venugopal, R.; Rudramuni, A.S.; Khan, N.; Hosthor, S.S. Grading of Oral Leukoplakia: Can It Be Improvised Using Immunohistochemical Markers P63 and CD31. Contemp. Clin. Dent. 2021, 12, 37–43.
- Truong, A.B.; Kretz, M.; Ridky, T.W.; Kimmel, R.; Khavari, P.A. P63 Regulates Proliferation and Differentiation of Developmentally Mature Keratinocytes. Genes Dev. 2006, 20, 3185–3197.
- Bergholz, J.; Xiao, Z.-X. Role of P63 in Development, Tumorigenesis and Cancer Progression. Cancer Microenviron. Off. J. Int. Cancer Microenviron. Soc. 2012, 5, 311–322.
- Patel, S.B.; Manjunatha, B.S.; Shah, V.; Soni, N.; Sutariya, R. Immunohistochemical Evaluation of P63 and Cyclin D1 in Oral Squamous Cell Carcinoma and Leukoplakia. J. Korean Assoc. Oral Maxillofac. Surg. 2017, 43, 324–330.
- Gupta, S.; Gupta, V.; Tyagi, N.; Vij, R.; Vij, H.; Sharma, E. Analysis of Role of Angiogenesis in Epithelial Dysplasia: An Immunohistochemical Study. J. Clin. Diagn. Res. 2017, 11, EC29–EC34.
- Tawara, M.; Suzuki, H.; Goto, N.; Tanaka, T.; Kaneko, M.K.; Kato, Y. A Novel Anti-CD44 Variant 9 Monoclonal Antibody C44Mab-1 Was Developed for Immunohistochemical Analyses against Colorectal Cancers. Curr. Issues Mol. Biol. 2023, 45, 3658–3673.
- Venkat Naga, S.K.S.; Shekar, P.C.; Kattappagari, K.K.; Prakash Chandra, K.L.; Reddy, G.S.; Ramana Reddy, B.V. Expression of Cluster Differentiation-44 Stem Cell Marker in Grades of Oral Epithelial Dysplasia: A Preliminary Study. J. Oral Maxillofac. Pathol. JOMFP 2019, 23, 203–207.
- Aravind, T.; Janardhanan, M.; Rakesh, S.; Savithri, V.; Unnikrishnan, U.G. Immunolocalization of Osteopontin in Dysplasias and Squamous Cell Carcinomas Arising from Oral Epithelium. J. Oral Maxillofac. Pathol. JOMFP 2017, 21, 18–23.
- Sodek, J.; Ganss, B.; McKee, M.D. Osteopontin. Crit. Rev. Oral Biol. Med. Off. Publ. Am. Assoc. Oral Biol. 2000, 11, 279–303.
- Mrochem, J.; Bartnik, W. Osteopontin—A New Marker in Neoplastic Diseases. Contemp. Oncol. Onkol. 2008, 12, 349–353.
- Ozaki, T.; Nakagawara, A. Role of P53 in Cell Death and Human Cancers. Cancers 2011, 3, 994–1013.
- Williams, A.B.; Schumacher, B. P53 in the DNA-Damage-Repair Process. Cold Spring Harb. Perspect. Med. 2016, 6, a026070.
- Babamohamadi, M.; Babaei, E.; Ahmed Salih, B.; Babamohammadi, M.; Jalal Azeez, H.; Othman, G. Recent Findings on the Role of Wild-Type and Mutant P53 in Cancer Development and Therapy. Front. Mol. Biosci. 2022, 9, 903075.
- Borrero, L.J.H.; El-Deiry, W.S. Tumor Suppressor P53: Biology, Signaling Pathways, and Therapeutic Targeting. Biochim. Biophys. Acta Rev. Cancer 2021, 1876, 188556.
- Pandya, J.A.; Boaz, K.; Natarajan, S.; Manaktala, N.; Nandita, K.P.; Lewis, A.J. A Correlation of Immunohistochemical Expression of TP53 and CDKN1A in Oral Epithelial Dysplasia and Oral Squamous Cell Carcinoma. J. Cancer Res. Ther. 2018, 14, 666–670.
- Patil, S.; Gawande, M.; Chaudhari, M.; Sharma, P.; Hande, A.; Sonone, A. Prognostic Significance of P53 Expression in Various Grades of Epithelial Dysplasia. J. Datta Meghe Inst. Med. Sci. Univ. 2022, 17, 306–310.
- Sawada, K.; Momose, S.; Kawano, R.; Kohda, M.; Irié, T.; Mishima, K.; Kaneko, T.; Horie, N.; Okazaki, Y.; Higashi, M.; et al. Immunohistochemical Staining Patterns of P53 Predict the Mutational Status of TP53 in Oral Epithelial Dysplasia. Mod. Pathol. Off. J. US Can. Acad. Pathol. Inc. 2022, 35, 177–185.
- Imaizumi, T.; Matsuda, K.; Tanaka, K.; Kondo, H.; Ueki, N.; Kurohama, H.; Otsubo, C.; Matsuoka, Y.; Akazawa, Y.; Miura, S.; et al. Detection of Endogenous DNA Double-Strand Breaks in Oral Squamous Epithelial Lesions by P53-Binding Protein 1. Anticancer Res. 2021, 41, 4771–4779.
- Napoli, M.; Wu, S.J.; Gore, B.L.; Abbas, H.A.; Lee, K.; Checker, R.; Dhar, S.; Rajapakshe, K.; Tan, A.C.; Lee, M.G.; et al. ΔNp63 Regulates a Common Landscape of Enhancer Associated Genes in Non-Small Cell Lung Cancer. Nat. Commun. 2022, 13, 614.
- Abylkassov, R.; Xie, Y. Role of Yes-Associated Protein in Cancer: An Update. Oncol. Lett. 2016, 12, 2277–2282.
- Ono, S.; Nakano, K.; Takabatake, K.; Kawai, H.; Nagatsuka, H. Immunohistochemistry of YAP and dNp63 and Survival Analysis of Patients Bearing Precancerous Lesion and Oral Squamous Cell Carcinoma. Int. J. Med. Sci. 2019, 16, 766–773.
- Sun, X.; Kaufman, P.D. Ki-67: More than a Proliferation Marker. Chromosoma 2018, 127, 175–186.
- Booth, D.G.; Takagi, M.; Sanchez-Pulido, L.; Petfalski, E.; Vargiu, G.; Samejima, K.; Imamoto, N.; Ponting, C.P.; Tollervey, D.; Earnshaw, W.C.; et al. Ki-67 Is a PP1-Interacting Protein That Organises the Mitotic Chromosome Periphery. eLife 2014, 3, e01641.
- Liang, Y.; Ma, C.; Li, F.; Nie, G.; Zhang, H. The Role of Contactin 1 in Cancers: What We Know So Far. Front. Oncol. 2020, 10, 574208.
- Iqbal, A.; Tamgadge, S.; Tamgadge, A.; Pereira, T.; Kumar, S.; Acharya, S.; Jadhav, A. Evaluation of Ki-67 Expression in Oral Submucous Fibrosis and Its Correlation with Clinical and Histopathological Features. J. Microsc. Ultrastruct. 2019, 8, 20–24.
- Kim, C.-H.; Lee, H.S.; Park, J.-H.; Choi, J.-H.; Jang, S.-H.; Park, Y.-B.; Lee, M.G.; Hyun, I.G.; Kim, K.I.; Kim, H.S.; et al. Prognostic Role of P53 and Ki-67 Immunohistochemical Expression in Patients with Surgically Resected Lung Adenocarcinoma: A Retrospective Study. J. Thorac. Dis. 2015, 7, 822–833.
- Lalkota, B.P.; Srinivasa, B.J.; Swamy, M.V.; Hazarika, D.; Jeet, B.M.; Jyothi, K.; Ghosh, M.; Sayeed, S.M.; Nasiruddin, M.; Naik, R. The Role of P53 and Ki67 in Predicting Clinical Outcome in Breast Cancer Patients. J. Cancer Res. Ther. 2023, 19, 208.
- Humayun, S.; Prasad, V.R. Expression of P53 Protein and Ki-67 Antigen in Oral Premalignant Lesions and Oral Squamous Cell Carcinomas: An Immunohistochemical Study. Natl. J. Maxillofac. Surg. 2011, 2, 38–46.
- Kumar, P.; Kane, S.; Rathod, G.P. Coexpression of P53 and Ki 67 and Lack of C-erbB2 Expression in Oral Leukoplakias in India. Braz. Oral Res. 2012, 26, 228–234.
- Kamala, K.A.; Kanetkar, S.R.; Datkhile, K.D.; Sankethguddad, S. Expression of Ki67 Biomarker in Oral Submucous Fibrosis with Clinico-Pathological Correlations: A Prospective Study. Asian Pac. J. Cancer Prev. APJCP 2022, 23, 253–259.
- Dash, K.C.; Mahapatra, N.; Bhuyan, L.; Panda, A.; Behura, S.S.; Mishra, P. An Immunohistochemical Study Showing Ki-67 as an Analytical Marker in Oral Malignant and Premalignant Lesions. J. Pharm. Bioallied Sci. 2020, 12, S274–S278.
- Mondal, K.; Mandal, R.; Sarkar, B.C. Importance of Ki-67 Labeling in Oral Leukoplakia with Features of Dysplasia and Carcinomatous Transformation: An Observational Study over 4 Years. S. A. J. Cancer 2020, 9, 99–104.
- Takkem, A.; Barakat, C.; Zakaraia, S.; Zaid, K.; Najmeh, J.; Ayoub, M.; Seirawan, M.Y. Ki-67 Prognostic Value in Different Histological Grades of Oral Epithelial Dysplasia and Oral Squamous Cell Carcinoma. Asian Pac. J. Cancer Prev. APJCP 2018, 19, 3279–3286.
- Swain, S.; Nishat, R.; Ramachandran, S.; Raghuvanshi, M.; Behura, S.S.; Kumar, H. Comparative Evaluation of Immunohistochemical Expression of MCM2 and Ki67 in Oral Epithelial Dysplasia and Oral Squamous Cell Carcinoma. J. Cancer Res. Ther. 2022, 18, 997–1002.
- Gadbail, A.R.; Chaudhary, M.; Sarode, S.C.; Gondivkar, S.; Tekade, S.A.; Zade, P.; Hande, A.; Sarode, G.S.; Patil, S. Ki67, CD105, and α-SMA Expression Supports the Transformation Relevant Dysplastic Features in the Atrophic Epithelium of Oral Submucous Fibrosis. PLoS ONE 2018, 13, e0200171.
- Gadbail, A.R.; Chaudhary, M.S.; Sarode, S.C.; Gawande, M.; Korde, S.; Tekade, S.A.; Gondivkar, S.; Hande, A.; Maladhari, R. Ki67, CD105, and α-SMA Expressions Better Relate the Binary Oral Epithelial Dysplasia Grading System of World Health Organization. J. Oral Pathol. Med. Off. Publ. Int. Assoc. Oral Pathol. Am. Acad. Oral Pathol. 2017, 46, 921–927.
- Suwasini, S.; Chatterjee, K.; Purkait, S.K.; Samaddar, D.; Chatterjee, A.; Kumar, M. Expression of P53 Protein and Ki-67 Antigen in Oral Leukoplakia with Different Histopathological Grades of Epithelial Dysplasia. J. Int. Soc. Prev. Community Dent. 2018, 8, 513–522.
- Leung, E.Y.; McMahon, J.D.; McLellan, D.R.; Syyed, N.; McCarthy, C.E.; Nixon, C.; Orange, C.; Brock, C.; Hunter, K.D.; Adams, P.D. DNA Damage Marker Phosphorylated Histone H2AX Is a Potential Predictive Marker for Progression of Epithelial Dysplasia of the Oral Cavity. Histopathology 2017, 71, 522–528.
- Monteiro, L.; Silva, P.; Delgado, L.; Amaral, B.; Garcês, F.; Salazar, F.; Pacheco, J.-J.; Lopes, C.; Bousbaa, H.; Warnakulasuriya, S. Expression of Spindle Assembly Checkpoint Proteins BubR1 and Mad2 Expression as Potential Biomarkers of Malignant Transformation of Oral Leukoplakia: An Observational Cohort Study. Med. Oral Patol. Oral Cirugia Bucal 2021, 26, e719–e728.
- Rubin, C.I.; Atweh, G.F. The Role of Stathmin in the Regulation of the Cell Cycle. J. Cell. Biochem. 2004, 93, 242–250.
- Feng, S.; Song, Y.; Shen, M.; Xie, S.; Li, W.; Lu, Y.; Yang, Y.; Ou, G.; Zhou, J.; Wang, F.; et al. Microtubule-Binding Protein FOR20 Promotes Microtubule Depolymerization and Cell Migration. Cell Discov. 2017, 3, 17032.
- Vadla, P.; Deepthi, G.; Kumar, C.A.; Bashamalla, R.; Syeda, N.; Naramala, S. Immunohistochemical Expression of Stathmin in Oral Dysplasia: An Original Study with an Insight of Its Action on Microtubules. J. Oral Maxillofac. Pathol. JOMFP 2021, 25, 247–252.
- Blanpain, C.; Horsley, V.; Fuchs, E. Epithelial Stem Cells: Turning over New Leaves. Cell 2007, 128, 445–458.
- Kalluri, R.; Weinberg, R.A. The Basics of Epithelial-Mesenchymal Transition. J. Clin. Investig. 2009, 119, 1420–1428.
- Dongre, A.; Weinberg, R.A. New Insights into the Mechanisms of Epithelial-Mesenchymal Transition and Implications for Cancer. Nat. Rev. Mol. Cell Biol. 2019, 20, 69–84.
- Ding, D.-C.; Shyu, W.-C.; Lin, S.-Z. Mesenchymal Stem Cells. Cell Transplant. 2011, 20, 5–14.
- Huang, Y.; Hong, W.; Wei, X. The Molecular Mechanisms and Therapeutic Strategies of EMT in Tumor Progression and Metastasis. J. Hematol. Oncol. 2022, 15, 129.
- Chen, T.; You, Y.; Jiang, H.; Wang, Z.Z. Epithelial-Mesenchymal Transition (EMT): A Biological Process in the Development, Stem Cell Differentiation, and Tumorigenesis. J. Cell. Physiol. 2017, 232, 3261–3272.
- Toriumi, K.; Berto, S.; Koike, S.; Usui, N.; Dan, T.; Suzuki, K.; Miyashita, M.; Horiuchi, Y.; Yoshikawa, A.; Asakura, M.; et al. Combined Glyoxalase 1 Dysfunction and Vitamin B6 Deficiency in a Schizophrenia Model System Causes Mitochondrial Dysfunction in the Prefrontal Cortex. Redox Biol. 2021, 45, 102057.
- Krisanaprakornkit, S.; Iamaroon, A. Epithelial-Mesenchymal Transition in Oral Squamous Cell Carcinoma. ISRN Oncol. 2012, 2012, 681469.
- Ling, Z.; Cheng, B.; Tao, X. Epithelial-to-Mesenchymal Transition in Oral Squamous Cell Carcinoma: Challenges and Opportunities. Int. J. Cancer 2021, 148, 1548–1561.
- Hardwick, J.M.; Soane, L. Multiple Functions of BCL-2 Family Proteins. Cold Spring Harb. Perspect. Biol. 2013, 5, a008722.
- Chao, D.T.; Korsmeyer, S.J. BCL-2 Family: Regulators of Cell Death. Annu. Rev. Immunol. 1998, 16, 395–419.
- Hua, C.; Zorn, S.; Jensen, J.P.; Coupland, R.W.; Ko, H.S.; Wright, J.J.; Bakhshi, A. Consequences of the t(14;18) Chromosomal Translocation in Follicular Lymphoma: Deregulated Expression of a Chimeric and Mutated BCL-2 Gene. Oncogene Res. 1988, 2, 263–275.
- Pathak, A.; Shetty, D.C.; Dhanapal, R.; Kaur, G. To Analyse the Mitotic and Keratinisation Correlation with Bcl-2 Expression in Varying Grades of Oral Epithelial Dysplasia and Squamous Cell Carcinoma. J. Oral Maxillofac. Pathol. JOMFP 2022, 26, 316–321.
- Pallavi, N.; Nalabolu, G.R.K.; Hiremath, S.K.S. Bcl-2 and c-Myc Expression in Oral Dysplasia and Oral Squamous Cell Carcinoma: An Immunohistochemical Study to Assess Tumor Progression. J. Oral Maxillofac. Pathol. JOMFP 2018, 22, 325–331.
- Han, Y.; Liu, D.; Li, L. PD-1/PD-L1 Pathway: Current Researches in Cancer. Am. J. Cancer Res. 2020, 10, 727–742.
- Kujan, O.; Agag, M.; Smaga, M.; Vaishnaw, Y.; Idrees, M.; Shearston, K.; Farah, C.S. PD-1/PD-L1, Treg-Related Proteins, and Tumour-Infiltrating Lymphocytes Are Associated with the Development of Oral Squamous Cell Carcinoma. Pathology 2022, 54, 409–416.
- Wang, Q.; Yang, H.-S. The Role of Pdcd4 in Tumor Suppression and Protein Translation. Biol. Cell 2018, 110, 169–177.
- Desai, K.M.; Kale, A.D. Immunoexpression of Programmed Cell Death 4 Protein in Normal Oral Mucosa, Oral Epithelial Dysplasia and Oral Squamous Cell Carcinoma. J. Oral Maxillofac. Pathol. JOMFP 2017, 21, 462.
- Ferns, G.; Shams, S.; Shafi, S. Heat Shock Protein 27: Its Potential Role in Vascular Disease. Int. J. Exp. Pathol. 2006, 87, 253–274.
- Vidyasagar, A.; Wilson, N.A.; Djamali, A. Heat Shock Protein 27 (HSP27): Biomarker of Disease and Therapeutic Target. Fibrogenesis Tissue Repair 2012, 5, 7.
- Karri, R.L.; Subramanyam, R.V.; Venigella, A.; Babburi, S.; Pinisetti, S.; Rudraraju, A. Differential Expression of Heat Shock Protein 27 in Oral Epithelial Dysplasias and Squamous Cell Carcinoma. J. Microsc. Ultrastruct. 2020, 8, 62–68.
- Yagui-Beltran, A.; Craig, A.L.; Lawrie, L.; Thompson, D.; Pospisilova, S.; Johnston, D.; Kernohan, N.; Hopwood, D.; Dillon, J.F.; Hupp, T.R. The Human Oesophageal Squamous Epithelium Exhibits a Novel Type of Heat Shock Protein Response. Eur. J. Biochem. 2001, 268, 5343–5355.
- Chen, K.; Li, Y.; Dai, Y.; Li, J.; Qin, Y.; Zhu, Y.; Zeng, T.; Ban, X.; Fu, L.; Guan, X.-Y. Characterization of Tumor Suppressive Function of Cornulin in Esophageal Squamous Cell Carcinoma. PLoS ONE 2013, 8, e68838.
- Santosh, N.; McNamara, K.K.; Beck, F.M.; Kalmar, J.R. Expression of Cornulin in Oral Premalignant Lesions. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2019, 127, 526–534.
- Lee, J.-W.; Bae, S.-H.; Jeong, J.-W.; Kim, S.-H.; Kim, K.-W. Hypoxia-Inducible Factor (HIF-1)Alpha: Its Protein Stability and Biological Functions. Exp. Mol. Med. 2004, 36, 1–12.
- Patel, N.R.; Jain, L.; Mahajan, A.M.; Hiray, P.V.; Shinde, S.S.; Patel, P.A. An Immunohistochemical Study of HIF-1 Alpha in Oral Epithelial Dysplasia and Oral Squamous Cell Carcinoma. Indian J. Otolaryngol. Head Neck Surg. Off. Publ. Assoc. Otolaryngol. India 2019, 71, 435–441.
- Kleinert, H.; Forstermann, U. Inducible Nitric Oxide Synthase. In xPharm: The Comprehensive Pharmacology Reference; Enna, S.J., Bylund, D.B., Eds.; Elsevier: New York, NY, USA, 2007; pp. 1–12. ISBN 978-0-08-055232-3.
- Singh, D.N.; Srivastava, K.C.; Potsangbam, A.D.; Shrivastava, D.; Nandini, D.B.; Singh, W.T.; Singh, K.S. A Case-Control Study Comparing and Correlating iNOS Expression among Various Clinicopathological Variants of Oral Leukoplakia and Oral Squamous Cell Carcinoma: A Immunohistochemistry Study. J. Pharm. Bioallied Sci. 2020, 12, S324–S331.
- Turini, M.E.; DuBois, R.N. Cyclooxygenase-2: A Therapeutic Target. Annu. Rev. Med. 2002, 53, 35–57.
- Sharada, P.; Swaminathan, U.; Nagamalini, B.; Vinod Kumar, K.; Ashwini, B. Histoscore and Discontinuity Score—A Novel Scoring System to Evaluate Immunohistochemical Expression of COX-2 and Type IV Collagen in Oral Potentially Malignant Disorders and Oral Squamous Cell Carcinoma. J. Orofac. Sci. 2021, 13, 96–104.
- Schaller, M.D. Paxillin: A Focal Adhesion-Associated Adaptor Protein. Oncogene 2001, 20, 6459–6472.
- Alam, S.; Astekar, M.S.; Sapra, G.; Agarwal, A.; Agarwal, A.M.; Vishnu Rao, S.G. Immunohistochemical Expression of Paxillin in Potentially Malignant Disorders and Squamous Cell Carcinoma Patients. J. Oral Maxillofac. Pathol. JOMFP 2022, 26, 322–329.
- Soonthornthum, T.; Arias-Pulido, H.; Joste, N.; Lomo, L.; Muller, C.; Rutledge, T.; Verschraegen, C. Epidermal Growth Factor Receptor as a Biomarker for Cervical Cancer. Ann. Oncol. 2011, 22, 2166–2178.
- Kim, J.W.; Kim, Y.T.; Kim, D.K.; Song, C.H.; Lee, J.W. Expression of Epidermal Growth Factor Receptor in Carcinoma of the Cervix. Gynecol. Oncol. 1996, 60, 283–287.
- Fakurnejad, S.; van Keulen, S.; Nishio, N.; Engelen, M.; van den Berg, N.S.; Lu, G.; Birkeland, A.; Baik, F.; Colevas, A.D.; Rosenthal, E.L.; et al. Fluorescence Molecular Imaging for Identification of High-Grade Dysplasia in Patients with Head and Neck Cancer. Oral Oncol. 2019, 97, 50–55.
- Kawai, R.; Sugita, Y.; Suzumura, T.; Hattori, T.; Yoshida, W.; Kubo, K.; Maeda, H. Melanoma Inhibitory Activity and Melanoma Inhibitory Activity 2 as Novel Immunohistochemical Markers of Oral Epithelial Dysplasia. J. Clin. Med. 2021, 10, 3661.
- Ekblom, P.; Lonai, P.; Talts, J.F. Expression and Biological Role of Laminin-1. Matrix Biol. J. Int. Soc. Matrix Biol. 2003, 22, 35–47.
- Vageli, D.; Doukas, P.G.; Zacharouli, K.; Kakanis, V.; Strataki, M.; Zioga, A.; Skoulakis, C.; Koukoulis, G.; Ioannou, M. Laminin Immunostaining in Biopsies as a Useful Biomarker of Early Invasion in Actinic Cheilitis and Differential Diagnosis Between Actinic Cheilitis and Lip Cancer: New Insights. Head Neck Pathol. 2022, 17, 331–338.
- Nguyen, C.T.K.; Okamura, T.; Morita, K.-I.; Yamaguchi, S.; Harada, H.; Miki, Y.; Izumo, T.; Kayamori, K.; Yamaguchi, A.; Sakamoto, K. LAMC2 Is a Predictive Marker for the Malignant Progression of Leukoplakia. J. Oral Pathol. Med. Off. Publ. Int. Assoc. Oral Pathol. Am. Acad. Oral Pathol. 2017, 46, 223–231.
- Debta, P.; Sarode, G.; Siddhartha, S.; Sarode, S.; Debta, F.M.; Swain, S.K.; Sahu, M.C.; Patro, S.; Patil, S. GLUT-1 Expression: An Aid in Complementing the WHO Oral Epithelial Dysplasia Grading System. J. Contemp. Dent. Pract. 2020, 21, 951–955.
- Patlolla, P.; Shyam, N.D.V.; Kumar, G.K.; Narayen, V.; Konda, P.; Mudududla, P. Evaluation of Glucose Transporter-1 Expression in Oral Epithelial Dysplasia and Oral Squamous Cell Carcinoma: An Immunohistochemical Study. J. Oral Maxillofac. Pathol. JOMFP 2020, 24, 578.
- Udompatanakorn, C.; Taebunpakul, P. The Expression of Methyltransferase-Like 3 in Oral Precancerous Lesions and Oral Squamous Cell Carcinoma. Eur. J. Dent. 2022, 17, 349–356.
- Singh, D.; Nishi, K.; Khambata, K.; Balasinor, N.H. Introduction to Epigenetics: Basic Concepts and Advancements in the Field. In Epigenetics and Reproductive Health; Tollefsbol, T., Ed.; Translational Epigenetics; Academic Press: Cambridge, MA, USA, 2020; Volume 21, pp. xxv–xliv.
- Takisawa, H.; Mimura, S.; Kubota, Y. Eukaryotic DNA Replication: From Pre-Replication Complex to Initiation Complex. Curr. Opin. Cell Biol. 2000, 12, 690–696.
- Zakaria, S.H.; Farag, H.A.; Khater, D.S. Immunohistochemical Expression of MCM-2 in Oral Epithelial Dysplasias. Appl. Immunohistochem. Mol. Morphol. AIMM 2018, 26, 509–513.
