Photocatalysis, a promising semiconductor-based technology activated by free and eternal solar energy, has great potential for addressing environmental remediation and energy conversion challenges. Concentrated solar power (CSP) technologies, namely parabolic trough reflectors, solar power towers, parabolic dish reflectors and linear Fresnel reflectors, exhibited excellent feasibility for boosting solar-driven photocatalytic processes. Based on the structural characteristics of CSP technologies, the CSP-based photocatalytic reactors could be divided into concentrated types and non/low-concentrated types.
- concentrated solar power (CSP) technology
- photocatalysis
- solar energy
1. Introduction
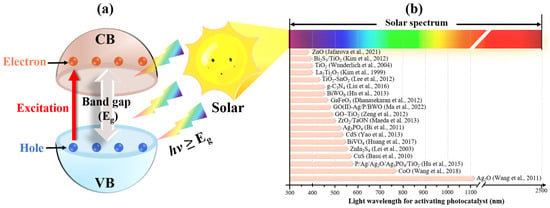
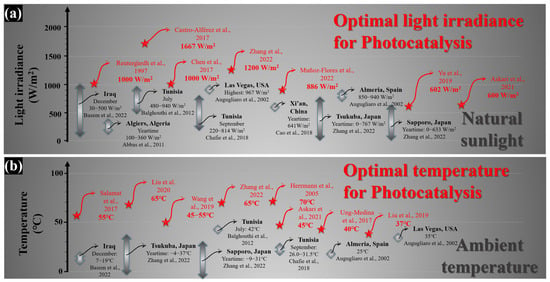
2. Feasibility of Solar Energy for Photocatalytic Applications
3. Brief Overview of Concentrated Solar Power Technologies
3.1. Parabolic Trough Reflectors
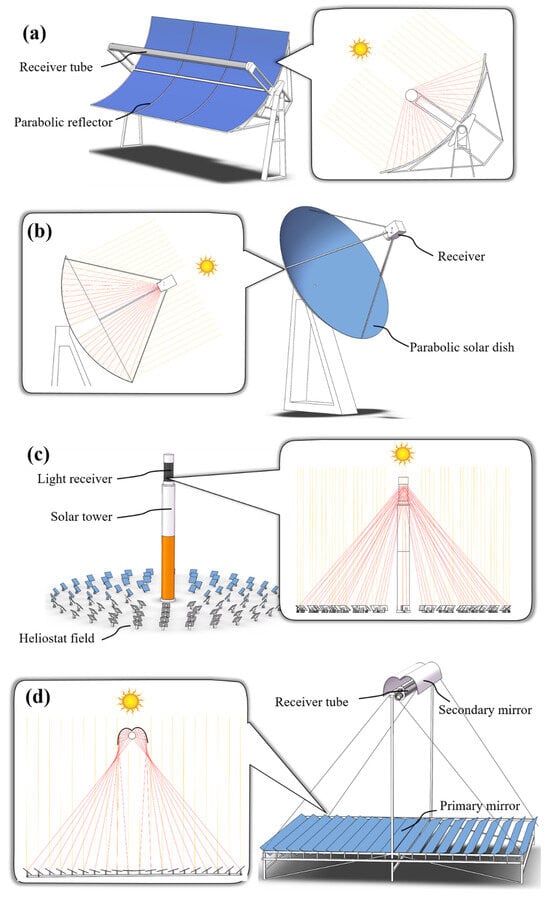
3.2. Parabolic Dish Reflectors
3.3. Solar Power Towers
3.4. Linear Fresnel Reflectors
4. CSP Technologies Applied in Photocatalysis
4.1. Solar Concentrated Type
4.1.1. Parabolic Trough Reflector-Based Photoreactors
4.1.2. Parabolic Dish Reflector-Based Photoreactors
4.1.3. Fresnel Condenser-Based Photoreactors
4.1.4. Problems in Solar-Concentrated Photoreactors

4.2. Non/Low-Concentrated Photoreactors
4.2.1. V-Groove-Based Photoreactors
4.2.2. Compound Parabolic Collector-Based Photoreactors
5. Relevant Problems in CSP-Based Photoreactors
5.1. Instability of Real Weather
5.2. Nighttime Operation
5.3. Configuration of Photocatalyst Immobilization Substrate
5.4. Economic Analysis
6. Conclusions
This entry is adapted from the peer-reviewed paper 10.3390/en17020463
References
- Zhang, C.; Ming, J.; Sun, X.; Zhu, Y.; An, G.; Chen, G.; Yang, Y. Development of a Green and Efficient Photocatalytic Mesh Microalgae Biorefinery (PMMB) System for Sustainable Biomass Conversion under Real Solar Light. Chem. Eng. J. 2023, 466, 143260.
- Akpan, J.O.O. Sustainable Energy Development: History and Recent Advances. Energies 2023, 16, 7049.
- Ming, J.; Sun, X.; Ma, Q.; Liu, N.; Zhang, C.; Kawazoe, N.; Chen, G.; Yang, Y. Advanced Photocatalytic Sterilization for Recalcitrant Enterococcus Sp. Contaminated Water by Newly Developed Z-Scheme Bi2WO6 Based Composites under Solar Light. Chemosphere 2023, 310, 136912.
- Ma, Q.; Hu, X.; Liu, N.; Sharma, A.; Zhang, C.; Kawazoe, N.; Chen, G.; Yang, Y. Polyethylene Glycol (PEG)-Modified Ag/Ag2O/Ag3PO4/Bi2WO6 Photocatalyst Film with Enhanced Efficiency and Stability under Solar Light. J. Colloid Interface Sci. 2020, 569, 101–113.
- Zhu, Q.; Liu, N.; Ma, Q.; Sharma, A.; Nagai, D.; Sun, X.; Zhang, C.; Yang, Y. Sol-Gel/Hydrothermal Two-Step Synthesis Strategy for Promoting Ag Species–Modified TiO2-Based Composite Activity toward H2 Evolution under Solar Light. Mater. Today Energy 2021, 20, 100648.
- Hu, X.; Ma, Q.; Wang, X.; Yang, Y.; Liu, N.; Zhang, C.; Kawazoe, N.; Chen, G.; Yang, Y. Layered Ag/Ag2O/BiPO4/Bi2WO6 Heterostructures by Two-Step Method for Enhanced Photocatalysis. J. Catal. 2020, 387, 28–38.
- Zhang, C.; Liu, N.; Ming, J.; Sharma, A.; Ma, Q.; Liu, Z.; Chen, G.; Yang, Y. Development of a Novel Solar Energy Controllable Linear Fresnel Photoreactor (LFP) for High-Efficiency Photocatalytic Wastewater Treatment under Actual Weather. Water Res. 2022, 208, 117880.
- Bi, Y.; Ouyang, S.; Umezawa, N.; Cao, J.; Ye, J. Facet Effect of Single-Crystalline Ag3PO4 Sub-Microcrystals on Photocatalytic Properties. J. Am. Chem. Soc. 2011, 4, 6490–6492.
- Portela, R.; Suárez, S.; Tessinari, R.F.; Hernández-Alonso, M.D.; Canela, M.C.; Sánchez, B. Solar/Lamp-Irradiated Tubular Photoreactor for Air Treatment with Transparent Supported Photocatalysts. Appl. Catal. B Environ. 2011, 105, 95–102.
- Liu, N.; Zhu, Q.; Zhang, N.; Zhang, C.; Kawazoe, N.; Chen, G.; Negishi, N.; Yang, Y. Superior Disinfection Effect of Escherichia Coli by Hydrothermal Synthesized TiO2-Based Composite Photocatalyst under LED Irradiation: Influence of Environmental Factors and Disinfection Mechanism. Environ. Pollut. 2019, 247, 847–856.
- Zhang, G.; Lan, Z.A.; Lin, L.; Lin, S.; Wang, X. Overall Water Splitting by Pt/g-C3N4 Photocatalysts without Using Sacrificial Agents. Chem. Sci. 2016, 7, 3062–3066.
- Liu, J.; Liu, Y.; Liu, N.; Han, Y.; Zhang, X.; Huang, H.; Lifshitz, Y.; Lee, S.T.; Zhong, J.; Kang, Z. Metal-Free Efficient Photocatalyst for Stable Visible Water Splitting via a Two-Electron Pathway. Science 2015, 347, 970–974.
- Reli, M.; Edelmannová, M.; Šihor, M.; Praus, P.; Svoboda, L.; Mamulová, K.K.; Otoupalíková, H.; Čapek, L.; Hospodková, A.; Obalová, L.; et al. Photocatalytic H2 Generation from Aqueous Ammonia Solution Using ZnO Photocatalysts Prepared by Different Methods. Int. J. Hydrogen Energy 2015, 40, 8530–8538.
- Kominami, H.; Nishimune, H.; Ohta, Y.; Arakawa, Y.; Inaba, T. Photocatalytic Hydrogen Formation from Ammonia and Methyl Amine in an Aqueous Suspension of Metal-Loaded Titanium(IV) Oxide Particles. Appl. Catal. B Environ. 2012, 111–112, 297–302.
- Vidyasagar, D.; Ghugal, S.G.; Kulkarni, A.; Mishra, P.; Shende, A.G.; Jagannath; Umare, S.S.; Sasikala, R. Silver/Silver(II) Oxide (Ag/AgO) Loaded Graphitic Carbon Nitride Microspheres: An Effective Visible Light Active Photocatalyst for Degradation of Acidic Dyes and Bacterial Inactivation. Appl. Catal. B Environ. 2018, 221, 339–348.
- Deng, J.; Liang, J.; Li, M.; Tong, M. Enhanced Visible-Light-Driven Photocatalytic Bacteria Disinfection by g-C3N4-AgBr. Colloids Surf. B Biointerfaces 2017, 152, 49–57.
- Braham, R.J.; Harris, A.T. Review of Major Design and Scale-up Considerations for Solar Photocatalytic Reactors. Ind. Eng. Chem. Res. 2009, 48, 8890–8905.
- Ajbar, W.; Hernández, J.A.; Parrales, A.; Torres, L. Thermal Efficiency Improvement of Parabolic Trough Solar Collector Using Different Kinds of Hybrid Nanofluids. Case Stud. Therm. Eng. 2023, 42, 102759.
- Desai, N.B.; Bandyopadhyay, S. Optimization of Concentrating Solar Thermal Power Plant Based on Parabolic Trough Collector. J. Clean. Prod. 2015, 89, 262–271.
- Ma, Q.; Ming, J.; Sun, X.; Liu, N.; Chen, G.; Yang, Y. Visible Light Active Graphene Oxide Modified Ag/Ag2O/BiPO4/Bi2WO6 for Photocatalytic Removal of Organic Pollutants and Bacteria in Wastewater. Chemosphere 2022, 306, 135512.
- Jafarova, V.N.; Orudzhev, G.S. Structural and Electronic Properties of ZnO: A First-Principles Density-Functional Theory Study within LDA(GGA) and LDA(GGA)+U Methods. Solid State Commun. 2021, 325, 114166.
- Huang, C.K.; Wu, T.; Huang, C.W.; Lai, C.Y.; Wu, M.Y.; Lin, Y.W. Enhanced Photocatalytic Performance of BiVO4 in Aqueous AgNO3 Solution under Visible Light Irradiation. Appl. Surf. Sci. 2017, 399, 10–19.
- Lee, S.S.; Bai, H.; Liu, Z.; Sun, D.D. Electrospun TiO2/SnO2 Nanofibers with Innovative Structure and Chemical Properties for Highly Efficient Photocatalytic H2 Generation. Int. J. Hydrogen Energy 2012, 37, 10575–10584.
- Yao, W.; Song, X.; Huang, C.; Xu, Q.; Wu, Q. Enhancing Solar Hydrogen Production via Modified Photochemical Treatment of Pt/CdS Photocatalyst. Catal. Today 2013, 199, 42–47.
- Zeng, P.; Zhang, Q.; Zhang, X.; Peng, T. Graphite Oxide-TiO2 Nanocomposite and Its Efficient Visible-Light-Driven Photocatalytic Hydrogen Production. J. Alloys Compd. 2012, 516, 85–90.
- Kim, J.; Kang, M. High Photocatalytic Hydrogen Production over the Band Gap-Tuned Urchin-like Bi2S3-Loaded TiO2 Composites System. Int. J. Hydrogen Energy 2012, 37, 8249–8256.
- Liu, J.; Cheng, B.; Yu, J. A New Understanding of the Photocatalytic Mechanism of the Direct Z-Scheme g-C3N4/TiO2 Heterostructure. Phys. Chem. Chem. Phys. 2016, 18, 31175–31183.
- Wunderlich, W.; Oekermann, T.; Miao, L.; Hue, N.T.; Tanemura, S.; Tanemura, M. Electronic Properties of Nano-Porous TiO2- and ZnO-Thin Films-Comparison of Simulations and Experiments. J. Ceram. Process. Res. 2004, 5, 343–354.
- Lei, Z.; You, W.; Liu, M.; Zhou, G.; Takata, T.; Hara, M.; Domen, K.; Li, C. Photocatalytic Water Reduction under Visible Light on a Novel ZnIn2S4 Catalyst Synthesized by Hydrothermal Method. Chem. Commun. 2003, 3, 2142–2143.
- Wang, X.; Li, S.; Yu, H.; Yu, J.; Liu, S. Ag2O as a New Visible-Light Photocatalyst: Self-Stability and High Photocatalytic Activity. Chem. A Eur. J. 2011, 17, 7777–7780.
- Hu, X.; Zhu, Q.; Wang, X.; Kawazoe, N.; Yang, Y. Nonmetal-Metal-Semiconductor-Promoted P/Ag/Ag2O/Ag3PO4/TiO2 Photocatalyst with Superior Photocatalytic Activity and Stability. J. Mater. Chem. A 2015, 3, 17858–17865.
- Maeda, K.; Lu, D.; Domen, K. Direct Water Splitting into Hydrogen and Oxygen under Visible Light by Using Modified Taon Photocatalysts with D0 Electronic Configuration. Chem. A Eur. J. 2013, 19, 4986–4991.
- Kim, H.G.; Hwang, D.W.; Kim, J.; Kim, Y.G.; Lee, J.S. Highly Donor-Doped (110) Layered Perovskite Materials as Novel Photocatalysts for Overall Water Splitting. Chem. Commun. 1999, 2, 1077–1078.
- Dhanasekaran, P.; Gupta, N.M. Factors Affecting the Production of H2 by Water Splitting over a Novel Visible-Light-Driven Photocatalyst GaFeO3. Int. J. Hydrogen Energy 2012, 37, 4897–4907.
- Wang, Y.; Ge, H.X.; Chen, Y.P.; Meng, X.Y.; Ghanbaja, J.; Horwat, D.; Pierson, J.F. Wurtzite CoO: A Direct Band Gap Oxide Suitable for a Photovoltaic Absorber. Chem. Commun. 2018, 54, 13949–13952.
- Hu, S.P.; Xu, C.Y.; Zhen, L. Solvothermal Synthesis of Bi2WO6 Hollow Structures with Excellent Visible-Light Photocatalytic Properties. Mater. Lett. 2013, 95, 117–120.
- Basu, M.; Sinha, A.K.; Pradhan, M.; Sarkar, S.; Negishi, Y.; Govind; Pal, T. Evolution of Hierarchical Hexagonal Stacked Plates of CuS from Liquid—Liquid Interface and Its Photocatalytic Application for Oxidative Degradation of Different Dyes under Indoor Lighting. Environ. Sci. Technol. 2010, 44, 6313–6318.
- Muñoz-Flores, P.; Poon, P.S.; Ania, C.O.; Matos, J. Performance of a C-Containing Cu-Based Photocatalyst for the Degradation of Tartrazine: Comparison of Performance in a Slurry and CPC Photoreactor under Artificial and Natural Solar Light. J. Colloid Interface Sci. 2022, 623, 646–659.
- Reutergårdh, L.B.; Iangphasuk, M. Photocatalytic Decolourization of Reactive Azo Dye: A Comparison between TiO2 and CdS Photocatalysis. Chemosphere 1997, 35, 585–596.
- Askari, N.; Beheshti, M.; Mowla, D.; Farhadian, M. Facile Construction of Novel Z-Scheme MnWO4/Bi2S3 Heterojunction with Enhanced Photocatalytic Degradation of Antibiotics. Mater. Sci. Semicond. Process. 2021, 127, 105723.
- Yu, J.; Kiwi, J.; Zivkovic, I.; Rønnow, H.M.; Wang, T.; Rtimi, S. Quantification of the Local Magnetized Nanotube Domains Accelerating the Photocatalytic Removal of the Emerging Pollutant Tetracycline. Appl. Catal. B Environ. 2019, 248, 450–458.
- Castro-Alférez, M.; Polo-López, M.I.; Marugán, J.; Fernández-Ibáñez, P. Mechanistic Model of the Escherichia Coli Inactivation by Solar Disinfection Based on the Photo-Generation of Internal ROS and the Photo-Inactivation of Enzymes: CAT and SOD. Chem. Eng. J. 2017, 318, 214–223.
- Chen, Y.; Wang, Y.; Li, W.; Yang, Q.; Hou, Q.; Wei, L.; Liu, L.; Huang, F.; Ju, M. Enhancement of Photocatalytic Performance with the Use of Noble-Metal-Decorated TiO2 Nanocrystals as Highly Active Catalysts for Aerobic Oxidation under Visible-Light Irradiation. Appl. Catal. B Environ. 2017, 210, 352–367.
- Abbas, M.; Boumeddane, B.; Said, N.; Chikouche, A. Dish Stirling Technology: A 100 MW Solar Power Plant Using Hydrogen for Algeria. Int. J. Hydrogen Energy 2011, 36, 4305–4314.
- Balghouthi, M.; Chahbani, M.H.; Guizani, A. Investigation of a Solar Cooling Installation in Tunisia. Appl. Energy 2012, 98, 138–148.
- Augugliaro, V.; Baiocchi, C.; Prevot, A.B.; García-López, E.; Loddo, V.; Malato, S.; Marcí, G.; Palmisano, L.; Pazzi, M.; Pramauro, E. Azo-Dyes Photocatalytic Degradation in Aqueous Suspension of TiO2 under Solar Irradiation. Chemosphere 2002, 49, 1223–1230.
- Chafie, M.; Ben Aissa, M.F.; Guizani, A. Energetic End Exergetic Performance of a Parabolic Trough Collector Receiver: An Experimental Study. J. Clean. Prod. 2018, 171, 285–296.
- Cao, F.; Wei, Q.; Liu, H.; Lu, N.; Zhao, L.; Guo, L. Development of the Direct Solar Photocatalytic Water Splitting System for Hydrogen Production in Northwest China: Design and Evaluation of Photoreactor. Renew. Energy 2018, 121, 153–163.
- Basem, A.; Moawed, M.; Abbood, M.H.; El-Maghlany, W.M. The Energy and Exergy Analysis of a Combined Parabolic Solar Dish—Steam Power Plant. Renew. Energy Focus 2022, 41, 55–68.
- Salamat, S.; Younesi, H.; Bahramifar, N. Synthesis of Magnetic Core-Shell Fe3O4@TiO2 Nanoparticles from Electric Arc Furnace Dust for Photocatalytic Degradation of Steel Mill Wastewater. RSC Adv. 2017, 7, 19391–19405.
- Ung-Medina, F.; Caudillo-Flores, U.; Correa-González, J.C.; Maya-Yescas, R.; Chávez-Parga, M.D.C.; Cortés, J.A. Use of an Annular Non-Sleeve Photoreactor for Photocatalytic Dye Degradation: Study of Temperature and Light Intensity Effects. Environ. Prog. Sustain. Energy 2017, 36, 1083–1088.
- Wang, H.; Sun, Y.; Wu, Y.; Tu, W.; Wu, S.; Yuan, X.; Zeng, G.; Xu, Z.J.; Li, S.; Chew, J.W. Electrical Promotion of Spatially Photoinduced Charge Separation via Interfacial-Built-in Quasi-Alloying Effect in Hierarchical Zn2In2S5/Ti3C2(O, OH)x Hybrids toward Efficient Photocatalytic Hydrogen Evolution and Environmental Remediation. Appl. Catal. B Environ. 2019, 245, 290–301.
- Liu, B.; Wu, H.; Parkin, I.P. New Insights into the Fundamental Principle of Semiconductor Photocatalysis. ACS Omega 2020, 5, 14847–14856.
- Herrmann, J.M. Heterogeneous Photocatalysis: State of the Art and Present Applications In honor of Pr. R.L. Burwell Jr. (1912–2003), Former Head of Ipatieff Laboratories, Northwestern University, Evanston (Ill). Top. Catal. 2005, 34, 49–65.
- Al-Soud, M.S.; Hrayshat, E.S. A 50 MW Concentrating Solar Power Plant for Jordan. J. Clean. Prod. 2009, 17, 625–635.
- Balzar, A.; Stumpf, P.; Eckhoff, S.; Ackermann, H.; Grupp, M. A Solar Cooker Using Vacuum-Tube Collectors with Integrated Heat Pipes. Sol. Energy 1996, 58, 63–68.
- Wu, S.Y.; Xiao, L.; Cao, Y.; Li, Y.R. A Parabolic Dish/AMTEC Solar Thermal Power System and Its Performance Evaluation. Appl. Energy 2010, 87, 452–462.
- Imran Khan, M.; Asfand, F.; Al-Ghamdi, S.G. Progress in Technology Advancements for next Generation Concentrated Solar Power Using Solid Particle Receivers. Sustain. Energy Technol. Assess. 2022, 54, 102813.
- Wang, Q.; Yao, Y.; Shen, Z.; Hu, M.; Yang, H. Concentrated Solar Power Tower Systems Coupled Locally with Spectrally Selective Coatings for Enhancement of Solar-Thermal Conversion and Economic Performance. Green Energy Resour. 2023, 1, 100001.
- González-Mora, E.; Dolores Durán García, M. Methodology for an Opto-Geometric Optimization of a Linear Fresnel Reflector for Direct Steam Generation. Energies 2020, 13, 355.
- Sebastián, A.; Abbas, R.; Valdés, M.; Casanova, J. Innovative Thermal Storage Strategies for Fresnel-Based Concentrating Solar Plants with East-West Orientation. Appl. Energy 2018, 230, 983–995.
- Mills, D.R.; Morrison, G.L. Compact Linear Fresnel Reflector Solar Thermal Powerplants. Sol. Energy 2000, 68, 263–283.
- Ochoa-Gutiérrez, K.S.; Tabares-Aguilar, E.; Mueses, M.Á.; Machuca-Martínez, F.; Li Puma, G. A Novel Prototype Offset Multi Tubular Photoreactor (OMTP) for Solar Photocatalytic Degradation of Water Contaminants. Chem. Eng. J. 2018, 341, 628–638.
- Anderson, J.V.; Link, H.; Bohn, M.; Gupta, B. Development of Solar Detoxification Technology in the USA—An Introduction. Sol. Energy Mater. 1991, 24, 538–549.
- Minero, C.; Pelizzetti, E.; Malato, S.; Blanco, J. Large Solar Plant Photocatalytic Water Decontamination: Degradation of Pentachlorophenol. Chemosphere 1993, 26, 2103–2119.
- Malato, S.; Blanco, J.; Richter, C.; Curco, D.; Gimenez, J. Low-Concentrating CPC Collectors for Photocatalytic Water Detoxification: Comparison with a Medium Concentrating Solar Collector. Water Sci. Technol. 1997, 35, 157–164.
- Klare, M.; Scheen, J.; Vogelsang, K.; Jacobs, H.; Broekaert, J.A.C. Degradation of Short-Chain Alkyl- and Alkanolamines by TiO2- and Pt/TiO2-Assisted Photocatalysis. Chemosphere 2000, 41, 353–362.
- Sano, T.; Negishi, N.; Takeuchi, K.; Matsuzawa, S. Degradation of Toluene and Acetaldehyde with Pt-Loaded TiO2 Catalyst and Parabolic Trough Concentrator. Sol. Energy 2004, 77, 543–552.
- Bandala, E.R.; Arancibia-Bulnes, C.A.; Orozco, S.L.; Estrada, C.A. Solar Photoreactors Comparison Based on Oxalic Acid Photocatalytic Degradation. Sol. Energy 2004, 77, 503–512.
- McLoughlin, O.A.; Kehoe, S.C.; McGuigan, K.G.; Duffy, E.F.; Al Touati, F.; Gernjak, W.; Oller Alberola, I.; Malato Rodríguez, S.; Gill, L.W. Solar Disinfection of Contaminated Water: A Comparison of Three Small-Scale Reactors. Sol. Energy 2004, 77, 657–664.
- Barzegar, M.H.; Sabzehmeidani, M.M.; Ghaedi, M.; Avargani, V.M.; Moradi, Z.; Roy, V.A.L.; Heidari, H. S-Scheme Heterojunction g-C3N4/TiO2 with Enhanced Photocatalytic Activity for Degradation of a Binary Mixture of Cationic Dyes Using Solar Parabolic Trough Reactor. Chem. Eng. Res. Des. 2021, 174, 307–318.
- Minero, C.; Pelizzetti, E.; Malato, S.; Blanco, J. Large Solar Plant Photocatalytic Water Decontamination: Degradation of Atrazine. Sol. Energy 1996, 56, 411–419.
- Oyama, T.; Aoshima, A.; Horikoshi, S.; Hidaka, H.; Zhao, J.; Serpone, N. Solar Photocatalysis, Photodegradation of a Commercial Detergent in Aqueous TiO2 Dispersions under Sunlight Irradiation. Sol. Energy 2004, 77, 525–532.
- Malato, S.; Blanco, J.; Vidal, A.; Richter, C. Photocatalysis with Solar Energy at a Pilot-Plant Scale: An Overview. Appl. Catal. B Environ. 2002, 37, 1–15.
- Fernández-García, A.; Rojas, E.; Pérez, M.; Silva, R.; Hernández-Escobedo, Q.; Manzano-Agugliaro, F. A Parabolic-Trough Collector for Cleaner Industrial Process Heat. J. Clean. Prod. 2015, 89, 272–285.
- Cao, F.; Pang, J.; Gu, X.; Wang, M.; Shangguan, Y. Performance Simulation of Solar Trough Concentrators: Optical and Thermal Comparisons. Energies 2023, 16, 1673.
- Mussard, M.; Nydal, O.J. Charging of a Heat Storage Coupled with a Low-Cost Small-Scale Solar Parabolic Trough for Cooking Purposes. Sol. Energy 2013, 95, 144–154.
- Stanek, B.; Węcel, D.; Bartela, Ł.; Rulik, S. Solar Tracker Error Impact on Linear Absorbers Efficiency in Parabolic Trough Collector—Optical and Thermodynamic Study. Renew. Energy 2022, 196, 598–609.
- Beltran, R.; Velazquez, N.; Espericueta, A.C.; Sauceda, D.; Perez, G. Mathematical Model for the Study and Design of a Solar Dish Collector with Cavity Receiver for Its Application in Stirling Engines. J. Mech. Sci. Technol. 2012, 26, 3311–3321.
- Nepveu, F.; Ferriere, A.; Bataille, F. Thermal Model of a Dish/Stirling Systems. Sol. Energy 2009, 83, 81–89.
- El Ouederni, A.R.; Salah, M.B.; Askri, F.; Nasrallah, M.B.; Aloui, F. Experimental Study of a Parabolic Solar Concentrator. J. Renew. Energ. 2009, 12, 395–404.
- Abbas, R.; Montes, M.J.; Piera, M.; Martínez-Val, J.M. Solar Radiation Concentration Features in Linear Fresnel Reflector Arrays. Energy Convers. Manag. 2012, 54, 133–144.
- Geyer, M.; Lüpfert, E.; Osuna, R.; Nava, P.; Langenkamp, J.; Mandelberg, E. EUROTROUGH-Parabolic Trough Collector Developed for Cost Efficient Solar Power Generation. In Proceedings of the 11th International Symposium on Concentrating Solar Power and Chemical Energy Technologies, Zurich, Switzerland, 4–6 September 2002; Volume 7.
- Sharma, A.K.; Sharma, C.; Mullick, S.C.; Kandpal, T.C. GHG Mitigation Potential of Solar Industrial Process Heating in Producing Cotton Based Textiles in India. J. Clean. Prod. 2017, 145, 74–84.
- Palenzuela, P.; Zaragoza, G.; Alarcón-Padilla, D.C.; Guillén, E.; Ibarra, M.; Blanco, J. Assessment of Different Configurations for Combined Parabolic-Trough (PT) Solar Power and Desalination Plants in Arid Regions. Energy 2011, 36, 4950–4958.
- Peiró, G.; Prieto, C.; Gasia, J.; Jové, A.; Miró, L.; Cabeza, L.F. Two-Tank Molten Salts Thermal Energy Storage System for Solar Power Plants at Pilot Plant Scale: Lessons Learnt and Recommendations for Its Design, Start-up and Operation. Renew. Energy 2018, 121, 236–248.
- de Risi, A.; Milanese, M.; Laforgia, D. Modelling and Optimization of Transparent Parabolic Trough Collector Based on Gas-Phase Nanofluids. Renew. Energy 2013, 58, 134–139.
- Linares, J.I.; Montes, M.J.; Cantizano, A.; Sánchez, C. A Novel Supercritical CO2 Recompression Brayton Power Cycle for Power Tower Concentrating Solar Plants. Appl. Energy 2020, 263, 114644.
- Zhuang, X.; Wang, H.; Lu, H.; Yang, Z.; Guo, H. Numerical Investigation of Heat Transfer and Flow Characteristics of Supercritical CO2 in Solar Tower Microchannel Receivers at High Temperature. Energies 2023, 16, 6445.
- Pino, F.J.; Caro, R.; Rosa, F.; Guerra, J. Experimental Validation of an Optical and Thermal Model of a Linear Fresnel Collector System. Appl. Therm. Eng. 2013, 50, 1463–1471.
- Beltagy, H.; Semmar, D.; Lehaut, C.; Said, N. Theoretical and Experimental Performance Analysis of a Fresnel Type Solar Concentrator. Renew. Energy 2017, 101, 782–793.
- Mills, D.R.; Morrison, G.L. Modelling Study for Compact Fresnel Reflector Power Plant. Le J. De Phys. IV 1999, 9, Pr3-159–Pr3-165.
- Vidal, A.; Díaz, A.I.; El Hraiki, A.; Romero, M.; Muguruza, I.; Senhaji, F.; González, J. Solar Photocatalysis for Detoxification and Disinfection of Contaminated Water: Pilot Plant Studies. Catal. Today 1999, 54, 283–290.
- Kositzi, M.; Poulios, I.; Malato, S.; Caceres, J.; Campos, A. Solar Photocatalytic Treatment of Synthetic Municipal Wastewater. Water Res. 2004, 38, 1147–1154.
- Fernández, P.; Blanco, J.; Sichel, C.; Malato, S. Water Disinfection by Solar Photocatalysis Using Compound Parabolic Collectors. Catal. Today 2005, 101, 345–352.
- Augugliaro, V.; García-López, E.; Loddo, V.; Malato-Rodríguez, S.; Maldonado, I.; Marcì, G.; Molinari, R.; Palmisano, L. Degradation of Lincomycin in Aqueous Medium: Coupling of Solar Photocatalysis and Membrane Separation. Sol. Energy 2005, 79, 402–408.
- Villén, L.; Manjón, F.; García-Fresnadillo, D.; Orellana, G. Solar Water Disinfection by Photocatalytic Singlet Oxygen Production in Heterogeneous Medium. Appl. Catal. B Environ. 2006, 69, 1–9.
- Sichel, C.; Tello, J.; de Cara, M.; Fernández-Ibáñez, P. Effect of UV Solar Intensity and Dose on the Photocatalytic Disinfection of Bacteria and Fungi. Catal. Today 2007, 129, 152–160.
- Alrousan, D.M.A.; Polo-López, M.I.; Dunlop, P.S.M.; Fernández-Ibáñez, P.; Byrne, J.A. Solar Photocatalytic Disinfection of Water with Immobilised Titanium Dioxide in Re-Circulating Flow CPC Reactors. Appl. Catal. B Environ. 2012, 128, 126–134.
- Quiñones, D.H.; Álvarez, P.M.; Rey, A.; Beltrán, F.J. Removal of Emerging Contaminants from Municipal WWTP Secondary Effluents by Solar Photocatalytic Ozonation. A Pilot-Scale Study. Sep. Purif. Technol. 2015, 149, 132–139.
- Colina-Márquez, J.; Machuca-Martínez, F.; Puma, G.L. Modeling the Photocatalytic Mineralization in Water of Commercial Formulation of Estrogens 17-β Estradiol (E2) and Nomegestrol Acetate in Contraceptive Pills in a Solar Powered Compound Parabolic Collector. Molecules 2015, 20, 13354–13373.
- Otálvaro-Marín, H.L.; Mueses, M.A.; Crittenden, J.C.; Machuca-Martinez, F. Solar Photoreactor Design by the Photon Path Length and Optimization of the Radiant Field in a TiO2-Based CPC Reactor. Chem. Eng. J. 2017, 315, 283–295.
- Aguas, Y.; Hincapie, M.; Fernández-Ibáñez, P.; Polo-López, M.I. Solar Photocatalytic Disinfection of Agricultural Pathogenic Fungi (Curvularia Sp.) in Real Urban Wastewater. Sci. Total Environ. 2017, 607–608, 1213–1224.
- Haranaka-Funai, D.; Didier, F.; Giménez, J.; Marco, P.; Esplugas, S.; Machulek-Junior, A. Photocatalytic Treatment of Valproic Acid Sodium Salt with TiO2 in Different Experimental Devices: An Economic and Energetic Comparison. Chem. Eng. J. 2017, 327, 656–665.
- Moreira, N.F.F.; Narciso-da-Rocha, C.; Polo-López, M.I.; Pastrana-Martínez, L.M.; Faria, J.L.; Manaia, C.M.; Fernández-Ibáñez, P.; Nunes, O.C.; Silva, A.M.T. Solar Treatment (H2O2, TiO2-P25 and GO-TiO2 Photocatalysis, Photo-Fenton) of Organic Micropollutants, Human Pathogen Indicators, Antibiotic Resistant Bacteria and Related Genes in Urban Wastewater. Water Res. 2018, 135, 195–206.
- López, N.; Marco, P.; Giménez, J.; Esplugas, S. Photocatalytic Diphenhydramine Degradation under Different Radiation Sources: Kinetic Studies and Energetic Comparison. Appl. Catal. B Environ. 2018, 220, 497–505.
- Vela, N.; Calín, M.; Yáñez-Gascón, M.J.; Garrido, I.; Pérez-Lucas, G.; Fenoll, J.; Navarro, S. Solar Reclamation of Wastewater Effluent Polluted with Bisphenols, Phthalates and Parabens by Photocatalytic Treatment with TiO2/Na2S2O8 at Pilot Plant Scale. Chemosphere 2018, 212, 95–104.
- Luna-Sanguino, G.; Ruíz-Delgado, A.; Tolosana-Moranchel, A.; Pascual, L.; Malato, S.; Bahamonde, A.; Faraldos, M. Solar Photocatalytic Degradation of Pesticides over TiO2-RGO Nanocomposites at Pilot Plant Scale. Sci. Total Environ. 2020, 737, 140286.
- Zheng, Q.; Aiello, A.; Choi, Y.S.; Tarr, K.; Shen, H.; Durkin, D.P.; Shuai, D. 3D Printed Photoreactor with Immobilized Graphitic Carbon Nitride: A Sustainable Platform for Solar Water Purification. J. Hazard. Mater. 2020, 399, 123097.
