The mortality rate of acute respiratory distress syndrome (ARDS) is still very high, and the remission and treatment of ARDS are still the focus of research. The causes of acute respiratory distress syndrome are varied, with pneumonia and non-pulmonary sepsis being the most common. Trauma and blood transfusion can also cause acute respiratory distress syndrome. In ARDS, the accumulation and infiltration of neutrophils in the lungs have a great influence on the development of the disease. Neutrophils regulate inflammatory responses through various pathways, and neutrophils release via neutrophilic extracellular traps (NETs) is considered to be one of the most important mechanisms.
- ARDS
- ALI
- NETs
- NETosis
- COVID-19
- inflammatory storm
1. Introduction
Acute respiratory distress syndrome (ARDS) is the acute onset of respiratory disease characterized by bilateral pulmonary edema and hypoxemia of noncardiac origin caused by damage to the pulmonary endothelial barrier and excessive permeability of alveolar capillaries, which is clinically manifested as bilateral pulmonary infiltrates and respiratory failure [1]. The Berlin definition classifies ARDS as mild, moderate, or severe based on the level of pulmonary oxygenation, and removes the definition of acute lung injury (ALI) [2]. The most common causes of ARDS include pneumonia and non-pulmonary sepsis, as well as the aspiration of the stomach contents, trauma, pancreatitis, burns, inhalation injury, drug overdose, multiple blood transfusions or shock, e-cigarettes, chemotherapy and immunotherapy (including checkpoint inhibitors), etc. [3,4]. A 2016 observational study covering 459 intensive care units (ICU) in 50 countries showed that 10% of ICU patients and 23% of mechanically ventilated patients had ARDS, with a 28-day mortality rate of 35% and a mortality rate of more than 40% for patients with severe ARDS [5]. In addition to a high incidence and mortality, patients with ARDS may have a sequela of physical, cognitive, and mental health conditions after recovery [3]. Alveoli neutrophil infiltration, which causes persistent inflammation, is one of the of ARDS [6] (Figure 1).
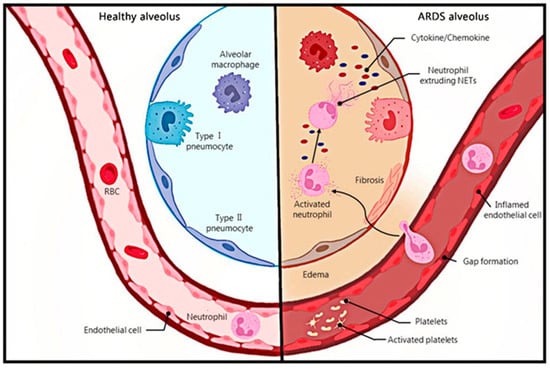
Neutrophils, a major component of innate immunity and the host’s first line of defense against infectious pathogens, are recruited at the onset of infection and destroy pathogens through mechanisms such as phagocytosis, degranulation, production of reactive oxygen species, release of antimicrobial peptides, and the recently discovered formation of neutrophil extracellular traps (NET) [7]. NETs are produced by activated neutrophils and consist of DNA, histones, granular proteins such as neutrophil elastase, cathepsin G, and myeloperoxidase [8]. The process of NET formation by neutrophils, known as NETosis, is a novel mechanism of programmed cell death of neutrophils, and can be divided into NADPH oxidase 2 (NOX)-dependent and independent NETosis [9](Figure 2A). NETs can be induced by IL-8, LPS, etc., and have strong antibacterial effects [8]. In addition, NETs can also mediate tissue damage [10], cancer [11], inflammation [12], and autoimmune diseases [13]. Recently, the association between NETs and ARDS has been confirmed by a number of studies. This review describes the interaction between NETs and ARDS, and discusses the relationship between NETs and inflammatory storms and immune balance, while focusing on the production of NETs and the effect on the progression of ARDS disease.
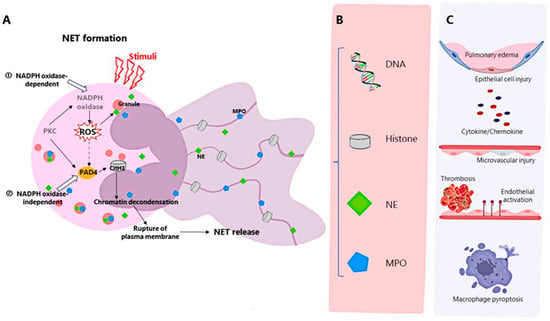
Formation of NETs, components of NETs and mechanisms affecting ARDS. (A) The formation of NETs can be divided into NADPH oxidase-dependent and independent formations. ① Activation of PKC leads to activation of NADPH oxidase and the production of ROS. ROS stimulates MPO and NE to migrate to the nucleus, NE triggers histone degradation, and MPO collaborates with NE to promote chromatin decondensation. ② Activated PAD4-mediated histone citrullination leads to chromatin decondensation[9]. (B) The major components of NETs include DNA, histones, NE and MPO. (C) NETs and their components can cause pulmonary tissue edema, damage alveolar epithelial cells and microvessels, cause the formation of immune thrombosis and activation of endothelial cells, and mediate the pyroptosis of macrophages and the release of downstream cytokines.
2. Evidence on the Influence of NETs on ARDS
3. The Mechanism of NETs Influencing ARDS
3.1. Histones
3.2. NE
3.3. DNA
3.4. MPO
4. The Mechanism of NETs Production in ARDS and the Substances That Affect the Production
5. Relationship between the Inflammatory Storm and NETs
6. Correlation between Immune Balance and Regulation in ARDS and NETs
7. Production and Role of NETs in COVID-19 ARDS
8. Clinical Research
9. Treatment
11. Conclusions
This entry is adapted from the peer-reviewed paper 10.3390/ijms25031464
