Chitosan is a biopolymer derived from chitin that gained much attention due to its biological activities. Chitosan can be produced by the exoskeleton of arthropods (crustaceans and insects) and structural membranes and spores of fungi. Its application has spread to many sectors, including pharmaceutical, medical, veterinary, food and agricultural. In the latter one, chitosan is applied to improve the interaction between plants and microorganisms and metabolisms of plants, fruits and germination. In addition, chitosan is demonstrated to enhance the availability and stabilization of insecticides and essential oils. Several chitosan formulations were studied as tools for insect pest control.
- biopolymer
- chitin
- insect control
- chitosan formulations
- chitosan nanoparticles
1. Introduction
2. Use of Chitosan against Insects
| Chitosan Formulation | Insect Order | Insect Species/Family | Target Instars |
References |
|---|---|---|---|---|
| Plain chitosan incorporated into a diet |
Diptera | Musca domestica (Muscidae) | Adults | [22] |
| Tabanus nigrovittatus (Tabanidae) | Adults | [22] | ||
| Phormia regina (Calliphoridae) | Adults | [22] | ||
| Plain chitosan added to paper and wood | Blattodea | Reticulitermes virginicus (Rhinotermitidae) | Adults | [23] |
| Reticulitermes flavipes (Rhinotermitidae) | Adults | [24] | ||
| Plain chitosan coating | Diptera | Anastrepha ludens (Tephritidae) | Larvae | [25] |
| Anastrepha obliqua (Tephritidae) | Adults | [26] | ||
| Chitosan coating with EOs | Lepidoptera | Plutella xylostella (Plutellidae) | Adults | [27] |
| Hemiptera | Myzus persicae (Aphididae) | Adults | [27] | |
| Diptera | Bactrocera carambolae (Tephritidae) |
Adults | [28] | |
| Calliphora vomitoria (Calliphoridae) |
Adults | [29] | ||
| Coleoptera | Acanthoscelides obtectus (Bruchidae) | Adults | [30] | |
| Sitophilus oryzae (Curculionidae) | Adults | [31] | ||
| Chitosan coating in active packaging |
Coleoptera | Sitophilus zeamais (Curculionidae) |
Adults | [32] |
| Chitosan with nematodes |
Coleoptera | Ryncgophorus ferrugineus (Curculionidae) |
Larvae | [33][34] |
| Chitosan oligosaccharides |
Lepidoptera | Helicoverpa armigera (Noctuidae) | Larvae | [35] |
| Plutella xylostella (Plutellidae) | Larvae | [35] | ||
| Hemiptera | Sitobion avenae (Aphididae) | Adults | [33] | |
| Metopolophium dirhodum (Aphididae) | Adults | [35] | ||
| Myzus persicae (Aphididae) | Adults | [35] | ||
| Hyalopterus pruni (Aphididae) | Adults | [35] | ||
| Aphis gossypii (Aphididae) | Adults | [35] | ||
| Rhopalosiphum padi (Aphididae) | Adults | [35] | ||
| Chitosan derivatives | Lepidoptera | Spodoptera littoralis (Noctuidae) | Larvae | [36][37][38] |
| Chitosan–metal complexes |
Lepidoptera | Spodoptera littoralis (Noctuidae) | Larvae | [39] |
| Hemiptera | Aphis nerii (Aphididae) | Adults | [39] | |
| Chitosan nanoparticles loaded with EOs | Coleoptera | Tribolium castaneum (Tenebrionidae) | Adults | [20][21][40][41][42] |
| Sitophilus oryzae (Curculionidae) | Adults | [21] | ||
| Oryzaephilus surinamensis (Silvanidae) |
Adults | [43] | ||
| Carpophilus hemipterus (Nitidulidae) |
Adults | [44] | ||
| Diptera | Anopheles stephensi (Culicidae) | Larvae | [45] | |
| Culex pipiens (Culicidae) | Larvae | [46] | ||
| Aedes aegypti (Culicidae) | Larvae | [19][47] | ||
| Musca domestica (Muscidae) | Adults | [48] | ||
| Hemiptera | Bemisia tabaci (Aleyrodidae) | Adults | [49] | |
| Chitosan nanoparticles loaded with agrochemicals |
Lepidoptera | Helicoverpa armigera (Noctuidae) | Larvae | [18] |
| Spodoptera litura (Noctuidae) | Larvae | [50] | ||
| Diptera | Drosophila melanogaster (Drosophilidae) |
Adults/ larvae |
[51] | |
| Chitosan-g-poly acrylic acid nanoparticles | Hemiptera | Aphis gossypii (Aphididae) | Adults | [52] |
| Coleoptera | Cassida vittata (Chrysomelidae) | Larvae | [53] | |
| Myristic acid chitosan nanoparticles | Coleoptera | Sitophilus granarius (Curculionidae) |
Adults | [54][55] |
| Tribolium confusum (Tenebrionidae) |
Adults | [54][55] | ||
| Chitosan nanoparticles RNAi | Diptera | Anopheles gambie (Culicidae) | Larvae | [56][57] |
| Aedes aegypti (Culicidae) | Larvae | [57][58] |
2.1. Plain Chitosan
2.2. Chitosan Coating
2.3. Chitosan Coating with Essential Oils
2.4. Chitosan Coating in Active Packaging
2.5. Chitosan and Nematodes
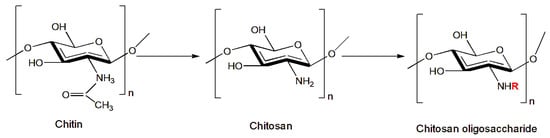
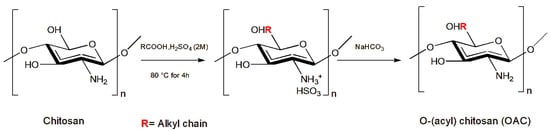
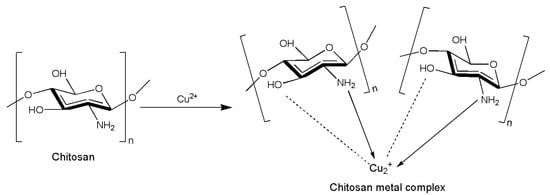
2.6. Chitosan Chemical Modification
2.7. Chitosan Nanoparticles
3. Conclusions and Future Perspectives
This entry is adapted from the peer-reviewed paper 10.3390/insects14120949
References
- Ravi Kumar, M.N.V. A review of chitin and chitosan applications. React. Funct. Polym. 2000, 46, 1–27.
- Bakshi, P.S.; Selvakumar, D.; Kadirvelu, K.; Kumar, N.S. Chitosan as an environment friendly biomaterial—A review on recent modifications and applications. Int. J. Biol. Macromol. 2020, 150, 1072–1083.
- Odier, A. Mémoire sur la composition chimique des parties cornées des insectes. Mem. Soc. Hist. Paris 1823, 1, 29–42.
- Muzzarelli, R.A.A. Chitin, 1937. In EPST, 2nd ed.; Mark, H.F., Bikales, N.M., Overberger, C.G., Menges, G., Eds.; Wiley: New York, NY, USA, 1985; Volume 3, pp. 430–449.
- Kou, S.; Peters, L.M.; Mucalo, M.R. Chitosan: A review of sources and preparation methods. Int. J. Biol. Macromol. 2021, 169, 85–94.
- Gillett, R. Global study of shrimp fisheries. FAO Fish Tech. Pap. 2008, 475, 25–29.
- Hahn, T.; Tafi, E.; Paul, A.; Salvia, R.; Falabella, P.; Zibek, S. Current state of chitin purification and chitosan production from insects. J. Chem. Technol. Biotechnol. 2020, 95, 2775–2795.
- Spranghers, T.; Ottoboni, M.; Klootwijk, C.; Ovyn, A.; Deboosere, S.; De Meulenaer, B.; Michiels, J.; Eeckhout, M.; De Clercq, P.; De Smet, S. Nutritional composition of black soldier fly (Hermetia illucens) prepupae reared on different organic waste substrates. J. Sci. Food Agric. 2017, 97, 2594–2600.
- Mohan, K.; Ganesan, A.R.; Muralisankar, T.; Jayakumar, R.; Sathishkumar, P.; Uthayakumar, V.; Chandirasekar, R.; Revathi, N. Recent insights into the extraction, characterization, and bioactivities of chitin and chitosan from insects. Trends Food Sci. 2020, 105, 17–42.
- Kong, M.; Chen, X.G.; Xing, K.; Park, H.J. Antimicrobial properties of chitosan and mode of action: A state of the art review. Int. J. Food Microbiol. 2010, 144, 51–63.
- Vinsova, J.; Vavrikova, E. Chitosan Derivatives with Antimicrobial, Antitumour and Antioxidant Activities—A Review. Curr. Pharm. Des. 2011, 17, 3596–3607.
- Sharif, R.; Mujtaba, M.; Ur Rahman, M.; Shalmani, A.; Ahmad, H.; Anwar, T.; Tianchan, D.; Wang, X. The Multifunctional Role of Chitosan in Horticultural Crops; A Review. Molecules 2018, 23, 872.
- Jiménez-Gómez, C.P.; Cecilia, J.A. Chitosan: A Natural Biopolymer with a Wide and Varied Range of Applications. Molecules 2020, 25, 3981.
- Ma, J.; Faqir, Y.; Tan, C.; Khaliq, G. Terrestrial insects as a promising source of chitosan and recent developments in its application for various industries. Food Chem. 2022, 373, 131407.
- Kashyap, P.L.; Xiang, X.; Heiden, P. Chitosan nanoparticle based delivery systems for sustainable agriculture. Int. J. Biol. Macromol. 2015, 77, 36–51.
- Žabka, M.; Pavela, R. The dominance of chitosan hydrochloride over modern natural agents or basic substances in efficacy against Phytophthora infestans, and its safety for the non-target model species Eisenia fetida. Horticulturae 2021, 7, 366.
- Orzali, L.; Allagui, M.B.; Chaves-Lopez, C.; Molina-Hernandez, J.B.; Moumni, M.; Mezzalama, M.; Romanazzi, G. Basic Substances and Potential Basic Substances: Key Compounds for a Sustainable Management of Seedborne Pathogens. Horticulturae 2023, 9, 1220.
- Paulraj, G.M.; Ignacimuthu, S.; Gandhi, M.R.; Shajahan, A.; Ganesan, P.; Packiam, S.M.; Al-Dhabi, N.A. Comparative studies of tripolyphosphate and glutaraldehyde cross-linked chitosan-botanical pesticide nanoparticles and their agricultural applications. Int. J. Biol. Macromol. 2017, 104, 1813–1819.
- Ferreira, T.P.; Haddi, K.H.; Corrêa, F.T.; Zapata, V.L.B.; Piau, T.B.; Souza, L.F.N.; Santos, S.G.; Oliveira, E.E.; Jumbo, L.O.; Ribeiro, B.M.; et al. Prolonged mosquitocidal activity of Siparuna guianensis essential oil encapsulated in chitosan nanoparticles. PLoS Negl. Trop. Dis. 2019, 13, e0007624.
- Rajkumar, V.; Gunasekaran, C.; Dharmaraj, J.; Chinnaraj, P.; Paul, C.A.; Kanithachristy, I. Structural characterization of chitosan nanoparticle loaded with Piper nigrum essential oil for biological efficacy against the stored grain pest control. Pestic. Biochem. Phys. 2020, 166, 104566.
- Rajkumar, V.; Gunasekaran, C.; Paul, C.A.; Dharmaraj, J. Development of encapsulated peppermint essential oil in chitosan nanoparticles: Characterization and biological efficacy against stored-grain pest control. Pestic. Biochem. Phys. 2020, 170, 104679.
- Stoffolano, J.; Wong, R.; Lo, T.; Ford, B.; Geden, C.J. Effect of chitosan on adult longevity when fed, in no-choice experiments, to Musca domestica L., Tabanus nigrovittatus Macquart, and Phormia regina (Meigen) adults and its consumption in adult Musca domestica L. Pest Manag. Sci. 2020, 76, 4293–4300.
- Telmadarrehei, T.; Tang, D.J.; Raji, O.; Rezazadeh, A.; Jeremic, D. Effect of chitosan on diversity and number of protists in subterranean termites. In Proceedings of the 114th Annual Meeting of the American Wood Protection, Seattle, WA, USA, 22–24 April 2018; pp. 22–24.
- Raji, O.; Tang, J.D.; Telmadarrehei, T.; Jeremic, D. Termiticidal activity of chitosan against the subterranean termites Reticulitermes flavipes and Reticulitermes virginicus. Pest Manag. Sci. 2018, 74, 1704–1710.
- Salvador-Figueroa, M.; Hernández-Ortiz, E.; Ventura-González, C.; Ovando-Medina, I.; Adriano-Anaya, L. Effect of chitosan coatings on the development of Anastrepha ludens (Loew) in mango fruits (Mangifera indica L.) cv. Ataulfo. Rev. Iberoam. De Tecnol. Postcosecha 2013, 14, 14–20.
- Limon, T.; Birke, A.; Monribot-Villanueva, J.L.; Guerrero-Analco, J.A.; Altúzar-Molina, A.; Carrión, G.; Goycoolea, F.M.; Moerschbacher, B.M.; Aluja, M. Chitosan coatings reduce fruit fly (Anastrepha obliqua) infestation and development of the fungus Colletotrichum gloeosporioides in Manila mangoes. J. Sci. Food Agric. 2021, 101, 2756–2766.
- Haas, J.; Lozano, E.R.; Haida, K.S.; Mazaro, S.M.; de Souza Vismara, E.; Poppy, G.M. Getting ready for battle: Do cabbage seeds treated with jasmonic acid and chitosan affect chewing and sap-feeding insects? Entomol. Exp. Appl. 2018, 166, 412–419.
- Perwita, M.G.C.S.; Wahyuningsih, T.D.; Astuti, E.; Pranowo, D. Synthesis Edible Film Chitosan/Polyethylene Glycol/Carboxymethylcellulose with Lemongrass Oils as Insect Ovipositing Repellent. Key Eng. Mater. 2020, 840, 142–148.
- Farina, P.; Ascrizzi, R.; Bedini, S.; Castagna, A.; Flamini, G.; Macaluso, M.; Mannucci, A.; Pieracci, Y.; Ranieri, A.; Sciampagna, M.C.; et al. Chitosan and Essential Oils Combined for Beef Meat Protection against the oviposition of Calliphora vomitoria, Water Loss, Lipid Peroxidation, and Colour Changes. Foods 2022, 11, 3994.
- Ascrizzi, R.; Flamini, G.; Bedini, S.; Tani, C.; Giannotti, P.; Lombardi, T.; Conti, B.; Fraternale, D. Ferulago campestris Essential Oil as Active Ingredient in Chitosan Seed-Coating: Chemical Analyses, Allelopathic Effects, and Protective Activity against the Common Bean Pest Acanthoscelides obtectus. Agronomy 2021, 11, 1578.
- Hossain, F.; Follett, P.; Salmieri, S.; Vu, K.D.; Harich, M.; Lacroix, M. Synergistic effects of nanocomposite films containing essential oil nanoemulsions in combination with ionizing radiation for control of rice weevil Sitophilus oryzae in stored grains. J. Food Sci. 2019, 84, 1439–1446.
- De Fátima Silva, M.; Maciel, V.B.V.; Noletto, A.P.R.; Venturini, A.C.; de Carvalho, R.A.; Yoshida, C.M.P. Chitosan active coating on paperboard surface forming an anti-insect grain-based food packaging. Packag. Technol. Sci. 2022, 35, 361–372.
- Llácer, E.; Martínez de Altube, M.M.; Jacas, J.A. Evaluation of the efficacy of Steinernema carpocapsae in a chitosan formulation against the red palm weevil, Rhynchophorus ferrugineus, in Phoenix canariensis. BioControl 2009, 54, 559–565.
- Dembilio, O.; Llacer, E.; Martínez de Altube, M.D.M.; Jacas, J.A. Field efficacy of imidacloprid and Steinernema carpocapsae in a chitosan formulation against the red palm weevil Rhynchophorus ferrugineus (Coleoptera: Curculionidae) in Phoenix canariensis. Pest Manag. Sci. 2010, 66, 365–370.
- Zhang, M.I.; Tan, T.; Yuan, H.; Rui, C. Insecticidal and fungicidal activities of chitosan and oligo-chitosan. J. Bioact. Compat. Polym. 2003, 18, 391–400.
- Rabea, E.I.; Badawy, M.E.; Rogge, T.M.; Stevens, C.V.; Höfte, M.; Steurbaut, W.; Smagghe, G. Insecticidal and fungicidal activity of new synthesized chitosan derivatives. Pest Manag. Sci. 2005, 61, 951–960.
- Rabea, E.I.; Badawy, M.E.; Rogge, T.M.; Stevens, C.V.; Steurbaut, W.; Höfte, M.; Smagghe, G. Enhancement of fungicidal and insecticidal activity by reductive alkylation of chitosan. Pest Manag. Sci. 2006, 62, 890–897.
- Badawy, M.E.I.; Rabea, E.I.; Rogge, T.M.; Stevens, C.V.; Steurbaut, W.; Höfte, M.; Smagghe, G. Fungicidal and Insecticidal Activity of O-Acyl Chitosan Derivatives. Polym. Bull. 2005, 54, 279–289.
- Badawy, M.E.I.; El-Aswad, A.F. Insecticidal activity of chitosans of different molecular weights and chitosan-metal complexes against cotton leafworm Spodoptera littoralis and oleander aphid Aphis nerii. Plant Prot. Sci. 2012, 48, 131–141.
- Rajkumar, V.; Gunasekaran, C.; Christy, I.K.; Dharmaraj, J.; Chinnaraj, P.; Paul, C.A. Toxicity, antifeedant and biochemical efficacy of Mentha piperita L. essential oil and their major constituents against stored grain pest. Pestic. Biochem. Phys. 2019, 156, 138–144.
- Upadhyay, N.; Singh, V.K.; Dwivedy, A.K.; Das, S.; Chaudhari, A.K.; Dubey, N.K. Assessment of Melissa officinalis L. essential oil as an eco-friendly approach against biodeterioration of wheat flour caused by Tribolium castaneum Herbst. Environ. Sci. Pollut. Res. 2019, 26, 14036–14049.
- Soltani, A.; Labidi, A.; Ben Jemaa, J. Development of formulation based on essential oils of rosemary to manage pests of stored cereal foodstuffs. In Proceedings of the 1st International Electronic Conference on Entomology, 1–15 July 2021; p. 10407.
- Soltani, A.; Ncibi, S.; Djebbi, T.; Sadraoui, I.A.; Abada, M.B.; Chargui, H.; Hassine, K.; Laabidi, A.; Mahmoudi, H.; Majdoub, H.; et al. Microencapsulation of rosemary Rosmarinus officinalis essential oil by Chitosan—Gum Arabic and its application for the control of two secondary pests of stored cereals. Res. Sq. 2022. Preprint.
- Soltani, A.; Haouel-Hamdi, S.; Sadraoui Ajmi, I.; Djebbi, T.; Ben Abada, M.; Yangui, I.; Chouachi, N.; Hassine, K.; Majdoub, H.; Messaoud, C.; et al. Insights for the control of dried-fruit beetle Carpophilus hemipterus (Nitidulidae) using rosemary essential oil loaded in chitosan nanoparticles. Int. J. Environ. Health Res. 2022, 33, 1243–1253.
- Sanei-Dehkordi, A.; Moemenbellah-Fard, M.D.; Sereshti, H.; Shahriari-Namadi, M.; Zarenezhad, E.; Osanloo, M. Chitosan nanoparticles containing Elettaria cardamomum and Cinnamomum zeylanicum essential oils; repellent and larvicidal effects against a malaria mosquito vector, and cytotoxic effects on a human skin normal cell line. Chem. Pap. 2021, 75, 6545–6556.
- Werdin González, J.O.; Jesser, E.N.; Yeguerman, C.A.; Ferrero, A.A.; Fernández Band, B. Polymer nanoparticles containing essential oils: New options for mosquito control. Environ. Sci. Pollut. Res. 2017, 24, 17006–17015.
- Paula, H.C.; Sombra, F.M.; Abreu, F.O.; Paul, R. Lippia sidoides essential oil encapsulation by angico gum/chitosan nanoparticles. J. Braz. Chem. Soc. 2010, 21, 2359–2366.
- El-Monairy, O.M.; Abdel-Meguid, A.D.; Emara, M.M. Efficacy of Methanol Leaf Extract, Biosynthesized Silver and Chitosan Nanoparticles Using Nerium oleander against Musca domestica. Egypt. Acad. J. Biol. Sci. F. Toxicol. Pest Control 2020, 12, 35–45.
- De Oliveira, J.L.; Campos, E.V.R.; Pereira, A.E.; Nunes, L.E.; Da Silva, C.C.; Pasquoto, T.; Lima, R.; Smaniotto, G.; Polanczyk, R.A.; Fraceto, L.F. Geraniol encapsulated in chitosan/gum arabic nanoparticles: A promising system for pest management in sustainable agriculture. J. Agric. Food Chem. 2018, 66, 5325–5334.
- Namasivayam, K.R.S.; Arvind Bharani, R.S.; Karunamoorthy, K. Insecticidal fungal metabolites fabricated chitosan nanocomposite (IM-CNC) preparation for the enhanced larvicidal activity—An effective strategy for green pesticide against economic important insect pests. Int. J. Biol. Macromol. 2018, 120, 921–944.
- Sharma, A.; Sood, K.; Kaur, J.; Khatri, M. Agrochemical loaded biocompatible chitosan nanoparticles for insect pest management. Biocatal. Agric. Biotechnol. 2019, 18, 101079.
- Sahab, A.F.; Waly, A.I.; Sabbour, M.M.; Nawar, L.S. Synthesis, antifungal and insecticidal potential of Chitosan (CS)-g-poly (acrylic acid) (PAA) nanoparticles against some seed borne fungi and insects of soybean. Int. J. ChemTech Res. 2015, 8, 589–598.
- Sabbour, M.M.; Abdel-Hakim, E.A. Control of Cassida vittata (Vill) (Coleoptera: Chrysomelidae) using chitosan and nano chitosan. Sciences 2018, 8, 141–144.
- Ziaee, M.; Moharramipour, S.; Mohsenifar, A. Toxicity of Carum copticum essential oil-loaded nanogel against Sitophilus granarius and Tribolium confusum. J. Appl. Entomol. 2014, 138, 763–771.
- Ziaee, M.; Moharramipour, S.; Mohsenifar, A. MA-chitosan nanogel loaded with Cuminum cyminum essential oil for efficient management of two stored product beetle pests. J. Pest Sci. 2014, 87, 691–699.
- Zhang, X.; Zhang, J.; Zhu, K.Y. Chitosan/double-stranded RNA nanoparticle-mediated RNA interference to silence chitin synthase genes through larval feeding in the African malaria mosquito (Anopheles gambiae). Insect Mol. Biol. 2010, 19, 683–693.
- Zhang, X.; Mysore, K.; Flannery, E.; Michel, K.; Severson, D.W.; Zhu, K.Y.; Duman-Scheel, M. Chitosan/interfering RNA nanoparticle mediated gene silencing in disease vector mosquito larvae. J. Vis. Exp. 2015, 97, e52523.
- Kumar, D.; Saravana Kumar, P.; Gandhi, M.R.; Al-Dhabi, N.A.; Paulraj, M.G.; Ignacimuthu, S. Delivery of chitosan/dsRNA nanoparticles for silencing of wing development vestigial (vg) gene in Aedes aegypti mosquitoes. Int. J. Biol. Macromol. 2016, 86, 89–95.
- Muryeti, M.; Pratiwi, F.E.; Yuniastuti, R.T.; Mulyani, E.B. Termiticidal activity of chitosan on paper. Prog. Chem. Appl. Chitin Deriv. 2020, 25, 164–173.
- Liibert, L.; Treu, A.; Meier, P. A two-step wood protection process using alternative wood protection agents in combination with an oil treatment. In Proceedings of the 7th meeting of the Nordic-Baltic Network in Wood Material Science & Engineering (WSE), Oslo, Norway, 27–28 October 2011.
- Jianglian, D.; Shaoying, Z. Application of Chitosan Based Coating in Fruit and Vegetable Preservation: A Review. J. Food Process. 2013, 4, 227.
- Shiekh, R.A.; Malik, M.A.; Al-Thabaiti, S.A.; Shiekh, M.A. Chitosan as a novel edible coating for fresh fruits. Food Sci. Technol. Res. 2013, 19, 139–155.
- Reddy, M.B.; Belkacemi, K.; Corcuff, R.; Castaigne, F.; Arul, J. Effect of pre-harvest chitosan sprays on post-harvest infection by Botrytis cinerea and quality of strawberry fruit. Postharvest Biol. Technol. 2000, 20, 39–51.
- Salgado-Cruz, M.D.L.P.; Salgado-Cruz, J.; García-Hernández, A.B.; Calderón-Domínguez, G.; Gómez-Viquez, H.; Oliver-Espinoza, R.; Fernández-Marínez, C.M.; Yáñez-Fernández, J. Chitosan as a coating for biocontrol in postharvest products: A bibliometric review. Membranes 2021, 11, 421.
- Baldrick, P. The safety of chitosan as a pharmaceutical excipient. Regul. Toxicol. Pharmacol. 2010, 56, 290–299.
- Shiri, M.A.; Bakhshi, D.; Ghasemnezhad, M.; Dadi, M.; Papachatzis, A.; Kalorizou, H. Coating improves the shelf life and postharvest quality of table grape (Vitis vinifera) cultivar Shahroudi. Turk. J. Agric. For. 2013, 37, 148–156.
- Sikder, M.B.H.; Islam, M.M. Effect of shrimp chitosan coating on physico-chemical properties and shelf-life extension of banana. Int. J. Eng. Technol. Sci. 2019, 6, 41–54.
- Li, K.; Xing, R.; Liu, S.; Li, P. Chitin and chitosan fragments responsible for plant elicitor and growth stimulator. J. Agric. Food Chem. 2020, 68, 12203–12211.
- Aluja, M.; Mangan, R.L. Fruit Fly (Diptera: Tephritidae) Host Status Determination: Critical Conceptual, Methodological, and Regulatory Considerations. Annu. Rev. Entomol. 2008, 53, 473–502.
- Bedini, S.; Flamini, G.; Cosci, F.; Ascrizzi, R.; Echeverria, M.C.; Guidi, L.; Landi, M.; Lucchi, A.; Conti, B. Artemisia spp. Essential oils against the disease-carrying blowfly Calliphora vomitoria. Parasites Vectors 2017, 10, 80.
- Bedini, S.; Cosci, F.; Girardi, J.; Bocchino, R.; Conti, C. Aromatic plant essential oils for the control of blowflies in the production of dry-cured meat. In Proceedings of the IOBC-WPRS Bulletin Bulletin OILB-SROP Proceeding of the meeting at Ljubljana (Slovenia), Ljubljana, Slovenia, 3–5 July 2017; Volume 130, pp. 75–80.
- Bedini, S.; Flamini, G.; Cosci, F.; Ascrizzi, R.; Echeverria, M.C.; Gomez, E.V.; Guidi, L.; Landi, M.; Lucchi, A.; Conti, B. Toxicity and oviposition deterrence of essential oils of Clinopodium nubigenum and Lavandula angustifolia against the myiasis inducing blowfly Lucilia sericata. PLoS ONE 2019, 14, e0212576.
- Bedini, S.; Guarino, S.; Echeverria, M.C.; Flamini, G.; Ascrizzi, R.; Loni, A.; Conti, B. Allium sativum, Rosmarinus officinalis, and Salvia officinalis essential oils: A spiced shield against blowflies. Insects 2020, 11, 143.
- Bedini, S.; Farina, P.; Napoli, E.; Flamini, G.; Ascrizzi, R.; Verzera, A.; Conti, B.; Zappalà, L. Bioactivity of different chemotypes of oregano essential oil against the blowfly Calliphora vomitoria vector of foodborne pathogens. Insects 2021, 12, 52.
- Farina, P.; Venturi, F.; Ascrizzi, R.; Flamini, G.; Chiriboga Ortega, R.D.; Echeverría, M.C.; Ortega, S.; Zinnai, A.; Bedini, S.; Conti, B. Andean plants essential oils: A scented alternative to synthetic insecticides for the control of blowflies. Insects 2021, 12, 894.
- Pierattini, E.C.; Bedini, S.; Venturi, F.; Ascrizzi, R.; Flamini, G.; Bocchino, R.; Girardi, J.; Giannotti, P.; Ferroni, G.; Conti, B. Sensory Quality of Essential Oils and Their Synergistic Effect with Diatomaceous Earth, for the Control of Stored Grain Insects. Insects 2019, 10, 114.
- Kumar, N.; Kaur, P.; Bhatia, S. Advances in bio-nanocomposite materials for food packaging: A review. Nutr. Food Sci. 2017, 47, 591–606.
- Hadwiger, L.A.; Loschke, D.C. Molecular Communication in Host-Parasite Interactions: Hexosamine Polymers (Chitosan) as Regulator Compounds in Race-Specific and Other Interactions. Phytopathology 1981, 71, 756–762.
- Ait Barka, E.; Eullaffroy, P.; Clément, C.; Vernet, G. Chitosan improves development and protects Vitis vinifera L. against Botrytis cinerea. Plant Cell Rep. 2004, 22, 608–614.
- Smart, G.C., Jr. Entomopathogenic nematodes for the biological control of insects. J. Nematol. 1995, 27, 529.
- Lacey, L.A.; Georgis, R. Entomopathogenic nematodes for control of insect pests above and below ground with comments on commercial production. J. Nematol. 2012, 44, 218.
- Shapiro-Ilan, D.; Dolinski, C. Entomopathogenic nematode application technology. In Nematode Pathogenesis of Insects and Other Pests: Ecology and Applied Technologies for Sustainable Plant and Crop Protection; Springer: Berlin/Heidelberg, Germany, 2015; pp. 231–254.
- Abbas, M.S.T.; Saleh, M.M.E.; Akil, A.M. Laboratory and field evaluation of the pathogenicity of entomopathogenic nematodes to the red palm weevil, Rhynchophorus ferrugineus (Oliv.) (Col.: Curculionidae). Anz. Schädlingskunde 2001, 74, 167–168.
- Peña, A.M. Biological Pesticide Based on Chitosan and Entomopathogenic Nematodes. WO Patent 037966, 16 May 2002.
- Wang, W.; Meng, Q.; Li, Q.; Liu, J.; Zhou, M.; Jin, Z.; Zhao, K. Chitosan derivatives and their application in biomedicine. Int. J. Mol. Sci. 2020, 21, 487.
- No, H.K.; Park, N.Y.; Lee, S.H.; Meyers, S.P. Antibacterial activity of chitosans and chitosan oligomers with different molecular weights. Int. J. Food Microbiol. 2002, 74, 65–72.
- Zou, P.; Yang, X.; Wang, J.; Li, Y.; Yu, H.; Zhang, Y.; Liu, G. Advances in characterisation and biological activities of chitosan and chitosan oligosaccharides. Food Chem. 2016, 190, 1174–1181.
- Xia, W.; Liu, P.; Zhang, J.; Chen, J. Biological activities of chitosan and chitooligosaccharides. Food Hydrocoll. 2011, 25, 170–179.
- Sahariah, P.; Másson, M. Antimicrobial chitosan and chitosan derivatives: A review of the structure–activity relationship. Biomacromolecules 2017, 18, 3846–3868.
- Wang, X.; Du, Y.; Liu, H. Preparation, characterization and antimicrobial activity of chitosan–Zn complex. Carbohydr. Polym. 2004, 56, 21–26.
- Varma, A.J.; Deshpande, S.V.; Kennedy, J.F. Metal complexation by chitosan and its derivatives: A review. Carbohydr. Polym. 2004, 55, 77–93.
- Wang, X.; Du, Y.; Fan, L.; Liu, H.; Hu, Y. Chitosan-metal complexes as antimicrobial agent: Synthesis, characterization and Structure-activity study. Polym. Bull. 2005, 55, 105–113.
- Athanassiou, C.G.; Kavallieratos, N.G.; Benelli, G.; Losic, D.; Usha Rani, P.; Desneux, N. Nanoparticles for pest control: Current status and future perspectives. J. Pest Sci. 2018, 91, 1–15.
- An, C.; Sun, C.; Li, N.; Huang, B.; Jiang, J.; Shen, Y.; Wang, C.; Zhao, X.; Cui, B.; Wang, C.; et al. Nanomaterials and nanotechnology for the delivery of agrochemicals: Strategies towards sustainable agriculture. J. Nanobiotechnology 2022, 20, 11.
- Ghormade, V.; Deshpande, M.V.; Paknikar, K.M. Perspectives for nano-biotechnology enabled protection and nutrition of plants. Biotechnol. Adv. 2011, 29, 792–803.
- Divya, K.; Jisha, M.S. Chitosan nanoparticles preparation and applications. Environ. Chem. Lett. 2018, 16, 101–112.
- Shoueir, K.R.; El-Desouky, N.; Rashad, M.M.; Ahmed, M.K.; Janowska, I.; El-Kemary, M. Chitosan based-nanoparticles and nanocapsules: Overview, physicochemical features, applications of a nanofibrous scaffold, and bioprinting. Int. J. Biol. Macromol. 2021, 167, 1176–1197.
- Maganga, M.E.; Gries, G.; Cries, R. Repellency of various oils and pine oil constituents to house flies (Diptera: Muscidae). Environ. Entomol. 1996, 25, 1182–1187.
- Katerinopoulos, H.E.; Pagona, G.; Afratis, A.; Stratigakis, N.; Roditakis, N. Composition and insect attracting activity of the essential oil of Rosmarinus officinalis. J. Chem. Ecol. 2005, 31, 111–122.
- Lacotte, V.; Rey, M.; Peignier, S.; Mercier, P.E.; Rahioui, I.; Sivignon, C.; Razy, L.; Benhamou, S.; Livi, S.; da Silva, P. Bioactivity and chemical composition of forty plant essential oils against the pea aphid Acyrthosiphon pisum revealed peppermint oil as a promising biorepellent. Ind. Crops Prod. 2023, 197, 116610.
- Lahlali, R.; El Hamss, H.; Mediouni-Ben Jemâa, J.; Barka, E.A. The Use of Plant Extracts and Essential Oils as Biopesticides. Front. Agron. 2022, 4, 921965.
- Maluin, F.N.; Hussein, M.Z. Chitosan-based agronanochemicals as a sustainable alternative in crop protection. Molecules 2020, 25, 1611.
- Werdin González, J.O.; Gutiérrez, M.M.; Ferrero, A.A.; Fernández Band, B. Essential oils nanoformulations for stored-product pest control—Characterization and biological properties. Chemosphere 2014, 100, 130–138.
