Gangliosides are sialic acid containing complex glycolipids, anchored and enriched in the outer leaflet of neuronal plasma membranes, with their glycan chains facing the extracellular space. Undegradeable gangliosides and related glycosphingolipids and oligosaccharides accumulate progressively in fatal lysosomal storage diseases, originally described as infantile amaurotic idiocy. Their lysosomal storage is caused by specific monogenic defects of catabolic hydrolyses or ancillary lipid-binding and -transfer proteins, essential for specific steps in their lysosomal catabolism.
However, small gangliosides can also accumulate as secondary material in other lysosomal storage diseases without a known defect in their catabolic pathway. Primary storage material of such diseases, sphingomyelin, lysosphingolipids, cholesterol and chondroitin sulfate are efficient inhibitors of specific steps of ganglioside catabolic pathway. They can attenuate ganglioside turnover, assisted by lipid binding proteins, the GM2 activator protein (GM2AP) and saposin B
- Glycosphingolipids
- Lysosomal storage diseases
- Intra-lysosomal vesicles (ILVs)
- Topology of ganglioside catabolism at ILVs
- GM1 Gangliosidosis
- GM2 Gangliosidoses
- Tay-Sachs disease
- Sandhoff disease
- GM2 Activator Protein Deficiency (AB Variant)
Primary Storage Compounds in Gangliosidoses and Historical Aspects
| Disease |
Affected Protein
|
Affected Gene | Storage Compound | References |
|---|---|---|---|---|
| GM2 Gangliosidoses | ||||
| Tay–Sachs disease (B variant) | Hex A1, Hex S2 | HEXA | GM2, SM2a, lyso-GM2, GA2 | [6] |
| B1 variant | Hex A1 | HEXA | GM2 | [6] |
| Sandhoff disease | Hex A1, Hex B3 | HEXB | GM2, globoside, oligosaccharides, lyso-GM2 | [6,7,8] |
| GM2AP deficiency (AB variant) | GM2AP | GM2A | GM2 | [6] |
| GM1 Gangliosidosis | acid β-galactosidase | GLB1 | GM1, GA1, GM2, GM3, GA1a, lyso GM1 GlcCer Laccer, oligosaccharides, keratan sulfate | [9,10] |
GM2 Gangliosidoses
Tay–Sachs Disease (B Variant) and B1 Variant
Sandhoff Disease (SD) (0 Variant)
GM2 Activator Protein Deficiency (AB Variant)
GM1 Gangliosidosis
Topology and Regulation of GG Catabolism
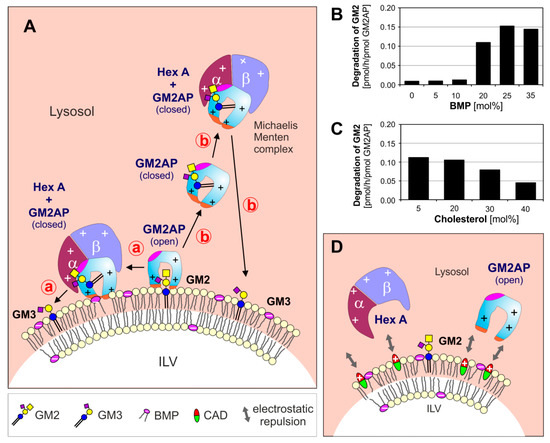
Due to their lipid and amphiphilic nature and their poor solubility in aqueous solutions, GGs and other amphiphilic sphingolipids are membrane components of eukaryotic cells. GGs are especially enriched in neuronal plasma-membranes of mammalian cells. GGs are degraded after endocytosis and internalization into intralysosomal luminal vesicles (ILVs). Functional defects of any catabolic step cause an accumulation of membrane-bound-GGs in the lysosome. Based on experimental evidence obtained in vitro and by in vivo studies using murine and human cell cultures, we assume that the surface of ILVs, carrying GGs and other complex lipids, is the main location of their catabolism in the lysosomal compartment [4,34,35].
At low pH-values, most lysosomal hydrolases are protonated and positively charged, whereas the surfaces of the ILV membranes are negatively charged, mainly due to their high content of the anionic lysolipid bis(monoacylglycero)phosphate (BMP) and the possible presence of other anionic phospholipids in the ILV membranes [36]. BMP is an intermediate of the phosphatidylglycerol catabolism and can enrich in ILV membranes up to 40–60 mol% of their phospholipid content [4,37,38], mainly due to its slow catabolism. It can generate a negative zeta potential on the surfaces of ILVs [36], which electrostatically attracts positively charged hydrolases and SAPs to the sphingolipid-substrate carrying ILV-membranes, speeding up their catabolic rates [4,39,40] (Figure 1).
For diagnosis of LSDs, most lysosomal hydrolases are usually assayed in vitro with the help of synthetic and soluble fluorogenic substrates [41,42]. These convenient assays are an easy way to detect the presence of a lysosomal hydrolase in patients’ samples and to determine its activity in vitro. The activity measured in vitro with soluble synthetic substrates, however, does not indicate, in any way, the level of the sphingolipid-substrate cleaving activity of the patient’s hydrolase in vivo [43], since the sphingolipid cleaving activity of a lysosomal hydrolase can be strongly regulated and modified in vivo by genetic and by post translational modifiers which do not affect its activity against soluble substrates in vitro.
As genetic modifiers we consider the SAPs, small lipid-binding, lipid-transfer and/or vesicle-fusion glycoproteins of the lysosomal compartment. These cofactors are essential for the glycosphingolipid and sphingolipid cleaving activity of lysosomal hydrolases to reach physiologically relevant levels. Their inherited deficiencies can cause fatal storage diseases [4 (see Suppl.),34,35,44] despite the presence of fully active hydrolases, detectable with soluble fluorogenic substrates in patient`s cultivated cells or blood samples.
As posttranslational modifiers of the lipid-cleaving activity of lysosomal hydrolases we consider the strong inhibitory action of chondroitin-sulfate (accumulating in the lysosomes of Hurler, Hunter, Sanfilippo and Sly disease [45]) on the GM2 catabolism, as well as the collective properties of the sphingolipid-substrate carrying vesicle-membranes (e.g., extent of negative surface charge of ILVs to attract and bind protonated and positively charged hydrolases and the lipid composition of the vesicular membranes, especially the presence of stimulatory (e.g., BMP, ceramide) or inhibitory lipids (cholesterol, sphingomyelin)). These genetic and posttranslational modifiers strongly regulate the lipid-cleaving activity of lysosomal hydrolases, but rarely affect their activity against soluble, synthetic substrates used in vitro to diagnose lysosomal lipid storage diseases, as recently detailed in reconstitution experiments for the regulation of GM2 cleavage by Hex A in comparison to the unaffected cleavage of the soluble substrate MUGS (4-methylumbelliferyl-6-sulfo-2-acetamido-2-deoxy-β-D-glycopyranoside) [43].
Organellar membranes of eukaryotic cells maintain an organelle specific protein and lipid composition [46]. Cellular plasma membranes are rich in stabilizing lipids like cholesterol (up to 40 mol% of their lipid content), and maintain in their outer leaflet high levels of sphingomyelin and complex glycosphingolipids. Both, cholesterol and sphingomyelin, were identified as major inhibitors of key steps of lysosomal sphingolipid catabolism [4,34]. Therefore, the conversion of inhibitory sphingomyelin into stimulatory ceramide by acid sphingomyelinase (ASM) along the endocytose pathway at the level of late endosomes, and the removal of inhibitory cholesterol from nascent ILVs by two sterol-binding and -transfer proteins, Niemann–Pick disease protein C type 1 (NPC1) and NPC2, are essential to allow a physiological GG GM2, sphingolipid and GSL turnover [35,29,47,48].
Outlook
Lysosomal GG and GSL accumulation had been also observed in LSDs without a genetic defect in the GG catabolism. While the secondary accumulation of GG GM2 in Niemann-Pick diseases and in some mucopolysaccaridoses [49,50],could be based on a strong inhibitory effect of the primary storage compounds in these diseases (SM,cholesterol and chondroitin sulfate [43,45]) on the GG GM2 catabolism [35], the molecular mechanism causing a secondary accumulation of GG GM2, small GGs and glycosphingolipids in many other LSDs remains to be analysed: In sphingolipidoses, prosaposin deficiency, mucolipidoses (MLs), glycoproteinoses, neuronal ceroid lipofuscinoses (NCLs) and hereditary spastic paraplegia (HSP). In these diseases, mainly the GGs, GM2 and GM3, are accumulated. They are minor compounds (1–2% of the total gangliosides) of the healthy human brain and their proportion is even smaller in mice. A secondary accumulation of GM2 and GM3 in many LSDs, however, is associated with neuropathology. Therefore, the molecular basis of their secondary accumulation in these diseases should be investigated.
Though many ganglioside storage diseases are known for a long time, there is still no cure available. Enzyme replacement therapy (ERT) has been established for some LSDs not involving the central nervous system. In recent years, gene-replacement therapy has been successfully studied in animal models of gangliosidoses and is now applied to patients. It carries the promise of a cure, but still needs a long way to really cure progressive neurodegenerative diseases like gangliosidoses.
[2] Sachs, B. A family form of idiocy, generally fatal, associated with early blindness (amaurotic family idiocy). J. Nerv. Ment. Dis. 1896, 21, 475–479. [Google Scholar]
[3] Klenk, E. Niemann–Pick’sche Krankheit und Amaurotische Idiotie. Hoppe-Seyler’s Z Physiol. Chem. 1939, 262, 128–143. [Google Scholar] [CrossRef]
[4] Breiden, B.; Sandhoff, K. Lysosomal glycosphingolipid storage diseases. Annu. Rev. Biochem. 2019, 88, 461–485.
[5] Sandhoff, K.; Harzer, K. Gangliosides and gangliosidoses: Principles of molecular and metabolic pathogenesis. J. Neurosci. 2013, 33, 10195–10208. [Google Scholar] [CrossRef] [PubMed]
[6] Gravel, R.; Kaback, M.M.; Proia, R.L.; Sandhoff, K.; Suzuki, K.; Suzuki, K. The GM2 gangliosidoses. In The Metabolic and Molecular Bases of Inherited Disease, 8th ed.; Scriver, C.R., Beaudet, A.L., Sly, W.S., Valle, D., Eds.; McGraw-Hill: New York, NY, USA, 2001; pp. 3827–3876. [Google Scholar]
[7] Sandhoff, K.; Andreae, U.; Jatzkewitz, H. Deficient hexosaminidase activity in an exceptional case of Tay–Sachs disease with additional storage of kidney globoside in visceral organs. Pathol Eur. 1968, 3, 278–285. [Google Scholar] [CrossRef]
[8] Pilz, H.; Müller, D.; Sandhoff, K.; ter Meulen, V. Tay–Sachssche Krankheit mit Hexosaminidase-Defekt. Klinische, morphologische und biochemische Befunde bei einem Fall mit viszeraler Speicherung von Nierenglobosid. Dtsch Med. Wochenschr 1968, 93, 1833–1839. [Google Scholar] [CrossRef]
[9] Suzuki, K.; Chen, G.C. GM1-gangliosidosis (generalized gangliosidosis). Morphology and chemical pathology. Pathol Eur. 1968, 3, 389–408. [Google Scholar]
[10] Siegel, D.A.; Walkley, S.U. Growth of ectopic dendrites on cortical pyramidal neurons in neuronal storage diseases correlates with abnormal accumulation of GM2 ganglioside. J. Neurochem. 1994, 62, 1852–1862. [Google Scholar] [CrossRef]
[11] Tay, W. Symmetrical changes in the region of the yellow spot in each eye of an infant. Trans. Ophthalmol. Soc. 1881, 1, 55–57. [Google Scholar] [CrossRef]
[12] Jatzkewitz, H.; Sandhoff, K. On a biochemically special form of infantile amaturotic idiocy. Biochim. Biophys. Acta. 1963, 70, 354–356. [Google Scholar] [CrossRef]
[13] Sandhoff, K. Die Amaurotische Idiotie des Menschen als Störung im Glykosphingolipidstoffwechsel. Doctoral Thesis, University of Munich, Munich, Germany, 1965. [Google Scholar]
[14] Kuhn, R.; Wiegandt, H. Die Konstitution der Ganglio-N-tetraose und des Gangliosids GI. Chem. Ber. 1963, 96, 866–880. [Google Scholar] [CrossRef]
[15] Sandhoff, K. The GM2-gangliosidoses and the elucidation of the beta-hexosaminidase system. Adv. Genet. 2001, 44, 67–91. [Google Scholar]
[16] Sandhoff, K. Variation of beta-N-acetylhexosaminidase-pattern in Tay–Sachs disease. FEBS Lett. 1969, 4, 351–354. [Google Scholar] [CrossRef]
[17] Sandhoff, K.; Harzer, K.; Wässle, W.; Jatzkewitz, H. Enzyme alterations and lipid storage in three variants of Tay–Sachs disease. J. Neurochem. 1971, 18, 2469–2489. [Google Scholar] [CrossRef]
[18] Conzelmann, E.; Sandhoff, K. AB variant of infantile GM2 gangliosidosis: Deficiency of a factor necessary for stimulation of hexosaminidase A-catalyzed degradation of ganglioside GM2 and glycolipid GA2. Proc. Natl. Acad. Sci. USA 1978, 75, 3979–3983. [Google Scholar] [CrossRef]
[19] Okada, S.; O’Brien, J.S. Tay–Sachs disease: Generalized absence of a beta-D-N-acetylhexosaminidase component. Science 1969, 165, 698–700. [Google Scholar] [CrossRef
[20] Sandhoff, K. The hydrolysis of Tay–Sachs ganglioside (TSG) by human N-acetyl-beta-D-hexosaminidase A. FEBS Lett. 1970, 11, 342–344. [Google Scholar] [CrossRef]
[21] Sandhoff, R.; Schulze, H.; Sandhoff, K. Ganglioside metabolism in health and disease. Prog. Mol. Biol. Transl. Sci. 2018, 156, 1–62. [Google Scholar] [CrossRef] [PubMed]
[22] Sandhoff, K.; Jatzkewitz, H.; Peters, G. Die infantile amaurotische Idiotie und verwandte Formen als Gangliosid-Speicherkrankheiten. Naturwissenschaften 1969, 56, 356–362. [Google Scholar] [CrossRef] [PubMed]
[23] Hepbildikler, S.T.; Sandhoff, R.; Kölzer, M.; Proia, R.L.; Sandhoff, K. Physiological substrates for human lysosomal beta -hexosaminidase S. J. Biol. Chem. 2002, 277, 2562–2572. [Google Scholar] [CrossRef] [PubMed]
[24] Kytzia, H.J.; Hinrichs, U.; Maire, I.; Suzuki, K.; Sandhoff, K. Variant of GM2-gangliosidosis with hexosaminidase A having a severely changed substrate specificity. Embo J. 1983, 2, 1201–1205. [Google Scholar] [CrossRef]
[25] Kytzia, H.J.; Sandhoff, K. Evidence for two different active sites on human beta-hexosaminidase A. Interaction of GM2 activator protein with beta-hexosaminidase A. J. Biol. Chem. 1985, 260, 7568–7572. [Google Scholar]
[26] Sango, K.; Yamanaka, S.; Hoffmann, A.; Okuda, Y.; Grinberg, A.; Westphal, H.; McDonald, M.P.; Crawley, J.N.; Sandhoff, K.; Suzuki, K.; et al. Mouse models of Tay–Sachs and Sandhoff diseases differ in neurologic phenotype and ganglioside metabolism. Nat. Genet. 1995, 11, 170–176. [Google Scholar] [CrossRef]
[27] Jeyakumar, M.; Smith, D.; Eliott-Smith, E.; Cortina-Borja, M.; Reinkensmeier, G.; Butters, T.D.; Lemm, T.; Sandhoff, K.; Perry, V.H.; Dwek, R.A.; et al. An inducible mouse model of late onset Tay–Sachs disease. Neurobiol. Dis. 2002, 10, 201–210. [Google Scholar] [CrossRef] [PubMed]
[28] Sango, K.; McDonald, M.P.; Crawley, J.N.; Mack, M.L.; Tifft, C.J.; Skop, E.; Starr, C.M.; Hoffmann, A.; Sandhoff, K.; Suzuki, K.; et al. Mice lacking both subunits of lysosomal [beta]-hexosaminidase display gangliosidosis and mucopolysaccharidosis. Nat. Genet. 1996, 14, 348–352. [Google Scholar]
[29] Anheuser, S.; Breiden, B.; Schwarzmann, G.; Sandhoff, K. Membrane lipids regulate ganglioside GM2 catabolism and GM2 activator protein activity. J. Lipid Res. 2015, 56, 1747–1761. [Google Scholar] [CrossRef] [PubMed]
[30] O’Brien, J.S.; Stern, M.B.; Landing, B.H.; O’Brien, J.K.; Donnell, G.N. Generalized gangliosidosis: Another inborn error of ganglioside metabolism? Am. J. Dis. Child. 1965, 109, 338–346. [Google Scholar] [CrossRef]
[31] Lawrence, R.; Van Vleet, J.L.; Mangini, L.; Harris, A.; Martin, N.; Clark, W.; Chandriani, S.; LeBowitz, J.H.; Giugliani, R.; d’Azzo, A.; et al. Characterization of glycan substrates accumulating in GM1 gangliosidosis. Mol. Genet. Metab. Rep. 2019, 21, 100524. [Google Scholar] [CrossRef]
[32] Neufeld, E.; Muenzer, J. The mucopolysaccharidoses. In The Metabolic and Molecular Bases of Inherited Disease, 8 ed.; Scriver, C.R., Beaudet, A.L., Sly, W.S., Valle, D., Eds. McGraw-Hill: New York, NY, USA, 2001; pp. 3421–3452.
[33] Wilkening, G.; Linke, T.; Uhlhorn-Dierks, G.; Sandhoff, K. Degradation of membrane-bound ganglioside GM1. Stimulation by bis(monoacylglycero)phosphate and the activator proteins SAP-B and GM2-AP. J. Biol. Chem. 2000, 275, 35814–35819. [Google Scholar] [CrossRef]
[34] Sandhoff, R.; Sandhoff, K. Emerging concepts of ganglioside metabolism. FEBS Lett. 2018, 592, 3835–3864.
[35] Breiden, B.; Sandhoff, K. Mechanism of secondary ganglioside and lipid accumulation in lysosomal disease. Int J Mol Sci 2020, 21, 2566.
[36] Oninla, V.O.; Breiden, B.; Babalola, J.O.; Sandhoff, K. Acid sphingomyelinase activity is regulated by membrane lipids and facilitates cholesterol transfer by NPC2. J. Lipid Res. 2014, 55, 2606–2619.
[37] Kobayashi, T.; Beuchat, M.H.; Lindsay, M.; Frias, S.; Palmiter, R.D.; Sakuraba, H.; Parton, R.G.; Gruenberg, J. Late endosomal membranes rich in lysobisphosphatidic acid regulate cholesterol transport. Nat. Cell Biol. 1999, 1, 113–118.
[38] Möbius, W.; van Donselaar, E.; Ohno-Iwashita, Y.; Shimada, Y.; Heijnen, H.F.; Slot, J.W.; Geuze, H.J. Recycling compartments and the internal vesicles of multivesicular bodies harbor most of the cholesterol found in the endocytic pathway. Traffic (Copenhagen, Denmark) 2003, 4, 222–231.
[39] Graf, C.G.F.; Schulz, C.; Schmälzlein, M.; Heinlein, C.; Mönnich, M.; Perkams, L.; Püttner, M.; Boos, I.;Hessefort, M.; Lombana Sanchez, J.N., et al. Synthetic glycoforms reveal carbohydrate-dependent bioactivity of human saposin D. Angew Chem. Int. Ed. Engl. 2017, 56, 5252–5257.
[40] Kölzer, M.; Werth, N.; Sandhoff, K. Interactions of acid sphingomyelinase and lipid bilayers in the presence of the tricyclic antidepressant desipramine. FEBS Lett. 2004, 559, 96–98.
[41] Yu, C.; Sun, Q.; Zhou, H. Enzymatic screening and diagnosis of lysosomal storage diseases. N. Am. J. Med. Sci. 2013, 6, 186–193.
[42] Galjaard, H. Genetic metabolic diseases: Early diagnosis and prenatal analysis; Elsevier-Noth Holland Biomedical Press: Amsterdam, The Netherlands, 1980.
[43] Anheuser, S.; Breiden, B.; Sandhoff, K. Membrane lipids and their degradation compounds control GM2 catabolism at intralysosomal luminal vesicles. J. Lipid Res. 2019, 60, 1099–1111.
[44] Kolter, T.; Sandhoff, K. Lysosomal degradation of membrane lipids. FEBS Letters 2010, 584, 1700-1712.
[45] Anheuser, S.; Breiden, B.; Sandhoff, K. Ganglioside GM2 catabolism is inhibited by storage compounds of mucopolysaccharidoses and by cationic amphiphilic drugs. Mol. Genet. Metab 2019, 128, 75–83.
[46] van Meer, G.; Voelker, D.R.; Feigenson, G.W. Membrane lipids: Where they are and how they behave. Nat. Rev. Mol. Cell Biol. 2008, 9, 112–124.
[47] Abdul-Hammed, M.; Breiden, B.; Adebayo, M.A.; Babalola, J.O.; Schwarzmann, G.; Sandhoff, K. Role of endosomal membrane lipids and NPC2 in cholesterol transfer and membrane fusion. J. Lipid Res. 2010, 51, 1747–1760.
[48] Wang, M.L.; Motamed, M.; Infante, R.E.; Abi-Mosleh, L.; Kwon, H.J.; Brown, M.S.; Goldstein, J.L. Identification of surface residues on Niemann–Pick C2 essential for hydrophobic handoff of cholesterol to NPC1 in lysosomes. Cell Metab. 2010, 12, 166–173.
[49] Jatzkewitz, H.; Pilz, H.; Sandhoff, K. Quantitative Bestimmungen von Gangliosiden und ihren Neuraminsäurefreien Derivaten bei infantilen, juvenilen und adulten Formen der amaurotischen Idiotie und einer spätinfantilen biochemischen Sonderform. J. Neurochem. 1965, 12, 135–144.
[50] Walkley, S.U.; Vanier, M.T. Secondary lipid accumulation in lysosomal disease. Biochim. Biophys. Acta. 2009, 1793, 726–736.
This entry is adapted from the peer-reviewed paper 10.3390/ijms21072566
