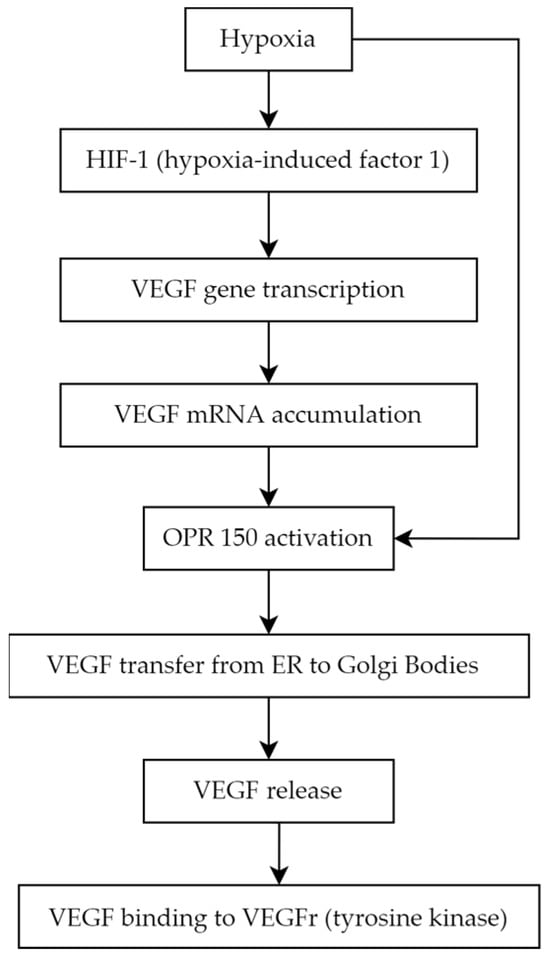
VEGFR1 can be secreted in the soluble form (sFlt1—expressed in the placenta during gestation) to prevent endothelial overproliferation due to VEGF accumulation. There are known VEGF coreceptors as well: the neuropilins, neuropilin-1 and neuropilin-2. Their main role is to enhance the binding of VEGF to the VEGFR2 and the migration of endothelial cells stimulated by VEGF [
]. Heparan sulfate and integrins can also modulate the signal from VEGFR. VEGFR1 is a crucial factor in providing a proper level of VEGF-A, and it plays an important role in the negative regulation of vascular modification. VEGFR1 is also necessary for the process of monocyte migration. PGF bound to VEGFR1 is responsible for initiating inflammatory-related angiogenesis, which is vital in the pathogenesis of various diseases. VEGF-B characteristically binds to VEGFR1 in tissues with high metabolic activity such as myocardial cells. VEGFR2, the most comprehensively studied among the VEGF receptors, binds with VEGF-A, VEGF-C, and VEGF-D. It is known that the activation of VEGFR2 kinase through VEGF-induced VEGFR2 homodimerization is responsible for the majority, if not all, of the known VEGF-related processes such as mitosis stimulation, migration, and survival of endothelial cells, ultimately leading to the formation of new blood vessels [
]. The crucial functions of VEGFR2 in endothelial biology are evident from the various processes involved in its tight regulation such as internalization to early endosomes for the activation of specific pathways and the involvement of phosphatases like vascular endothelial protein tyrosine phosphatase. VEGFR3 has an affinity to VEGF-C and VEGF-D. Although the main function of VEGFR3 was initially identified as the regulation of lymphatic endothelial development and biology, it has also been observed to be present in blood vascular endothelial cells. Angiogenesis-involved endothelial cells exhibit the expression of both VEGFR2 and VEGFR3. VEGFR3 activation is not solely dependent on VEGF-C/VEGF-D binding, as it can also be activated through integrin-mediated mechanisms, it leads to lymphatic vessel expansion and the absorption of interstitial fluid [
]. In addition, VEGF is critical for ensuring the proper morphology and function of vascular structures [
]. Notably, mutations that result in the loss of the VEGF gene allele have been found to be lethal [
]. The development of PDR is linked to relative retinal ischemia, which creates a hypoxic environment, which favors HIF-1 activation and VEGF production. The pathological revascularization in the retina has been attributed to VEGF-A165, a specific splice variant of VEGF-A [
].
Arginine is an amino acid essential for normal growth and development. Endogenous synthesis is adequate in healthy people but might be deficient in many pathological states [
63]. The earliest sign of vascular complications is endothelial dysfunction [
64]. Nitric oxide (NO) is an important vasodilator that is crucial in maintaining the health of the vascular endothelium. Studies demonstrate that endothelial dysfunction plays a critical role in the development of diabetes-associated microvascular complications and often precedes advanced diabetic retinopathy (DR) [
65,
66,
67]. NO is synthesized from the guanidine group of arginine by the enzyme family NO synthases (NOSs), which consist of three isoforms [
68,
69]. Asymmetric dimethylarginine (ADMA) is an active endogenous methylated amino acid, a structural analogue of L-arginine, which inhibits the activity of all isoforms of NOS, inhibiting the formation of nitric oxide in tissues and blood plasma [
70,
71]. ADMA is synthesized by the protein arginine N-methyltransferase 1 (PRMT1), mainly metabolized by the dimethylarginine dimethylaminohydrolases (DDAHs) pathway, and eliminated from the body by kidneys [
72,
73]. ADMA enters cells through cationic amino-acid transporters (CATs) [
74]. Plasma levels of ADMA in healthy people vary between 0.3 and 0.5 μmol/L [
75], but in pathological states, it may increase even tenfold [
76]. ADMA has a negative effect on cells, contributing to oxidative stress, shortening telomeres, inhibiting the release of NO, and increasing the secretion of interleukin-8 and monocyte chemotaxis factor 1 [
75]. Under normal conditions, endothelial NOS is inhibited by 10%, but in pathological situations, even by 30–70% [
76]. When the plasma ADMA level increases, the NO synthesis in the environment decreases, vascular homeostasis degrades due to vasoconstriction, and endothelial dysfunction begins [
69]. Endothelial dysfunction and impaired ocular hemodynamics prime diabetic retinopathy development are associated with decreased NOS activity and NO bioavailability, thus resulting in increased reactive oxygen species (ROS) and vasoconstriction [
76,
77]. Oxidative stress is closely related to DDAH activity, which further affects ADMA concentrations in patients with diabetes [
78,
79]. Increased oxidative stress contributes to elevated ADMA, and by the upregulation of circulating markers of oxidative stress, increased serum ADMA concentration is associated with increased vascular oxidative stress [
80,
81,
82]. ADMA accumulation was first reported in patients characterized by endothelial dysfunction including hyperglycemia, hypercholesterolemia, and hypertension [
83,
84]. Impaired liver or renal function could also have an impact on the plasma concentration of ADMA. The significance of ADMA in the inhibition of vascular endothelial growth factor-mediated angiogenesis has been demonstrated in numerous studies. Some evidence suggests that diabetes mellitus with microvascular complications has increased serum levels of ADMA [
85,
86,
87,
88]. Elevated ADMA was detected in aqueous humor in diabetic patients, especially those with severe retinopathy [
89]. The plasma ADMA level is elevated in patients with diabetic microangiopathy such as DR [
66,
86,
90,
91,
92,
93]. Lowering ADMA levels may delay the progression of DR by reducing the formation of neovascularization, providing protective advantages for the blood–retinal barrier [
92].
4.3. MicroRNAs
MicroRNAs (miRNAs) are single-strengthened, non-coding RNA, which affect gene expression regulation. Their suppressor interaction with mRNA usually is associated with 3′ untranslated regions (3′ UTRs), although data claim as well its interaction potential according to different sequences such as gene promoters. Moreover, they also have a regulatory role in transcription and translation processes [
103]. The creation process of those micromolecules goes from DNA transcription to primary miRNA (pri-mRNA) through precursor miRNA (pre-miRNA) leading to mature miRNA formation [
104]. The role of miRNA in signalization pathways is studied nowadays excessively because of those particles’ multiplicity.
Molecular bases of miRNA mechanisms of action are distinct for different miRNAs, and it is possible to distinguish which particles affect which pathway leading to DR, such as affecting cell proliferation, angiogenesis, apoptosis, or basement membrane thickening [
106]. It has been proven that directly or indirectly particles such as miRNA-9, miRNA-152, miRNA-15b, miRNA-29b-3p, miRNA-199a-3p, miRNA-203a-3p, miRNA-200b-3p, and miRNA-30a-3p downregulate VEGF expression, which lowers the range of active cell-cycle-related proteins and by that protects RMECs (retinal microvascular endothelial cells) from abnormal proliferation [
107]. In addition, from previously mentioned biomolecules, the alternative pathway to downregulate VEGF is SIRT1 (nicotinamide adenosine dinucleotide (NAD+)-dependent deacetylase) upregulation, which is possible by miRNA-29b-3p and miRNA-34a inhibition, moreover, causing an increase in proinflammatory cytokines [
107]. MiRNA-34a was evaluated to be an interesting therapeutic target, as in rats with induced DR, its silencing was observed as an apoptosis regulation [
108].
MiRNA-20a and miRNA-20b were revealed to downregulate VEGF as well but in different mechanisms—first act by Yse-associated protein (YAP)/hypoxia-inducible factor 1α (HIF1α)/VEGF axis, and second was revealed in the study on rats to be correlated with downregulation of AKT3, lowering VEGF expression [
111,
112]. Moreover, it was assessed that Resolvin D1 modulates the intracellular VEGF-related miRNAs—miRNA-20a-3p, miRNA-20a-5p, miRNA-106a-5p, and miRNA-20b—expression of retinal photoreceptors challenged with high glucose [
113].
The role of the miRNA was investigated as a DR biomarker using different sample types and designs compared to various groups according to diabetes type 1 or 2, T1DM or T2DM, patients with DM and healthy individuals, as well studies referring to DR progression. In blood serum samples in T1DM patients with DR and those without retinopathy, the most significant was miRNA-211. Then, miRNA-18b and miRNA-19b were revealed as upregulated; additionally, miRNA-29a, miRNA-148a, miRNA-181a, and miRNA-200a were revealed to have such an impact [
117,
118].
According to T2DM, a study was performed and the differences in the following particles were noted: hsa-let-7a-5p, hsa-miRNA-novel-chr5_15976, hsa-miRNA-28-3p, hsa-miRNA-151a-5p, and hsa-miRNA-148a-3p were upregulated compared to DM group with no retinopathy; however, a panel of the first three of them were the closest to help in assessing the diagnosis as its sensitivity and specificity were as follows: 0.92 and 0.94 [
121]. Another study showed that in T2DM patients, DR was associated with increased circulating levels of miRNA-25-3p and miRNA-320b and decreased levels of miRNA-495-3p [
122].
Plasma results among T2DM patients gave an insight into lower levels of miRNA-29b in the DR group and miRNA-21 as biomarkers that were significantly associated with PDR. Other parameters that were increased in T2DM patients with DR were miRNA-93 via SIRT1 and miRNA-21, as well as miRNA-152 [
126,
127]. On the contrary, miRNA-15a, miRNA-20b, miRNA-21, miRNA-24, miRNA-320, miRNA-486, and miRNA-150, miRNA-126, miRNA-191, miRNA-197 are downregulated in that group of patients’ plasma samples [
128]. Importantly, miRNA-150 is observed in both T1DM and T2DM patients’ circulation and in the neutral retina. That factor by Elk1 upregulation stimulates proinflammatory, pro-angiogenic, and apoptotic influences. Otherwise, a lower range of miRNA-150 in serum impacts Elk1 and Myb overexpression, resulting in the same as the previously mentioned pathway in microvascular complications and neovascularization leading to DR; so, according to that analysis, it is not only a diagnostic biomarker but as well is significantly involved in DR pathogenesis [
129].
4.4. Endothelin-1
Endothelin-1 (ET-1) in its active form is a 21-amino acid hormone that helps to maintain basal vascular tone and metabolic function in healthy individuals [
132]. ET-1 is an endothelium-derived factor with proliferative, profibrotic, and proinflammatory properties [
133], and it is the most abundantly expressed member of the endothelin family of proteins (ET-1, ET-2, and ET-3). Immature ET-1 undergoes extensive post-transcriptional processing that concludes with cleavage by endothelin converting enzymes (ECEs) and subsequent release of mature ET-1 primarily toward the interstitial space, and in smaller proportion, into the circulation [
132]. ET-1 works on two different ET-1 receptor subtypes, ETA and ETB, to produce its various biological effects [
134]. The first subtype, ETA receptors, is predominantly localized on vascular smooth muscle cells (VSMCs) of blood vessels where they mediate contractile and proliferative response to ET-1, whereas ETB receptors have a more composite relation to vascular regulation. ETB receptors can lead to vasodilation via the release of relaxing factors if they are present on endothelial cells or vasoconstriction when they are located on VSMCs in certain vascular beds [
133]. Therefore, the overall effect of ET-1 on different tissues is largely dependent on the expression and relative densities of individual receptor subtypes. ET-1 is one of the important markers of endothelial dysfunction, a state characterized by disturbed balance between vasoconstrictors and vasodilators [
135]. Due to its vasoconstrictive properties, ET-1 has been widely studied in terms of its role in hypertension and proved clinically significant, e.g., with the use of endothelin receptor antagonists for the treatment of patients with pulmonary arterial hypertension [
136]. The vasoconstrictive and in turn hypertensive properties of ET-1 can explain a possible link between elevated plasma ET-1 level and retinopathy under ischemia, a finding relevant to diabetic retinopathy, which is thought to be the consequence of retinal ischemia. Animal models have shown that administration of ET-1 into the posterior vitreous body or the optic nerve leads to physiological and cellular damages of ischemic origin, including obstruction of retinal blood flow, elevated scotopic b-wave in electroretinogram, and apoptosis of cells in ganglion cell layer of the retina [
137].
4.5. Advanced Glycation End Products
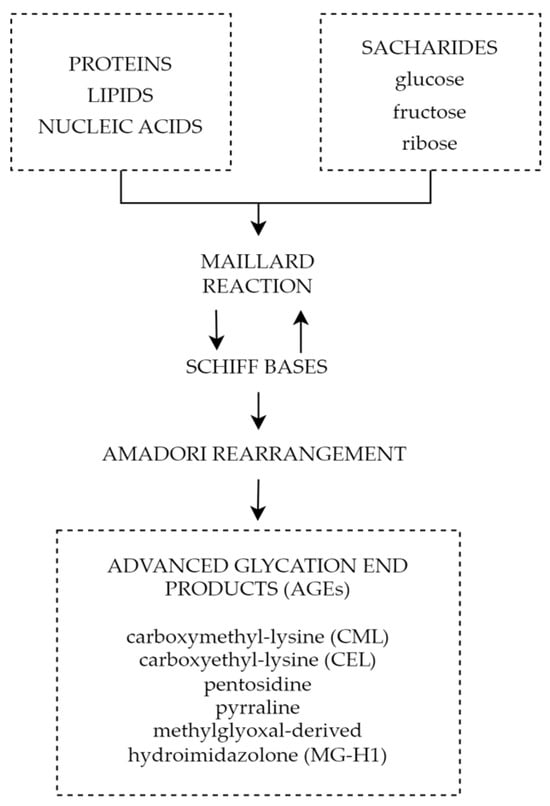
One of the mechanisms connecting chronic hyperglycemia with diabetic retinopathy is the formation and accumulation of advanced glycation end products (AGEs). Advanced glycation end products are heterogeneous groups of molecules formed from post-translational non-enzymatic modifications of proteins, lipids, or nucleic acids by saccharides including glucose, fructose, and pentose through the Maillard reaction represented by
Figure 5 [
148,
149]. There are over 20 AGEs identified in human tissues, but some of the most common ones are carboxymethyl-lysine (CML), carboxyethyl-lysine (CEL), pentosidine, pyrraline, and methylglyoxal-derived hydroimidazolone (MG-H1) [
150]. The characteristic factor of AGEs that distinguishes them from early glycation products, such as glycohemoglobin A1c (HbA1c), is the lack of spontaneous reversion ability, which once derived results in the accumulation in tissues over time [
151]. Even though the discovery of AGEs dates to the early 20th century, not until the 1980s, the role of AGEs in aging and chronic diseases was recognized [
152]. The first mention of AGEs and their accumulation in human tissues and their potential role in diabetic complications appeared in 1988 in a scientific article published by Helen Vlassara et al. [
153]. Since then, AGEs and their involvement in pathophysiological processes have been the subject of extensive research.
Figure 5. Forming of AGEs through Maillard reaction.
5. Summary
ADMA inhibits the activity of NOS, which results in decreased levels of NO and leads to vasoconstriction and endothelial dysfunction. Increased ADMA levels may be considered an early prognostic factor of diabetes complications such as PDR. The use of ADMA as a biomarker may help in early diagnosis, monitoring, and effective therapeutic management of the disease. Reducing ADMA levels in patients with diabetes may be a new therapeutic target to prevent the development of diabetic retinopathy. Endothelin-1 is another factor with an undoubted relationship to diabetic retinopathy. Increased serum and aqueous humor levels are observed in patients with ET-1 elevation dependent on the severity of the progression of the disease. This, juxtaposed with promising results of ET-1 receptor antagonist animal studies, showcases the potential of ET-1 as a possible target for future therapy. It is important to note that miRNAs are not only supposed to be an innovative predictive biomarker and progression indicator in DR but also a potential therapeutic target. Different miRNAs can be found in T1DM and T2DM as well depending on sample type, moreover, some of them differ depending on DR type. The variety of miRNAs and frequently high amounts of particles involved in several pathogenesis pathways can be at the same time the advantage and disadvantage of that prospective novel biomarkers group; hence, miRNAs panels are more adequate than a single biomarker rating. Finally, advanced glycation end products play a significant role in the pathophysiology of diabetic retinopathy causing impairment of the neurovascular units through reactive oxygen species, inflammatory reactions, and cell death pathways. All the above mechanisms play a significant role not only in diabetic retinal disorders, but also other chronic oxidative-based diseases; therefore, a thorough understanding of their properties and mechanisms will allow advances in the diagnosis and treatment of chronic diseases and most importantly diabetic retinopathy. The above factors and signaling pathways can help to create multimodal and highly specified therapies for patients suffering from DR. It is crucial to investigate molecular agents participating in DR pathogenesis. Hopefully, it will provide the ability to inhibit this progressive disease at its early stage.