Teeth begin to grow in the jaws before birth and continue to appear in an overlapping sequence until each is complete in length. Subsequently, the central pulp (nerve) chamber of each tooth slowly fills in with dentine and the root of the tooth continues to accumulate thin layers of cementum until the individual dies. Each of the tooth tissues, the hard enamel cap, the dentine core of the tooth and the root cementum grow incrementally and incorporate small quantities of blood-born trace elements ingested from our diet into their structure. A chronological record of zinc incorporation exists in each tooth tissue and can be visualised in thin sections, or slices, of teeth using a beam of synchrotron light. Zinc markings in teeth are especially useful and occur at birth in enamel and dentine and annually in the cementum layers. This work shows that zinc is consistently concentrated within surface enamel and in the dentine surrounding the central pulp chamber. Knowing where to sample Zn in modern and fossil teeth enables us to reconstruct a chronology of growth and to determine something about diet in the past from the remnants of different Zn isotopes contained in different foodstuffs.
- trace elements
- neonatal line
- enamel
- SXRF
- enamel thickness
- mineralisation process
1. Introduction
Zinc is an essential biological trace element. Besides this, Zn also has an affinity for bone and the three mineralised tooth tissues, enamel, dentine and cementum. Not only does the distribution of Zn vary among the three tooth tissues, but its origins also derive from different physiological, developmental and chemical processes. Ultimately, the Zn source is either from the intestinal absorption of ingested dietary Zn, or from the mother perinatally, via a direct-blood-borne maternal–placental transfer to the fetus. Zinc retrieved from tooth tissues in an archaeological and palaeontological context offers strong potential for revealing aspects of an individual’s life history, for example, as a marker of birth or of annual increments of tissue laid down during life, or as an indicator of diet. Reviewing the role and function of Zn in the body, and of how it comes to distribute differently in each of the tooth tissues, provides some insight into how and where it might best be sampled and of where its incorporation into each of the mineralised tooth tissues is likely to be tightly chronologically circumscribed or not. Fossil tooth tissues are often, if not always, altered through diagenesis, and a comparison of Zn distribution and preservation in fossils from contrasting sites may point to similarities or differences with modern teeth and so indicate where within the tooth tissues Zn of biogenic origin persists for longest.
2. Zinc as an Essential Trace Element in the Body
Zinc is an essential trace element involved in many physiological processes as both a catalyst in biochemical reactions, in the maintenance of protein quaternary structure and as a component of many essential enzymes [1]. Zinc is required, among other things, for cell division, tissue growth and wound healing, but also for intestinal electrolyte absorption, neurotransmission, the immune response, thymus activity and vision [1,2]. It is the most abundant trace element found in bone mineral, being present in concentrations of between 200–300 ppm [2,3]. Zinc is also present in muscle, red blood cells and skin as well as in bone and the mineralised tooth tissues [2,4]. Zinc is required for the DNA binding proteins involved in regulating gene transcription and expression [5]. It is also a critical component of several hundred essential enzymes and proteins [4]. At least 10% of human proteins contain Zn as a cofactor [6]. These include many metalloproteins and enzymes that are involved in most of the major metabolic pathways [1].
Among metalloenzymes, Zn is a component of carbonic anhydrase, alcohol dehydrogenase [7] and of alkaline phosphatase that is crucially involved in the process of mineralisation through the generation of free phosphate groups that are then taken up by newly forming bone and tooth tissues [8,9,10,11]. The zinc-finger proteins that control a variety of fundamental cellular activities require Zn to maintain their precise quaternary structure and functional integrity [1]. Some zinc-finger proteins have been identified as specific regulators of skeletal development and mineralised tissues formation [5,11]. Zinc in bone is concentrated at the sites of mineralisation and has been shown to stimulate osteoblast differentiation and proliferation [9,10,12]. Kim et al. [13] have also demonstrated that a Zn finger-containing transcription factor (Osterix; Osx) is an essential site-specific regulator of odontoblast differentiation, maturation and of tooth root elongation.
3. Zinc in Enamel Secretion and Maturation
Matrix metalloproteinase 20, or MMP-20, (sometimes also referred to as enamelysin), is a protease that is secreted with enamel proteins during the early (secretory) stage of amelogenesis, when the enamel crystallites are growing predominantly in length [11,14]. MMP-20 cleaves the enamel proteins surrounding crystallites to allow their slow growth in width, after which these cleavage products are reabsorbed by secretory ameloblasts and degraded. The more aggressive serine protease, kallikrein 4, or KLK-4, is a protease that is secreted during the later transition and maturation stages of amelogenesis. KLK-4 degrades what remains of the enamel organic matrix after enamel secretion is completed [11,14]. The principal functions of MMP-20 and KLK-4 in dental enamel formation are to facilitate the orderly replacement of organic matrix with mineral to form hard dense enamel. This occurs in a low-calcium environment that favours the slow controlled formation of hydroxyapatite that is maintained by the tight barrier filter of the secretory ameloblast sheet; this excludes or removes excess Ca [15]. MMP-20 itself contains Zn, but Zn in enamel is a potent inhibitor of serine proteases and so may have an important controlling influence, especially on KLK-4 during the maturation phase [11]. For these reasons, it has been suggested that Zn enrichment in the outer enamel [16,17], but also at the EDJ [18] and in dentine [19], may result from metalloprotease activity and degradation that results in the retention and sequestration of Zn originally involved in the mineralisation and/or enamel maturation process.
4. Zinc Absorption and Maternal–Fetal Transfer
Zinc absorption from the gut involves first binding to a surface receptor on the gut wall enterocytes and then subsequently being taken up into the enterocytes [10,20]. Zn has five stable isotopes of which the most abundant are 64Zn and 66Zn. The detection and distribution of Zn isotopes in dental tissues and their relation to diet are discussed more fully below. However, the intestinal absorption of Zn is inhibited and undergoes isotopic fractionation by, for example, plant phytates which precipitate dietary Zn, and because there is a preferential precipitation of the lighter Zn isotopes (64Zn) with plant phytates, this then favours heavier 66Zn absorption and enrichment relative to 64Zn [21]. Thus, the isotopic composition of Zn found in body tissues may not directly reflect the dietary isotopic composition [21,22]. A proportion of absorbed Zn then remains bound to metallothionein within the enterocyte, and as this accumulates, it inhibits further Zn absorption from the gut, thus regulating Zn uptake. It follows that as dietary Zn intake increases beyond the threshold required, Zn absorption decreases relative to intake [20]. The proportion of Zn that remains bound to metallothionein within the cell, eventually returns to the bowel lumen when the enterocyte is shed. Consequently, faecal loss of unabsorbed Zn increases with excess Zn uptake as the Zn-containing enterocytes are shed. Zn is also lost through urinary excretion and through sweat; this has practical implications when performing Synchrotron X-ray fluorescence (SXRF) experiments to map Zn distribution in tooth and bone samples, as surfaces under investigation must remain completely clean and untouched. Zn is an important essential metal ion such that recent research has identified metallochaperone proteins that escort or direct Zn to specific crucial cellular enzymes, ensuring the correct Zn allocation when levels are scarce [6].
Some Zn becomes bound in serum to albumin or an alpha-2 macroglobulin after absorption; this may be transported to the liver [2,20], but beyond the Zn component in bone mineral, there are no real functional reserves of Zn in the body [1]. The exception is in neonates where, at term, Zn accumulated during gestation is stored in the liver and then released postnatally. Most Zn is transferred to the fetus from the mother after the 24th week of gestation and stored in the fetal liver [1]. Pre-term birth, therefore, reduces the amount of Zn that can accumulate with the risk of Zn deficiency in the neonate [1,23]. Maternal-fetal placental transfer of Zn is an active process and fetal Zn concentrations are maintained constantly higher than maternal levels. While Zn in human milk varies in concentration between 0.7–1.6 mg/L, and declines with time, colostrum contains 8–12 mg/L, falling to 3–6 mg/L within a week [1].
5. The Distribution of Zinc in Mineralised Tissues
6. Zinc Incorporation into Hydroxyapatite
7. Zinc Distribution in Modern and Fossil Teeth
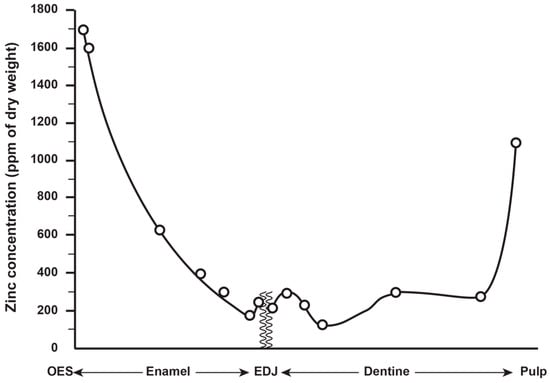
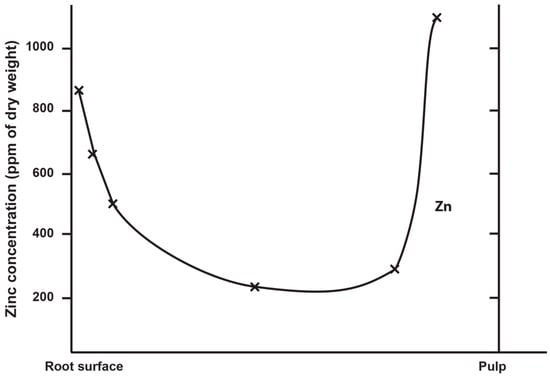
Zinc concentration in outer enamel is universally high across all the deciduous, permanent and fossil hominoid teeth studied so far. Where secondary dentine has formed, or is preserved, Zn concentrations are also higher than in primary dentine. Besides enamel and dentine, cementum layers also contain Zn at greater concentrations than the underlying root dentine. Figure 3 shows synchrotron X-ray fluorescence (SXRF) maps of the Zn distribution in seven permanent teeth of varying tooth types.

Figure 3. Zinc intensity maps revealed with synchrotron X-ray fluorescence (SXRF) of permanent modern great ape and human teeth, (a) Pan lower first molar tooth, (b) Pan lower third molar tooth, (c) Pongo female canine, (d) modern human upper canine (e) Gorilla lower second molar tooth, (f) Gorilla unerupted lower third molar tooth from the same individual as (e), (g) modern human lower third molar tooth. In all teeth, including the unerupted lower third molar, surface enamel is Zn-rich. In all cases, where it has formed, secondary dentine and cementum are Zn-rich. The Zn distribution in dentine in each tooth broadly follows the incremental formation pattern. Images are not to scale.
The thickness of the Zn-enriched surface enamel layer was observed not to be proportionate in thickness to the regional linear enamel thickness and varied considerably between and within teeth in both the maximum Zn concentration measured in outer enamel (197–1743 ppm), and in the rate of the exponential rise of concentration towards the surface. Zinc levels at the cusp, mid-crown and cervix of the same tooth may vary, but mid-crown measurements are as good a representation of the overall average concentrations as any, and perhaps the most consistently high. The Zn concentration range and maximum values in outer enamel overlap in the modern human, great ape and fossil hominoid deciduous and permanent tooth samples studied here. Zinc is also laid down in teeth prenatally and at the birth line (or neonatal line) in deciduous teeth and along the enamel-dentine junction (Figure 4). Figure 4 shows a thin section of a modern deciduous tooth cusp with the neonatal line as it appears in transmitted light and with synchrotron X-ray fluorescence (SXRF) to reveal the Zn distribution corresponding with the neonatal line in both enamel and dentine.

Figure 4. (a) Transmitted light micrograph and (b) matching Zn SXRF intensity image of the cuspal region of a modern human second deciduous molar tooth showing the neonatal line and enamel-dentine junction. In the SXRF Zn map, the neonatal line in both enamel (white arrows) and dentine (black arrows) is Zn-rich as is the prenatal dentine (black asterisks) and enamel-dentine junction. In prenatal enamel (white asterisks), the SXRF intensity map suggests this is Zn-depleted relative to postnatal enamel. Adapted from Figs. 2e and h in Dean et al [19].
Zinc is also tenacious compared to other trace elements contained in fossil tooth tissues and distributes within many fossil teeth exactly as it does in modern teeth. It can even be preserved at the neonatal line in fossil teeth for millions of years (Figure 5) and used as a marker of birth, with the caveat that Zn lines in enamel and dentine may also be indicative of other stress events in life.

Figure 5. (a) Transmitted light micrograph (TLM) and (b) matching Zn SXRF intensity image of the cuspal region of a fossil first molar tooth showing the neonatal line (white arrows) and enamel-dentine junction. This fossil hominoid tooth is attributed to Ekembo heseloni and dates to ~17.5 million years from Rusinga Island, Kenya.
The consistent distribution of Zn in both modern and fossil hominoid teeth allows us to take samples with greater confidence and precision. New high-resolution sampling and analytical techniques now enable access to the chronological record of Zn laid down in dentine and cementum layers through life with good prospects for tracking dietary shifts, stress events and seasonal changes far back into the archaeological and fossil records.
This entry is adapted from the peer-reviewed paper 10.3390/biology12121455
References
- Greenberg, S.R. The Histopathology of Clinically-Important Metals. A Review. Histol. Histopathol. 1989, 4, 375–380.
- Jeejeebhoy, K. Zinc: An Essential Trace Element for Parenteral Nutrition. Gastroenterology 2009, 137, S7–S12.
- Terrin, G.; Berni Canani, R.; Di Chiara, M.; Pietravalle, A.; Aleandri, V.; Conte, F.; De Curtis, M. Zinc in Early Life: A Key Element in the Fetus and Preterm Neonate. Nutrients 2015, 7, 10427–10446.
- Donangelo, C.M.; King, J.C. Maternal Zinc Intakes and Homeostatic Adjustments during Pregnancy and Lactation. Nutrients 2012, 4, 782–798.
- Brudevold, F.; Steadman, L.T.; Spinelli, M.A.; Amdur, B.H.; Grøn, P. A Study of Zinc in Human Teeth. Arch. Oral Biol. 1963, 8, 135–144.
- Takeuchi, K.; Nakagaki, H.; Toyama, Y.; Kimata, N.; Ito, F.; Robinson, C.; Weatherell, J.A.; Stösser, L.; Künzel, W. Fluoride Concentrations and Distribution in Premolars of Children from Low and Optimal Fluoride Areas. Caries Res. 1996, 30, 76–82.
- Lynch, R.J.M. Zinc in the Mouth, Its Interactions with Dental Enamel and Possible Effects on Caries; a Review of the Literature. Int. Dent. J. 2011, 61, 46–54.
- Ide-Ektessabi, A.; Shirasawa, K.; Koizumi, A.; Azechi, M. Application of Synchrotron Radiation Microbeams to Environmental Monitoring. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. At. 2004, 213, 761–765.
- Brozou, A.; Mannino, M.A.; Van Malderen, S.J.M.; Garrevoet, J.; Pubert, E.; Fuller, B.T.; Dean, M.C.; Colard, T.; Santos, F.; Lynnerup, N.; et al. Using SXRF and LA-ICP-TOFMS to Explore Evidence of Treatment and Physiological Responses to Leprosy in Medieval Denmark. Biology 2023, 12, 184.
- Obtel, N.; Le Cabec, A.; Nguyen, T.N.; Giabicani, E.; Van Malderen, S.J.M.; Garrevoet, J.; Percot, A.; Paris, C.; Dean, C.; Hadj-Rabia, S.; et al. Impact of Claudin-10 Deficiency on Amelogenesis: Lesson from a HELIX Tooth. Ann. N. Y. Acad. Sci. 2022, 1516, 197–211.
- Sánchez-Quevedo, M.; Crespo, P.; García, J.; Campos, A. X-ray Histochemistry of Zinc in Dental Tissues. Eur. Arch. Biol. 1992, 103, 47–49.
- Humphrey, L.T.; Jeffries, T.E.; Dean, M.C. Micro Spatial Distributions of Lead and Zinc in Human Deciduous Tooth Enamel. In Technique and Application in Dental Anthropology; Irish, J.D., Nelson, G.C., Eds.; Studies in Biological Anthropology; Cambridge University Press: Cambridge, UK, 2008; Volume 53, pp. 87–110.
- Humphrey, L.T.; Hassett, B.R.; Jeffries, T.E.; Dean, C.M. Trace Element Distributions in Early and Late Forming Tooth Enamel. In Proceedings of the 4th Meeting of the European Society for the Study of Human Evolution, Florence, Italy, 18–20 September 2014; p. 88.
- Humphrey, L.T.; Hassett, B.R.; Jeffries, T.E.; Dean, C.M. Micro-spatial Patterning of Lead and Zinc in Deciduous Tooth Enamel. In Proceedings of the 17th International Symposium on Dental Morphology, Bordeaux, France, 4–7 October 2017; p. 170.
- Müller, W.; Nava, A.; Evans, D.; Rossi, P.F.; Alt, K.W.; Bondioli, L. Enamel Mineralization and Compositional Time-Resolution in Human Teeth Evaluated via Histologically-Defined LA-ICPMS Profiles. Geochim. Cosmochim. Acta 2019, 255, 105–126.
- Bourgon, N.; Jaouen, K.; Bacon, A.-M.; Jochum, K.P.; Dufour, E.; Duringer, P.; Ponche, J.-L.; Joannes-Boyau, R.; Boesch, Q.; Antoine, P.-O.; et al. Zinc Isotopes in Late Pleistocene Fossil Teeth from a Southeast Asian Cave Setting Preserve Paleodietary Information. Proc. Natl. Acad. Sci. USA 2020, 117, 4675.
- Klimuszko, E.; Orywal, K.; Sierpinska, T.; Sidun, J.; Golebiewska, M. The Evaluation of Zinc and Copper Content in Tooth Enamel without Any Pathological Changes—An in Vitro Study. Int. J. Nanomed. 2018, 13, 1257–1264.
- Kang, D.; Amarasiriwardena, D.; Goodman, A.H. Application of Laser Ablation-Inductively Coupled Plasma-Mass Spectrometry (LA-ICP-MS) to Investigate Trace Metal Spatial Distributions in Human Tooth Enamel and Dentine Growth Layers and Pulp. Anal. Bioanal. Chem. 2004, 378, 1608–1615.
- Dean, M.C.; Spiers, K.M.; Garrevoet, J.; Le Cabec, A. Synchrotron X-ray Fluorescence Mapping of Ca, Sr and Zn at the Neonatal Line in Human Deciduous Teeth Reflects Changing Perinatal Physiology. Arch. Oral Biol. 2019, 104, 90–102.
- Stock, S.R.; Deymier-Black, A.C.; Veis, A.; Telser, A.; Lux, E.; Cai, Z. Bovine and Equine Peritubular and Intertubular Dentin. Biomineralization 2014, 10, 3969–3977.
- Dean, M.C.; Le Cabec, A.; Spiers, K.; Zhang, Y.; Garrevoet, J. Incremental Distribution of Strontium and Zinc in Great Ape and Fossil Hominin Cementum Using Synchrotron X-ray Fluorescence Mapping. J. R. Soc. Interface 2018, 15, 20170626.
- Brudevold, F.; Steadman, L.T.; Smith, F.A. Inorganic and Organic Components of Tooth Structure. Ann. N. Y. Acad. Sci. 1960, 85, 110–132.
- Martin, R.R.; Naftel, S.J.; Nelson, A.J.; Feilen, A.B.; Narvaez, A. Synchrotron X-ray Fluorescence and Trace Metals in the Cementum Rings of Human Teeth. J. Environ. Monit. 2004, 6, 783–786.
- Martin, R.R.; Naftel, S.J.; Nelson, A.J.; Sapp III, W.D. Comparison of the Distributions of Bromine, Lead, and Zinc in Tooth and Bone from an Ancient Peruvian Burial Site by X-ray Fluorescence. Can. J. Chem. 2007, 85, 831–836.
- Stock, S.R.; Finney, L.A.; Telser, A.; Maxey, E.; Vogt, S.; Okasinski, J.S. Cementum Structure in Beluga Whale Teeth. Acta Biomater. 2017, 48, 289–299.
- Stock, S.R.; Veis, A.; Telser, A.; Cai, Z. Near Tubule and Intertubular Bovine Dentin Mapped at the 250 Nm Level. J. Struct. Biol. 2011, 176, 203–211.
- Djomehri, S.I.; Candell, S.; Case, T.; Browning, A.; Marshall, G.W.; Yun, W.; Lau, S.H.; Webb, S.; Ho, S.P. Mineral Density Volume Gradients in Normal and Diseased Human Tissues. PLoS ONE 2015, 10, e0121611.
- Klevezal, G.A.; Kirillova, I.V.; Shishlina, N.I.; Sokolov, A.A.; Trunova, Y.E. Growth Layers in Tooth Dentin and Cementum: Problems and Perspectives of Their Use in the Study of Fossil and Subfossil Mammal Remains Including Humans. Doc. Archaeobiol. 2006, 4, 113–124.
- Le Cabec, A.; Tang, N.K.; Ruano Rubio, V.; Hillson, S. Nondestructive Adult Age at Death Estimation: Visualizing Cementum Annulations in a Known Age Historical Human Assemblage Using Synchrotron X-ray Microtomography. Am. J. Phys. Anthropol. 2019, 168, 25–44.
- Cerrito, P.; Bailey, S.E.; Hu, B.; Bromage, T.G. Parturitions, Menopause and Other Physiological Stressors Are Recorded in Dental Cementum Microstructure. Sci. Rep. 2020, 10, 5381.
- Gomez, S.; Rizzo, R.; Pozzi-Mucelli, M.; Bonucci, E.; Vittur, F. Zinc Mapping in Bone Tissues by Histochemistry and Synchrotron Radiation–Induced X-ray Emission: Correlation with the Distribution of Alkaline Phosphatase. Bone 1999, 25, 33–38.
- Yamaguchi, M.; Inamoto, K.; Suketa, Y. Effect of Essential Trace Metals on Bone Metabolism in Weanling Rats: Comparison with Zinc and Other Metals’ Actions. Res. Exp. Med. (Berl.) 1986, 186, 337–342.
- Seo, H.-J.; Cho, Y.-E.; Kim, T.; Shin, H.-I.; Kwun, I.-S. Zinc May Increase Bone Formation through Stimulating Cell Proliferation, Alkaline Phosphatase Activity and Collagen Synthesis in Osteoblastic MC3T3-E1 Cells. Nutr. Res. Pract. 2010, 4, 356.
- Mansell, R.E.; Hendershot, L.C. The Spectrochemical Analysis of Metals in Rat Molar Enamel, Femurs and Incisors. Arch. Oral Biol. 1960, 2, 31–37.
- Sasaki, T.; Takagi, M.; Yanagisawa, T. Structure and Function of Secretory Ameloblasts in Enamel Formation. In Proceedings of the Ciba Foundation Symposium 205; Chadwick, D.J., Cardew, G., Eds.; Novartis Foundation Symposia; John Wiley & Sons, Ltd.: Chichester, UK, 1997; pp. 32–53. ISBN 978-0-470-51530-3.
- Moonga, B.S.; Dempster, D.W. Zinc Is a Potent Inhibitor of Osteoclastic Bone Resorption in Vitro. J. Bone Miner. Res. 1995, 10, 453–457.
- Featherstone, J.D.B.; Nelson, D.G.A. The Effect of Fluoride, Zinc, Strontium, Magnesium and Iron on the Crystal-Structural Disorder in Synthetic Carbonated Apatites. Aust. J. Chem. 1980, 33, 2363.
- Crawford, A.W.; De Bruin, H.J. Concentration Changes in Surface Ca, P, F, Zn, Fe, and Sr During White Spot Formation. J. Dent. Res. 1983, 62, 964–968.
- Mohammed, N.R.; Mneimne, M.; Hill, R.G.; Al-Jawad, M.; Lynch, R.J.M.; Anderson, P. Physical Chemical Effects of Zinc on in Vitro Enamel Demineralization. J. Dent. 2014, 42, 1096–1104.
- Yamaguchi, M. Role of Nutritional Zinc in the Prevention of Osteoporosis. Mol. Cell. Biochem. 2010, 338, 241–254.
- Mayer, I.; Apfelbaum, F.; Featherstone, J.D.B. Zinc Ions in Synthetic Carbonated Hydroxyapatites. Arch. Oral Biol. 1994, 39, 87–90.
- Fuierer, T.A.; LoRe, M.; Puckett, S.A.; Nancollas, G.H. A Mineralization Adsorption and Mobility Study of Hydroxyapatite Surfaces in the Presence of Zinc and Magnesium Ions. Langmuir 1994, 10, 4721–4725.
