1. Piperine Reduces Inflammation
Inflammation has become a target for cancer prevention and therapy. Many studies have evaluated the anti-inflammatory activity of piperine. In fact, in vitro studies using a variety of isolated cells have been used to screen the anti-inflammatory activity of piperine [
12,
25]. In a model of LPS-induced inflammation of nucleus pulposus cells, piperine significantly inhibited multiple inflammatory factors and oxidative stress-associated genes [
26]. In human peripheral blood mononuclear cells (PBMCs), piperine inhibits IL-2 and interferon-gamma (IFN-γ) production [
27]. Additionally, in a model of oxidative stress induced by UV-B in HaCaT keratinocyte cells, piperine also inhibits ROS/RNS production, leading to a decrease in inflammatory mediators such as p38, JNK, AP-1, iNOS (nitric oxide synthase), and COX-2 (cyclooxygenase-2) protein expression [
12]. Piperine also inhibits LPS-induced tumor necrosis factor (TNF)-α, IL-6, IL-1β, and prostaglandin E
2 (PGE
2) production in BV2 microglial cells [
28]. Furthermore, piperine down-regulates pathways associated with IL-1β and nuclear factor-κB (NF-κB) [
29], activates the Nrf2/keap1 pathway, and inhibits TNF-α-induced expressions of cell adhesion molecules such as ICAM-1, VCAM1, and E-selectin. This leads to the blocking of neutrophil adhesion to the endothelium in a time- and concentration-dependent manner [
13,
14]. Similarly, in vivo studies using a wide range of acute and chronic experimental models revealed the anti-inflammatory activity of piperine in a dose-dependent way. In experiments involving carrageenan-induced paw edema, formalin-induced arthritis, croton oil-induced granuloma pouch, and cotton pellet-induced granuloma, piperine demonstrated a 56%, 40%, 40%, and 10% inhibition of inflammation, respectively [
30,
31]. In a research study conducted on fibroblast-like synoviocytes stimulated with IL-1β, derived from patients having rheumatoid arthritis and in an animal model of arthritis, it was found that piperine inhibits IL-6, MMP-13, and AP-1. It also reduces PGE
2 levels in a dose-dependent manner. Furthermore, piperine-treated rats showed a significant reduction in nociceptive and arthritic symptoms [
32].
In summary, piperine consistently proves its potent anti-inflammatory properties across diverse models. Its efficacy is evident in inhibiting inflammatory factors, modulating cytokine production, and down-regulating key pathways. The dose-dependent inhibition of inflammation in various experimental models underscores piperine’s robust potential for addressing inflammatory-related conditions in future research.
2. Piperine Induces Various Cell Death Types
2.1. Apoptosis
Apoptosis can be caused by two main pathways: the intrinsic and extrinsic pathways [
33]. Apoptosis can be triggered by different factors and the way the signal is transmitted also varies, but ultimately, they converge onto the same execution pathway. This results in several changes, such as DNA fragmentation, cytoskeletal and nuclear protein degradation, protein cross-linking, the formation of apoptotic bodies, the expression of phagocytic cell receptor ligands, and finally, uptake by phagocytic cells [
34].
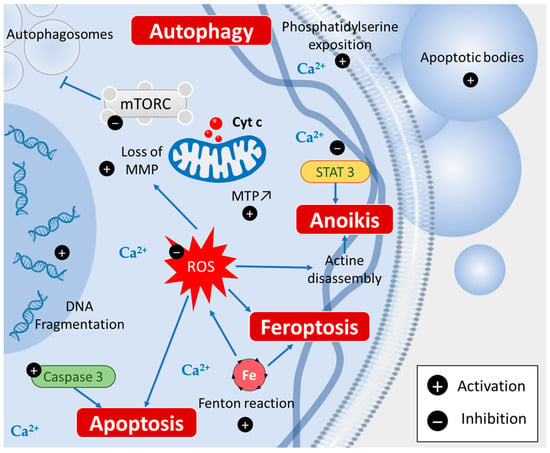
Apoptosis is known to be induced by many chemopreventive agents and piperine can activate both the intrinsic and extrinsic pathways of apoptosis. A study performed by Tawani et al. reported the cytotoxic effects of piperine against four different human cancer cell lines (i.e., breast carcinoma (MCF-7), liver carcinoma (HepG2), cervical carcinoma (HeLa), and prostate cancer cells (PC3)), showing apoptotic characteristics such as cytoplasmic and nuclear condensation, where the externalization of membrane phospholipid phosphatidylserine and DNA cleavage were also observed [
32]. Furthermore, piperine has been shown to target human G-quadruplex DNA sequences, a structure playing a vital role in regulating cellular processes that might contribute to cancer development [
32]. These results were confirmed by Jafri et al., showing that piperine induces apoptosis in a dose-dependent manner by increasing reactive oxygen species (ROS) generation, nuclear condensation, the disruption of mitochondrial membrane potential, DNA fragmentation, and finally, the activation of caspase-3 [
35] (
Figure 3). Furthermore, in vivo studies showed that piperine reduced tumors in an osteosarcoma xenograft mouse model by up-regulating both Bax and p53 expression, as well as reducing Bcl-2 expression [
36].
Figure 3. Piperine induces various types of cell death: apoptosis via caspase 3 activation, ferroptosis via Fenton reaction, autophagy by inhibiting mTORC signaling, and anoikis via STAT3 inhibition.
2.2. Autophagy
Autophagy is also an interesting way by which some chemopreventive agents could act. It is a fundamental cellular process that eliminates molecules and subcellular elements via lysosome-mediated degradation to promote homeostasis, differentiation, development, and survival [
37]. In the context of cancer, autophagy prevents tissue damage and cell death, which can cause cancer initiation and progression [
38]. Furthermore, it is known that the central pathway governing autophagy is led by PI3K/Akt/mTOR signaling [
32,
39]. Strikingly, piperine has been shown to induce autophagy in cancer cells by inhibiting mTORC kinase activity, allowing the formation of autophagosomes [
40,
41]. In this context, piperine inhibited thioredoxin reductase (TrxR) activity, increased ROS levels, reduced mitochondrial membrane potential, and induced autophagy in Bel-7402/5-FU cells via the regulation of autophagy-related proteins LC3, p62, and beclin-1 [
15] (
Figure 3). In vivo experiments demonstrated that piperine induces autophagy by inhibiting PI3K signaling, leading to a decrease in oral cancer tumor growth [
42].
2.3. Ferroptosis
Ferroptosis is also an interesting target of chemopreventive agents. Ferroptosis is characterized by iron and lipidic ROS/peroxides accumulation due to the Fenton reaction and via the loss of balance in ROS production and cell glutathione (GSH)-dependent antioxidants, which protect cells from lipid peroxidation [
43]. Piperine could increase the intracellular Ca
2+ level and ROS in cancer cells also activate the Fenton reaction at high concentrations [
24,
43,
44,
45] (
Figure 3).
2.4. Anoikis
Anoikis, a particular programmed cell death, could be induced by a piperine structural analogous named piperlongumine in melanoma cells in vitro by inhibiting STAT3 [
46] (
Figure 3).
3. Piperine Inhibits Cancer Stem Cells
In a tumor, cancer stem cells (CSCs) are cells that can undergo continuous self-renewal and generate heterogeneous lines of cancer cells. CSCs also contribute to tumor initiation and the relapse of cancers [
47,
48,
49]. CSCs are characterized by the deregulation of cellular energetics, promoting an inflammatory state in the tumor, evading apoptosis, avoiding immune destruction, and resisting anticancer drugs [
49]. At a molecular level, Wnt/β-catenin, Hedgehog, Notch, JAK-STAT (Janus kinase/signal transducers and activators of transcription), NF-κB, PI3K/Akt/mTOR, and TGF/SMAD are fundamental signaling pathways regulating self-renewal and differentiation in CSCs [
50,
51]. Therefore, finding chemopreventive agents capable of targeting those signaling pathways could help prevent tumor formation. Piperine influences all these pathways directly or indirectly.
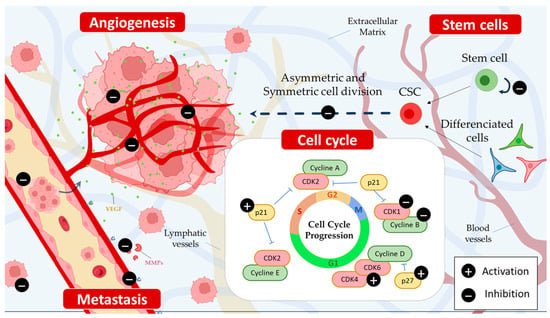
On colorectal and breast cancer cell lines, piperine has been shown to inhibit the Wnt/β-catenin signaling pathway [
51], and PI3K/Akt/mTOR signaling pathways could also be influenced by piperine [
32,
39]. Furthermore, human cervical cancer treated with piperine and mitomycin-C resulted in inactivating STAT3/NF-κB, leading to suppression of the Bcl-2 signaling pathway [
52] (
Figure 4).
Figure 4. Piperine inhibits angiogenesis and metastasis by reducing the expression of vascular endothelial growth factor (VEGF) and down-regulating matrix metalloproteinase (MMP). Additionally, it hinders the self-renewal capacity of cancer stem cells (CSCs) via both direct and indirect mechanisms, targeting critical pathways such as Wnt/β-catenin, Hedgehog, and Notch. Moreover, piperine induces cell cycle arrest at various phases, including G1, G1/S, or G2/M.
4. Piperine Induces Cell Cycle Arrest
The dysregulation of cell cycle control, a fundamental cellular process responsible for maintaining proper cell proliferation and safeguarding cellular integrity, is commonly associated with cancer development [
53,
54]. Two groups of proteins, called cyclins and cyclin-dependent kinases (CDKs), are responsible for the progression of the cell through various checkpoints.
Piperine has shown an inhibition capacity of different protein regulators and checkpoints. Fofaria et al. showed that piperine treatment inhibits the growth of melanoma cells SKMEL 28 and B16F0 in a dose- and time-dependent manner by arresting the cell cycle of both cell lines in the G1 phase [
55]. Their results correlated with the down-regulation of cyclin D1 and the induction of p21 [
55]. Furthermore, the phosphorylation of H2AX at Ser139 supposed that intracellular ROS formation induced DNA damage, leading to apoptosis [
55]. The antiproliferative effect of piperine was also achieved in HT-29 colon carcinoma cells by causing G1 phase cell cycle arrest via the inhibition of cyclins D1 and D3 and their activating partner, cyclin-dependent kinases 4 and 6. A reduction in retinoblastoma protein phosphorylation and an up-regulation of p21/WAF1 and p27/KIP1 was also promoted by piperine [
55]. Piperine can also inhibit the cell cycle at the G2/M phase in cancer cells via the down-regulation of G2-associated (cyclin B, CDK1, Cdc25C) proteins, the induction of p21, and the enhancement of the phosphorylation of both CDK1 and checkpoint kinase 2 (Chk2) [
18,
56,
57] (
Figure 4).
5. Piperine Selectively Inhibits the Growth of Cancer Cells
One of the main limitations of anticancer drugs is their frequent and severe toxic side effects, which are caused by their inability to selectively target cancer cells [
58]. In an attempt to address this limitation, piperine has emerged as a promising candidate due to its selective action on cancer cells while sparing non-transformed cells in vitro. In a study by Si et al., normal ovarian cells exposed to 20 µM piperine did not show significant effects on viability. However, exposure to 8 µM piperine caused a significant decrease in ovarian cancer cell viability after 48 h [
16]. Similarly, when evaluating prostate cells, piperine displayed no cytotoxicity up to 80 µM on prostate epithelial cells, in contrast to two out of the three prostate cancer cell lines tested [
41]. In addition, a 200 µM dose of piperine had a weaker growth inhibitory effect on human hFOB osteoblasts after 72 h of incubation than on human osteosarcoma cells [
18] (
Figure 4). Nevertheless, in vivo studies are needed to confirm and better understand the selectivity of piperine for cancer cells, as these studies will provide insights into whether piperine’s preferential action against cancer cells observed in vitro can be replicated within living organisms.
6. Piperine Inhibits Cancer Invasion and Metastasis Process
Piperine inhibits cancer invasion and metastasis process by having a dual therapeutic potential in cancer by manifesting anti-angiogenic effects, inhibiting the formation of new blood vessels, and displaying anti-metastatic activity, impeding cancer cells from invading other tissues. This is explored in two distinct sections below.
6.1. Anti-Angiogenic Effects of Piperine
Tumor progression is a step characterized by the activation of pathways that promote cell migration and the creation of further blood vessels (angiogenesis) that will allow them to self-sustain via the angiogenic factor release, such as the vascular endothelial growth factor (VEGF) [
59]. In this regard, an in vivo and in vitro experimental study demonstrated that piperine inhibited tubule formation, which is a crucial step in angiogenesis [
60], and inhibited angiogenic activity induced by collagen, caused by breast cancer cells. Piperine also blocked the phosphorylation of Ser 473 and Thr 308 residues of Akt involved in regulating endothelial cells and, therefore, angiogenesis [
17]. To assess the effect of piperine on angiogenesis, another study evaluated VEGF expression levels in the vicinity of piperine. This study found that piperine treatment reduced VEGF expression in a dose-dependent manner, suggesting that the alkaloid negatively regulates this key growth factor in cancer cell migration [
61]. The effect of piperine on crucial angiogenic factors involved in angiogenesis and tumor progression was elucidated in a study conducted in a human breast cancer cell line [
61]. Piperine down-regulated MMP-9 and VEGF mRNA expressions in a dose-dependent manner. In addition, piperine treatment increased the expression levels of E-cadherin, a cell adhesion molecule required to maintain extracellular matrix integrity and cell-to-cell contact, thereby supporting the anti-metastatic potential of this alkaloid [
62] (
Figure 4).
6.2. Anti-Metastatic Activity of Piperine
Metastasis is a process by which cancer cells escape and invade other tissues. Although not all cancer cells reach this stage, metastasis is the leading cause of death from cancer [
63]. An experimental test on a murine lung metastasis model also demonstrated piperine’s anti-metastatic activity. Indeed, piperine effectively reduced the size of tumor nodules and the levels of uronic acid and hexosamine, both involved in the metastasis pathway. Hydroxyproline, a collagen metabolite and a potential marker of tumor cell infiltration into the bones, indicating the presence of metastasis, was also quantitatively reduced via the treatment with piperine [
64]. Another research study investigating the effect of piperine on a human gastric cancer cell line showed that it inhibits the expression of IL-6, which has a prominent role in cancer cell invasion and metastasis via the c-Src/RhoA/ROCK signaling pathway [
56,
65]. Further experiments found that piperine inhibited tumor migration and progression by lowering the expression of MMP-13 and -9 and that high doses of piperine significantly reduced lung metastasis [
63,
64]. Accordingly, experimental studies have elucidated the anti-metastatic activity of piperine by regulating the pathways that involve matrix metalloproteinases [
18].
Table 1. Piperine mechanisms and targets in in vitro and in vivo models for cancer therapy.
| Targets |
Model |
Mechanisms |
References |
| In vitro |
| Inflammation |
|
-
Inhibition of IL-2 and IFN-γ production;
-
Inhibition of ROS/RNS production;
-
Inhibition of the expression of proinflammatory mediators (e.g., p38, iNOS);
-
Down-regulation of IL-1β, NF-κB, and oxidative stress-associated genes.
|
[26,27,28,29] |
| Cell death |
-
MCF-7, HepG2, HeLa, and PC3, Bel-7402/5-FU cell lines
|
-
Induces nuclear condensation, externalization of membrane phospholipid phosphatidylserine, DNA cleavage, increases intracellular ROS, disrupts mitochondrial membrane potential, and activates caspase-3.
-
Inhibits mTORC kinase and TrxR activity, regulates LC3, p62, and beclin-1 and induces formation of autophagosomes.
-
Increase in intracellular Ca2+, ROS, and induction of Fenton reaction.
-
Inhibition of STAT3.
|
[15,32,35,40,41,43,44,45,46] |
| Cancer stem cells |
|
-
Inhibition of Wnt/β-catenin signaling pathway;
-
Inhibition of PI3K/Akt/mTOR signaling pathway;
-
Inactivation of STAT3/NF-κB;
-
Inhibition of Bcl-2 signaling pathway.
|
[32,39,51,52] |
| Cell cycle |
|
-
G1 and G2 phases cell cycle arrest;
-
Down-regulation of cyclin D1 and D3;
-
Inhibition of CDK4 and 6;
-
Up-regulation of p21/WAF1 and p27/KIP1;
-
Phosphorylation of H2AX;
-
Down-regulation of cyclin B, CDK1, and Cdc25C proteins.
|
[18,55,56,57] |
| Cancer cells growth |
|
|
[16,18,41] |
| Invasion and metastasis |
|
-
Inhibition of tubule formation;
-
Down-regulation of MMP-9 and VEGF mRNA expression;
-
Inhibition IL-6 expression;
-
Down-regulation of expression of MMP-13 and MMP-9.
|
[56,60,61,62,63,64,65] |
| In vivo |
| Inflammation |
-
Carrageenan-induced paw edema;
-
Formalin-induced arthritis;
-
Croton oil-induced gran-uloma pouch;
-
Cotton pellet-induced granuloma.
|
-
Significant inhibition of inflammation and arthritic symptoms (e.g., pain, edema);
-
Inhibition of IL-6, MMP-13, and AP-1.
|
[30,31,32] |
| Cell death (apoptosis, autophagy) |
|
-
Up-regulation of Bax and p53 expression;
-
Reduction in Bcl-2 expression;
-
Modulation of Wnt/β-catenin pathway;
-
Synergistically inhibits cancer cell proliferation with Celecoxib.
|
[36,42,66] |
| Invasion and metastasis |
|
|
[63,64] |
This entry is adapted from the peer-reviewed paper 10.3390/cancers15225488