Metabolic-associated Fatty Liver Disease is one of the outstanding challenges in gastroenterology. The increasing incidence of the disease is undoubtedly connected with the ongoing obesity pandemic. The lack of specific symptoms in the early phases and the grave complications of the disease require an active approach to prompt diagnosis and treatment. Therapeutic lifestyle changes should be introduced in a great majority of patients; but, in many cases, the adherence is not satisfactory. There is a great need for an effective pharmacological therapy for Metabolic-Associated Fatty Liver Disease, especially before the onset of steatohepatitis.
- metabolic-associated fatty liver disease
- incretins
- diabetes
1. Introduction
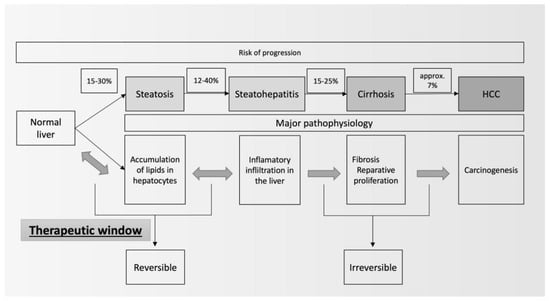
2. Therapeutic Lifestyle Changes
3. Drugs Affecting Carbohydrate Metabolism
3.1. Sodium/Glucose Cotransporter-2 Inhibitors
3.2. Agonists of Glucagon-like Peptide-1 Receptors
3.3. Dual Agonists of Incretin Receptors (Glucagon-like Peptide-1 and Glucose-Dependent Insulinotropic Polypeptide)
3.4. Dual Agonists of Incretin Receptors (Glucagon-like Peptide-1 and Glucagon)
3.5. Triple Incretin Receptor Agonists
3.6. Dipeptidyl Peptidase-4 Inhibitors
3.7. Glitazones
3.8. Ketohexokinase Inhibitors
3.9. Metformin
4. Drugs Affecting Lipid Metabolism
4.1. Core Cholesterol-Lowering Therapy (Statins or/and Ezetimibe)
4.2. PCSK9 Inhibitors
4.3. Peroxisome Proliferator-Activated Receptors Alpha Agonists
4.4. Selective Peroxisome Proliferator-Activated Receptor Modulators
4.5. Multiple Peroxisome Proliferator-Activated Receptor Agonists
4.6. Acetyl-CoA Carboxylase Inhibitors
4.7. Fatty-acid Synthase Inhibitors
4.8. Diacylglycerol Acyltransferase Inhibitors
4.9. Fibroblast Growth Factor 21
5. The Role of Ursodeoxycholic Acid in Metabolic-Associated Fatty Liver Disease
6. The Role of the Microbiota in the Pathogenesis and Management of Metabolic-Associated Fatty Liver Disease
6.1. The Role of Helicobacter pylori in the Pathogenesis and Management of Metabolic-Associated Fatty Liver Disease
6.2. Modification of Microbiota
7. Antioxidants
Vitamin E (Alpha-Tocopherol)
8. Interactions between the Endocrine System and NAFLD
8.1. Thyroid Hormones
8.2. Testosterone
8.3. Estradiol
8.4. Growth Hormone
9. Other Therapies
9.1. Xanthine Oxidase Inhibitors
9.2. Lubiprostone
9.3. Pentoxifylline
9.4. Other Drugs
10. Summary
This entry is adapted from the peer-reviewed paper 10.3390/medicina59101789
References
- Rinella, M.E.; Lazarus, J.V.; Ratziu, V.; Francque, S.M.; Sanyal, A.J.; Kanwal, F.; Romero, D.; Abdelmalek, M.F.; Anstee, Q.M.; Arab, J.P.; et al. A multi-society Delphi consensus statement on new fatty liver disease nomenclature. J. Hepatol. 2023, 2023, 101133.
- Lieverse, R.J.; Jansen, J.B.M.J.; Masclee, A.A.M.; Lamers, C.B.H.W. Gastrointestinal Disturbances with Obesity. Scand. J. Gastroenterol. 1993, 28, 53–58.
- Basha, A.; May, S.C.; Anderson, R.M.; Samala, N.; Mirmira, R.G. Non-Alcoholic Fatty Liver Disease: Translating Disease Mechanisms into Therapeutics Using Animal Models. Int. J. Mol. Sci. 2023, 24, 9996.
- Fernández, T.; Viñuela, M.; Vidal, C.; Barrera, F. Lifestyle changes in patients with non-alcoholic fatty liver disease: A systematic review and meta-analysis. PLoS ONE 2022, 17, e0263931.
- Kim, H.; Lee, D.S.; An, T.H.; Park, H.-J.; Kim, W.K.; Bae, K.-H.; Oh, K.-J. Metabolic Spectrum of Liver Failure in Type 2 Diabetes and Obesity: From NAFLD to NASH to HCC. Int. J. Mol. Sci. 2021, 22, 4495.
- Basu, R.; Noureddin, M.; Clark, J.M. Nonalcoholic Fatty Liver Disease: Review of Management for Primary Care Providers. Mayo Clin. Proc. 2022, 97, 1700–1716.
- European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J. Hepatol. 2016, 64, 1388–1402.
- Mátis, D.; Hegyi, P.; Teutsch, B.; Tornai, T.; Erőss, B.; Pár, G.; Váncsa, S. Improved body composition decreases the fat content in non-alcoholic fatty liver disease, a meta-analysis and systematic review of longitudinal studies. Front. Med. 2023, 10, 1114836.
- Rinella, M.E.; Neuschwander-Tetri, B.A.; Siddiqui, M.S.; Abdelmalek, M.F.; Caldwell, S.; Barb, D.; Kleiner, D.E.; Loomba, R. AASLD Practice Guidance on the clinical assessment and management of nonalcoholic fatty liver disease. Hepatology 2023, 77, 1797–1835.
- Gofton, C.; Upendran, Y.; Zheng, M.-H.; George, J. MAFLD: How is it different from NAFLD? Clin. Mol. Hepatol. 2023, 29, S17–S31.
- Hazlehurst, J.M.; Tomlinson, J.W. Mechanisms in endocrinology: Non-alcoholic fatty liver disease in common endocrine disorders. Eur. J. Endocrinol. 2013, 169, R27–R37.
- Ranjbar, G.; Mikhailidis, D.P.; Sahebkar, A. Effects of newer antidiabetic drugs on nonalcoholic fatty liver and steatohepatitis: Think out of the box! Metabolism 2019, 101, 154001.
- Rieg, T.; Vallon, V. Development of SGLT1 and SGLT2 inhibitors. Diabetologia 2018, 61, 2079–2086.
- Jasleen, B.; Vishal, G.K.; Sameera, M.; Fahad, M.; Brendan, O.; Deion, S.; Pemminati, S. Sodium-Glucose Cotransporter 2 (SGLT2) Inhibitors: Benefits Versus Risk. Cureus 2023, 15, 1–11.
- Van Der Aart-van Der Beek, A.B.; De Boer, R.A.; Heerspink, H.J.L. Kidney and heart failure outcomes associated with SGLT2 inhibitor use. Nat. Rev. Nephrol. 2022, 18, 294–306.
- Nasiri-Ansari, N.; Nikolopoulou, C.; Papoutsi, K.; Kyrou, I.; Mantzoros, C.S.; Kyriakopoulos, G.; Chatzigeorgiou, A.; Kalotychou, V.; Randeva, M.S.; Chatha, K.; et al. Empagliflozin Attenuates Non-Alcoholic Fatty Liver Disease (NAFLD) in High Fat Diet Fed ApoE(-/-) Mice by Activating Autophagy and Reducing ER Stress and Apoptosis. Int. J. Mol. Sci. 2021, 22, 818.
- Shaaban, H.H.; Alzaim, I.; El-Mallah, A.; Aly, R.G.; El-Yazbi, A.F.; Wahid, A. Metformin, pioglitazone, dapagliflozin and their combinations ameliorate manifestations associated with NAFLD in rats via anti-inflammatory, anti-fibrotic, anti-oxidant and anti-apoptotic mechanisms. Life Sci. 2022, 308, 120956.
- Sun, L.; Deng, C.; Gu, Y.; He, Y.; Yang, L.; Shi, J. Effects of dapagliflozin in patients with nonalcoholic fatty liver disease: A systematic review and meta-analysis of randomized controlled trials. Clin. Res. Hepatol. Gastroenterol. 2022, 46, 101876.
- Bellanti, F.; Buglio, A.L.; Dobrakowski, M.; Kasperczyk, A.; Kasperczyk, S.; Aich, P.; Singh, S.P.; Serviddio, G.; Vendemiale, G. Impact of sodium glucose cotransporter-2 inhibitors on liver steatosis/fibrosis/inflammation and redox balance in non-alcoholic fatty liver disease. WJG 2022, 28, 3243–3257.
- Buldak, L.; Machnik, G.; Skudrzyk, E.; Boldys, A.; Maliglowka, M.; Kosowski, M.; Basiak, M.; Buldak, R.J.; Okopien, B. Exenatide prevents statin-related LDL receptor increase and improves insulin secretion in pancreatic beta cells (1.1E7) in a protein kinase A-dependent manner. J. Appl. Biomed. 2022, 20, 130–140.
- Bułdak, Ł.; Skudrzyk, E.; Machnik, G.; Bołdys, A.; Bułdak, R.J.; Okopień, B. Exenatide improves antioxidant capacity and reduces the expression of LDL receptors and PCSK9 in human insulin-secreting 1.1E7 cell line subjected to hyperglycemia and oxidative stress. Postępy Hig. I Med. Doświadczalnej 2022, 76, 16–23.
- Bułdak, Ł.; Machnik, G.; Skudrzyk, E.; Bołdys, A.; Okopień, B. The impact of exenatide (a GLP-1 agonist) on markers of inflammation and oxidative stress in normal human astrocytes subjected to various glycemic conditions. Exp. Ther. Med. 2019, 17, 2861–2869.
- Armstrong, M.J.; Gaunt, P.; Aithal, G.P.; Barton, D.; Hull, D.; Parker, R.; Hazlehurst, J.M.; Guo, K.; Abouda, G.; Aldersley, M.A.; et al. Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): A multicentre, double-blind, randomised, placebo-controlled phase 2 study. Lancet 2016, 387, 679–690.
- Study of Semaglutide for Non-Alcoholic Fatty Liver Disease (NAFLD), a Metabolic Syndrome With Insulin Resistance, Increased Hepatic Lipids, and Increased Cardiovascular Disease Risk (The SLIM LIVER Study). Available online: https://classic.clinicaltrials.gov/ct2/show/NCT04216589 (accessed on 10 August 2023).
- Non-Alcoholic Fatty Liver Disease, the HEpatic Response to Oral Glucose, and the Effect of Semaglutide (NAFLD HEROES). Available online: https://classic.clinicaltrials.gov/ct2/show/NCT03884075 (accessed on 10 August 2023).
- Semaglutide in Nonalcoholic Fatty Liver Disease. Available online: https://classic.clinicaltrials.gov/ct2/show/NCT05813249 (accessed on 10 August 2023).
- A Multiple Dose Study of LY3502970 in Healthy Overweight and Obese Participants. Available online: https://ichgcp.net/clinical-trials-registry/NCT05841238 (accessed on 11 August 2023).
- A Study of LY3502970 in Participants With Obesity or Overweight With Weight-related Comorbidities. Available online: https://classic.clinicaltrials.gov/ct2/show/NCT05051579 (accessed on 11 August 2023).
- Pratt, E.; Ma, X.; Liu, R.; Robins, D.; Coskun, T.; Sloop, K.W.; Haupt, A.; Benson, C. Orforglipron (LY3502970), a novel, oral non-peptide glucagon-like peptide-1 receptor agonist: A Phase 1b, multicentre, blinded, placebo-controlled, randomized, multiple-ascending-dose study in people with type 2 diabetes. Diabetes Obes. Metab. 2023, 25, 2642–2649.
- El, K.; Douros, J.D.; Willard, F.S.; Novikoff, A.; Sargsyan, A.; Perez-Tilve, D.; Wainscott, D.B.; Yang, B.; Chen, A.; Wothe, D.; et al. The incretin co-agonist tirzepatide requires GIPR for hormone secretion from human islets. Nat. Metab. 2023, 5, 945–954.
- Thomas, M.K.; Nikooienejad, A.; Bray, R.; Cui, X.; Wilson, J.; Duffin, K.; Milicevic, Z.; Haupt, A.; Robins, D.A. Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-cell Function and Insulin Sensitivity in Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2021, 106, 388–396.
- A Study of Tirzepatide (LY3298176) in Participants With Nonalcoholic Steatohepatitis (NASH) (SYNERGY-NASH). Available online: https://classic.clinicaltrials.gov/ct2/show/NCT04166773 (accessed on 11 August 2023).
- Hartman, M.L.; Sanyal, A.J.; Loomba, R.; Wilson, J.M.; Nikooienejad, A.; Bray, R.; Karanikas, C.A.; Duffin, K.L.; Robins, D.A.; Haupt, A. Effects of Novel Dual GIP and GLP-1 Receptor Agonist Tirzepatide on Biomarkers of Nonalcoholic Steatohepatitis in Patients With Type 2 Diabetes. Diabetes Care 2020, 43, 1352–1355.
- Romero-Gómez, M.; Lawitz, E.; Shankar, R.R.; Chaudhri, E.; Liu, J.; Lam, R.L.H.; Kaufman, K.D.; Engel, S.S.; Bruzone, S.O.; Coronel, M.J.; et al. A phase IIa active-comparator-controlled study to evaluate the efficacy and safety of efinopegdutide in patients with non-alcoholic fatty liver disease. J. Hepatol. 2023, 79, 888–897.
- Nahra, R.; Wang, T.; Gadde, K.M.; Oscarsson, J.; Stumvoll, M.; Jermutus, L.; Hirshberg, B.; Ambery, P. Effects of Cotadutide on Metabolic and Hepatic Parameters in Adults With Overweight or Obesity and Type 2 Diabetes: A 54-Week Randomized Phase 2b Study. Diabetes Care 2021, 44, 1433–1442.
- Study to Evaluate Efficacy, Safety and Tolerability of HM15211 in Subjects. Available online: https://clinicaltrials.gov/study/NCT04505436 (accessed on 12 August 2023).
- A Study of Multiple Doses of HM15211 in Obese Subjects with NAFLD. Available online: https://classic.clinicaltrials.gov/ct2/show/NCT03744182 (accessed on 12 August 2023).
- A Study of LY3437943 in Participants Who Have Obesity or Are Overweight. Available online: https://clinicaltrials.gov/study/NCT04881760 (accessed on 12 August 2023).
- Jastreboff, A.M.; Kaplan, L.M.; Frías, J.P.; Wu, Q.; Du, Y.; Gurbuz, S.; Coskun, T.; Haupt, A.; Milicevic, Z.; Hartman, M.L. Triple–Hormone-Receptor Agonist Retatrutide for Obesity—A Phase 2 Trial. N. Engl. J. Med. 2023, 389, 514–526.
- Kongmalai, T.; Srinonprasert, V.; Anothaisintawee, T.; Kongmalai, P.; McKay, G.; Attia, J.; Thakkinstian, A. New anti-diabetic agents for the treatment of non-alcoholic fatty liver disease: A systematic review and network meta-analysis of randomized controlled trials. Front. Endocrinol. 2023, 14, 1182037.
- Cusi, K.; Isaacs, S.; Barb, D.; Basu, R.; Caprio, S.; Garvey, W.T.; Kashyap, S.; Mechanick, J.I.; Mouzaki, M.; Nadolsky, K.; et al. American Association of Clinical Endocrinology Clinical Practice Guideline for the Diagnosis and Management of Nonalcoholic Fatty Liver Disease in Primary Care and Endocrinology Clinical Settings. Endocr. Pract. 2022, 28, 528–562.
- Valenzuela-Vallejo, L.; Guatibonza-García, V.; Mantzoros, C.S. Recent guidelines for Non-Alcoholic Fatty Liver disease (NAFLD)/ Fatty Liver Disease (FLD): Are they already outdated and in need of supplementation? Metabolism 2022, 136, 155248.
- Dos Santos, L.R.; Duarte, M.L.; Peccin, M.S.; Gagliardi, A.R.D.T.; Melnik, T. Dipeptidyl Peptidase IV Inhibitors for Nonalcoholic Fatty Liver Disease—Systematic Review and Metanalysis. CDR 2021, 17, e101120187811.
- Alam, S.; Ghosh, J.; Mustafa, G.; Kamal, M.; Ahmad, N. Effect of sitagliptin on hepatic histological activity and fibrosis of nonalcoholic steatohepatitis patients: A 1-year randomized control trial. HMER 2018, 10, 23–31.
- Aktaş, A.; Ozan, Z. The Efficacy and safety of vildagliptin treatment for nonalcoholic fatty liver disease in type 2 diabetes mellitus. Cumhur. Med. J. 2020, 42, 491–499.
- Jin, D.; Cui, Z.; Jin, S.; Zhou, T.; Guo, B.; Gao, P.; Li, G. Comparison of efficacy of anti-diabetics on non-diabetic NAFLD: A network meta-analysis. Front. Pharmacol. 2023, 13, 1096064.
- Hattori, S.; Nomoto, K.; Suzuki, T.; Hayashi, S. Beneficial effect of omarigliptin on diabetic patients with non-alcoholic fatty liver disease/non-alcoholic steatohepatitis. Diabetol. Metab. Syndr. 2021, 13, 28.
- Gupta, V.K. 1029-P: Teneligliptin Significantly Reduces Liver Fat Content (LFC) and Delays Progression of NASH in Type 2 Diabetes Mellitus Patients. Diabetes 2019, 68 (Suppl. S1), 1029-P.
- Chen, L.; Zhang, X.; Zhang, L.; Zheng, D. Effect of Saxagliptin, a Dipeptidyl Peptidase 4 Inhibitor, on Non-Alcoholic Fatty Liver Disease. Diabetes Metab. Syndr. Obes. Targets Ther. 2020, 13, 3507–3518.
- Li, J.-J.; Zhang, P.; Fan, B.; Guo, X.-L.; Zheng, Z.-S. The efficacy of saxagliptin in T2DM patients with non-alcoholic fatty liver disease: Preliminary data. Rev. Assoc. Med. Bras. 2019, 65, 33–37.
- Sanyal, A.J.; Chalasani, N.; Kowdley, K.V.; McCullough, A.; Diehl, A.M.; Bass, N.M.; Neuschwander-Tetri, B.A.; Lavine, J.E.; Tonascia, J.; Unalp, A.; et al. Pioglitazone, Vitamin E, or Placebo for Nonalcoholic Steatohepatitis. N. Engl. J. Med. 2010, 362, 1675–1685.
- Aithal, G.P.; Thomas, J.A.; Kaye, P.V.; Lawson, A.; Ryder, S.D.; Spendlove, I.; Austin, A.S.; Freeman, J.G.; Morgan, L.; Webber, J. Randomized, Placebo-Controlled Trial of Pioglitazone in Nondiabetic Subjects with Nonalcoholic Steatohepatitis. Gastroenterology 2008, 135, 1176–1184.
- Belfort, R.; Harrison, S.A.; Brown, K.; Darland, C.; Finch, J.; Hardies, J.; Balas, B.; Gastaldelli, A.; Tio, F.; Pulcini, J.; et al. A Placebo-Controlled Trial of Pioglitazone in Subjects with Nonalcoholic Steatohepatitis. N. Engl. J. Med. 2006, 355, 2297–2307.
- Cusi, K.; Orsak, B.; Bril, F.; Lomonaco, R.; Hecht, J.; Ortiz-Lopez, C.; Tio, F.; Hardies, J.; Darland, C.; Musi, N.; et al. Long-Term Pioglitazone Treatment for Patients With Nonalcoholic Steatohepatitis and Prediabetes or Type 2 Diabetes Mellitus: A Randomized Trial. Ann. Intern. Med. 2016, 165, 305.
- Bril, F.; Biernacki, D.M.; Kalavalapalli, S.; Lomonaco, R.; Subbarayan, S.K.; Lai, J.; Tio, F.; Suman, A.; Orsak, B.K.; Hecht, J.; et al. Role of Vitamin E for Nonalcoholic Steatohepatitis in Patients With Type 2 Diabetes: A Randomized Controlled Trial. Diabetes Care 2019, 42, 1481–1488.
- Della Pepa, G.; Russo, M.; Vitale, M.; Carli, F.; Vetrani, C.; Masulli, M.; Riccardi, G.; Vaccaro, O.; Gastaldelli, A.; Rivellese, A.A.; et al. Pioglitazone even at low dosage improves NAFLD in type 2 diabetes: Clinical and pathophysiological insights from a subgroup of the TOSCA.IT randomised trial. Diabetes Res. Clin. Pract. 2021, 178, 108984.
- Ndakotsu, A.; Vivekanandan, G. The Role of Thiazolidinediones in the Amelioration of Nonalcoholic Fatty Liver Disease: A Systematic Review. Cureus 2022, 14, 1–9.
- Kazierad, D.J.; Chidsey, K.; Somayaji, V.R.; Bergman, A.J.; Birnbaum, M.J.; Calle, R.A. Inhibition of ketohexokinase in adults with NAFLD reduces liver fat and inflammatory markers: A randomized phase 2 trial. Med 2021, 2, 800–813.e3.
- Pinyopornpanish, K.; Leerapun, A.; Pinyopornpanish, K.; Chattipakorn, N. Effects of Metformin on Hepatic Steatosis in Adults with Nonalcoholic Fatty Liver Disease and Diabetes: Insights from the Cellular to Patient Levels. Gut Liver 2021, 15, 827–840.
- Li, Y.; Liu, L.; Wang, B.; Wang, J.; Chen, D. Metformin in non-alcoholic fatty liver disease: A systematic review and meta-analysis. Biomed. Rep. 2013, 1, 57–64.
- Musso, G.; Cassader, M.; Rosina, F.; Gambino, R. Impact of current treatments on liver disease, glucose metabolism and cardiovascular risk in non-alcoholic fatty liver disease (NAFLD): A systematic review and meta-analysis of randomised trials. Diabetologia 2012, 55, 885–904.
- Loomba, R.; Lutchman, G.; Kleiner, D.E.; Ricks, M.; Feld, J.J.; Borg, B.B.; Modi, A.; Nagabhyru, P.; Sumner, A.E.; Liang, T.J.; et al. Clinical trial: Pilot study of metformin for the treatment of non-alcoholic steatohepatitis. Aliment. Pharmacol. Ther. 2009, 29, 172–182.
- Bugianesi, E.; Gentilcore, E.; Manini, R.; Natale, S.; Vanni, E.; Villanova, N.; David, E.; Rizzetto, M.; Marchesini, G. A Randomized Controlled Trial of Metformin versus Vitamin E or Prescriptive Diet in Nonalcoholic Fatty Liver Disease. Am. J. Gastroenterol. 2005, 100, 1082–1090.
- Ioannou, G.N. The Role of Cholesterol in the Pathogenesis of NASH. Trends Endocrinol. Metab. 2016, 27, 84–95.
- Bułdak, Ł. The treatment of heterozygous familial hypercholesterolemia—A local perspective. Endokrynol. Pol. 2021, 72, 189–190.
- Okopień, B.; Bułdak, Ł.; Bołdys, A. Current and future trends in the lipid lowering therapy. Pharmacol. Rep. 2016, 68, 737–747.
- Boutari, C.; Pappas, P.D.; Anastasilakis, D.; Mantzoros, C.S. Statins’ efficacy in non-alcoholic fatty liver disease: A systematic review and meta-analysis. Clin. Nutr. 2022, 41, 2195–2206.
- Ahsan, F.; Oliveri, F.; Goud, H.K.; Mehkari, Z.; Mohammed, L.; Javed, M.; Althwanay, A.; Rutkofsky, I.H. Pleiotropic Effects of Statins in the Light of Non-Alcoholic Fatty Liver Disease and Non-Alcoholic Steatohepatitis. Cureus 2020, 12, 1–12.
- Athyros, V.G.; Mikhailidis, D.P.; Didangelos, T.P.; Giouleme, O.I.; Liberopoulos, E.N.; Karagiannis, A.; Kakafika, A.I.; Tziomalos, K.; Burroughs, A.K.; Elisaf, M.S. Effect of multifactorial treatment on non-alcoholic fatty liver disease in metabolic syndrome: A randomised study. Curr. Med. Res. Opin. 2006, 22, 873–883.
- Athyros, V.G.; Tziomalos, K.; Gossios, T.D.; Griva, T.; Anagnostis, P.; Kargiotis, K.; Pagourelias, E.D.; Theocharidou, E.; Karagiannis, A.; Mikhailidis, D.P. Safety and efficacy of long-term statin treatment for cardiovascular events in patients with coronary heart disease and abnormal liver tests in the Greek Atorvastatin and Coronary Heart Disease Evaluation (GREACE) Study: A post-hoc analysis. Lancet 2010, 376, 1916–1922.
- Pastori, D.; Pani, A.; Di Rocco, A.; Menichelli, D.; Gazzaniga, G.; Farcomeni, A.; D’Erasmo, L.; Angelico, F.; Del Ben, M.; Baratta, F. Statin liver safety in non-alcoholic fatty liver disease: A systematic review and metanalysis. Brit J. Clin. Pharma 2022, 88, 441–451.
- Zhang, J.; Fu, S.; Liu, D.; Wang, Y.; Tan, Y. Statin can reduce the risk of hepatocellular carcinoma among patients with nonalcoholic fatty liver disease: A systematic review and meta-analysis. Eur. J. Gastroenterol. Hepatol. 2023, 35, 353–358.
- Wargny, M.; Ducluzeau, P.-H.; Petit, J.-M.; Le May, C.; Smati, S.; Arnaud, L.; Pichelin, M.; Bouillet, B.; Lannes, A.; Blanchet, O.; et al. Circulating PCSK9 levels are not associated with the severity of hepatic steatosis and NASH in a high-risk population. Atherosclerosis 2018, 278, 82–90.
- Di Filippo, M.; Vokaer, B.; Seidah, N.G. A case of hypocholesterolemia and steatosis in a carrier of a PCSK9 loss-of-function mutation and polymorphisms predisposing to nonalcoholic fatty liver disease. J. Clin. Lipidol. 2017, 11, 1101–1105.
- Emma, M.R.; Giannitrapani, L.; Cabibi, D.; Porcasi, R.; Pantuso, G.; Augello, G.; Giglio, R.V.; Re, N.L.; Capitano, A.R.; Montalto, G.; et al. Hepatic and circulating levels of PCSK9 in morbidly obese patients: Relation with severity of liver steatosis. Biochim. Et. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2020, 1865, 158792.
- El-Haggar, S.M.; Mostafa, T.M. Comparative clinical study between the effect of fenofibrate alone and its combination with pentoxifylline on biochemical parameters and liver stiffness in patients with non-alcoholic fatty liver disease. Hepatol. Int. 2015, 9, 471–479.
- Araki, E.; Yamashita, S.; Arai, H.; Yokote, K.; Satoh, J.; Inoguchi, T.; Nakamura, J.; Maegawa, H.; Yoshioka, N.; Tanizawa, Y.; et al. Effects of Pemafibrate, a Novel Selective PPARα Modulator, on Lipid and Glucose Metabolism in Patients With Type 2 Diabetes and Hypertriglyceridemia: A Randomized, Double-Blind, Placebo-Controlled, Phase 3 Trial. Diabetes Care 2018, 41, 538–546.
- Ikeda, S.; Sugihara, T.; Hoshino, Y.; Matsuki, Y.; Nagahara, T.; Okano, J.; Kitao, S.; Fujioka, Y.; Yamamoto, K.; Isomoto, H. Pemafibrate Dramatically Ameliorated the Values of Liver Function Tests and Fibrosis Marker in Patients with Non-Alcoholic Fatty Liver Disease. Yonago Acta Med. 2020, 63, 188–197.
- Seko, Y.; Yamaguchi, K.; Umemura, A.; Yano, K.; Takahashi, A.; Okishio, S.; Kataoka, S.; Okuda, K.; Moriguchi, M.; Okanoue, T.; et al. Effect of pemafibrate on fatty acid levels and liver enzymes in non-alcoholic fatty liver disease patients with dyslipidemia: A single-arm, pilot study. Hepatol. Res. 2020, 50, 1328–1336.
- Shinozaki, S.; Tahara, T.; Lefor, A.K.; Ogura, M. Pemafibrate decreases markers of hepatic inflammation in patients with non-alcoholic fatty liver disease. Clin. Exp. Hepatol. 2020, 6, 270–274.
- Roy, S. Clinical Case Series of Decrease in Shear Wave Elastography Values in Ten Diabetic Dyslipidemia Patients Having NAFLD with Saroglitazar 4 mg: An Indian Experience. Case Rep. Med. 2020, 2020, 4287075.
- Mitra, A. An Observational Study of Reduction in Glycemic Parameters and Liver Stiffness by Saroglitazar 4 mg in Patients With Type 2 Diabetes Mellitus and Nonalcoholic Fatty Liver Disease. Cureus 2020, 12, 1–7.
- Fabbrini, E.; Sullivan, S.; Klein, S. Obesity and nonalcoholic fatty liver disease: Biochemical, metabolic, and clinical implications. Hepatology 2010, 51, 679–689.
- Lambert, J.E.; Ramos-Roman, M.A.; Browning, J.D.; Parks, E.J. Increased de novo lipogenesis is a distinct characteristic of individuals with nonalcoholic fatty liver disease. Gastroenterology 2014, 146, 726–735.
- Kim, C.-W.; Addy, C.; Kusunoki, J.; Anderson, N.N.; Deja, S.; Fu, X.; Burgess, S.C.; Li, C.; Ruddy, M.; Chakravarthy, M.; et al. Acetyl CoA Carboxylase Inhibition Reduces Hepatic Steatosis but Elevates Plasma Triglycerides in Mice and Humans: A Bedside to Bench Investigation. Cell Metab. 2017, 26, 394–406.e6.
- Loomba, R.; Kayali, Z.; Noureddin, M.; Ruane, P.; Lawitz, E.J.; Bennett, M.; Wang, L.; Harting, E.; Tarrant, J.M.; McColgan, B.J.; et al. GS-0976 Reduces Hepatic Steatosis and Fibrosis Markers in Patients With Nonalcoholic Fatty Liver Disease. Gastroenterology 2018, 155, 1463–1473.e6.
- O’Farrell, M.; Duke, G.; Crowley, R.; Buckley, D.; Martins, E.B.; Bhattacharya, D.; Friedman, S.L.; Kemble, G. FASN inhibition targets multiple drivers of NASH by reducing steatosis, inflammation and fibrosis in preclinical models. Sci. Rep. 2022, 12, 15661.
- Syed-Abdul, M.M.; Parks, E.J.; Gaballah, A.H.; Bingham, K.; Hammoud, G.M.; Kemble, G.; Buckley, D.; McCulloch, W.; Manrique-Acevedo, C. Fatty Acid Synthase Inhibitor TVB-2640 Reduces Hepatic de Novo Lipogenesis in Males With Metabolic Abnormalities. Hepatology 2020, 72, 103–118.
- Calle, R.A.; Amin, N.B.; Carvajal-Gonzalez, S.; Ross, T.T.; Bergman, A.; Aggarwal, S.; Crowley, C.; Rinaldi, A.; Mancuso, J.; Aggarwal, N.; et al. ACC inhibitor alone or co-administered with a DGAT2 inhibitor in patients with non-alcoholic fatty liver disease: Two parallel, placebo-controlled, randomized phase 2a trials. Nat. Med. 2021, 27, 1836–1848.
- Woo, Y.C.; Xu, A.; Wang, Y.; Lam, K.S.L. Fibroblast growth factor 21 as an emerging metabolic regulator: Clinical perspectives. Clin. Endocrinol. 2013, 78, 489–496.
- Loomba, R.; Lawitz, E.J.; Frias, J.P.; Ortiz-Lasanta, G.; Johansson, L.; Franey, B.B.; Morrow, L.; Rosenstock, M.; Hartsfield, C.L.; Chen, C.-Y.; et al. Safety, pharmacokinetics, and pharmacodynamics of pegozafermin in patients with non-alcoholic steatohepatitis: A randomised, double-blind, placebo-controlled, phase 1b/2a multiple-ascending-dose study. Lancet Gastroenterol. Hepatol. 2023, 8, 120–132.
- Loomba, R.; Sanyal, A.J.; Kowdley, K.V.; Bhatt, D.L.; Alkhouri, N.; Frias, J.P.; Bedossa, P.; Harrison, S.A.; Lazas, D.; Barish, R.; et al. Randomized, Controlled Trial of the FGF21 Analogue Pegozafermin in NASH. N. Engl. J. Med. 2023, 389, 998–1008.
- To Evaluate the Efficacy and Safety of Pegozafermin in Subjects with Severe Hypertriglyceridemia (ENTRUST). Available online: https://classic.clinicaltrials.gov/ct2/show/NCT05852431 (accessed on 24 September 2023).
- Zhang, W.; Tang, Y.; Huang, J.; Hu, H. Efficacy of ursodeoxycholic acid in nonalcoholic fatty liver disease: An updated meta-analysis of randomized controlled trials. Asia Pac. J. Clin. Nutr. 2020, 29, 696–705.
- Ferro, D.; Baratta, F.; Pastori, D.; Cocomello, N.; Colantoni, A.; Angelico, F.; Del Ben, M. New Insights into the Pathogenesis of Non-Alcoholic Fatty Liver Disease: Gut-Derived Lipopolysaccharides and Oxidative Stress. Nutrients 2020, 12, 2762.
- An, L.; Wirth, U.; Koch, D.; Schirren, M.; Drefs, M.; Koliogiannis, D.; Nieß, H.; Andrassy, J.; Guba, M.; Bazhin, A.V.; et al. The Role of Gut-Derived Lipopolysaccharides and the Intestinal Barrier in Fatty Liver Diseases. J. Gastrointest. Surg. 2022, 26, 671–683.
- Carpi, R.Z.; Barbalho, S.M.; Sloan, K.P.; Laurindo, L.F.; Gonzaga, H.F.; Grippa, P.C.; Zutin, T.L.M.; Girio, R.J.S.; Repetti, C.S.F.; Detregiachi, C.R.P.; et al. The Effects of Probiotics, Prebiotics and Synbiotics in Non-Alcoholic Fat Liver Disease (NAFLD) and Non-Alcoholic Steatohepatitis (NASH): A Systematic Review. Int. J. Mol. Sci. 2022, 23, 8805.
- Sharpton, S.R.; Maraj, B.; Harding-Theobald, E.; Vittinghoff, E.; Terrault, N.A. Gut microbiome-targeted therapies in nonalcoholic fatty liver disease: A systematic review, meta-analysis, and meta-regression. Am. J. Clin. Nutr. 2019, 110, 139–149.
- Davani-Davari, D.; Negahdaripour, M.; Karimzadeh, I.; Seifan, M.; Mohkam, M.; Masoumi, S.J.; Berenjian, A.; Ghasemi, Y. Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications. Foods 2019, 8, 92.
- Mavilia-Scranton, M.G.; Wu, G.Y.; Dharan, M. Impact of Helicobacter pylori Infection on the Pathogenesis and Management of Nonalcoholic Fatty Liver Disease. J. Clin. Transl. Hepatol. 2023, 11, 670.
- Dong, Y.; Sun, D.; Wang, Y.; Du, Q.; Zhang, Y.; Han, R.; Teng, M.; Zhang, T.; Shi, L.; Zheng, G.; et al. Evaluation of the current guidelines for antibacterial therapy strategies in patients with cirrhosis or liver failure. BMC Infect. Dis. 2022, 22, 23.
- Gangarapu, V.; Ince, A.T.; Baysal, B.; Kayar, Y.; Kılıç, U.; Gök, Ö.; Uysal, Ö.; Şenturk, H. Efficacy of rifaximin on circulating endotoxins and cytokines in patients with nonalcoholic fatty liver disease. Eur. J. Gastroenterol. Hepatol. 2015, 27, 840–845.
- Abdel-Razik, A.; Mousa, N.; Shabana, W.; Refaey, M.; Elzehery, R.; Elhelaly, R.; Zalata, K.; Abdelsalam, M.; Eldeeb, A.A.; Awad, M.; et al. Rifaximin in nonalcoholic fatty liver disease: Hit multiple targets with a single shot. Eur. J. Gastroenterol. Hepatol. 2018, 30, 1237–1246.
- Podszun, M.C.; Alawad, A.S.; Lingala, S.; Morris, N.; Huang, W.-C.A.; Yang, S.; Schoenfeld, M.; Rolt, A.; Ouwerkerk, R.; Valdez, K.; et al. Vitamin E treatment in NAFLD patients demonstrates that oxidative stress drives steatosis through upregulation of de-novo lipogenesis. Redox Biol. 2020, 37, 101710.
- Podszun, M.C.; Frank, J. Impact of vitamin E on redox biomarkers in non-alcoholic fatty liver disease. Redox Biol. 2021, 42, 101937.
- Luo, Q.; Wei, R.; Cai, Y.; Zhao, Q.; Liu, Y.; Liu, W.J. Efficacy of Off-Label Therapy for Non-alcoholic Fatty Liver Disease in Improving Non-invasive and Invasive Biomarkers: A Systematic Review and Network Meta-Analysis of Randomized Controlled Trials. Front Med. 2022, 9, 793203.
- An, J.; Sohn, J.H. Pharmacological advances in the treatment of nonalcoholic fatty liver diseases: Focused on global results of randomized controlled trials. Clin. Mol. Hepatol. 2023, 29, S268–S275.
- Tanase, D.M.; Gosav, E.M.; Neculae, E.; Costea, C.F.; Ciocoiu, M.; Hurjui, L.L.; Tarniceriu, C.C.; Floria, M. Hypothyroidism-Induced Nonalcoholic Fatty Liver Disease (HIN): Mechanisms and Emerging Therapeutic Options. Int. J. Mol. Sci. 2020, 21, 5927.
- Harrison, S.A.; Bashir, M.R.; Guy, C.D.; Zhou, R.; Moylan, C.A.; Frias, J.P.; Alkhouri, N.; Bansal, M.B.; Baum, S.; Neuschwander-Tetri, B.A.; et al. Resmetirom (MGL-3196) for the treatment of non-alcoholic steatohepatitis: A multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 2019, 394, 2012–2024.
- Polyzos, S.A.; Mousiolis, A.; Mintziori, G.; Goulis, D.G. Nonalcoholic fatty liver disease in males with low testosterone concentrations. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 1571–1577.
- Yassin, A.A.; Alwani, M.; Talib, R.; Almehmadi, Y.; Nettleship, J.E.; Alrumaihi, K.; Albaba, B.; Kelly, D.M.; Saad, F. Long-term testosterone therapy improves liver parameters and steatosis in hypogonadal men: A prospective controlled registry study. Aging Male 2020, 23, 1553–1563.
- Patil, V.; Jothimani, D.; Harika, K.; Hakeem, A.R.; Sachan, D.; Vij, M.; Rela, M. Versatility of Anabolic Androgenic Steroid-Induced Hepatotoxicity. J. Clin. Exp. Hepatol. 2022, 12, 216–221.
- Palmisano, B.T.; Zhu, L.; Stafford, J.M. Role of Estrogens in the Regulation of Liver Lipid Metabolism. Adv. Exp. Med. Biol. 2017, 1043, 227–256.
- Gutierrez-Grobe, Y.; Ponciano-Rodríguez, G.; Ramos, M.H.; Uribe, M.; Méndez-Sánchez, N. Prevalence of non alcoholic fatty liver disease in premenopausal, posmenopausal and polycystic ovary syndrome women. The role of estrogens. Ann. Hepatol. 2010, 9, 402–409.
- Cao, W.; Xu, Y.; Shen, Y.; Wang, Y.; Ma, X.; Bao, Y. Associations between sex hormones and metabolic-associated fatty liver disease in a middle-aged and elderly community. Endocr. J. 2022, 69, 1007–1014.
- Polyzos, S.A.; Lambrinoudaki, I.; Goulis, D.G. Menopausal hormone therapy in women with dyslipidemia and nonalcoholic fatty liver disease. Hormones 2022, 21, 375–381.
- Oxley, M.; Francis, H.; Sato, K. Growth Hormone Signaling in Liver Diseases: Therapeutic Potentials and Controversies. Semin. Liver Dis. 2023, 43, 024–030.
- Hwang, Y.; Lee, H.W.; Ahn, S.H.; Lee, E.J.; Ku, C.R.; Kim, S.U. Positive association between nonalcoholic fatty liver disease and growth hormone deficiency in patients with nonfunctioning pituitary adenoma. Front. Endocrinol. 2023, 13, 1057769.
- Dichtel, L.E.; Corey, K.E.; Haines, M.S.; Chicote, M.L.; Kimball, A.; Colling, C.; Simon, T.G.; Long, M.T.; Husseini, J.; Bredella, M.A.; et al. The GH/IGF-1 Axis Is Associated with Intrahepatic Lipid Content and Hepatocellular Damage in Overweight/Obesity. J. Clin. Endocrinol. Metab. 2022, 107, e3624–e3632.
- Liu, Z.; Que, S.; Zhou, L.; Zheng, S. Dose-response Relationship of Serum Uric Acid with Metabolic Syndrome and Non-alcoholic Fatty Liver Disease Incidence: A Meta-analysis of Prospective Studies. Sci. Rep. 2015, 5, 14325.
- Nishikawa, T.; Nagata, N.; Shimakami, T.; Shirakura, T.; Matsui, C.; Ni, Y.; Zhuge, F.; Xu, L.; Chen, G.; Nagashimada, M.; et al. Xanthine oxidase inhibition attenuates insulin resistance and diet-induced steatohepatitis in mice. Sci. Rep. 2020, 10, 815.
- Kakimoto, M.; Fujii, M.; Sato, I.; Honma, K.; Nakayama, H.; Kirihara, S.; Fukuoka, T.; Ran, S.; Hirohata, S.; Kitamori, K.; et al. Antioxidant action of xanthine oxidase inhibitor febuxostat protects the liver and blood vasculature in SHRSP5/Dmcr rats. J. Appl. Biomed. 2023, 21, 80–90.
- NCT05474560. Available online: https://classic.clinicaltrials.gov/ct2/show/NCT05474560 (accessed on 9 June 2023).
- Kessoku, T.; Kobayashi, T.; Tanaka, K.; Yamamoto, A.; Takahashi, K.; Iwaki, M.; Ozaki, A.; Kasai, Y.; Nogami, A.; Honda, Y.; et al. The Role of Leaky Gut in Nonalcoholic Fatty Liver Disease: A Novel Therapeutic Target. Int. J. Mol. Sci. 2021, 22, 8161.
- Wilson, N.; Schey, R. Lubiprostone in constipation: Clinical evidence and place in therapy. Ther. Adv. Chronic Dis. 2015, 6, 40–50.
- El-Deen, R.M.; Heeba, G.H.; Abdel-latif, R.G.; Khalifa, M.M.A. Comparative effectiveness of phosphodiesterase 3, 4, and 5 inhibitors in amelioration of high-fat diet-induced nonalcoholic fatty liver in rats. Fundam. Clin. Pharmacol. 2020, 34, 353–364.
- Vachliotis, I.D.; Polyzos, S.A. The Role of Tumor Necrosis Factor-Alpha in the Pathogenesis and Treatment of Nonalcoholic Fatty Liver Disease. Curr. Obes. Rep. 2023, 12, 191–206.
- Malladi, N.; Alam, M.J.; Maulik, S.K.; Banerjee, S.K. The role of platelets in non-alcoholic fatty liver disease: From pathophysiology to therapeutics. Prostaglandins Other Lipid Mediat. 2023, 169, 106766.
- Meng, C.; Song, Z.; Zhang, L.; Geng, Y.; Sun, J.; Miao, G.; Liu, P. Effects of losartan in patients with NAFLD: A meta-analysis of randomized controlled trial. Open Life Sci. 2023, 18, 20220583.
