Arginine vasopressin (AVP) is a non-catecholamine hormone produced in the hypothalamus and released into the circulation through the posterior pituitary gland. It was isolated it in 1895 [
1] from the extract of the posterior pituitary. After the identification of the molecule, its potent antidiuretic effects and its benefits in diabetes insipidus (DI) led it to be renamed as antidiuretic hormone (ADH) [
2]. Du Vigneaud synthesized AVP and described its structure in early 1950s, earning him the Nobel Prize in 1955 [
3]. The AVP used as a therapeutic agent is of synthetic origin and is structurally identical to the human peptide hormone. Until about 30 years ago, AVP was employed to manage polyuria in patients with DI [
4] and minimize blood loss in gastrointestinal bleeding [
5]. It was not until the early 1990s that its clinical vasopressor effect began to be used, establishing the drug as potentially useful in the treatment of vasodilatory shock [
6].
2. Physiology
2.1. Synthesis and Release
Arginine vasopressin is a little peptide composed of nine amino acids, with arginine occupying the eighth position [
8]. It is produced by magnocellular neurosecretory neurons located in the anterior hypothalamus that directly function as osmoreceptors. AVP subsequently migrates (as a prohormone) along the supraoptic-hypophyseal tract to the posterior pituitary gland (neurohypophysis), where it is stored in vesicles and is released into the circulation in response to appropriate stimuli. The most important stimuli producing AVP release are mainly an increase in plasma osmolarity and/or decreased blood volume [
9]. Its half-life is 5–15 min [
10].
2.2. AVP Receptors and Signal Transduction
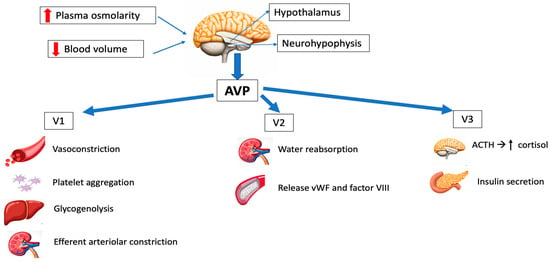
Three different types of AVP receptors have been identified (Figure 1): V1 (previously known as V1a, mainly vascular), V2 (mainly renal) and V3 (previously V1b, mainly central). Its location and function are summarized in Table 1.
Figure 1. Schematic representation of the main physiological effects of vasopressin. AVP: arginine vasopressin; ACTH: adrenocorticotropic hormone; vWF: von Willebrand factor.
Table 1. Vasopressin agonists.
V1 receptors are mainly expressed on vascular smooth muscle, and their activation leads to vasoconstriction. Other effects of V1 stimulation include the endothelial release of nitric oxide (NO), which causes vasodilation of the coronary and pulmonary vessels [
11,
12] and platelet aggregation.
V2 receptors are found in the distal tubal and collecting ducts, regulating the antidiuretic effects of AVP, and on vascular endothelium, releasing coagulation factor VIII and von Willebrand factor (vWF), which are important for blood clot formation on bleeding [
13].
V3 receptors are found in the anterior pituitary, inducing the secretion of adrenocorticotropic hormone (ACTH), and in the pancreas, resulting in insulin secretion [
13,
14].
2.3. Physiological Functions
AVP has a significant role in osmoregulation, cardiovascular stability and overall homeostasis. Additionally, it acts as a corticotropin secretagogue and has an impact on cognition, learning and memory. However, since AVP does not go through the blood–brain barrier, its central nervous system effects are not relevant in the case of intravenous administration of the drug.
- -
-
Osmoregulation
AVP maintains plasma osmolality between 275 and 290 mOsm/kg H
2O. The most important stimuli of AVP release are an increase in osmolarity or decreased blood volume [
9].
DI is caused by a lack of AVP effect. This can be of renal origin (mutations of the V2 receptor) [
15] or central origin, with reduced vasopressin release (idiopathic or secondary to brain tumors, brain ischemia or head injuries) [
16]. The osmoregulatory functions of AVP can be replaced with a synthetic selective V2 receptor agonist known as desmopressin (DDAVP) [
16].
- -
-
Cardiovascular control
The maintenance of arterial blood pressure involves the interaction of sympathetic, renin–angiotensin and AVP systems. Under physiological conditions, the role of AVP in regulating arterial blood pressure is minimal, with the influence of vasopressin on vasomotor tone being minimal in healthy subjects. However, if the two other systems are compromised, AVP can play a more significant role, mainly in clinical scenarios of relative vasopressin deficiency such as sepsis and other vasodilatory shock states [
17].
- -
-
Corticotropin secretion
AVP induces corticotropic axis stimulation (increase in ACTH and cortisol) via the V3 receptors [
18].
- -
-
Hemostasis
AVP promotes blood clotting, with the release of coagulation factor VIII and von Willebrand factor (vWF) [
12]. Desmopressin (DDAVP) is commonly used for bleeding disorders due to having fewer side effects than AVP. In perioperative situations, patients with mild hemophilia A, type 1 von Willebrand disease or congenital or acquired platelet disorders could find advantages in the hemostatic properties of DDAVP [
19].
3. Pharmacology
AVP requires parenteral administration, since trypsin rapidly hydrolyzes the molecule. It is not protein-bound, and the plasma half-life is 5–15 min, so continuous infusion is necessary. The clearance of AVP is mainly mediated by renal and liver vasopressinases [
7,
13], and a little part is eliminated in urine without undergoing changes. Normal plasma concentrations are less than 4 pg/mL.
Throughout the years, many efforts have been made to modify AVP and develop analogs with different pharmacological characteristics that could overcome its limitations [
13,
20]. These analogs involve the alteration of one or more amino acids in the sequence of the molecule, aiming for longer half-lives and better receptor selectivity (
Table 1):
-
Arginine vasopressin (AVP) acts on the V1, V2 and V3 receptors, and has been employed in the management of refractory vasodilatory hypotension, cardiac arrest and septic shock. It has the great advantage of having a short half-life, so the dose can be easily titrated.
-
Desmopressin acetate (DDAVP) is a synthetic agonist with V2 receptor specificity and was first used in management of central diabetes insipidus. By directly affecting the endothelial V2 receptors, DDAVP also raises the plasma factor VIII and vWF concentrations in healthy subjects.
-
Terlipressin (TP) has a greater selectivity for the V1 receptor than AVP. It is a prodrug of AVP and undergoes metabolism by exopeptidases to yield the active metabolite lysine vasopressin in the circulation, producing a “slow release” effect and affording a longer biological half-life (6 h). Terlipressin has been used as treatment for bleeding gastric second to esophageal varices, portal hypertension and septic shock. The drug increases blood pressure and improves the outcomes of hepatorenal syndrome (contracting the mesenteric arteries, resulting in decreased portal venous inflow and subsequently lowering portal pressure) [
5]. The latest Surviving Sepsis Campaign (SSC) recommendations [
21] do not advise its use in patients with septic shock, due to greater undesirable effects (more serious adverse events than NE, especially digital ischemia) [
22,
23].
-
Selepressin, another synthetic vasopressin analog, is a short-acting selective V1 receptor agonist. It may present benefits compared to AVP due its ability to induce pure vasoconstriction; it also has reduced antidiuretic effects, a lower risk of thrombotic complications (because of reduced release of vWF) and affords superior protection from increased permeability. However, recently, a trial was stopped due to futility criteria, because no difference was observed in vasopressor- and ventilator-free days [
24]. The drug is currently not approved for clinical use.
-
Ornipressin exhibits a particular affinity for V1 receptors, thus mimicking the vascular effects of AVP. It has been reported to reduce blood loss during laparoscopic myomectomy [
25] and proved useful in cirrhosis with hepatorenal syndrome [
26].
4. Vasopressin in Septic Shock
Septic shock is the most frequent cause of vasodilatory shock. In 2016, the Third International Consensus Definition for Sepsis and Septic Shock (Sepsis-3) defined sepsis as life-threatening organ dysfunction resulting from dysregulated host responses to infection. Septic shock was defined as a subgroup of sepsis in which circulatory and cellular metabolic abnormalities are severe enough to significantly elevate the risk of mortality: despite adequate fluid resuscitation, patients experience hypotension requiring the use of vasopressors and have a raised serum lactate concentration of over 2 mmol/L [
27]. Sepsis mortality remains higher (25–30%) [
28], and even 40–50% when shock is present [
29].
The Surviving Sepsis Campaign (SSC) [
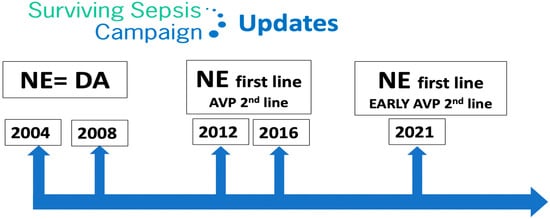
21] is a global initiative aimed at improving the management of sepsis. The campaign was launched in 2002 by the Society of Critical Care Medicine and the European Society of Intensive Care Medicine. The SSC initially focused on creating guidelines for the treatment of sepsis, which were first published in 2004 and were updated in 2008, 2012, 2016 and 2021. These guidelines have become widely accepted as the standard of care for sepsis, to be undertaken as a medical emergency. The SSC guidelines provide evidence-based recommendations for the management of septic shock which include source control, antibiotic therapy, fluid resuscitation, vasopressor therapy, supportive care and monitoring and follow-up.
Regarding vasopressor therapy, since 2012 the SSC recommended NE as the first-line vasopressor agent (it previously recommended either NE or dopamine) and suggested adding AVP. However, since 2021 the SSC suggests adding AVP early [
21], rather than increasing the NE dose (
Figure 2).
Figure 2. Surviving Sepsis Campaign updates. NE: norepinephrine; DA: dopamine; AVP: arginine vasopressin.
The authors of the SSC recognize that certain evidence implies that AVP might be superior to NE in terms of clinical outcomes. However, due to its higher costs and lesser availability, they consider NE as a first-line agent instead of AVP. A recent Cochrane review found that there was insufficient mortality-related evidence to consider any vasopressor as being superior to others [
30]. NE is the first-line vasopressor in septic shock because it has been found to be superior to dopamine and equivalent to AVP and epinephrine in randomized controlled trials (RCTs) [
31,
32,
33,
34,
35,
36] (
Table 2).
Table 2. Pivotal trials of vasopressors in septic shock.
|
TRIAL
|
Intervention
|
Control
|
Intervention 28 Day Mortality
|
Control 28 Day Mortality
|
Absolute Difference (95%CI)
p-Value
|
|
VASST [32]
|
Norepinephrine
|
Vasopressin
|
35.4%
|
39.3%
|
3.9 (−2.9–10.7)
0.26
|
|
VANISH [33]
|
Norepinephrine
|
Vasopressin
|
30.9%
|
27.5%
|
3.4 (−5.4–12.3)
|
|
SOAP II [34]
|
Norepinephrine
|
Dopamine
|
48.5%
|
52.5%
|
1.17 (0.97–1.42)
0.10
|
|
ATHOS-3 [35]
|
Angiotensin II
|
Placebo
|
46%
|
54%
|
Hazard ratio 0.78 (0.57–1.07)
0.12
|
|
CAT [36]
|
Epinephrine
|
Norepinephrine
|
23%
|
27%
|
Hazard ratio 0.87 (0.48–1.58)
0.65
|
|
CATS [37]
|
Epinephrine
|
Norepinephrine + dobutamine
|
40%
|
34%
|
Relative risk 0.86 (0.65–1.14)
0.31
|
5. AVP in Vasodilatory Shock in Heart Surgery
Vasodilatory shock in heart surgery is a well-recognized syndrome occurring in 9–44% of all patients undergoing cardiopulmonary bypass (CPB) procedures, with an associated mortality rate of up to 25% [
106]. It is also known as vasoplegic syndrome, and like the early stages of septic shock, it is characterized by low mean arterial pressure (<60–65 mmHg), markedly low systemic vascular resistance, a normal or elevated cardiac index and a poor or insufficient response to fluid or catecholamine administration. Among other risk factors (
Table 4), vasoplegic syndrome occurs more frequently in populations with congenital heart disease undergoing heart surgery and in patients with heart failure requiring the implantation of a ventricular assist device or heart transplant [
107,
108].
Table 4. Risk factors associated with vasoplegia in heart surgery.
|
Predominant and independent risk factors
|
-
-
-
Type of procedure (OHT, LVAD, CHD, combined surgery)
-
-
-
Reduced ejection fraction < 35%
-
-
-
Thyroid disease
-
-
-
VAD before surgery
-
-
-
Preoperative use of intravenous heparin, ACEIs or beta-blockers
|
|
Other risk factors
|
-
-
-
Duration of CPB
-
-
-
Higher body mass index
-
-
-
Pre-existing endothelial cell activation reflected by higher baseline von Willebrand factor propeptide and sP-selectin levels
-
-
-
High preoperative plasma copeptin concentration
-
-
-
Low AVP concentration
-
-
-
Increased adenosine levels
-
-
-
Ischemia-modified albumin
|
OHT: orthotopic heart transplant; LVAD: left ventricular assist device; CHD: congenital heart disease; VAD: ventricular assist device; ACEIs: angiotensin-converting enzyme inhibitors; CPB: cardiopulmonary bypass; AVP: arginine vasopressin [102].
6. AVP in Cardiac Arrest
Historically, both epinephrine and AVP have been used in the context of circulatory arrest. However, the present evidence shows no benefit in using AVP in this setting, and the current international resuscitation guidelines do not recommend AVP as vasopressor therapy [
124].
A systematic review of vasopressors in adult cardiac arrest was conducted by the International Liaison Committee on Resuscitation Advanced Life Support Task Force and published in 2019. Three RCTs comparing AVP versus epinephrine during out-of-hospital cardiac arrest (OHCA) were included. The results showed no significant differences between the groups in terms of return to spontaneous circulation (ROSC), survival to hospital admission, survival to hospital discharge or survival to hospital discharge with favorable neurological outcome. Subgroup analysis based on initial rhythm also did not reveal statistically significant differences. Additionally, this review included three RCTs comparing the combined use of epinephrine and AVP to epinephrine alone during OHCA, and no significant differences were found in terms of ROSC, survival to hospital admission or survival to discharge [
125].
At present, epinephrine is the only recommended vasopressor during cardiac arrest, with a strong grade of recommendation both in situations of shockable rhythm after an unsuccessful first shock and in situations of non-shockable rhythms. There are recommendations against the administration of AVP instead of or in addition to epinephrine during cardiac arrest [
124].
7. Authorized Indications and Dosages in Europe and the United States of America (USA)
The European Medicines Agency (EMA) authorized AVP for the management of catecholamine refractory hypotension following septic shock in patients older than 18 years. AVP should be administered through continuous intravenous infusion of 0.01 IU per minute using a motor pump. Dependent on the clinical response, the dose may be increased every 15–20 min up to a maximum of 0.03 IU/min.
On the other hand, the USA authorized AVP for vasodilatory shock in adults (sepsis and post-cardiotomy); for septic shock, it recommends starting with a dose of 0.01 IU/min (maximum 0.07 IU/min), and for post-cardiotomy shock, starting with a dose of 0.03 units/minute (maximum 0.1 IU/min).
8. Adverse Effects of AVP
Due to its potent vasoconstrictor effect, there have been concerns about the potential impact of AVP on splanchnic circulation, with a fear of splanchnic ischemia and liver dysfunction [
13]. Experimental studies have refuted these concerns, demonstrating that with adequate fluid resuscitation, mesenteric blood flow and ileal microcirculation remain preserved [
126]. Furthermore, clinical trials have not reported any adverse effects upon splanchnic circulation with the use of AVP [
126].
The rate of serious adverse effects was comparable between the AVP and NE groups in the VASST study [
81]. However, there were more cardiac arrests in patients in the NE group and higher occurrence of digital ischemia and hyponatremia with the AVP group. Other side effects of both AVP and NE include reduced cardiac output, skin necrosis and intestinal ischemia.
AVP induces vasoconstriction in cutaneous blood vessels, and this effect is dose-dependent. A retrospective study found that nearly one-third of patients exposed to AP experienced ischemic skin lesions [
127]. Risk factors associated with the development of ischemic cutaneous lesions included being overweight, receiving a high dose of NE, receiving platelets and fresh frozen plasma transfusions, having a history of peripheral arterial occlusive disease and the occurrence of septic shock. After conducting a multivariate analysis, only the latter two factors remained associated with the occurrence of cutaneous complications.