The embryonic epicardium originates from the proepicardium, an extracardiac primordium constituted by a cluster of mesothelial cells. In early embryos, the embryonic epicardium is characterized by a squamous cell epithelium resting on the myocardium surface. Subsequently, it invades the subepicardial space and thereafter the embryonic myocardium by means of an epithelial–mesenchymal transition. Within the myocardium, epicardial-derived cells present multilineage potential, later differentiating into smooth muscle cells and contributing both to coronary vasculature and cardiac fibroblasts in the mature heart.
1. Origin of the Embryonic Epicardium
During cardiac development, the epicardium originates from an extracardiac primordium, the proepicardium (PE), which is constituted by a cluster of mesothelial cells located on the cephalic and ventral surfaces of the liver-
sinus venosus limit in avian embryos [
1,
2,
3,
4,
5,
6,
7,
8] and the pericardial side of the
septum transversum in mammalian embryos [
9]. In early embryos, the epicardium acquires the form of a squamous cell epithelium that either rests directly on the surface of the myocardium or covers a subepicardial space that appears to be densely populated by mesenchymal cells [
10]. It is also assumed that the epicardium is not a simple mesothelium but that it is made up of discrete clusters of heterogeneous cell types which include a hematopoietic contribution from distinct origins, encased by an extracellular matrix (ECM) that anatomically resembles a stem or progenitor cell ‘niche’ [
11].
It was reported that PE originates in the periphery of the heart forming fields in the lateral plate mesoderm (LPM) as part of an early cardiac progenitor lineage [
12]. A single PE bud is formed during zebrafish cardiogenesis [
13] while in other fish, such as the sturgeons, bilateral primordia are formed which subsequently converge into a single PE structure in the embryonic midline [
14]. Noticeably, in mice, bilateral PE anlage is also established and further develops similarly to sturgeons while in chick embryos only the right-side anlage develops [
15]. Interestingly, chicken PE arises both from the splanchnic layer of the LPM and the somatic mesoderm, which also contributes to the mesothelial portion of the PE that forms the typical villous protrusions [
16,
17,
18]. The above observations suggest that the embryonic left–right signal might play a significant role during PE development. Furthermore, while all proepicardial cells in a given species (e.g., in mice, zebrafish, and chicks) are morphologically similar, they exhibit a distinct differentiation potential due to various marker expressions [
19,
20,
21]. Therefore, the detailed composition of the embryonic epicardium is not well known.
After PE formation, cells translocate to the myocardial surface of the looping heart, where they adhere, migrate, and proliferate to form a squamous epithelial layer: the embryonic epicardium [
22]. It has been described that PE translocation to the myocardium takes place through distinct mechanisms among species, including direct contact and/or the release of free-floating cell clusters (or cysts) into the pericardial cavity or PE cell migration from the
sinus venosus towards the heart along the surface of the inflow tract [
23,
24,
25,
26]. After attachment to the myocardial surface, the cells start to migrate laterally until the complete heart is enveloped by the epicardium [
5]. It has been described that the epicardium surrounding the arterial pole does not originate from the PE but from the coelomic/pericardial mesothelium at the area where the aortic sac leaves the pericardial cavity [
27,
28]. This cell population will contribute to the outer mesenchymal layer of the arterial pole within the pericardial cavity, contributing to the arterial epicardium formation, whereas the PE-derived epicardium will cover the myocardial outflow tract (
Figure 1A).
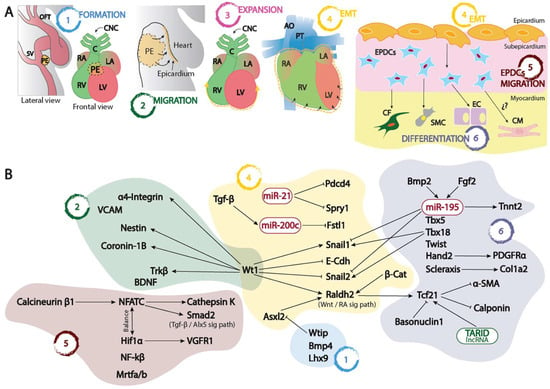
Figure 1. (A) Schematic representation of the proepicardium (PE) and embryonic epicardium formation since its origin at the sinus venosus–septum transversum (1), its migration (2) and expansion (3) into the naked myocardium, its epithelial to mesenchymal transition leading to the formation of the EPCDs (4), and finally the migration and invasion (5) of the embryonic myocardium differentiating into distinct cell types (6). (B) Schematic representation of the distinct transcriptional and post-transcriptional regulatory mechanisms involved in each of the distinct processes depicted in (A). OFT, outflow tract; SV, sinus venosus; PE, proepicardium; RA, right atrium; LA, left atrium; RV, right ventricle; LV, left ventricle; CNC, cardiac neural crest, C, conus; AO, aorta; PT, pulmonary trunk; EPDCs, epicardial derived cells; CF, cardiac fibroblasts; SMC, smooth muscle cells; EC, endothelial cells; CM, cardiomyocytes; EMT, epithelial to mesenchymal transition.
2. Derivates of the Embryonic Epicardium
Once the epicardium is established, epicardial cells will be directly involved in the formation of the myocardium. A group of epithelial cells will undergo an epithelial–mesenchymal transition (EMT), giving rise to epicardium-derived cells (EPDCs), and then migrate into the matrix in the subepicardial layer to form the subepicardium [
22]. The subepicardium thickness will eventually vary according to the underlying heart structure to be covered and may vary among species. In particular, in chick embryos, the subepicardium is relatively thin in the atrial and ventricular myocardium. However, in the atrioventricular sulcus, the subepicardium is thicker in order to provide those EPDCs needed for coronary formation [
29,
30].
From the subepicardium, mesenchymal EPDCs will form migratory processes and invade the myocardium in a spatio-temporarily regulated fashion where several factors expressed in the underlying myocardium will define the permissiveness for EPDCs. These migrate into the underlying myocardium in a tangential pattern and most of them are retained directly underneath the local area of the subepicardium [
5,
30,
31].
Within the myocardium, EPDCs present multi-lineage potential, differentiating into smooth muscle cells (SMCs) and contributing to the coronary vasculature and cardiac fibroblasts (CF) of the mature heart [
22,
26] in line with reports linking EMT and the acquisition of stem cell properties [
32]. At present, it is still controversial as to whether these distinct cell lineages are already established in the proepicardium or if EPDCs acquire pluripotentiality during or after EMT [
33].
Most EPDCs reach their final positions (i) around the coronary arteries as smooth muscle cells (SMCs) and adventitial fibroblasts [
27,
29,
34,
35,
36]; (ii) in the atrioventricular cushions [
27,
31,
35]; (iii) in the subendocardium of the ventricular trabeculae and atria [
27,
30,
31]; and (iv) in the ventricular myocardium as interstitial fibroblasts [
31]. Other contributions of EPDCs to cardiac endothelial cells (ECs) [
37,
38,
39,
40] and cardiomyocytes (CMs) [
41,
42] have also been described although this issue needs further research [
43,
44]. Therefore, both the
sinus venosus and ventricular endocardium are considered major contributors to ECs [
45] while EPDCs have a low contribution, if any [
46]. With respect to epicardial-derived cardiomyocytes, lineage tracing studies, by using Scleraxis, WT1, and TBX18, have indicated possible epicardial-derived cardiomyocyte labeling, although their contribution is still controversial [
41,
43,
44].
Additionally, after the embryonic epicardium has covered the developing heart, the epicardial cells and the EPDCs will produce cytokines and growth factors in order to induce the myocardial development [
25]. These factors and the regulation of their expression are still rather unknown but a number of Fgfs, particularly Fgf-9 and Igf-2, have been proposed to be mitogenic factors for the developing cardiomyocytes [
47,
48,
49]. In this sense, impaired embryonic epicardium development and/or cytokines and growth factor delivery results in deficient ventricular chamber maturation [
47,
50,
51,
52,
53,
54].
In contrast to the embryonic epicardium, the postnatal mammalian epicardium seems to be a dormant single-cell layer since most genes involved in epicardial activation, such as WT1, Tbx18, and Raldh2 are rapidly downregulated postnatally, being scarcely detectable only during the first three months in mice [
55]. This is an interesting issue since regeneration of the injured myocardium in non-mammalian species is dependent on epicardial activation and re-expression of genes characteristic of the embryonic epicardium [
56]. Even the murine heart, which has a very limited regenerative potential, shows a re-expression of the embryonic epicardial gene program and generation of Wt1-lineage positive subepicardial mesenchyme after myocardial infarction [
57].
This entry is adapted from the peer-reviewed paper 10.3390/jcdd10110440