Emerging energy storage systems have received significant attention along with the development of renewable energy, thereby creating a green energy platform for humans. Lithium-ion batteries (LIBs) are commonly used, such as in smartphones, tablets, earphones, and electric vehicles. However, lithium has certain limitations including safety, cost-effectiveness, and environmental issues. Sodium is believed to be an ideal replacement for lithium owing to its infinite abundance, safety, low cost, environmental friendliness, and energy storage behavior similar to that of lithium. Inhered in the achievement in the development of LIBs, sodium-ion batteries (SIBs) have rapidly evolved to be commercialized. Among the cathode, anode, and electrolyte, the cathode remains a significant challenge for achieving a stable, high-rate, and high-capacity device.
- sodium-ion batteries
- cathode materials
- inorganic cathodes
- organic cathodes
1. Introduction
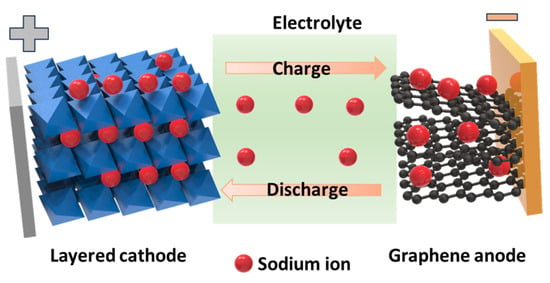
2. SIB Cathode Materials
2.1. Inorganic Compounds
2.1.1. Layered Oxide Materials (NaxMO2)
2.1.2. Tunnel Oxides
2.1.3. Polyanionic Compounds
Phosphate-Based Compound
NASICON
2.1.4. Pyrophosphates
2.1.5. Silicates
2.2. Organic Compounds
2.3. Metal–Organic Compounds: Prussian Blue Analogs
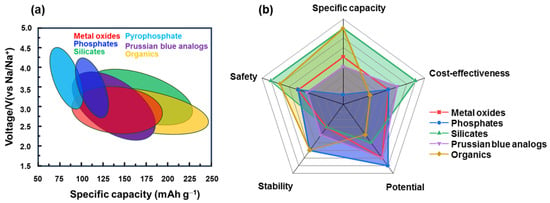
3. Summary
- Mahmud, S.; Rahman, M.; Kamruzzaman, M.; Ali, M.O.; Emon, M.S.A.; Khatun, H.; Ali, M.R. Recent advances in lithium-ion battery materials for improved electrochemical performance: A review. Results Eng. 2022, 15, 100472. [Google Scholar] [CrossRef]
- Liu, J.; Bao, Z.N.; Cui, Y.; Dufek, E.J.; Goodenough, J.B.; Khalifah, P.; Li, Q.Y.; Liaw, B.Y.; Liu, P.; Manthiram, A.; et al. Pathways for practical high-energy long-cycling lithium metal batteries. Nat. Energy 2019, 4, 180–186. [Google Scholar] [CrossRef]
- Boebinger, M.G.; Yarema, O.; Yarema, M.; Unocic, K.A.; Unocic, R.R.; Wood, V.; McDowell, M.T. Spontaneous and reversible hollowing of alloy anode nanocrystals for stable battery cycling. Nat. Nanotechnol. 2020, 15, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Masias, A.; Marcicki, J.; Paxton, W.A. Opportunities and Challenges of Lithium Ion Batteries in Automotive Applications. ACS Energy Lett. 2021, 6, 621–630. [Google Scholar] [CrossRef]
- Walter, M.; Kovalenko, M.V.; Kravchyk, K.V. Challenges and benefits of post-lithium-ion batteries. New J. Chem. 2020, 44, 1677–1683. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, F.; Ming, F.; Alshareef, H.N. Sodium-ion battery anodes: Status and future trends. EnergyChem 2019, 1, 100012. [Google Scholar] [CrossRef]
- Li, X.F.; Dhanabalan, A.; Gu, L.; Wang, C.L. Three-Dimensional Porous Core-Shell Sn@Carbon Composite Anodes for High-Performance Lithium-Ion Battery Applications. Adv. Energy Mater. 2012, 2, 238–244. [Google Scholar] [CrossRef]
- Hatzell, K.B.; Chen, X.C.; Cobb, C.L.; Dasgupta, N.P.; Dixit, M.B.; Marbella, L.E.; McDowell, M.T.; Mukherjee, P.P.; Verma, A.; Viswanathan, V.; et al. Challenges in Lithium Metal Anodes for Solid-State Batteries. ACS Energy Lett. 2020, 5, 922–934. [Google Scholar] [CrossRef]
- Velumani, D.; Bansal, A. Thermal Behavior of Lithium- and Sodium-Ion Batteries: A Review on Heat Generation, Battery Degradation, Thermal Runway—Perspective and Future Directions. Energy Fuels 2022, 36, 14000–14029. [Google Scholar] [CrossRef]
- Ponnada, S.; Kiai, M.S.; Krishnapriya, R.; Singhal, R.; Sharma, R.K. Lithium-Free Batteries: Needs and Challenges. Energy Fuels 2022, 36, 6013–6026. [Google Scholar] [CrossRef]
- Abraham, K.M. How Comparable Are Sodium-Ion Batteries to Lithium-Ion Counterparts? ACS Energy Lett. 2020, 5, 3544–3547. [Google Scholar] [CrossRef]
- Mosallanejad, B.; Malek, S.S.; Ershadi, M.; Daryakenari, A.A.; Cao, Q.; Boorboor Ajdari, F.; Ramakrishna, S. Cycling degradation and safety issues in sodium-ion batteries: Promises of electrolyte additives. J. Electroanal. Chem. 2021, 895, 115505. [Google Scholar] [CrossRef]
- Ellis, B.L.; Nazar, L.F. Sodium and sodium-ion energy storage batteries. Curr. Opin. Solid. State Mater. Sci. 2012, 16, 168–177. [Google Scholar] [CrossRef]
- Liu, Q.; Zhao, X.; Yang, Q.; Hou, L.; Mu, D.; Tan, G.; Li, L.; Chen, R.; Wu, F. The Progress in the Electrolytes for Solid State Sodium-Ion Battery. Adv. Mater. Technol. 2023, 8, 2200822. [Google Scholar] [CrossRef]
- Åvall, G.; Mindemark, J.; Brandell, D.; Johansson, P. Sodium-Ion Battery Electrolytes: Modeling and Simulations. Adv. Energy Mater. 2018, 8, 1703036. [Google Scholar] [CrossRef]
- Wang, B.; Wang, X.; Liang, C.; Yan, M.; Jiang, Y. An All-Prussian-Blue-Based Aqueous Sodium-Ion Battery. ChemElectroChem 2019, 6, 4848–4853. [Google Scholar] [CrossRef]
- Palomares, V.; Casas-Cabanas, M.; Castillo-Martínez, E.; Han, M.H.; Rojo, T. Update on Na-based battery materials: A growing research path. Energy Environ. Sci. 2013, 6, 2312–2337. [Google Scholar] [CrossRef]
- Li, Y.; Wu, F.; Li, Y.; Liu, M.; Feng, X.; Bai, Y.; Wu, C. Ether-based electrolytes for sodium ion batteries. Chem. Soc. Rev. 2022, 51, 4484–4536. [Google Scholar] [CrossRef]
- Wang, X.; Roy, S.; Shi, Q.; Li, Y.; Zhao, Y.; Zhang, J. Progress in and application prospects of advanced and cost-effective iron (Fe)-based cathode materials for sodium-ion batteries. J. Mater. Chem. A 2021, 9, 1938–1969. [Google Scholar] [CrossRef]
- Lee, J.M.; Singh, G.; Cha, W.; Kim, S.; Yi, J.; Hwang, S.-J.; Vinu, A. Recent Advances in Developing Hybrid Materials for Sodium-Ion Battery Anodes. ACS Energy Lett. 2020, 5, 1939–1966. [Google Scholar] [CrossRef]
- Tian, Y.; Zeng, G.; Rutt, A.; Shi, T.; Kim, H.; Wang, J.; Koettgen, J.; Sun, Y.; Ouyang, B.; Chen, T.; et al. Promises and Challenges of Next-Generation “Beyond Li-Ion” Batteries for Electric Vehicles and Grid Decarbonization. Chem. Rev. 2021, 121, 1623–1669. [Google Scholar] [CrossRef]
- Hwang, J.-Y.; Myung, S.-T.; Sun, Y.-K. Sodium-ion batteries: Present and future. Chem. Soc. Rev. 2017, 46, 3529–3614. [Google Scholar] [CrossRef]
- Wen, Y.; He, K.; Zhu, Y.; Han, F.; Xu, Y.; Matsuda, I.; Ishii, Y.; Cumings, J.; Wang, C. Expanded graphite as superior anode for sodium-ion batteries. Nat. Commun. 2014, 5, 4033. [Google Scholar] [CrossRef]
- He, J.; Wei, Y.; Zhai, T.; Li, H. Antimony-based materials as promising anodes for rechargeable lithium-ion and sodium-ion batteries. Mater. Chem. Front. 2018, 2, 437–455. [Google Scholar] [CrossRef]
- Yu, D.Y.W.; Prikhodchenko, P.V.; Mason, C.W.; Batabyal, S.K.; Gun, J.; Sladkevich, S.; Medvedev, A.G.; Lev, O. High-capacity antimony sulphide nanoparticle-decorated graphene composite as anode for sodium-ion batteries. Nat. Commun. 2013, 4, 2922. [Google Scholar] [CrossRef]
- Hwang, J.-Y.; Du, H.-L.; Yun, B.-N.; Jeong, M.-G.; Kim, J.-S.; Kim, H.; Jung, H.-G.; Sun, Y.-K. Carbon-Free TiO2 Microspheres as Anode Materials for Sodium Ion Batteries. ACS Energy Lett. 2019, 4, 494–501. [Google Scholar] [CrossRef]
- Ni, J.; Li, L.; Lu, J. Phosphorus: An Anode of Choice for Sodium-Ion Batteries. ACS Energy Lett. 2018, 3, 1137–1144. [Google Scholar] [CrossRef]
- Durai, L.; Gopalakrishnan, A.; Badhulika, S. Solid-state synthesis of β-NaAlO2 nanoflakes as an anode material for high-performance sodium-ion batteries. Mater. Chem. Front. 2022, 6, 2913–2920. [Google Scholar] [CrossRef]
- Nam, K.-H.; Hwa, Y.; Park, C.-M. Zinc Phosphides as Outstanding Sodium-Ion Battery Anodes. ACS Appl. Mater. Interfaces 2020, 12, 15053–15062. [Google Scholar] [CrossRef]
- Xu, H.; Chen, H.; Gao, C. Advanced Graphene Materials for Sodium/Potassium/Aluminum-Ion Batteries. ACS Mater. Lett. 2021, 3, 1221–1237. [Google Scholar] [CrossRef]
- He, M.; Davis, R.; Chartouni, D.; Johnson, M.; Abplanalp, M.; Troendle, P.; Suetterlin, R.-P. Assessment of the first commercial Prussian blue based sodium-ion battery. J. Power Sources 2022, 548, 232036. [Google Scholar] [CrossRef]
- Yadav, P.; Shelke, V.; Patrike, A.; Shelke, M. Sodium-based batteries: Development, commercialization journey and new emerging chemistries. Oxf. Open Mater. Sci. 2022, 3, itac019. [Google Scholar] [CrossRef]
- Wang, M.; Wang, Q.; Ding, X.; Wang, Y.; Xin, Y.; Singh, P.; Wu, F.; Gao, H. The prospect and challenges of sodium-ion batteries for low-temperature conditions. Interdiscip. Mater. 2022, 1, 373–395. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, T.; Li, W.; Li, T.; Zhang, L.; Zhang, X.; Wang, Z. Engineering of Sodium-Ion Batteries: Opportunities and Challenges. Engineering, 2022; in press. [Google Scholar] [CrossRef]
- Xie, J.; Gu, P.; Zhang, Q. Nanostructured Conjugated Polymers: Toward High-Performance Organic Electrodes for Rechargeable Batteries. ACS Energy Lett. 2017, 2, 1985–1996. [Google Scholar] [CrossRef]
- Zuo, W.; Innocenti, A.; Zarrabeitia, M.; Bresser, D.; Yang, Y.; Passerini, S. Layered Oxide Cathodes for Sodium-Ion Batteries: Storage Mechanism, Electrochemistry, and Techno-economics. Acc. Chem. Res. 2023, 56, 284–296. [Google Scholar] [CrossRef]
- Xiang, X.; Zhang, K.; Chen, J. Recent Advances and Prospects of Cathode Materials for Sodium-Ion Batteries. Adv. Mater. 2015, 27, 5343–5364. [Google Scholar] [CrossRef]
- Stansby, J.H.; Sharma, N.; Goonetilleke, D. Probing the charged state of layered positive electrodes in sodium-ion batteries: Reaction pathways, stability and opportunities. J. Mater. Chem. A 2020, 8, 24833–24867. [Google Scholar] [CrossRef]
- Jiang, L.; Dong, M.; Dou, Y.; Chen, S.; Liu, P.; Yin, H.; Zhao, H. Manganese oxides transformed from orthorhombic phase to birnessite with enhanced electrochemical performance as supercapacitor electrodes. J. Mater. Chem. A 2020, 8, 3746–3753. [Google Scholar] [CrossRef]
- Luo, J.; Huang, A.; Park, S.H.; Suib, S.L.; O’Young, C.-L. Crystallization of Sodium-Birnessite and Accompanied Phase Transformation. Chem. Mater. 1998, 10, 1561–1568. [Google Scholar] [CrossRef]
- Chen, S.; Liao, Z.; Kang, J.; Zhang, Y.; Zhi, S.; Cai, X.; Yang, W.; Zou, H.; Yang, W. Enhanced cyclic performance of O2-type Mn-based layered oxide via Al doping for lithium-ion battery. J. Alloys Compd. 2022, 910, 164793. [Google Scholar] [CrossRef]
- Song, T.; Chen, L.; Gastol, D.; Dong, B.; Marco, J.F.; Berry, F.; Slater, P.; Reed, D.; Kendrick, E. High-Voltage Stabilization of O3-Type Layered Oxide for Sodium-Ion Batteries by Simultaneous Tin Dual Modification. Chem. Mater. 2022, 34, 4153–4165. [Google Scholar] [CrossRef] [PubMed]
- Su, D.; Wang, C.; Ahn, H.-J.; Wang, G. Single Crystalline Na0.7MnO2 Nanoplates as Cathode Materials for Sodium-Ion Batteries with Enhanced Performance. Chem. Eur. J. 2013, 19, 10884–10889. [Google Scholar] [CrossRef] [PubMed]
- Shibata, T.; Fukuzumi, Y.; Kobayashi, W.; Moritomo, Y. Fast discharge process of layered cobalt oxides due to high Na+ diffusion. Sci. Rep. 2015, 5, 9006. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Zhu, W.; Liu, W. First-principles calculations of the Ti-doping effects on layered NaNiO2 cathode materials for advanced Na-ion batteries. J. Indian. Chem. Soc. 2022, 99, 100424. [Google Scholar] [CrossRef]
- Kanwade, A.; Gupta, S.; Kankane, A.; Tiwari, M.K.; Srivastava, A.; Kumar Satrughna, J.A.; Chand Yadav, S.; Shirage, P.M. Transition metal oxides as a cathode for indispensable Na-Ion batteries. RSC Adv. 2022, 12, 23284–23310. [Google Scholar] [CrossRef] [PubMed]
- Heubner, C.; Matthey, B.; Lein, T.; Wolke, F.; Liebmann, T.; Lämmel, C.; Schneider, M.; Herrmann, M.; Michaelis, A. Insights into the electrochemical Li/Na-exchange in layered LiCoO2 cathode material. Energy Stor. Mater. 2020, 27, 377–386. [Google Scholar] [CrossRef]
- Rai, A.K.; Anh, L.T.; Gim, J.; Mathew, V.; Kim, J. Electrochemical properties of NaxCoO2 (x~0.71) cathode for rechargeable sodium-ion batteries. Ceram. Int. 2014, 40, 2411–2417. [Google Scholar] [CrossRef]
- Vassilaras, P.; Ma, X.; Li, X.; Ceder, G. Electrochemical Properties of Monoclinic NaNiO2. J. Electrochem. Soc. 2013, 160, A207. [Google Scholar] [CrossRef]
- Rami Reddy, B.V.; Ravikumar, R.; Nithya, C.; Gopukumar, S. High performance NaxCoO2 as a cathode material for rechargeable sodium batteries. J. Mater. Chem. A 2015, 3, 18059–18063. [Google Scholar] [CrossRef]
- Park, K.; Yu, B.-C.; Goodenough, J.B. Electrochemical and Chemical Properties of Na2NiO2 as a Cathode Additive for a Rechargeable Sodium Battery. Chem. Mater. 2015, 27, 6682–6688. [Google Scholar] [CrossRef]
- Chen, T.; Ouyang, B.; Fan, X.; Zhou, W.; Liu, W.; Liu, K. Oxide cathodes for sodium-ion batteries: Designs, challenges, and perspectives. Carbon. Energy 2022, 4, 170–199. [Google Scholar] [CrossRef]
- Zhang, R.; Lu, Z.; Yang, Y.; Shi, W. First-principles investigation of the monoclinic NaMnO2 cathode material for rechargeable Na-ion batteries. Curr. Appl. Phys. 2018, 18, 1431–1435. [Google Scholar] [CrossRef]
- Palluzzi, M.; Silvestri, L.; Celeste, A.; Tuccillo, M.; Latini, A.; Brutti, S. Structural Degradation of O3-NaMnO2 Positive Electrodes in Sodium-Ion Batteries. Crystals 2022, 12, 885. [Google Scholar] [CrossRef]
- Ma, X.; Chen, H.; Ceder, G. Electrochemical Properties of Monoclinic NaMnO2. J. Electrochem. Soc. 2011, 158, A1307. [Google Scholar] [CrossRef]
- Billaud, J.; Clément, R.J.; Armstrong, A.R.; Canales-Vázquez, J.; Rozier, P.; Grey, C.P.; Bruce, P.G. β-NaMnO2: A High-Performance Cathode for Sodium-Ion Batteries. J. Am. Chem. Soc. 2014, 136, 17243–17248. [Google Scholar] [CrossRef]
- Kubota, K.; Miyazaki, M.; Kim, E.J.; Yoshida, H.; Barpanda, P.; Komaba, S. Structural change induced by electrochemical sodium extraction from layered O′3-NaMnO2. J. Mater. Chem. A 2021, 9, 26810–26819. [Google Scholar] [CrossRef]
- Xiao, J.; Li, X.; Tang, K.; Wang, D.; Long, M.; Gao, H.; Chen, W.; Liu, C.; Liu, H.; Wang, G. Recent progress of emerging cathode materials for sodium ion batteries. Mater. Chem. Front. 2021, 5, 3735–3764. [Google Scholar] [CrossRef]
- Gupta, P.; Pushpakanth, S.; Haider, M.A.; Basu, S. Understanding the Design of Cathode Materials for Na-Ion Batteries. ACS Omega 2022, 7, 5605–5614. [Google Scholar] [CrossRef]
- Kwon, M.-S.; Lim, S.G.; Park, Y.; Lee, S.-M.; Chung, K.Y.; Shin, T.J.; Lee, K.T. P2 Orthorhombic Na0.7[Mn1−xLix]O2+y as Cathode Materials for Na-Ion Batteries. ACS Appl. Mater. Interfaces 2017, 9, 14758–14768. [Google Scholar] [CrossRef]
- Nanthagopal, M.; Ho, C.W.; Shaji, N.; Sim, G.S.; Varun Karthik, M.; Kim, H.K.; Lee, C.W. Enhanced NaFe0.5Mn0.5O2/C Nanocomposite as a Cathode for Sodium-Ion Batteries. Nanomaterials 2022, 12, 984. [Google Scholar] [CrossRef]
- Liu, X.; Zuo, W.; Zheng, B.; Xiang, Y.; Zhou, K.; Xiao, Z.; Shan, P.; Shi, J.; Li, Q.; Zhong, G.; et al. P2-Na0.67AlxMn1−xO2: Cost-Effective, Stable and High-Rate Sodium Electrodes by Suppressing Phase Transitions and Enhancing Sodium Cation Mobility. Angew. Chem. Int. Ed. 2019, 58, 18086–18095. [Google Scholar] [CrossRef]
- Abou-Rjeily, J.; Bezza, I.; Laziz, N.A.; Neacsa, D.; Autret-Lambert, C.; Ghamouss, F. P2-Na0.67Mn0.85Al0.15O2 and NaMn2O4 Blend as Cathode Materials for Sodium-Ion Batteries Using a Natural β-MnO2 Precursor. ACS Omega 2021, 6, 1064–1072. [Google Scholar] [CrossRef]
- Shi, Q.; Qi, R.; Feng, X.; Wang, J.; Li, Y.; Yao, Z.; Wang, X.; Li, Q.; Lu, X.; Zhang, J.; et al. Niobium-doped layered cathode material for high-power and low-temperature sodium-ion batteries. Nat. Commun. 2022, 13, 3205. [Google Scholar] [CrossRef]
- Zuo, W.; Liu, X.; Qiu, J.; Zhang, D.; Xiao, Z.; Xie, J.; Ren, F.; Wang, J.; Li, Y.; Ortiz, G.F.; et al. Engineering Na+-layer spacings to stabilize Mn-based layered cathodes for sodium-ion batteries. Nat. Commun. 2021, 12, 4903. [Google Scholar] [CrossRef] [PubMed]
- Clément, R.J.; Bruce, P.G.; Grey, C.P. Review—Manganese-Based P2-Type Transition Metal Oxides as Sodium-Ion Battery Cathode Materials. J. Electrochem. Soc. 2015, 162, A2589. [Google Scholar] [CrossRef]
- Liu, Q.; Hu, Z.; Chen, M.; Zou, C.; Jin, H.; Wang, S.; Gu, Q.; Chou, S. P2-type Na2/3Ni1/3Mn2/3O2 as a cathode material with high-rate and long-life for sodium ion storage. J. Mater. Chem. A 2019, 7, 9215–9221. [Google Scholar] [CrossRef]
- Zheng, L.; Wang, Z.; Wu, M.; Xu, B.; Ouyang, C. Jahn–Teller type small polaron assisted Na diffusion in NaMnO2 as a cathode material for Na-ion batteries. J. Mater. Chem. A 2019, 7, 6053–6061. [Google Scholar] [CrossRef]
- Nathan, M.G.T.; Yu, H.; Kim, G.-T.; Kim, J.-H.; Cho, J.S.; Kim, J.; Kim, J.-K. Recent Advances in Layered Metal-Oxide Cathodes for Application in Potassium-Ion Batteries. Adv. Sci. 2022, 9, 2105882. [Google Scholar] [CrossRef]
- Xu, J.; Liu, H.; Meng, Y.S. Exploring Li substituted O3-structured layered oxides NaLixNi1/3−xMn1/3+xCo1/3−xO2 (x = 0.07, 0.13, and 0.2) as promising cathode materials for rechargeable Na batteries. Electrochem. Commun. 2015, 60, 13–16. [Google Scholar] [CrossRef]
- Liu, H.; Xu, J.; Ma, C.; Meng, Y.S. A new O3-type layered oxide cathode with high energy/power density for rechargeable Na batteries. Chem. Commun. 2015, 51, 4693–4696. [Google Scholar] [CrossRef]
- Kataoka, R.; Mukai, T.; Yoshizawa, A.; Sakai, T. Development of High Capacity Cathode Material for Sodium Ion Batteries Na0.95Li0.15(Ni0.15Mn0.55Co0.1)O2. J. Electrochem. Soc. 2013, 160, A933–A939. [Google Scholar] [CrossRef]
- Banik, T.; Bhattacharya, I. Novel P2-Type Na0.66Fe0.5-2xMn0.5TixVxO2 Cathode for High-Capacity. In Electrochemical Society Meeting Abstracts 240; MA2021-02; The Electrochemical Society, Inc.: Pennington, NJ, USA, 2021; p. 238. [Google Scholar] [CrossRef]
- Xu, J.; Lee, D.H.; Clément, R.J.; Yu, X.; Leskes, M.; Pell, A.J.; Pintacuda, G.; Yang, X.-Q.; Grey, C.P.; Meng, Y.S. Identifying the Critical Role of Li Substitution in P2–Nax[LiyNizMn1−y−z]O2 (0 < x, y, z < 1) Intercalation Cathode Materials for High-Energy Na-Ion Batteries. Chem. Mater. 2014, 26, 1260–1269. [Google Scholar] [CrossRef]
- Fu, F.; Liu, X.; Fu, X.; Chen, H.; Huang, L.; Fan, J.; Le, J.; Wang, Q.; Yang, W.; Ren, Y.; et al. Entropy and crystal-facet modulation of P2-type layered cathodes for long-lasting sodium-based batteries. Nat. Commun. 2022, 13, 2826. [Google Scholar] [CrossRef]
- Guo, Y.-J.; Wang, P.-F.; Niu, Y.-B.; Zhang, X.-D.; Li, Q.; Yu, X.; Fan, M.; Chen, W.-P.; Yu, Y.; Liu, X.; et al. Boron-doped sodium layered oxide for reversible oxygen redox reaction in Na-Ion battery cathodes. Nat. Commun. 2021, 12, 5267. [Google Scholar] [CrossRef]
- Kim, S.; Min, K.; Park, K. Y-doped P2-type Na0.67Ni0.33Mn0.67O2: A sodium-ion battery cathode with fast charging and enhanced cyclic performance. J. Alloys Compd. 2021, 874, 160027. [Google Scholar] [CrossRef]
- Clément, R.J.; Billaud, J.; Robert Armstrong, A.; Singh, G.; Rojo, T.; Bruce, P.G.; Grey, C.P. Structurally stable Mg-doped P2-Na2/3Mn1−yMgyO2 sodium-ion battery cathodes with high rate performance: Insights from electrochemical, NMR and diffraction studies. Energy Environ. Sci. 2016, 9, 3240–3251. [Google Scholar] [CrossRef]
- Yu, C.-Y.; Park, J.-S.; Jung, H.-G.; Chung, K.-Y.; Aurbach, D.; Sun, Y.-K.; Myung, S.-T. NaCrO2 cathode for high-rate sodium-ion batteries. Energy Environ. Sci. 2015, 8, 2019–2026. [Google Scholar] [CrossRef]
- Yabuuchi, N.; Yoshida, H.; Komaba, S. Crystal Structures and Electrode Performance of Alpha-NaFeO2 for Rechargeable Sodium Batteries. Electrochemistry 2012, 80, 716–719. [Google Scholar] [CrossRef]
- Ono, Y.; Yui, Y.; Hayashi, M.; Asakura, K.; Kitabayashi, H.; Takahashi, K.I. Electrochemical Properties of NaCuO2 for Sodium-Ion Secondary Batteries. ECS Trans. 2014, 58, 33–39. [Google Scholar] [CrossRef]
- Liang, J.; Liu, L.; Liu, X.; Meng, X.; Zeng, L.; Liu, J.; Li, J.; Shi, Z.; Yang, Y. O3-Type NaCrO2 as a Superior Cathode Material for Sodium/Potassium-Ion Batteries Ensured by High Structural Reversibility. ACS Appl. Mater. Interfaces 2021, 13, 22635–22645. [Google Scholar] [CrossRef]
- Myung, S.-T.; Park, J.s.; Jung, H.-G.; Chung, K.Y.; Aurbach, D.; Yu, C.-y.; Sun, Y.-K. NaCrO2 Cathode for High-Rate Sodium-Ionbatteries. In Electrochemical Society Meeting Abstracts 230; MA2016-02; The Electrochemical Society, Inc.: Pennington, NJ, USA, 2016; p. 664. [Google Scholar] [CrossRef]
- Wang, Z.; Shaw, L. Doping of NaCrO2 Cathode Material to Enhance Electrochemical Performance for Sodium-Ion Batteries. In Electrochemical Society Meeting Abstracts 239; MA2021-01; The Electrochemical Society, Inc.: Pennington, NJ, USA, 2021; p. 356. [Google Scholar] [CrossRef]
- Ono, Y. Structural Analysis of NaCuO2 Cathode at Various Charged/Discharged Stages and Its Reaction Mechanism. Electrochemistry 2018, 86, 309–314. [Google Scholar] [CrossRef]
- Lee, E.; Brown, D.E.; Alp, E.E.; Ren, Y.; Lu, J.; Woo, J.-J.; Johnson, C.S. New Insights into the Performance Degradation of Fe-Based Layered Oxides in Sodium-Ion Batteries: Instability of Fe3+/Fe4+ Redox in α-NaFeO2. Chem. Mater. 2015, 27, 6755–6764. [Google Scholar] [CrossRef]
- Feng, J.; Luo, S.; Cai, K.; Yan, S.; Wang, Q.; Zhang, Y.; Liu, X. Research progress of tunnel-type sodium manganese oxide cathodes for SIBs. Chin. Chem. Lett. 2022, 33, 2316–2326. [Google Scholar] [CrossRef]
- Byles, B.; Pomerantseva, E. Stabilization of Tunnel Manganese Oxide Electrodes in Li-Ion and Na-Ion Batteries. In Electrochemical Society Meeting Abstracts 233; MA2018-01; The Electrochemical Society, Inc.: Pennington, NJ, USA, 2018; p. 2581. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, J.; Lee, B.; Qiao, R.; Yang, Z.; Xu, S.; Yu, X.; Gu, L.; Hu, Y.-S.; Yang, W.; et al. Ti-substituted tunnel-type Na0.44MnO2 oxide as a negative electrode for aqueous sodium-ion batteries. Nat. Commun. 2015, 6, 6401. [Google Scholar] [CrossRef]
- Oz, E.; Altin, S.; Avci, S. Tunnel/Layer Composite Na0.44MnO2 Cathode Material with Enhanced Structural Stability via Cobalt Doping for Sodium-Ion Batteries. ACS Omega 2023, 8, 27170–27178. [Google Scholar] [CrossRef]
- Parant, J.-P.; Olazcuaga, R.; Devalette, M.; Fouassier, C.; Hagenmuller, P. Sur quelques nouvelles phases de formule NaxMnO2 (x ≤ 1). J. Solid. State Chem. 1971, 3, 1–11. [Google Scholar] [CrossRef]
- Hosono, E.; Matsuda, H.; Honma, I.; Fujihara, S.; Ichihara, M.; Zhou, H. Synthesis of single crystalline electro-conductive Na0.44MnO2 nanowires with high aspect ratio for the fast charge–discharge Li ion battery. J. Power Sources 2008, 182, 349–352. [Google Scholar] [CrossRef]
- Zhou, X.; Guduru, R.K.; Mohanty, P. Synthesis and characterization of Na0.44MnO2 from solution precursors. J. Mater. Chem. A 2013, 1, 2757–2761. [Google Scholar] [CrossRef]
- Shen, K.-Y.; Lengyel, M.; Wang, L.; Axelbaum, R.L. Spray pyrolysis and electrochemical performance of Na0.44MnO2 for sodium-ion battery cathodes. MRS Commun. 2017, 7, 74–77. [Google Scholar] [CrossRef]
- Zhang, J.; Yuan, H.; Huang, Y.; Kan, S.; Wu, Y.; Bu, M.; Liu, Y.; He, P.; Liu, H. Engineering sodium-rich manganese oxide with robust tunnel structure for high-performance sodium-ion battery cathode application. Chem. Eng. J. 2021, 417, 128097. [Google Scholar] [CrossRef]
- Chae, M.S.; Elias, Y.; Aurbach, D. Tunnel-Type Sodium Manganese Oxide Cathodes for Sodium-Ion Batteries. ChemElectroChem 2021, 8, 798–811. [Google Scholar] [CrossRef]
- Kim, D.J.; Ponraj, R.; Kannan, A.G.; Lee, H.-W.; Fathi, R.; Ruffo, R.; Mari, C.M.; Kim, D.K. Diffusion behavior of sodium ions in Na0.44MnO2 in aqueous and non-aqueous electrolytes. J. Power Sources 2013, 244, 758–763. [Google Scholar] [CrossRef]
- He, X.; Wang, J.; Qiu, B.; Paillard, E.; Ma, C.; Cao, X.; Liu, H.; Stan, M.C.; Liu, H.; Gallash, T.; et al. Durable high-rate capability Na0.44MnO2 cathode material for sodium-ion batteries. Nano Energy 2016, 27, 602–610. [Google Scholar] [CrossRef]
- Guo, S.; Yu, H.; Liu, D.; Tian, W.; Liu, X.; Hanada, N.; Ishida, M.; Zhou, H. A novel tunnel Na0.61Ti0.48Mn0.52O2 cathode material for sodium-ion batteries. Chem. Commun. 2014, 50, 7998–8001. [Google Scholar] [CrossRef]
- Xu, S.; Wang, Y.; Ben, L.; Lyu, Y.; Song, N.; Yang, Z.; Li, Y.; Mu, L.; Yang, H.-T.; Gu, L.; et al. Fe-Based Tunnel-Type Na0.61[Mn0.27Fe0.34Ti0.39]O2 Designed by a New Strategy as a Cathode Material for Sodium-Ion Batteries. Adv. Energy Mater. 2015, 5, 1501156. [Google Scholar] [CrossRef]
- Shi, W.-J.; Zheng, Y.-M.; Meng, X.-M.; Liu, S.-B.; Xu, S.-D.; Chen, L.; Wang, X.-M.; Zhang, D. Designing Sodium Manganese Oxide with 4 d-Cation Zr Doping as a High-Rate-Performance Cathode for Sodium-Ion Batteries. ChemElectroChem 2020, 7, 2545–2552. [Google Scholar] [CrossRef]
- Zheng, P.; Su, J.; Wang, Y.; Zhou, W.; Song, J.; Su, Q.; Reeves-McLaren, N.; Guo, S. A High-Performance Primary Nanosheet Heterojunction Cathode Composed of Na0.44MnO2 Tunnels and Layered Na2Mn3O7 for Na-Ion Batteries. ChemSusChem 2020, 13, 1793–1799. [Google Scholar] [CrossRef]
- Zhou, Y.-T.; Sun, X.; Zou, B.-K.; Liao, J.-Y.; Wen, Z.-Y.; Chen, C.-H. Cobalt-substituted Na0.44Mn1-xCoxO2: Phase evolution and a high capacity positive electrode for sodium-ion batteries. Electrochim. Acta 2016, 213, 496–503. [Google Scholar] [CrossRef]
- Han, D.-W.; Ku, J.-H.; Kim, R.-H.; Yun, D.-J.; Lee, S.-S.; Doo, S.-G. Aluminum Manganese Oxides with Mixed Crystal Structure: High-Energy-Density Cathodes for Rechargeable Sodium Batteries. ChemSusChem 2014, 7, 1870–1875. [Google Scholar] [CrossRef]
- Tang, W.; Song, X.; Du, Y.; Peng, C.; Lin, M.; Xi, S.; Tian, B.; Zheng, J.; Wu, Y.; Pan, F.; et al. High-performance NaFePO4 formed by aqueous ion-exchange and its mechanism for advanced sodium ion batteries. J. Mater. Chem. A 2016, 4, 4882–4892. [Google Scholar] [CrossRef]
- Ling, M.; Lv, Z.; Li, F.; Zhao, J.; Zhang, H.; Hou, G.; Zheng, Q.; Li, X. Revisiting of Tetragonal NaVPO4F: A High Energy Density Cathode for Sodium-Ion Batteries. ACS Appl. Mater. Interfaces 2020, 12, 30510–30519. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, A.; Kim, S.; Fister, T.T.; Johnson, C.S. Microwave-Assisted Synthesis of NaCoPO4 Red-Phase and Initial Characterization as High Voltage Cathode for Sodium-Ion Batteries. ACS Appl. Mater. Interfaces 2017, 9, 4391–4396. [Google Scholar] [CrossRef] [PubMed]
- Priyanka, V.; Savithiri, G.; Subadevi, R.; Sivakumar, M. An emerging electrochemically active maricite NaMnPO4 as cathode material at elevated temperature for sodium-ion batteries. Appl. Nanosci. 2020, 10, 3945–3951. [Google Scholar] [CrossRef]
- Mathew, V.; Kim, S.; Kang, J.; Gim, J.; Song, J.; Baboo, J.P.; Park, W.; Ahn, D.; Han, J.; Gu, L.; et al. Amorphous iron phosphate: Potential host for various charge carrier ions. NPG Asia Mater. 2014, 6, e138. [Google Scholar] [CrossRef]
- Wang, R.; Wu, S.; Zhang, F.; Zhao, X.; Lin, Z.; Wang, C.-Z.; Ho, K.-M. Stabilizing the crystal structures of NaFePO4 with Li substitutions. Phys. Chem. Chem. Phys. 2020, 22, 13975–13980. [Google Scholar] [CrossRef]
- Ali, G.; Lee, J.-H.; Susanto, D.; Choi, S.-W.; Cho, B.W.; Nam, K.-W.; Chung, K.Y. Polythiophene-Wrapped Olivine NaFePO4 as a Cathode for Na-Ion Batteries. ACS Appl. Mater. Interfaces 2016, 8, 15422–15429. [Google Scholar] [CrossRef]
- Priyanka, V.; Savithiri, G.; Rajkumar, P.; Meenatchi, T.; Subadevi, R.; Sivakumar, M. Tweaking the electrochemical activity of maricite NaMnPO4 in sodium batteries using different manganese precursors via polyol method. J. Solid. State Chem. 2020, 290, 121551. [Google Scholar] [CrossRef]
- Venkatachalam, P.; Ganesan, S.; Rengapillai, S.; Marimuthu, S. Gradual Development of Maricite NaMnPO4 with the Influence of Diol Chain Length on the Polyol Process of Surpassed Sodium Intercalation. Ind. Eng. Chem. Res. 2021, 60, 5861–5868. [Google Scholar] [CrossRef]
- Kosova, N.V.; Podugolnikov, V.R.; Devyatkina, E.T.; Slobodyuk, A.B. Structure and electrochemistry of NaFePO4 and Na2FePO4F cathode materials prepared via mechanochemical route. Mater. Res. Bull. 2014, 60, 849–857. [Google Scholar] [CrossRef]
- Jian, Z.; Yuan, C.; Han, W.; Lu, X.; Gu, L.; Xi, X.; Hu, Y.-S.; Li, H.; Chen, W.; Chen, D.; et al. Atomic Structure and Kinetics of NASICON NaxV2(PO4)3 Cathode for Sodium-Ion Batteries. Adv. Funct. Mater. 2014, 24, 4265–4272. [Google Scholar] [CrossRef]
- Goodenough, J.B.; Hong, H.Y.P.; Kafalas, J.A. Fast Na+-ion transport in skeleton structures. Mater. Res. Bull. 1976, 11, 203–220. [Google Scholar] [CrossRef]
- Hong, H.Y.P. Crystal structures and crystal chemistry in the system Na1+xZr2SixP3−xO12. Mater. Res. Bull. 1976, 11, 173–182. [Google Scholar] [CrossRef]
- Oh, J.A.S.; He, L.; Plewa, A.; Morita, M.; Zhao, Y.; Sakamoto, T.; Song, X.; Zhai, W.; Zeng, K.; Lu, L. Composite NASICON (Na3Zr2Si2PO12) Solid-State Electrolyte with Enhanced Na+ Ionic Conductivity: Effect of Liquid Phase Sintering. ACS Appl. Mater. Interfaces 2019, 11, 40125–40133. [Google Scholar] [CrossRef]
- Gopalakrishnan, J.; Rangan, K.K. Vanadium phosphate (V2(PO4)3): A novel NASICO N-type vanadium phosphate synthesized by oxidative deintercalation of sodium from sodium vanadium phosphate (Na3V2(PO4)3). Chem. Mater. 1992, 4, 745–747. [Google Scholar] [CrossRef]
- Zhu, Y.; Xu, H.; Ma, J.; Chen, P.; Chen, Y. The recent advances of NASICON-Na3V2(PO4)3 cathode materials for sodium-ion batteries. J. Solid. State Chem. 2023, 317, 123669. [Google Scholar] [CrossRef]
- Pandit, B.; Sougrati, M.T.; Fraisse, B.; Monconduit, L. Exploration of a Na3V2(PO4)3/C-Pb full cell Na-ion prototype. Nano Energy 2022, 95, 107010. [Google Scholar] [CrossRef]
- Zhang, X.; Rui, X.; Chen, D.; Tan, H.; Yang, D.; Huang, S.; Yu, Y. Na3V2(PO4)3: An advanced cathode for sodium-ion batteries. Nanoscale 2019, 11, 2556–2576. [Google Scholar] [CrossRef]
- Song, W.; Cao, X.; Wu, Z.; Chen, J.; Huangfu, K.; Wang, X.; Huang, Y.; Ji, X. A study into the extracted ion number for NASICON structured Na3V2(PO4)3 in sodium-ion batteries. Phys. Chem. Chem. Phys. 2014, 16, 17681–17687. [Google Scholar] [CrossRef]
- Cao, X.; Sun, Q.; Zhu, L.; Xie, L. Na3V2(PO4)3 nanoparticles confined in functional carbon framework towards high-rate and ultralong-life sodium storage. J. Alloys Compd. 2019, 791, 296–306. [Google Scholar] [CrossRef]
- Lim, S.-J.; Han, D.-W.; Nam, D.-H.; Hong, K.-S.; Eom, J.-Y.; Ryu, W.-H.; Kwon, H.-S. Structural enhancement of Na3V2(PO4)3/C composite cathode materials by pillar ion doping for high power and long cycle life sodium-ion batteries. J. Mater. Chem. A 2014, 2, 19623–19632. [Google Scholar] [CrossRef]
- Pal, S.K.; Thirupathi, R.; Chakrabarty, S.; Omar, S. Improving the Electrochemical Performance of Na3V2(PO4)3 Cathode in Na-Ion Batteries by Si-Doping. ACS Appl. Energy Mater. 2020, 3, 12054–12065. [Google Scholar] [CrossRef]
- Chen, Y.; Cheng, J.; Wang, Y.; Wang, C.; He, Z.; Li, D.; Guo, L. Insights into the elevated electrochemical performance and kinetic characteristics of magnesium-substituted Na3V2−xMgx(PO4)3/C with superior rate capability and long lifespan. J. Mater. Sci. 2020, 55, 13141–13156. [Google Scholar] [CrossRef]
- Song, W.; Liu, S. A sodium vanadium three-fluorophosphate cathode for rechargeable batteries synthesized by carbothermal reduction. Solid. State Sci. 2013, 15, 1–6. [Google Scholar] [CrossRef]
- Song, W.; Wu, Z.; Chen, J.; Lan, Q.; Zhu, Y.; Yang, Y.; Pan, C.; Hou, H.; Jing, M.; Ji, X. High-voltage NASICON Sodium Ion Batteries: Merits of Fluorine Insertion. Electrochim. Acta 2014, 146, 142–150. [Google Scholar] [CrossRef]
- Wei, P.; Chen, W.; Jing, Q.; Lee, M.-H.; Chen, Z. Effects of P2O7 clusters arrangement on second harmonic generation responses of pyrophosphates. J. Alloys Compd. 2020, 827, 153922. [Google Scholar] [CrossRef]
- Niu, Y.; Zhang, Y.; Xu, M. A review on pyrophosphate framework cathode materials for sodium-ion batteries. J. Mater. Chem. A 2019, 7, 15006–15025. [Google Scholar] [CrossRef]
- Uebou, Y.; Okada, S.; Yamaki, J.-I. Electrochemical insertion of lithium and sodium into (MoO2)2P2O7. J. Power Sources 2003, 115, 119–124. [Google Scholar] [CrossRef]
- Gabelica-Robert, M.; Goreaud, M.; Labbe, P.; Raveau, B. The pyrophosphate NaFeP2O7: A cage structure. J. Solid. State Chem. 1982, 45, 389–395. [Google Scholar] [CrossRef]
- Barpanda, P.; Lu, J.; Ye, T.; Kajiyama, M.; Chung, S.-C.; Yabuuchi, N.; Komaba, S.; Yamada, A. A layer-structured Na2CoP2O7 pyrophosphate cathode for sodium-ion batteries. RSC Adv. 2013, 3, 3857–3860. [Google Scholar] [CrossRef]
- Barpanda, P.; Ye, T.; Avdeev, M.; Chung, S.-C.; Yamada, A. A new polymorph of Na2MnP2O7 as a 3.6 V cathode material for sodium-ion batteries. J. Mater. Chem. A 2013, 1, 4194–4197. [Google Scholar] [CrossRef]
- Barpanda, P.; Liu, G.; Ling, C.D.; Tamaru, M.; Avdeev, M.; Chung, S.-C.; Yamada, Y.; Yamada, A. Na2FeP2O7: A Safe Cathode for Rechargeable Sodium-ion Batteries. Chem. Mater. 2013, 25, 3480–3487. [Google Scholar] [CrossRef]
- Kim, H.; Park, C.S.; Choi, J.W.; Jung, Y. Defect-Controlled Formation of Triclinic Na2CoP2O7 for 4 V Sodium-Ion Batteries. Angew. Chem. Int. Ed. 2016, 55, 6662–6666. [Google Scholar] [CrossRef] [PubMed]
- Ha, K.-H.; Woo, S.H.; Mok, D.; Choi, N.-S.; Park, Y.; Oh, S.M.; Kim, Y.; Kim, J.; Lee, J.; Nazar, L.F.; et al. Na4−αM2+α/2(P2O7)2 (2/3 ≤ α ≤ 7/8, M = Fe, Fe0.5Mn0.5, Mn): A Promising Sodium Ion Cathode for Na-ion Batteries. Adv. Energy Mater. 2013, 3, 770–776. [Google Scholar] [CrossRef]
- Erragh, F.; Boukhari, A.; Abraham, F.; Elouadi, B. Study of the Crystal Structures of Sodium Magnesium and Sodium Nickel Diphosphates. J. Solid. State Chem. 2000, 152, 323–331. [Google Scholar] [CrossRef]
- Liu, G.; Nishimura, S.-I.; Chung, S.C.; Fujii, K.; Yashima, M.; Yamada, A. Defect induced sodium disorder and ionic conduction mechanism in Na1.82Mg1.09P2O7. J. Mater. Chem. A 2014, 2, 18353–18359. [Google Scholar] [CrossRef]
- Kim, H.; Shakoor, R.A.; Park, C.; Lim, S.Y.; Kim, J.-S.; Jo, Y.N.; Cho, W.; Miyasaka, K.; Kahraman, R.; Jung, Y.; et al. Na2FeP2O7 as a Promising Iron-Based Pyrophosphate Cathode for Sodium Rechargeable Batteries: A Combined Experimental and Theoretical Study. Adv. Funct. Mater. 2013, 23, 1147–1155. [Google Scholar] [CrossRef]
- Liu, H.; Zhao, Y.; Zhang, H.; Lian, X.; Dong, Y.; Kuang, Q. Structural and electrochemical properties of Fe-doped Na2Mn3−xFex(P2O7)2 cathode material for sodium ion batteries. J. Power Sources 2017, 370, 114–121. [Google Scholar] [CrossRef]
- Kumar, S.; Singh, M.; Mondal, R.; Kumar, M.; Prakash, R.; Singh, P. Mo2P2O11: A Potential Cathode Material for Rechargeable Sodium-Ion Batteries. Energy Fuels 2023, 37, 1288–1296. [Google Scholar] [CrossRef]
- Pu, X.; Wang, H.; Yuan, T.; Cao, S.; Liu, S.; Xu, L.; Yang, H.; Ai, X.; Chen, Z.; Cao, Y. Na4Fe3(PO4)2P2O7/C nanospheres as low-cost, high-performance cathode material for sodium-ion batteries. Energy Stor. Mater. 2019, 22, 330–336. [Google Scholar] [CrossRef]
- Kundu, D.; Tripathi, R.; Popov, G.; Makahnouk, W.R.M.; Nazar, L.F. Synthesis, Structure, and Na-Ion Migration in Na4NiP2O7F2: A Prospective High Voltage Positive Electrode Material for the Na-Ion Battery. Chem. Mater. 2015, 27, 885–891. [Google Scholar] [CrossRef]
- Masese, T.; Orikasa, Y.; Tassel, C.; Kim, J.; Minato, T.; Arai, H.; Mori, T.; Yamamoto, K.; Kobayashi, Y.; Kageyama, H.; et al. Relationship between Phase Transition Involving Cationic Exchange and Charge-Discharge Rate in Li2FeSiO4. Chem. Mater. 2014, 26, 1380–1384. [Google Scholar] [CrossRef]
- Gao, S.; Zhao, J.; Zhao, Y.; Wu, Y.; Zhang, X.; Wang, L.; Liu, X.; Rui, Y.; Xu, J. Na2CoSiO4 as a novel positive electrode material for sodium-ion capacitors. Mater. Lett. 2015, 158, 300–303. [Google Scholar] [CrossRef]
- Treacher, J.C.; Wood, S.M.; Islam, M.S.; Kendrick, E. Na2CoSiO4 as a cathode material for sodium-ion batteries: Structure, electrochemistry and diffusion pathways. Phys. Chem. Chem. Phys. 2016, 18, 32744–32752. [Google Scholar] [CrossRef]
- Wang, J.; Hoteling, G.; Shepard, R.; Wahila, M.; Wang, F.; Smeu, M.; Liu, H. Reaction Mechanism of Na-Ion Deintercalation in Na2CoSiO4. J. Phys. Chem. C 2022, 126, 16983–16992. [Google Scholar] [CrossRef]
- Pedone, A.; Malavasi, G.; Menziani, M.C.; Cormack, A.N.; Segre, U. A New Self-Consistent Empirical Interatomic Potential Model for Oxides, Silicates, and Silica-Based Glasses. J. Phys. Chem. B 2006, 110, 11780–11795. [Google Scholar] [CrossRef]
- Wu, P.; Wu, S.Q.; Lv, X.; Zhao, X.; Ye, Z.; Lin, Z.; Wang, C.Z.; Ho, K.M. Fe–Si networks in Na2FeSiO4 cathode materials. Phys. Chem. Chem. Phys. 2016, 18, 23916–23922. [Google Scholar] [CrossRef]
- Bianchini, F.; Fjellvåg, H.; Vajeeston, P. First-principles study of the structural stability and electrochemical properties of Na2MSiO4 (M = Mn, Fe, Co and Ni) polymorphs. Phys. Chem. Chem. Phys. 2017, 19, 14462–14470. [Google Scholar] [CrossRef]
- Jin, T.; Li, H.; Zhu, K.; Wang, P.-F.; Liu, P.; Jiao, L. Polyanion-type cathode materials for sodium-ion batteries. Chem. Soc. Rev. 2020, 49, 2342–2377. [Google Scholar] [CrossRef]
- Kee, Y.; Dimov, N.; Staykov, A.; Okada, S. Investigation of metastable Na2FeSiO4 as a cathode material for Na-ion secondary battery. Mater. Chem. Phys. 2016, 171, 45–49. [Google Scholar] [CrossRef]
- Rangasamy, V.S.; Thayumanasundaram, S.; Locquet, J.-P. Solvothermal synthesis and electrochemical properties of Na2CoSiO4 and Na2CoSiO4/carbon nanotube cathode materials for sodium-ion batteries. Electrochim. Acta 2018, 276, 102–110. [Google Scholar] [CrossRef]
- Guan, W.H.; Lin, Q.Y.; Lan, Z.Y.; Pan, W.L.; Wei, X.; Sun, W.P.; Zheng, R.T.; Lu, Y.H.; Shu, J.; Pan, H.G.; et al. Approaching the theoretical capacity limit of Na2FeSiO4-based cathodes with fully reversible two-electron redox reaction for sodium-ion battery. Mater. Today Nano 2020, 12, 100098. [Google Scholar] [CrossRef]
- Law, M.; Ramar, V.; Balaya, P. Na2MnSiO4 as an attractive high capacity cathode material for sodium-ion battery. J. Power Sources 2017, 359, 277–284. [Google Scholar] [CrossRef]
- Schon, T.B.; McAllister, B.T.; Li, P.-F.; Seferos, D.S. The rise of organic electrode materials for energy storage. Chem. Soc. Rev. 2016, 45, 6345–6404. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.-G.; Yuan, S.; Ma, D.-L.; Huang, X.-L.; Meng, F.-L.; Zhang, X.-B. Tailored Aromatic Carbonyl Derivative Polyimides for High-Power and Long-Cycle Sodium-Organic Batteries. Adv. Energy Mater. 2014, 4, 1301651. [Google Scholar] [CrossRef]
- Wang, S.; Wang, L.; Zhu, Z.; Hu, Z.; Zhao, Q.; Chen, J. All Organic Sodium-Ion Batteries with Na4C8H2O6. Angew. Chem. 2014, 126, 6002–6006. [Google Scholar] [CrossRef]
- Kim, H.; Kwon, J.E.; Lee, B.; Hong, J.; Lee, M.; Park, S.Y.; Kang, K. High Energy Organic Cathode for Sodium Rechargeable Batteries. Chem. Mater. 2015, 27, 7258–7264. [Google Scholar] [CrossRef]
- Shen, Y.F.; Yuan, D.D.; Ai, X.P.; Yang, H.X.; Zhou, M. Poly(diphenylaminesulfonic acid sodium) as a cation-exchanging organic cathode for sodium batteries. Electrochem. Commun. 2014, 49, 5–8. [Google Scholar] [CrossRef]
- Wang, C.; Xu, Y.; Fang, Y.; Zhou, M.; Liang, L.; Singh, S.; Zhao, H.; Schober, A.; Lei, Y. Extended π-Conjugated System for Fast-Charge and -Discharge Sodium-Ion Batteries. J. Am. Chem. Soc. 2015, 137, 3124–3130. [Google Scholar] [CrossRef]
- Han, S.C.; Bae, E.G.; Lim, H.; Pyo, M. Non-crystalline oligopyrene as a cathode material with a high-voltage plateau for sodium ion batteries. J. Power Sources 2014, 254, 73–79. [Google Scholar] [CrossRef]
- Sakaushi, K.; Hosono, E.; Nickerl, G.; Gemming, T.; Zhou, H.; Kaskel, S.; Eckert, J. Aromatic porous-honeycomb electrodes for a sodium-organic energy storage device. Nat. Commun. 2013, 4, 1485. [Google Scholar] [CrossRef]
- Wang, D.-Y.; Liu, R.; Guo, W.; Li, G.; Fu, Y. Recent advances of organometallic complexes for rechargeable batteries. Coord. Chem. Rev. 2021, 429, 213650. [Google Scholar] [CrossRef]
- Zhao, R.; Liang, Z.; Zou, R.; Xu, Q. Metal-Organic Frameworks for Batteries. Joule 2018, 2, 2235–2259. [Google Scholar] [CrossRef]
- Li, X.; Yang, X.; Xue, H.; Pang, H.; Xu, Q. Metal–organic frameworks as a platform for clean energy applications. EnergyChem 2020, 2, 100027. [Google Scholar] [CrossRef]
- Du, M.; Li, Q.; Zhao, Y.; Liu, C.-S.; Pang, H. A review of electrochemical energy storage behaviors based on pristine metal–organic frameworks and their composites. Coord. Chem. Rev. 2020, 416, 213341. [Google Scholar] [CrossRef]
- Lu, Y.; Wang, L.; Cheng, J.; Goodenough, J.B. Prussian blue: A new framework of electrode materials for sodium batteries. Chem. Commun. 2012, 48, 6544–6546. [Google Scholar] [CrossRef] [PubMed]
- Yi, H.; Qin, R.; Ding, S.; Wang, Y.; Li, S.; Zhao, Q.; Pan, F. Structure and Properties of Prussian Blue Analogues in Energy Storage and Conversion Applications. Adv. Funct. Mater. 2020, 31, 2006970. [Google Scholar] [CrossRef]
- Luo, Y.; Peng, J.; Yin, S.; Xue, L.; Yan, Y. Acid-Assisted Ball Mill Synthesis of Carboxyl-Functional-Group-Modified Prussian Blue as Sodium-Ion Battery Cathode. Nanomaterials 2022, 12, 1290. [Google Scholar] [CrossRef]
- Xie, B.X.; Sun, B.Y.; Gao, T.Y.; Ma, Y.L.; Yin, G.P.; Zuo, P.J. Recent progress of Prussian blue analogues as cathode materials for nonaqueous sodium-ion batteries. Coord. Chem. Rev. 2022, 460, 214478. [Google Scholar] [CrossRef]
- Fu, H.; Liu, C.; Zhang, C.; Ma, W.; Wang, K.; Li, Z.; Lu, X.; Cao, G. Enhanced storage of sodium ions in Prussian blue cathode material through nickel doping. J. Mater. Chem. A 2017, 5, 9604–9610. [Google Scholar] [CrossRef]
- Qian, J.F.; Zhou, M.; Cao, Y.L.; Ai, X.P.; Yang, H.X. Nanosized Na4Fe(CN)6/C Composite as a Low-Cost and High-Rate Cathode Material for Sodium-Ion Batteries. Adv. Energy Mater. 2012, 2, 410–414. [Google Scholar] [CrossRef]
- Li, W.-J.; Chou, S.-L.; Wang, J.-Z.; Kang, Y.-M.; Wang, J.-L.; Liu, Y.; Gu, Q.-F.; Liu, H.-K.; Dou, S.-X. Facile Method To Synthesize Na-Enriched Na1+xFeFe(CN)6 Frameworks as Cathode with Superior Electrochemical Performance for Sodium-Ion Batteries. Chem. Mater. 2015, 27, 1997–2003. [Google Scholar] [CrossRef]
- Yang, D.; Liao, X.-Z.; Huang, B.; Shen, J.; He, Y.-S.; Ma, Z.-F. A Na4Fe(CN)6/NaCl solid solution cathode material with an enhanced electrochemical performance for sodium ion batteries. J. Mater. Chem. A 2013, 1, 13417–13421. [Google Scholar] [CrossRef]
- Jiao, S.; Tuo, J.; Xie, H.; Cai, Z.; Wang, S.; Zhu, J. The electrochemical performance of Cu3[Fe(CN)6]2 as a cathode material for sodium-ion batteries. Mater. Res. Bull. 2017, 86, 194–200. [Google Scholar] [CrossRef]
- Sun, H.; Sun, H.; Wang, W.; Jiao, H.; Jiao, S. Fe4[Fe(CN)6]3: A cathode material for sodium-ion batteries. RSC Adv. 2014, 4, 42991–42995. [Google Scholar] [CrossRef]
- Matsuda, T.; Takachi, M.; Moritomo, Y. A sodium manganese ferrocyanide thin film for Na-ion batteries. Chem. Commun. 2013, 49, 2750–2752. [Google Scholar] [CrossRef]
- Wang, L.; Lu, Y.; Liu, J.; Xu, M.; Cheng, J.; Zhang, D.; Goodenough, J.B. A Superior Low-Cost Cathode for a Na-Ion Battery. Angew. Chem. Int. Ed. 2013, 52, 1964–1967. [Google Scholar] [CrossRef]
- Chen, Z.-Y.; Fu, X.-Y.; Zhang, L.-L.; Yan, B.; Yang, X.-L. High-Performance Fe-Based Prussian Blue Cathode Material for Enhancing the Activity of Low-Spin Fe by Cu Doping. ACS Appl. Mater. Interfaces 2022, 14, 5506–5513. [Google Scholar] [CrossRef]
- Song, J.; Wang, L.; Lu, Y.; Liu, J.; Guo, B.; Xiao, P.; Lee, J.J.; Yang, X.Q.; Henkelman, G.; Goodenough, J.B. Removal of interstitial H2O in hexacyanometallates for a superior cathode of a sodium-ion battery. J. Am. Chem. Soc. 2015, 137, 2658–2664. [Google Scholar] [CrossRef]
- Hu, P.; Peng, W.; Wang, B.; Xiao, D.; Ahuja, U.; Réthoré, J.; Aifantis, K.E. Concentration-Gradient Prussian Blue Cathodes for Na-Ion Batteries. ACS Energy Lett. 2020, 5, 100–108. [Google Scholar] [CrossRef]
- Xu, Z.; Sun, Y.; Xie, J.; Nie, Y.; Xu, X.W.; Tu, J.; Zhang, J.; Qiu, L.C.; Zhu, T.J.; Zhao, X.B. Scalable Preparation of Mn/Ni Binary Prussian Blue as Sustainable Cathode for Harsh-Condition-Tolerant Sodium-Ion Batteries. ACS Sustain. Chem. Eng. 2022, 10, 13277–13287. [Google Scholar] [CrossRef]
- Nguyen, T.P.; Kim, I.T. Vanadium Ferrocyanides as a Highly Stable Cathode for Lithium-Ion Batteries. Molecules 2023, 28, 461. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.T.; He, Z.H.; Hou, J.F.; Kong, L.B. Sodium vanadium hexacyanoferrate as a high-rate capability and long-life cathode material for Na-ion batteries. J. Energy Storage 2022, 53, 105165. [Google Scholar] [CrossRef]
- Noor, N.; Parkin, I.P. Enhanced transparent-conducting fluorine-doped tin oxide films formed by Aerosol-Assisted Chemical Vapour Deposition. J. Mater. Chem. C 2013, 1, 984–996. [Google Scholar] [CrossRef]
- Wu, X.; Wu, C.; Wei, C.; Hu, L.; Qian, J.; Cao, Y.; Ai, X.; Wang, J.; Yang, H. Highly Crystallized Na2CoFe(CN)6 with Suppressed Lattice Defects as Superior Cathode Material for Sodium-Ion Batteries. ACS Appl. Mater. Interfaces 2016, 8, 5393–5399. [Google Scholar] [CrossRef]
- Takachi, M.; Matsuda, T.; Moritomo, Y. Cobalt Hexacyanoferrate as Cathode Material for Na+ Secondary Battery. Appl. Phys. Express 2013, 6, 025802. [Google Scholar] [CrossRef]
- Baster, D.; Kondracki, L.; Oveisi, E.; Trabesinger, S.; Girault, H.H. Prussian Blue Analogue-Sodium-Vanadium Hexacyanoferrate as a Cathode Material for Na-Ion Batteries. Acs Appl. Energy Mater. 2021, 4, 9758–9765. [Google Scholar] [CrossRef]
- Nguyen, T.P.; Kim, I.T. Iron-Vanadium Incorporated Ferrocyanides as Potential Cathode Materials for Application in Sodium-Ion Batteries. Micromachines 2023, 14, 521. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, S.; Chang, X.; Guo, H.; Li, Y.; Wang, M.; Li, W.; Jiao, L.; Wang, Y. MOF based on a longer linear ligand: Electrochemical performance, reaction kinetics, and use as a novel anode material for sodium-ion batteries. Chem. Commun. 2018, 54, 11793–11796. [Google Scholar] [CrossRef]
- Férey, G.; Millange, F.; Morcrette, M.; Serre, C.; Doublet, M.-L.; Grenèche, J.-M.; Tarascon, J.-M. Mixed-Valence Li/Fe-Based Metal–Organic Frameworks with Both Reversible Redox and Sorption Properties. Angew. Chem. Int. Ed. 2007, 46, 3259–3263. [Google Scholar] [CrossRef]
- Chen, T.; Liu, X.; Niu, L.; Gong, Y.; Li, C.; Xu, S.; Pan, L. Recent progress on metal–organic framework-derived materials for sodium-ion battery anodes. Inorg. Chem. Front. 2020, 7, 567–582. [Google Scholar] [CrossRef]
- Li, H.; Wang, T.; Wang, X.; Li, G.; Shen, J.; Chai, J. MOF-derived Al-doped Na2FePO4F/mesoporous carbon nanonetwork composites as high-performance cathode material for sodium-ion batteries. Electrochim. Acta 2021, 373, 137905. [Google Scholar] [CrossRef]
- Yu, T.-Y.; Sun, Y.-K. A fluorinated O3-type layered cathode for long-life sodium-ion batteries. J. Mater. Chem. A 2022, 10, 23639–23648. [Google Scholar] [CrossRef]
- Zhou, Q.; Wang, L.; Li, W.; Zhao, K.; Liu, M.; Wu, Q.; Yang, Y.; He, G.; Parkin, I.P.; Shearing, P.R.; et al. Sodium Superionic Conductors (NASICONs) as Cathode Materials for Sodium-Ion Batteries. Electrochem. Energy Rev. 2021, 4, 793–823. [Google Scholar] [CrossRef]
- Wang, Q.; Wu, X.; You, H.; Min, H.; Xu, X.; Hao, J.; Liu, X.; Yang, H. Template-directed Prussian blue nanocubes supported on Ni foam as the binder-free anode of lithium-ion batteries. Appl. Surf. Sci. 2022, 571, 151194. [Google Scholar] [CrossRef]
- Kim, H.; Sadan, M.K.; Kim, C.; Jo, J.; Seong, M.; Cho, K.-K.; Kim, K.-W.; Ahn, J.-H.; Ahn, H.-J. Enhanced reversible capacity of sulfurized polyacrylonitrile cathode for room-temperature Na/S batteries by electrochemical activation. Chem. Eng. J. 2021, 426, 130787. [Google Scholar] [CrossRef]
- Huang, Z.-X.; Zhang, X.-L.; Zhao, X.-X.; Zhao, Y.-Y.; Aravindan, V.; Liu, Y.-H.; Geng, H.; Wu, X.-L. Electrode/electrolyte additives for practical sodium-ion batteries: A mini review. Inorg. Chem. Front. 2023, 10, 37–48. [Google Scholar] [CrossRef]
- Hou, Y.; Jin, J.; Huo, C.; Liu, Y.; Deng, S.; Chen, J. New insights into the critical role of inactive element substitution in improving the rate performance of sodium oxide cathode material. Energy Stor. Mater. 2023, 56, 87–95. [Google Scholar] [CrossRef]
- Nanba, Y.; Iwao, T.; Boisse, B.M.D.; Zhao, W.; Hosono, E.; Asakura, D.; Niwa, H.; Kiuchi, H.; Miyawaki, J.; Harada, Y.; et al. Redox Potential Paradox in NaxMO2 for Sodium-Ion Battery Cathodes. Chem. Mater. 2016, 28, 1058–1065. [Google Scholar] [CrossRef]
This entry is adapted from the peer-reviewed paper 10.3390/ma16216869
