Another potential application for nanocarriers is as transdermal therapy for skin conditions such as psoriasis, ultraviolet damage, and aging. In an article, the nanospheres were prepared using the oil-in-water (o/w) technique. Cytotoxicity of the particles was assessed using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay (MTT) with L929 mouse fibroblasts, BALB/3T3 mouse embryo fibroblasts, and human HaCaT keratinocytes. The genotoxicity of the carriers was determined using the Ames test. The ability of the particles to penetrate the membrane of human osteosarcoma Saos 2 cells and accumulate in their cytoplasm was also evaluated. Additionally, penetration through human skin was investigated using samples obtained from a 42-year-old female donor after undergoing breast surgery. All tests had satisfactory results, indicating that the nanoparticles can be used for personalized treatment through topical administration [
58].
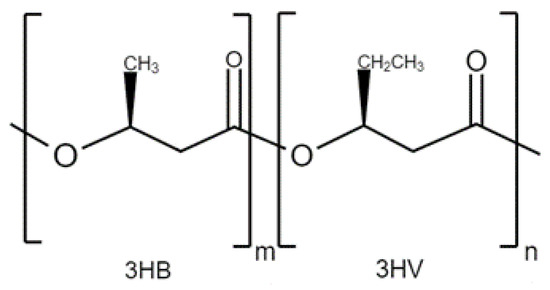
2.2. PHBV Composites for Tissue Engineering Applications
Tissue engineering is a discipline of biomedicine that aims to promote the regeneration of the structure and functionality of damaged tissues in the body by using biomaterials capable of guiding metabolic processes and cellular activity. In this field, PHAs have been widely used for the synthesis of three-dimensional cellular scaffolds with improved structural and functional properties for bone and cartilage regeneration [
60]. They can also be used as controlled-release systems of bioactive molecules and cellular signals to induce tissue regeneration. A specific example is the development of PLGA and PHBV nanoparticles incorporated into scaffolds to have a controlled release system of BMP-2 and BMP-7 morphogenetic proteins [
61].
Another method to create biodegradable PHBV scaffolds is using selective laser sintering (SLS). It is demonstrated that the unique microstructure formed by this technique has the potential for conducting in vitro and in vivo tests [
64]. In another publication, the degradation behavior of the scaffolds was investigated by incubating them in phosphate-buffered saline (PBS) for 6 weeks. SEM was used to characterize the microstructure of the scaffolds before and after incubation in PBS. The integrity of the structure was not affected and the molecular weight slightly decreased. The PHBV scaffolds fabricated with SLS generally showed adequate mechanical properties and good structural integrity after incubation [
65]. The same technique was used by another group, in this case, to fabricate a nanocomposite of CaP/PHBV. The in vitro studies revealed a high viability of SaOS-2 cells and normal morphology and phenotype after 3 and 7 days of cultures on all scaffolds. The release behavior of bovine serum albumin (BSA) in this nanostructure was also studied.
The PHBV biopolymer has also been used in neural-tissue regeneration, where it promotes cellular adhesion, proliferation, and differentiation. Its potential in treating various neural injuries and diseases has been demonstrated [
79]. With this aim, an electrospinning method was used to produce a chitosan-crosslinked nanofibrous biodegradable PHBV scaffold. The structure and cell culture assays using Schwann cells were microscopically, physically, and mechanically analyzed. The cells were able to grow fine on the created platform, indicating that this mixture of materials can be a promising candidate for applications in nerve conduits. The obtained results suggest that the generated material possesses suitable characteristics for further in vivo studies [
80].
There is another article that also discusses PHBV/PLA/collagen membranes for duroplasty after decompression in rats with SCI. The study focused on evaluating the material and the biological characteristics, subcutaneous implantation tests, and contusion SCI tests in rats to investigate the effects of the membranes on inflammasome activation and macrophage polarization. The results demonstrated that duroplasty with PHBV/PLA/Collagen membranes reduced glial scar formation and promoted axonal growth by inhibiting inflammasome activation and modulating macrophage polarization in acute SCI. Functional locomotor recovery improved 8 weeks after the injury [
82].
Various research groups are dedicated to exploring the most suitable materials and techniques for cartilage tissue engineering. One group of investigators used a conjugation of PHBV with type I collagen to produce a mechanically stable, biodegradable, and adhesive cell scaffold. The characterization of the scaffold was carried out using techniques such as SEM, Attenuated total reflection FTIR, atomic force microscopy, and electron spectroscopy for chemical analysis. Furthermore, the degradation and behavior of fibroblasts on nanofibrous scaffolds were studied. According to the article, electrospun PHBV/collagen composite nanofibrous scaffolds exhibited good mechanical properties, biocompatibility, and biodegradability. These scaffolds favored the growth and proliferation of fibroblast cells, and the cells demonstrated adequate adhesion and dispersion on the scaffolds. Results also revealed that the rate of degradation of the scaffolds could be controlled by adjusting the ratio of PHBV to collagen. This study suggested that electrospun PHBV/collagen composite nanofibrous scaffolds have potential applications in tissue engineering [
85]. In a publication in 2022, one group of investigators studied the influence of the addition of Bioglass into PHBV porous platforms. Cartilage progenitor cells (CPCs) were seeded into the control and the PHBV/10% Bioglass scaffolds. The CPC-constructs were exposed to a 6-week in vitro chondrogenic induction culture and then transplanted in vivo for another 6 weeks to see the difference between the CPC-PHBV and CPC-PHBV/10% Bioglass platforms. Compared to the control, the PHBV/ 10% Bioglass scaffold had better properties and results like hydrophilicity, a higher percentage of adherent cells, and significant production of cartilage-like tissues. Also, the polymerase chain reaction analysis showed that aggrecan, collagen II, and SOX-9 from the CPC-PHBV/10% Bioglass scaffolds were more expressed compared to the CPC-PHBV ones. All indicate that the addition of Bioglass to PHBV can improve the chondrogenic differentiation of CPCs [
86].
For wound healing, one group created a dressing made of a bilayer scaffold produced by electrospinning a hydrophilic PUL fibrous membrane (barrier layer) onto a wet electrospun hydrophobic PHBV fibrous mat (regenerative layer). The study optimized the production of PHBV using a bacterial strain called
Cupriavidus necator and characterized the resulting polymer using various techniques, including Proton nuclear magnetic resonance (
1H-NMR) and FTIR. The valerate molar percentage and the average molecular weight of the polymer were also determined using the
1H-NMR and SLS techniques, respectively. In vitro studies showed that the PUL membrane maintained L929 cell proliferation and prevented cells from migrating within the barrier phase, while the PHBV layer supported cell viability, proliferation, and migration, creating a regenerative 3D structure. The results showed that the new PUL/PHBV bilayer scaffold was a promising candidate for wound-healing applications [
90].
To fix abdominal wall defects, which can be caused by abdominal trauma or congenital rupture, silk fibroin and PHBV scaffolds were prepared. Characterization and cytotoxicity assays, in vitro tests to contemplate cell morphology and viability, and q-PCR to detect the gene expression of growth factor TGF-β1 and collagen I, were performed. In vivo studies were also achieved in Sprague Dawley rats. After 7 and 15 days of implanting the scaffold, an evaluation of their in vivo tissue regeneration capacity was performed. All the results pointed out that the hybrid nanofiber SF/PHBV scaffolds had a high efficiency and biocompatibility to repair abdominal wall defects [
93].
3. Conclusions
In conclusion, biopolymers of the PHA family are very promising in a wide range of applications due to their natural origin and their excellent properties, including biocompatibility and biodegradability. In the field of medicine, these biomaterials are particularly relevant as their degradation products do not produce toxic agents for the organism. This biocompatibility is essential for their use in biomedical applications such as tissue engineering, implants, sutures, and controlled drug-delivery systems.