Commercial applications of duckweeds include wastewater recycling and freshwater reclamation through removal of nitrogen- and phosphate-based fertilizer run-off and other waste-products, serving as feedstock for biofuels production (from starch or lipid), and production of food with superior nutritional quality (for humans and livestock), while requiring minimal space, light, and fertilizer.
- duckweed
- applications
1. Overview
Some duckweed plants (family Lemnaceae) are 100% edible, with much or all the plant (in rootless duckweeds) consisting of one or several fronds (small green, leaf-like organs). Fronds are largely responsible for nutrient uptake especially under ample nutrient supply. Only under low nutrient supply does the plant rely primarily on fine roots for taking up nutrients (except in rootless species). Lemnaceae possess the highest relative growth rates among plants [1], which may be related to the fact that aquatic floating plants reinvest their photosynthetic income predominantly into additional productive infrastructure (additional photosynthetic tissue), while having minimal need to invest in non-productive (non-photosynthetic) support structures (see [2]). Furthermore, duckweeds accumulate exceptionally large amounts of essential (= must be acquired from the diet) human micronutrients with antioxidative and anti-inflammatory effects (antioxidant vitamins A, C, E, carotenoids, and phenolics) [3][1][4][5][6]. Duckweed’s highly efficient uptake of ammonium (preferred over nitrate [7]) is likely related to the fact that ammonium is the most common form of nitrogenous waste produced by heterotrophic aquatic organisms. In addition, duckweeds receive ammonium from their endogenous N2-fixing Rhizobium symbionts and possess an unusual capacity for conversion of the (potentially toxic) ammonium to large amounts of vegetative storage protein. In addition to their exceptional potential to produce high-quality plant-based protein, duckweeds thus have a high fertilizer-use efficiency and capacity for phytoremediation [8], such as removal of fertilizer runoff from freshwater bodies and fertilizer retention in, e.g., rice paddies [9]. Finally, cultivation of duckweed could be further optimized using hydroponic vertical farms, where nutrients and water are recirculated, saving both resources, space, and energy to produce high-value products.
2. Commercial Applications of Duckweed
2.1.Human and Livestock Nutrition
Based on its nutritional composition, duckweed can provide high-quality nutrition for both humans and livestock. The propensity to accumulate large amounts of storage protein throughout the plant allows duckweed to produce between 20-40% of crude protein [4], which is equivalent to 20x the amount of protein produced by soybean per unit cultivated area [10] and much higher amounts of protein yet compared to those produced by cereals [11]. A key factor in duckweed’s superior protein production is protein accumulation in all of its tissues compared to only in the seeds of land plants. At the same time, duckweed-protein quality is as high as that of soybean or milk – with all essential amino acids that humans must obtain from their diet [12]. The main protein in duckweed is RuBisCO, a non-allergenic source of all nine essential amino acids for humans (and their livestock) with high digestibility, making it an ideal candidate for human food and as animal feed [13]. Duckweed also contains large amounts of the non-essential amino acid glutamic acid that has health benefits for humans and other animals [14]. However, like other plants (e.g., spinach or beet greens [15]), some duckweeds (e.g., genus Lemna) can contain high levels of anti-nutritive compounds such as oxalic acid [16]. This compound chelates calcium ions and other micronutrients, hindering their absorption in the intestine and compromising human health [17][18]. In contrast, duckweeds in the genus Wolffia contain minimal and thus safe amounts of anti-nutrients, which makes this genus suitable as a safe source of human food highly recommended for its superior nutritional characteristics regarding proteins, vitamins, and other micronutrients mentioned above. There are other potentially anti-nutritive compounds (nitrates, tannins, phytates, dioxins, and cyanogenic glycosides [19]) that require further investigation in duckweeds. Wolffia globosa (Mankai) has long been consumed as a high-protein source in Japan [20].
Sufficient intake of protein and other micronutrients (such as vitamins and antioxidants) is essential for optimal body function [50] and one of the current Global Challenges [21] facing human society and science. Currently, more than 15% of people worldwide cannot achieve the recommended daily protein intake and fall behind even further with respect to micronutrients (up to 25% deficit), leading to malnutrition [22][23]. The WHO “Global Strategy for Food Safety (2022–2030)” program (https://www.who.int/publications/i/item/9789240057685) aims to “ensure that all people, everywhere, consume safe and healthy food so as to reduce the burden of foodborne diseases” [24]. A resilient food system is thus needed that is fit for the future, with concomitant benefits for the health of individual people and societies as well as for the physical, natural environment including climate.
Plant-microbe interaction in duckweed also has multiple nutritional benefits, including minimizing loss of photosynthesis-associated micronutrients under elevated CO2 (see above; [25][26]. Furthermore, and in contrast to edible products from other plants, Mankai contains high levels of vitamin B12 – even after plants have been surface-sterilized [27]. The synthesis of vitamin B12 is carried out by bacteria and archaea absent in other plant-based food products [28]. Presence of vitamin B12 in Mankai is apparently due to as-yet undefined B12-producing bacteria that must be endophytic even in otherwise axenic Mankai cultures [27][12]. Because other plants are not a source of vitamin B12, this vitamin is currently obtained mainly from animal products (clams and beef liver are rich in vitamin B12; [29]). The current trend toward greater consumption of plant versus animal products [30] increases the risk of vitamin B12 deficiency and associated health problems [31]. To meet the need for B12 intake in diets low in animal products, algae were suggested as a source, but recent studies showed that vitamin B12 occurs in algae in a form (pseudo B12) not usable by humans and even capable of competing with the usable forms of B12 [32]. In contrast, Makai can be considered an excellent addition to meat-free diets. Moreover, the Mankai intake also positively modified human gut microbiota and increased, e.g, the diversity of genes (as assessed via 16S rRNA) related to vitamin B12 absorption [27]. Remarkably, a recent series of human clinical trials provided a comprehensive summary of these and additional health benefits of a diet including Mankai – with improvements in serum levels of vitamin B12, iron, and phenolics, gut microbiome composition, brain function, blood glucose control/insulin sensitivity, cardiovascular health, and weight management (e.g., decreased fat accumulation) [27][33][34][35][36][37][38][39][40]
2.2. Feedstock for Biofuels
Problems of current production systems for biofuel feedstocks include the food-fuel conflict and undesirable environmental impacts due to use of staple food crops with relatively high requirements for irrigation and fertilization. Production of maize, yucca, or sweet potato for biofuel increased the price of these basic foods and threatened food security [122]. In association with these crops’ high fertilizer requirement, excessive nutrient runoff caused environmental problems [41]. Therefore, it is urgent to identify new, sustainable feedstocks for biofuel production.
Due to its high starch content (see, e.g., [42]), duckweed is suitable for bioethanol production via starch hydrolysis and subsequent alcoholic fermentation. The yield of bioethanol from duckweed has been estimated to potentially reach 6,420 L ha-1, which is about 50% above that obtained for ethanol based on corn [43][41][44] and production could continue throughout the year. The ability of Lemna to accumulate starch without needing large amounts of light [42] and to respond to elevated CO2 with increased starch accumulation would support high biofuel-production yields [45]. Biofuel production could also be paired with wastewater treatment as previously shown for systems that combine wastewater recycling with livestock feed production [10]. Moreover, effective sequestration of atmospheric CO2 to starch by duckweed would contribute to CO2 mitigation and climate control. Duckweed-based systems could thus serve as a model for future regenerative life support systems providing both food and fuel on Earth as well as for human space exploration [46]
Starch accumulation by Lemna can be enhanced through restriction of nutrient supply with minimal impact on growth rate [41][47][48]. Restriction of sulfur supply showed the greatest promise with respect to stimulation of starch accumulation with negligible growth impact [47], while nitrogen limitation increased starch yield at the expense of growth and overall biomass production [47]. Recent progress has also been reported in engineering duckweed for enhanced accumulation of lipids [49][127] that could be used to produce biodiesel.
3. Agricultural Technology: Hydroponics and Vertical Farming for Improved Yield
Documented advantages of hydroponic crops include highly efficient use of water and nutrients [50] by crops such as tomato, cucumber, and other vegetables [50][51]. Recirculating hydroponic systems further increase resource-use efficiency [52]. Duckweed has a high potential for use in closed hydroponic systems consisting of multiple vertical layers (Figure 1), with controlled temperature, CO2, light intensity, quality, and photoperiod, as well as nutrient composition [53]. Vertical design would further optimize use of space [51][54] when growing a diminutive, 100% edible plant like duckweed. Control of environmental factors allows optimization of biomass quality with respect to micronutrients (vitamins, antioxidants) and proteins for use as food [55] or either starch or lipid content for use as biofuel feedstock. Recirculation of nutrients and water prevents nutrient release into the environment and resulting land and water pollution [56] as well as minimizing water loss [52] by reducing evaporation (proximity between different vertical levels could act as "semi-closed lids”). Duckweed, furthermore, thrives on a film of water [57]. Vertical, hydroponics-based farming is also suitable for continuous production throughout the year.
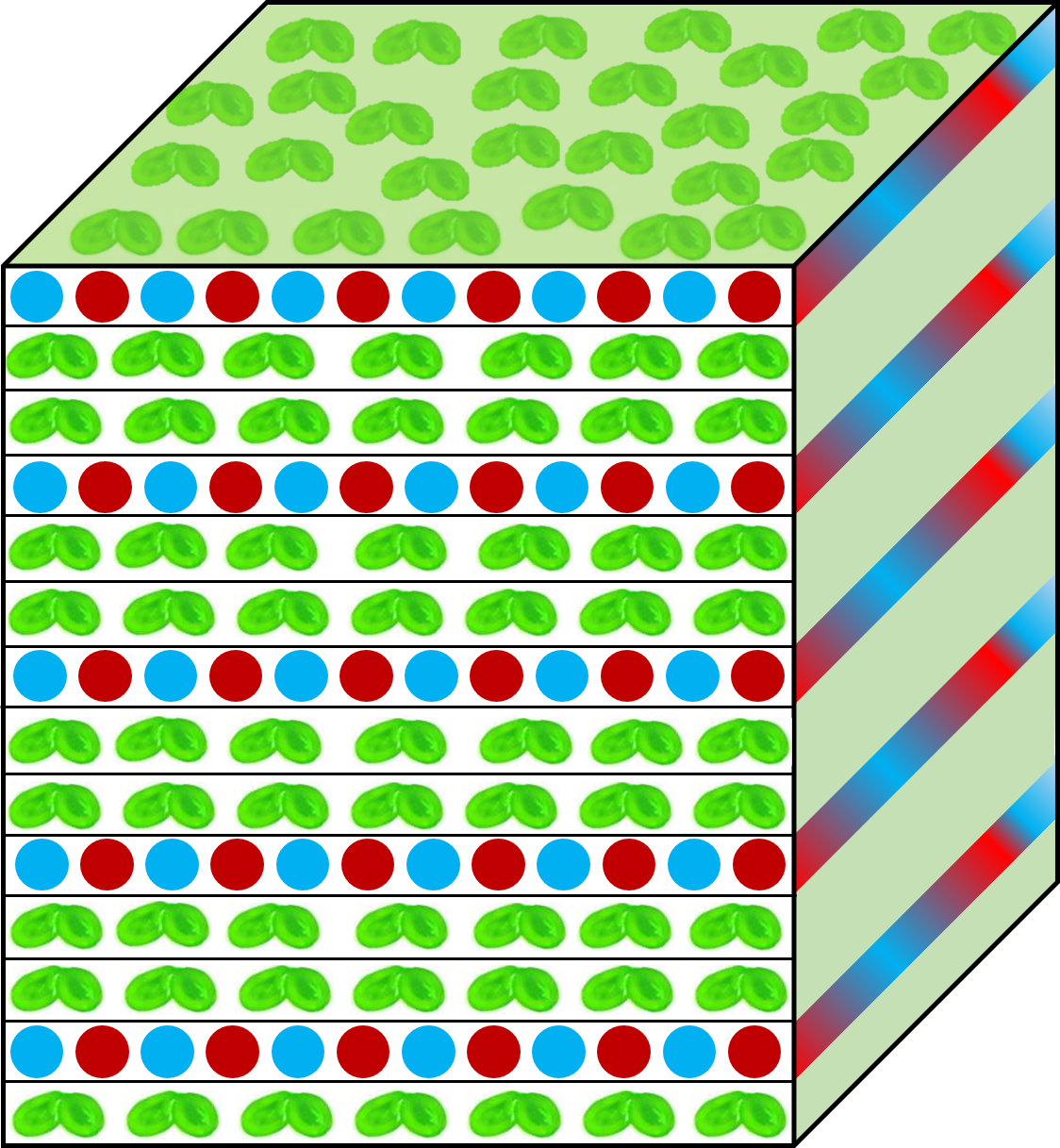
Figure 1: Schematic representation of a multi-level, closed vertical production system for duckweed with interspersed blue and red light emitting diodes supplying the low level of light that can saturate plant growth and food or biofuel feedstock production.
Figure 1 depicts a system where layers of duckweed are interspersed with layers of light-emitting diodes (LEDs), resulting in illumination of duckweed layers from both below and above, a design first suggested by Gale et al. [58]. Figure 1 features a mix of blue and red LEDs that, due to minimal self-shading within a single layer of fronds, may support maximal productivity with minimal energy input. We suggest using both red and blue wavelengths to support not only photosynthesis but also plant morphogenesis [59] and blue-light-triggered phenolics production [60].
4. Conclusions
Due to its high lipid- and starch-storage capacity, duckweed is a promising crop for production of biofuels, and its exceptionally high protein and human-micronutrient content makes it a food with high nutritional value. Duckweed’s outstanding ability to absorb ammonium (and turn it into protein) can furthermore serve in wastewater treatment.
All these characteristics make duckweed unusually productive in small culture areas or volumes. Combination of its diminutive size with exceptionally high relative growth rates, harvest index (no inedible parts), and nutrient density make this plant ideal for vertical growth in multiple stacked layers (Figure 1). Whereas multi-layer cultivation may also be attractive with terrestrial plants, it offers clear advantages in duckweed with highly efficient use of space, energy, nutrient supply, and gas exchange with the atmosphere.
This entry is adapted from the peer-reviewed paper 10.3390/plants12173090
References
- K. Sowjanya Sree; Sailendharan Sudakaran; Klaus-J. Appenroth; How fast can angiosperms grow? Species and clonal diversity of growth rates in the genus Wolffia (Lemnaceae). Acta Physiol. Plant. 2015, 37, 31-39, .
- Barbara Demmig-Adams; Marina López-Pozo; Stephanie K. Polutchko; Paul Fourounjian; Jared J. Stewart; Madeleine C. Zenir; William W. Adams; Growth and Nutritional Quality of Lemnaceae Viewed Comparatively in an Ecological and Evolutionary Context. Plants 2022, 11, 145, .
- Klaus-J. Appenroth; K. Sowjanya Sree; Volker Böhm; Simon Hammann; Walter Vetter; Matthias Leiterer; Gerhard Jahreis; Nutritional value of duckweeds (Lemnaceae) as human food. Food Chem. 2017, 217, 266-273, .
- Stephanie K. Polutchko; Jared J. Stewart; Maureen McNamara; Naiara Doherty Garcia; Marina López-Pozo; William W. Adams; Barbara Demmig-Adams; Lemna as a Sustainable, Highly Nutritious Crop: Nutrient Production in Different Light Environments. Nutraceuticals 2022, 2, 350-364, .
- Stephanie K. Polutchko; William W. Adams; Christine M. Escobar; Barbara Demmig-Adams; Conquering Space with Crops That Produce Ample Oxygen and Antioxidants. Oxyg. 2022, 2, 211-226, .
- K. Sowjanya Sree; Hans-Martin Dahse; Jima N. Chandran; Bernd Schneider; Gerhard Jahreis; Klaus J. Appenroth; Duckweed for Human Nutrition: No Cytotoxic and No Anti-Proliferative Effects on Human Cell Lines. Plant Foods Hum. Nutr. 2019, 74, 223-224, .
- Zhou, Y.; Kishchenko, O.; Stepanenko, A.; Chen, G.; Wang,W.; Zhou, J.; Pan, C.; Borisjuk, N.; The dynamics of NO3 and NH4 + uptake in duckweed are coordinated with the expression of major nitrogen assimilation genes. . Plants 2022, 11, 11, .
- GahYoung Baek; Maham Saeed; Hyung-Kyoon Choi; Duckweeds: their utilization, metabolites and cultivation. Appl. Biol. Chem. 2021, 64, 73, .
- Yuan Wang; Xiaodong Chen; Bin Guo; Chen Liu; Junli Liu; Gaoyang Qiu; Qinglin Fu; Hua Li; Alleviation of aqueous nitrogen loss from paddy fields by growth and decomposition of duckweed (Lemna minor L.) after fertilization. Chemosphere 2023, 311, 137073, .
- Rodrigo A. Mohedano; Rejane H.R. Costa; Flávia A. Tavares; Paulo Belli Filho; High nutrient removal rate from swine wastes and protein biomass production by full-scale duckweed ponds. Bioresour. Technol. 2012, 112, 98-104, .
- Myrthe F.A. de Beukelaar; Gertrude G. Zeinstra; Jurriaan J. Mes; Arnout R.H. Fischer; Duckweed as human food. The influence of meal context and information on duckweed acceptability of Dutch consumers. Food Qual. Preference 2019, 71, 76-86, .
- Kaplan, A.; Zelicha, H.; Tsaban, G.; Meir, A.Y.; Rinott, E.; Kovsan, J.; Novack, L.; Thiery, J.; Ceglarek, U.; Burkhardt, R.; et al. Protein bioavailability of Wolffia globosa duckweed, a novel aquatic plant—A randomized controlled trial. Clin. Nutr. 2019, 38, 2576–2582, .
- Rina Chakrabarti; William D. Clark; Jai Gopal Sharma; Ravi Kumar Goswami; Avanish Kumar Shrivastav; Douglas R. Tocher; Mass Production of Lemna minor and Its Amino Acid and Fatty Acid Profiles. Front. Chem. 2018, 6, 479, .
- Nazariyah Yahaya; Nabila Huda Hamdan; Atiqah Ruqayyah Zabidi; Ammar Mirza Mohamad; Mohammad Luqman Hakim Suhaimi; Muhammad Azhan Azfar Md Johari; Hanis Nadia Yahya; Hafiza Yahya; Duckweed as a future food: Evidence from metabolite profile, nutritional and microbial analyses. Futur. Foods 2022, 5, 100128, .
- S C Noonan; G P Savage; Oxalate content of foods and its effect on humans.. Asia Pac. J. Clin. Nutr. 1999, 8, 64-74, .
- Zhubin Hu; Yang Fang; Zhuolin Yi; Xueping Tian; Jinmeng Li; Yanling Jin; Kaize He; Penghui Liu; Anping Du; Yuhong Huang; et al. Determining the nutritional value and antioxidant capacity of duckweed (Wolffia arrhiza) under artificial conditions. LWT 2021, 153, 112477, .
- Vinay Kumar; Mohammad Irfan; Asis Datta; Manipulation of oxalate metabolism in plants for improving food quality and productivity. Phytochem. 2018, 158, 103-109, .
- Yang, J.; Fu, M.; Ji, C.; Huang, Y.; Wu, Y.; Maize oxalyl-coa decarboxylase1 degrades oxalate and affects the seed metabolome and nutritional quality.. Plant Cell 2018, 30, 2447-2462, .
- Hemmige Natesh N; Asiedu Sk; Abbey L; An overview of nutritional and anti nutritional factors in green leafy vegetables. Hortic. Int. J. 2017, 1, 58-65, .
- Klaus-J. Appenroth; K. Sowjanya Sree; Manuela Bog; Josef Ecker; Claudine Seeliger; Volker Böhm; Stefan Lorkowski; Katrin Sommer; Walter Vetter; Karla Tolzin-Banasch; et al. Nutritional Value of the Duckweed Species of the Genus Wolffia (Lemnaceae) as Human Food. Front. Chem. 2018, 6, 483, .
- Jhuma Sadhukhan; Tom I. J. Dugmore; Avtar Matharu; Elias Martinez-Hernandez; Jorge Aburto; Pattanathu K. S. M. Rahman; Jim Lynch; Perspectives on “Game Changer” Global Challenges for Sustainable 21st Century: Plant-Based Diet, Unavoidable Food Waste Biorefining, and Circular Economy. Sustain. 2020, 12, 1976, .
- Coughlan, N.E.; Walsh, É.; Bolger, P.; Burnell, G.; O’Leary, N.; O’Mahoney, M.; Paolacci, S.; Wall, D.; Jansen, M.A.K.; Duckweed bioreactors: Challenges and opportunities for large-scale indoor cultivation of Lemnaceae.. J. Clean. Prod. 2022, 336, 130285, .
- Giorgos Markou; Liang Wang; Jianfeng Ye; Adrian Unc; Using agro-industrial wastes for the cultivation of microalgae and duckweeds: Contamination risks and biomass safety concerns. Biotechnol. Adv. 2018, 36, 1238-1254, .
- World Health Organization.. WHO Global Strategy for Food Safety 2022–2030—Towards Stronger Food Safety Systems and Global Cooperation; World Health Organization, Eds.; WHO: Geneva, Switzerland, 2022; pp. 1-86.
- Madeleine C. Zenir; Marina López-Pozo; Stephanie K. Polutchko; Jared J. Stewart; William W. Adams; Adam Escobar; Barbara Demmig-Adams; Productivity and Nutrient Quality of Lemna minor as Affected by Microbiome, CO2 Level, and Nutrient Supply. Stress. 2022, 3, 69-85, .
- Effects of growth environment on Lemna minor . E Sch. Community Encycl. Retrieved 2023-9-21
- Ilan Sela; Anat Yaskolka Meir; Alexander Brandis; Rosa Krajmalnik-Brown; Lydia Zeibich; Debbie Chang; Blake Dirks; Gal Tsaban; Alon Kaplan; Ehud Rinott; et al. Wolffia globosa–Mankai Plant-Based Protein Contains Bioactive Vitamin B12 and Is Well Absorbed in Humans. Nutr. 2020, 12, 3067, .
- Green, R.; Miller, J.W.. Vitamin B12 deficiency. In Vitamins and Hormones; Litwack, G., Eds.; Academic Press: Cambridge, MA, USA, 2022; pp. 405-439.
- U.S. Department of Health & Human Services. National Institutes of Health. Division of Program Coordination, Planning, and S.I. Vitamin B12 Fact Sheet for Health Professionals. 2023. Available online: https://ods.od.nih.gov/factsheets/VitaminB12- HealthProfessional/ (accessed on 24 August 2023).
- Yoko Yokoyama; Susan M Levin; Neal D Barnard; Association between plant-based diets and plasma lipids: a systematic review and meta-analysis. Nutr. Rev. 2017, 75, 683-698, .
- Nithya Sukumar; Ponnusamy Saravanan; Investigating vitamin B12 deficiency. BMJ 2019, 365, l1865, .
- Watanabe, F.; Takenaka, S.; Kittaka-Katsura, H.; Ebara, S.; Miyamoto, E.; Characterization and bioavailability of vitamin B12- compounds from edible algae. J. Nutr. Sci. Vitaminol 2002, 48, 325-331, .
- Meir, A.Y.; Rinott, E.; Tsaban, G.; Zelicha, H.; Kaplan, A.; Rosen, P.; Shelef, I.; Youngster, I.; Shalev, A.; Blüher, M.; et al. Effect of green-Mediterranean diet on intrahepatic fat: The DIRECT plus randomised controlled trial. Gut 2021, 70, 2085–2095., .
- Meir, A.Y.; Tsaban, G.; Zelicha, H.; Rinott, E.; Kaplan, A.; Youngster, I.; Rudich, A.; Shelef, I.; Tirosh, A.; Brikner, D.; et al. Green-Mediterranean diet, supplemented with mankai duckweed, preserves iron-homeostasis in humans and is efficient in reversal of anemia in rats.. J. Nutr. 2019, 149, 1004-1011, .
- Ehud Rinott; Anat Yaskolka Meir; Gal Tsaban; Hila Zelicha; Alon Kaplan; Dan Knights; Kieran Tuohy; Matthias Uwe Scholz; Omry Koren; Meir J. Stampfer; et al. The effects of the Green-Mediterranean diet on cardiometabolic health are linked to gut microbiome modifications: a randomized controlled trial. Genome Med. 2022, 14, 29, .
- Gal Tsaban; Anat Yaskolka Meir; Ehud Rinott; Hila Zelicha; Alon Kaplan; Aryeh Shalev; Amos Katz; Assaf Rudich; Amir Tirosh; Ilan Shelef; et al. The effect of green Mediterranean diet on cardiometabolic risk; a randomised controlled trial. Hear. 2020, 107, 1054-1061, .
- Zelicha, H.; Kaplan, A.; Meir, A.Y.; Tsaban, G.; Rinott, E.; Shelef, I.; Tirosh, A.; Brikner, D.; Pupkin, E.; Qi, L.; et al. The effect of Wolffia globosa mankai, a green aquatic plant, on postprandial glycemic response: A randomized crossover controlled trial. Diabetes Care 2019, 42, 1162–1169, .
- Kaplan, A.; Zelicha, H.; Yaskolka Meir, A.; Rinott, E.; Tsaban, G.; Levakov, G.; Prager, O.; Salti, M.; Yovell, Y.; Ofer, J.; et al. The effect of a high-polyphenol Mediterranean diet (Green-MED) combined with physical activity on age-related brain atrophy: The Dietary Intervention Randomized Controlled Trial Polyphenols Unprocessed Study (DIRECT PLUS).. am. J. Clin. Nutr. 2022, 115, 1270-1281, .
- Anat Yaskolka Meir; Kieran Tuohy; Martin von Bergen; Rosa Krajmalnik-Brown; Uwe Heinig; Hila Zelicha; Gal Tsaban; Ehud Rinott; Alon Kaplan; Asaph Aharoni; et al. The Metabolomic-Gut-Clinical Axis of Mankai Plant-Derived Dietary Polyphenols. Nutr. 2021, 13, 1866, .
- Rinott, E.; Youngster, I.; Meir, A.Y.; Tsaban, G.; Zelicha, H.; Kaplan, A.; Knights, D.; Tuohy, K.; Fava, F.; Scholz, M.U.; et al. Effects of diet-modulated autologous fecal microbiota transplantation on weight gain.. Gastroenterology 2021, 160, 158-173, .
- W. Cui; J. J. Cheng; Growing duckweed for biofuel production: a review. Plant Biol. 2014, 17, 16-23, .
- Jared J. Stewart; William W. Adams; Christine M. Escobar; Marina López-Pozo; Barbara Demmig-Adams; Growth and Essential Carotenoid Micronutrients in Lemna gibba as a Function of Growth Light Intensity. Front. Plant Sci. 2020, 11, 480, .
- Jay J. Cheng; Anne-M. Stomp; Growing Duckweed to Recover Nutrients from Wastewaters and for Production of Fuel Ethanol and Animal Feed. CLEAN – Soil, Air, Water 2009, 37, 17-26, .
- Ya-Liang Xu; Yang Fang; Qi Li; Gui-Li Yang; Ling Guo; Guo-Ke Chen; Li Tan; Kai-Ze He; Yan-Ling Jin; Hai Zhao; et al. Turion, an innovative duckweed-based starch production system for economical biofuel manufacture. Ind. Crop. Prod. 2018, 124, 108-114, .
- Rodrigo A. Mohedano; Rejane H. R. Costa; Paulo Belli Filho; Effects of CO2 concentration on nutrient uptake and starch accumulation by duckweed used for wastewater treatment and bioethanol production. Rev. Latinoam. Biotecnol. Ambient. Algal 2016, 7, 30-41, .
- Polutchko, S.K.; Adams, W.W., III; Escobar, C.M.; Demmig-Adams, B.; Conquering space with crops that produce ample oxygen and antioxidants.. Oxygen 2022, 2, 211-226, .
- Sun, Z.; Guo,W.; Zhao, X.; Chen, Y.; Yang, J.; Xu, S.; Hou, H.; Sulfur limitation boosts more starch accumulation than nitrogen or phosphorus limitation in duckweed (Spirodela polyrhiza). Ind. Crops Prod. 2022, 185, 115098, .
- Li, J.M.; Du, A.P.; Liu, P.H.; Tian, X.P.; Jin, Y.L.; Yi, Z.L.; He, K.Z.; Fang, J.; Zhao, H.; High starch accumulation mechanism and phosphorus utilization efficiency of duckweed (Landoltia punctata) under phosphate starvation. Ind. Crops Prod. 2021, 167, 113529, .
- Liang, Y.; Yu, X.H.; Anaokar, S.; Shi, H.; Dahl, W.B.; Cai, Y.; Luo, G.; Chai, J.; Cai, Y.; Mollá-Morales, A.; et al. Engineering triacylglycerol accumulation in duckweed (Lemna japonica).. Plant Biotechnol. J. 2023, 21, 317-330, .
- Nisha Sharma; Somen Acharya; Kaushal Kumar; Narendra Singh; O.P. Chaurasia; Hydroponics as an advanced technique for vegetable production: An overview. J. Soil Water Conserv. 2018, 17, 364, .
- Finn Petersen; Johannes Demann; Jannis von Salzen; Hans-Werner Olfs; Heiner Westendarp; Petra Wolf; Klaus-Jürgen Appenroth; Andreas Ulbrich; Re-circulating indoor vertical farm: Technicalities of an automated duckweed biomass production system and protein feed product quality evaluation. J. Clean. Prod. 2022, 380, 134894, .
- Rodolfo De la Rosa-Rodríguez; Alfredo Lara-Herrera; Libia Iris Trejo-Téllez; Luz Evelia Padilla-Bernal; Luis Octavio Solis-Sánchez; José Manuel Ortiz-Rodríguez; Water and fertilizers use efficiency in two hydroponic systems for tomato production. Hortic. Bras. 2020, 38, 47-52, .
- Kurt Benke; Bruce Tomkins; Future food-production systems: vertical farming and controlled-environment agriculture. Sustain. Sci. Pr. Policy 2017, 13, 13-26, .
- Kozai, T.; Niu, G.; Takagaki, M.. Plant Factory: An Indoor Vertical Farming System for Efficient Quality Food Production; Kozai, T.; Niu, G.; Takagaki, M., Eds.; Academic Press:: Cambridge, MA, USA, 2015; pp. 1.
- AlShrouf, A; Hydroponics, aeroponic and aquaponic as compared with conventional farming. Am. Sci. Res. J. Eng. Technol. Sci. 2017, 27, 247-255, .
- Volkmar Keuter; Sebastian Deck; Heidi Giesenkamp; Denise Gonglach; Victor Takazi Katayama; Sica Liesegang; Finn Petersen; Sandra Schwindenhammer; Heidrun Steinmetz; Andreas Ulbrich; et al. Significance and Vision of Nutrient Recovery for Sustainable City Food Systems in Germany by 2050. Sustain. 2021, 13, 10772, .
- Escobar, C.M.; Escobar, A.C.; Power, G.J.; Nabity, J.A.; uG-LilyPondTM: Preliminary design of a floating plant pond for microgravity. Int. Conf. Environ. Syst. 2020, 1, 1-12, .
- J. Gale; D.T. Smernoff; B.A. Macler; R.D. MacElroy; Carbon balance and productivity of Lemna gibba, a candidate plant for CELSS. Adv. Space Res. 1989, 9, 43-52, .
- Cunying Fan; Abinaya Manivannan; Hao Wei; Light Quality-Mediated Influence of Morphogenesis in Micropropagated Horticultural Crops: A Comprehensive Overview. BioMed Res. Int. 2022, 2022, 4615079, .
- Mirai Endo; Naoya Fukuda; Hideo Yoshida; Naomichi Fujiuchi; Ryoichi Yano; Miyako Kusano; Effects of light quality, photoperiod, CO2 concentration, and air temperature on chlorogenic acid and rutin accumulation in young lettuce plants. Plant Physiol. Biochem. 2022, 186, 290-298, .
