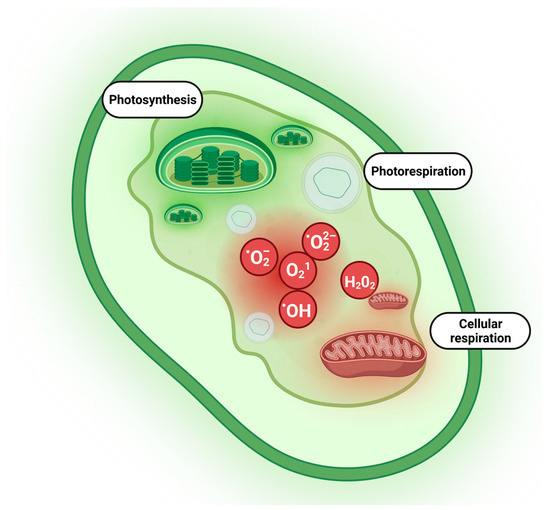
Reactive oxygen species (ROS) are excited or partially reduced forms of atmospheric oxygen, which are continuously produced during aerobic metabolism like many physiochemical processes operating throughout seed life. Previously, it was believed that ROS are merely cytotoxic molecules, however, now it has been established that they perform numerous beneficial functions in plants including many critical roles in seed physiology. ROS facilitate seed germination via cell wall loosening, endosperm weakening, signaling, and decreasing abscisic acid (ABA) levels. Most of the existing knowledge about ROS homeostasis and functions is based on the seeds of common plants or model ones.
- oxidative
- ROS scavenging
- salinity
- tolerance
1. Introduction
2. Halophytes: Importance, Classification, and Salt Tolerance Mechanisms
- (1)
-
Eu-halophytes: Growth of eu-halophytes is stimulated even in moderate salinities (such as Salicornia europaea and Suaeda maritima).
- (2)
-
Facultative halophytes: Growth of these halophytes is slightly stimulated at low salinity (such as Plantago maritima and Aster tripolium).
- (3)
-
Non-halophytes with low salinity tolerance: These plants are not halophytes and often include salt-tolerant crops and orchards. This group includes a wide range of economic plants such as barley (Hordeum vulgare), sorghum (Sorghum bicolor), cotton (Gossypium spp.), and pistachios (Pistacia vera).
- (4)
-
Halophobic: Plants that are sensitive to salinity and even at low salinity levels there is a significant reduction in their growth and yield, such as saffron (Crocus sativus), common bean (Phaseolus vulgaris), and most vegetables.
-
Recretohalophytes: Include halophytes that excrete salts on the outer surface (Exo-recretohalophytes) or the inside (Endo-recretohalophytes) of plant tissue.
-
Euhalophytes: Or true halophytes with succulent leaves or stems.
-
Pseudo-halophytes: Unreal halophytes that store salts in the parenchymal organs of the root.
-
Some halophytes have evolved salt glands or bladders that actively excrete sodium ions from their tissues (excretion of sodium ions.) By removing these ions from their cells, they effectively regulate salt concentration and protect themselves against saline stress [24].
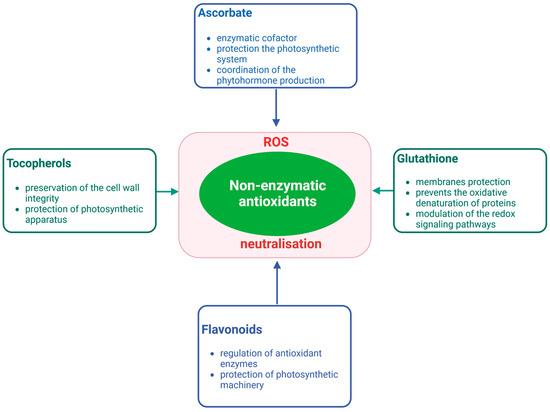
3. ROS and Antioxidants in Halophytes and Their Role in Salinity Tolerance
This entry is adapted from the peer-reviewed paper 10.3390/plants12173023
References
- Mittler, R. ROS are good. Trends Plant Sci. 2017, 22, 11–19.
- Shabala, S. Signalling by potassium: Another second messenger to add to the list? J. Exp. Bot. 2017, 68, 4003–4007.
- Pirasteh-Anosheh, H.; Ranjbar, G.; Akram, N.A.; Ghafar, M.A.; Panico, A. Forage potential of several halophytic species grown on saline soil in arid environments. Environ. Res. 2023, 219, 114954.
- Ozgur, R.; Uzilday, B.; Sekmen, A.H.; Turkan, I. Reactive oxygen species regulation and antioxidant defence in halophytes. Funct. Plant Biol. 2013, 40, 832–847.
- Bose, J.; Rodrigo-Moreno, A.; Shabala, S. ROS homeostasis in halophytes in the context of salinity stress tolerance. J. Exp. Bot. 2014, 65, 241–1257.
- Saed-Moucheshi, A.; Pakniyat, H.; Pirasteh-Anosheh, H.; Azooz, M.M. Role of ROS as signaling molecules in plants. In Oxidative Damage to Plants: Antioxidant Networks and Signaling; Ahmad, P., Ed.; Elsevier: Amsterdam, The Netherlands, 2014; pp. 585–620.
- Öztürk, M.; Altay, V.; Güvensen, A. Sustainable Use of Halophytic Taxa as Food and Fodder: An Important Genetic Resource in Southwest Asia. In Ecophysiology, Abiotic Stress Responses and Utilization of Halophytes; Hasanuzzaman, M., Nahar, K., Öztürk, M., Eds.; Springer: Singapore, 2019.
- Guvensen, A.; Gork, G.; Ozturk, M. An overview of the halophytes in Turkey. In Sabkha Ecosystems; Khan, M.A., Böer, B., Kust, G.S., Barth, H.J., Eds.; Tasks for Vegetation Science; Springer: Dordrecht, The Netherlands, 2006; Volume 42.
- Pirasteh-Anosheh, H.; Parvizi, H.; Parnian, A.; Esfahan, E.Z.; Ranjbar, G.; Bhardwaj, A.K. Relationship between soil salinity and alkalinity with Alhagi camelorum growth in hypersaline and hyperarid environments. J. Arid Environ. 2022, 206, 104830.
- Hnilickova, H.; Kraus, K.; Vachova, P.; Hnilicka, F. Salinity stress affects photosynthesis, malondialdehyde formation, and proline content in Portulaca oleracea L. Plants 2021, 10, 845.
- Shah, W.H.; Saleem, S.; Mushtaq, N.U.; Rasool, A.; Tahir, I.; Rehman, R.U. C4 and CAM Plants with Better Resilience to Environmental Stresses. In Photosynthesis and Respiratory Cycles during Environmental Stress Response in Plants; Apple Academic Press: Palm Bay, FL, USA, 2022; pp. 163–191.
- Pirasteh-Anosheh, H.; Saed-Moucheshi, A.; Pakniyat, H.; Pessarakli, M. Stomatal responses to drought stress. In Water Stress and Crop Plants: A Sustainable Approach; Ahmad, P., Ed.; John Wiley & Sons: London, UK, 2016; pp. 24–40.
- Pehlivan, F.E. Free radicals and antioxidant system in seed biology. In Advances in Seed Biology; InTech: London, UK, 2017; pp. 167–175.
- Polo-Ávila, A.; Infante-Izquierdo, M.D.; Sánchez-Gullón, E.; Castillo, J.M.; Muñoz-Rodríguez, A.F. Population Dynamic of the Annual Halophyte Salicornia ramosissima in Salt Pans: Towards a Sustainable Exploitation of Its Wild Populations. Plants 2022, 11, 1676.
- Hedayati-Firoozabadi, A.; Kazemeini, S.A.; Pirasteh-Anosheh, H.; Ghadiri, H.; Pessarakli, M. Forage yield and quality as affected by salt stress in different ratios of Sorghum bicolor-Bassia indica intercropping. J. Plant Nutr. 2020, 43, 2579–2589.
- Fekete, R.; Bak, H.; Vincze, O.; Süveges, K.; Molnár, V.A. Road traffic and landscape characteristics predict the occurrence of native halophytes on roadside verges. Sci. Rep. 2022, 12, 1298.
- Ranjbar, G.; Pirasteh-Anosheh, H.; Banakar, M.H.; Miri, H.R. Review on halophytes researches in Iran: Explanation of Challenges and Offer Approaches. J. Plant Ecophysiol. 2018, 10, 117–129.
- Ranjbar, G.; Pirasteh-Anosheh, H.; Dehghanie, F.; Keshtkar, S.; Race, M. Feasibility of growing Salicornia species in a coastal environment through planting date and density management in a direct seawater irrigation system. Environ. Sci. Pollut. Res. 2022, 29, 47800–47809.
- Prasad, M.N.V. Trace Elements as Contaminants and Nutrients: Consequences in Ecosystems and Human Health; John Wiley & Sons: Hoboken, NJ, USA, 2008.
- Kue Foka, I.C.; Ketehouli, T.; Zhou, Y.; Li, X.W.; Wang, F.W.; Li, H. The emerging roles of diacylglycerol kinase (DGK) in plant stress tolerance, growth, and development. Agronomy 2020, 10, 1375.
- Kefu, Z.; Hai, F.; Ungar, I.A. Survey of halophyte species in China. Plant Sci. 2002, 163, 491–498.
- Corwin, D.L. Climate change impacts on soil salinity in agricultural areas. Eur. J. Soil Sci. 2020, 72, 842–862.
- Mishra, A.; Tanna, B. Halophytes: Potential Resources for Salt Stress Tolerance Genes and Promoters. Front. Plant Sci. 2017, 8, 829.
- Meng, X.; Zhou, J.; Sui, N. Mechanisms of Salt Tolerance in Halophytes: Current Understanding and Recent Advances. Open Life Sci. 2018, 13, 149–154.
- Flowers, T.J.; Colmer, T.D. Plant salt tolerance: Adaptations in halophytes. Ann. Bot. 2015, 115, 327–331.
- Hafsi, C.; Romero-Puertas, M.C.; Gupta, D.K.; del Río, L.A.; Sandalio, L.M.; Abdelly, C. Moderate salinity enhances the antioxidative response in the halophyte Hordeum maritimum L. under potassium deficiency. Environ. Exp. Bot. 2010, 69, 129–136.
- Reginato, M.; Cenzano, A.M.; Arslan, I.; Furlan, A.; Varela, C.; Cavallin, V.; Papenbrock, J.; Luna, V. Na2SO4 and NaCl salts differentially modulate the antioxidant systems in the highly stress tolerant halophyte Prosopis strombulifera. Plant Physiol. Biochem. 2021, 167, 748–762.
- Inupakutika, M.A.; Sengupta, S.; Devireddy, A.R.; Azad, R.K.; Mittler, R. The evolution of reactive oxygen species metabolism. J. Exp. Bot. 2016, 67, 5933–5943.
- Sharma, V.; Anderson, D.; Dhawan, A. Zinc oxide nanoparticles induce oxidative DNA damage and ROS-triggered mitochondria mediated apoptosis in human liver cells. Apoptosis 2012, 17, 852–870.
- Pakar, N.; Pirasteh-Anosheh, H.; Emam, Y.; Pessarakli, M. Barley growth, yield, antioxidant enzymes, and ion accumulation affected by PGRs under salinity stress conditions. J. Plant Nutr. 2016, 39, 1372–1379.
- Alscher, R.G.; Erturk, N.; Heath, L.S. Role of superoxide dismutases (SODs) in controlling oxidative stress in plants. J. Exp. Bot. 2002, 53, 1331–1341.
- Ellouzi, H.; Ben Hamed, K.; Cela, J.; Munné-Bosch, S.; Abdelly, C. Early effects of salt stress on the physiological and oxidative status of Cakile maritima (halophyte) and Arabidopsis thaliana (glycophyte). Physiol. Plant. 2011, 142, 128–143.
- Yildiztugay, E.; Ozfidan-Konakci, C.; Kucukoduk, M. The role of antioxidant responses on the tolerance range of extreme halophyte Salsola crassa grown under toxic salt concentrations. Ecotoxicol. Environ. Saf. 2014, 110, 21–30.
- Ludwiczak, A.; Ciarkowska, A.; Rajabi Dehnavi, A.; Cárdenas-Pérez, S.; Piernik, A. Growth Stage-, Organ- and Time-Dependent Salt Tolerance of Halophyte Tripolium pannonicum (Jacq.) Dobrocz. Life 2023, 13, 462.
- Cárdenas-Pérez, S.; Rajabi Dehnavi, A.; Leszczynski, K.; Lubinska-Mielinska, S.; Ludwiczak, A.; Piernik, A. Salicornia europaea L. Functional Traits Indicate Its Optimum Growth. Plants 2022, 11, 1051.
- Anjum, N.A.; Sharma, P.; Gill, S.S.; Hasanuzzaman, M.; Khan, E.A.; Kachhap, K.; Sofo, A. Catalase and ascorbate peroxidase representative H2O2 detoxifying heme enzymes in plants. Environ. Sci. Pollut. Res. 2016, 23, 19002–19029.
- Shugaev, A.G.; Lashtabega, D.A.; Shugaeva, N.A.; Vyskrebentseva, E.I. Activities of antioxidant enzymes in mitochondria of growing and dormant sugar beet roots. Russ. J. Plant Physiol. 2011, 58, 387–393.
- Lombardi, T.; Bertacchi, A.; Pistelli, L.; Pardossi, A.; Pecchia, S.; Toffanin, A.; Sanmartin, C. Biological and Agronomic Traits of the Main Halophytes Widespread in the Mediterranean Region as Potential New Vegetable Crops. Horticulturae 2022, 8, 195.
- Ghanem, A.-M.F.M.; Mohamed, E.; Kasem, A.M.M.A.; El-Ghamery, A.A. Differential Salt Tolerance Strategies in Three Halophytes from the Same Ecological Habitat: Augmentation of Antioxidant Enzymes and Compounds. Plants 2021, 10, 1100.
- Kumar, A.; Mann, A.; Kumar, A.; Kumar, N.; Meena, B.L. Physiological response of diverse halophytes to high salinity through ionic accumulation and ROS scavenging. Int. J. Phytoremediat. 2021, 23, 1041–1051.
- Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Anee, T.I.; Parvin, K.; Nahar, K.; Mahmud, J.A.; Fujita, M. Regulation of Ascorbate-Glutathione Pathway in Mitigating Oxidative Damage in Plants under Abiotic Stress. Antioxidants 2019, 8, 384.
- Kohli, S.K.; Khanna, K.; Bhardwaj, R.; Abd Allah, E.F.; Ahmad, P.; Corpas, F.J. Assessment of subcellular ros and no metabolism in higher plants: Multifunctional signaling molecules. Antioxidants 2019, 8, 641.
- Mann, A.; Kumar, N.; Lata, C.; Kumar, A.; Meena, B.L.; Kumar, A. Physiological and differential gene expression reveals a trade-off between antioxidant capacity and salt tolerance in halophytes Urochondra setulosa and Dichanthium annulatum. Plant Growth Regul. 2023. preprint.
- Akram, N.A.; Shafiq, F.; Ashraf, M. Ascorbic acid-a potential oxidant scavenger and its role in plant development and abiotic stress tolerance. Front. Plant Sci. 2017, 8, 613.
- Xiao, M.; Li, Z.; Zhu, L.; Wang, J.; Zhang, B.; Zheng, F.; Zhao, B.; Zhang, H.; Wang, Y.; Zhang, Z. The multiple roles of ascorbate in the abiotic stress response of plants: Antioxidant, cofactor, and regulator. Front. Plant Sci. 2021, 12, 598173.
- Yildiztugay, E.; Ozfidan-Konakci, C.; Kucukoduk, M. Sphaerophysa kotschyana, an endemic species from Central Anatolia: Antioxidant system responses under salt stress. J. Plant Res. 2013, 126, 729–742.
- Hameed, A.; Rasheed, A.; Gul, B.; Khan, M.A. Salinity inhibits seed germination of perennial halophytes Limonium stocksii and Suaeda fruticosa by reducing water uptake and ascorbate dependent antioxidant system. Environ. Exp. Bot. 2014, 107, 32–38.
- Szarka, A.; Tomasskovics, B.; Bánhegyi, G. The ascorbate–glutathione–α-tocopherol triad in abiotic stress response. Int. J. Mol. Sci. 2012, 13, 4458–4483.
- Castagna, A.; Mariottini, G.; Gabriele, M.; Longo, V.; Souid, A.; Dauvergne, X.; Magné, C.; Foggi, G.; Conte, G.; Santin, M.; et al. Nutritional Composition and Bioactivity of Salicornia europaea L. Plants Grown in Monoculture or Intercropped with Tomato Plants in Salt-Affected Soils. Horticulturae 2022, 8, 828.
- Gechev, T.S.; Van Breusegem, F.; Stone, J.M.; Denev, I.; Laloi, C. Reactive oxygen species as signals that modulate plant stress responses and programmed cell death. Bioessays 2006, 28, 1091–1101.
