The use of hydrogels in the development of oil fields by the flooding method has firmly established itself in the oil industry, and this direction is constantly evolving in all oil-producing countries around the world, as evidenced by the growth of publication and patent activity on this topic. Operations for conformance control and flow diversion are impossible to imagine without the use of gel technologies today. Inorganic, organic, and hybrid gels, as well as foam gels, gel-forming and gel-dispersed systems are used. The ability to widely regulate structural-mechanical properties, thermal stability, and shear resistance through the introduction of micro- and nano-sized additives has made hydrogels indispensable tools for petroleum engineers.
1. Gels Based on Acrylamide Polymers
Historically, gels based on partially hydrolyzed polyacrylamide, as well as chromium and aluminum salts, were used as one of the first compositions for conformance control, and an interest in these gels has not decreased at the present time [
1,
82]. Due to the adjustable cross-linking time and the transition of the polymer solution into the gel, it is possible to place the gel screen at a predetermined distance from the injection well in such a way that, by changing the direction of the water flow, it leads to the displacement of oil from the bypassed parts of the reservoir (
Figure 17).
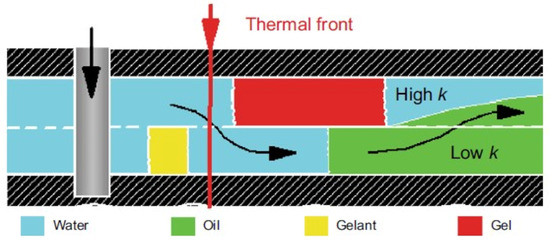
Figure 17. Illustration of in-depth profile modification [
1].
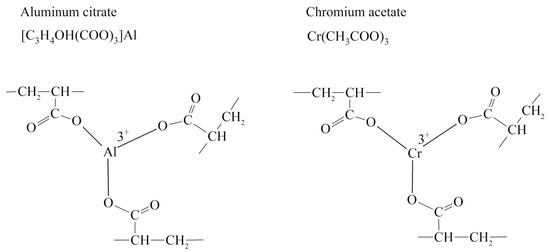
Trivalent metal salts act as cross-linkers in this technology: when they interact with the carboxyl group of the polymer, a cross-linked spatial structure is formed that prevents water filtration [
83] (
Figure 18).
Figure 18. Cross-linking of partially hydrolyzed polyacrylamide with ions of trivalent metals [
83].
Such hydrogels are called cross-linked polymer compositions (CLPC). The gelling time, as well as the structural and mechanical properties of the CLPC, is selected depending on the reservoir temperature by varying concentrations of components.
The CLPC treatment of layers with high layer-by-layer heterogeneity in permeability (≈10:1) and large values of the thickness ratio of the layers of different permeability (low-permeable intervals are ≈10 times thicker than high-permeable ones) is especially efficient. Deep treatment of the bottom-hole zone of the formation allows for the redirection of the water flow from a highly permeable interval to a low-permeable one (Figure 16). Due to the different filtration rates of the unformed gel (gelant) in the intervals of different permeability after gelling, the water flow bends around the barrier in the highly permeable part of the reservoir and displaces oil from the low-permeable part.
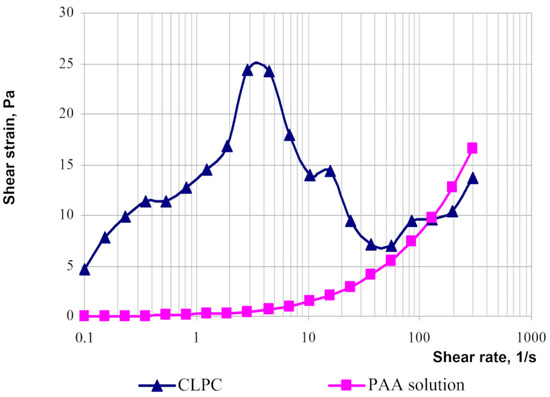
The rheological behaviors of solutions of linear polymers and CLPC-forming gels differ significantly [
23]. For instance, the rheological curves of the gels have an extreme character if the shear strain for polymer solutions increases parabolically with an increasing shear rate (
Figure 19).
Figure 19. Typical flow curves of polyacrylamide solution and cross-linked polymer composition based on PAM [
23].
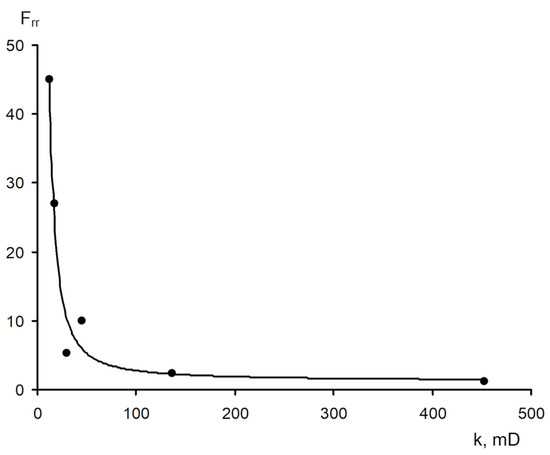
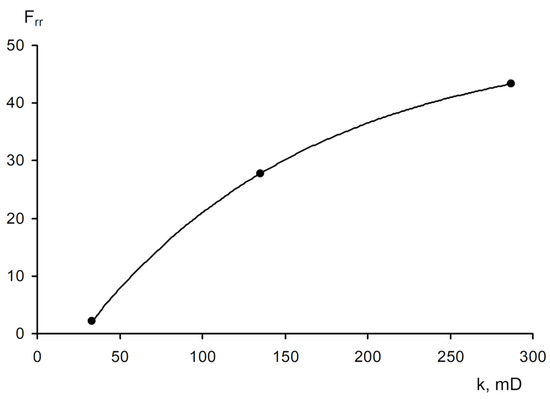
The area of shear strain growth to the maximum value corresponds to an unbroken cross-linked structure. The spatial structure is destroyed with a further increase of the shear rate. The strain at the inflection point represents the “ultimate strength”, and the corresponding shear rate represents the “critical deformation”. Such cardinal differences in rheological behavior in polymer solutions and gel structures also lead to a fundamental difference of their filtration characteristics [
23]. If the residual resistance factor, calculated as the ratio of the pressure drop in water after and before the injection of polymer and gel slug, decreases hyperbolically with increasing permeability in case of linear polymer solutions, then by contrast, it increases in the case of cross-linked gel structures [
23,
84,
85] (
Figure 20 and
Figure 21).
Figure 20. Relationship of the residual resistance factor (Frr) and permeability (polymer concentration with 0.06 wt.%) [
23].
Figure 21. Relationship of the residual resistance factor (Frr) and permeability in case of filtration of CLPC (the size of the slug is 0.3 pore. vol.) [
23].
A lot of theoretical and laboratory studies address the properties of CLPC of various nature and composition [
86,
87,
88]. However, polyacrylamide-based hydrogels with a Cr
3+ or Al
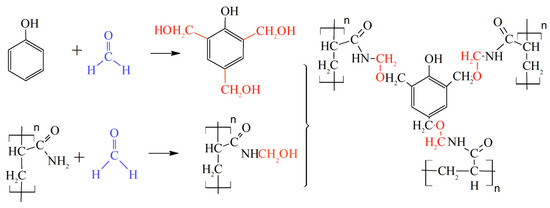
3+ cross-linking are still the most popular in commercial practice. It should be noted that the use of organic staplers—a mixture of phenol and formaldehyde—has allowed for the expansion of the boundaries of the applicability of gels in high-temperature layers, since the cross-linker protects the polymer from thermal oxidative destruction (
Figure 22, [
88]).
Figure 22. Covalent bonds formed by HPAM and phenol/formaldehyde. The cross-linking mechanism includes two steps: (1) Hydroxymethylation of the nitrogen on the amide functional group; and (2) Cross-linking with multiple alkylations on the phenol ring. (Reprinted (adapted) with permission from Zhu et al., Ref. [
88], Copyright 2017 American Chemical Society).
In commercial practice, the injection of sufficiently large volumes of polyacrylamide with a cross-linker (≈10 thousand m
3) in concentrations corresponding to the lower boundary of gel formation allows for the deep treatment of the bottom-hole zone of the formation and significant redistribution of filtration flows [
84,
89]. Therefore,
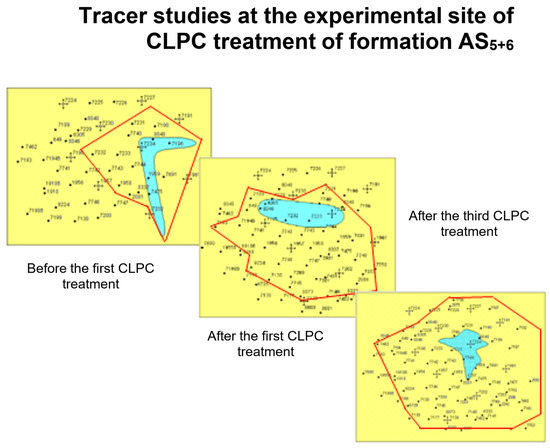
Figure 23 shows that there was actually a linear flow of water from the injection well in two perpendicular directions before CLPC treatment according to the tracer study data. There was a noticeable redistribution of the flow after the first injection of CLPC, and the flow became radial after the third treatment [
23].
Figure 23. Indicator studies in the experimental area of application of the CLPC in formation AS5+6.
Mixtures of water-soluble cellulose esters and polyacrylamide are also capable of forming gels under reservoir conditions under the impact of a cross layer with an adjustable gelling time [
90]. Gels based on hydrolyzed polyacrylonitrile with formalin and hydrochloric acid are used in carbonate fractured reservoirs. Moreover, in this technology, formalin is the cross layer of the polymer, and hydrochloric acid is the initiator of the gelling reaction [
90]. Hydrolyzed polyacrylonitrile in carbonate-fractured reservoirs is pumped without any additives. The polymer is cross-linked with the formation of gel deposits by reaction with ions of alkaline earth metals contained in reservoir water [
91]. In addition, already-formed polyacrylamide-based gels and a cross-linker are injected into carbonate reservoirs to isolate fractures from injection wells, which are not filtered into the pore matrix but selectively shut off only the fracture conductivity of the formation [
1].
Gel systems containing water-swellable yet insoluble particles of cross-linked polymers (gel-particle dispersions) are of great interest for flow-diversion purposes. The synthesis of water-swellable polymers can be carried out in various ways:
- −
-
At the polymerization stage, the bifunctional monomer methylene bisacrylamide is introduced into the composition of acrylamide and acrylic acid monomers [
92].
- −
-
Heat treatment of polyacrylamide at moderate temperatures when the cross-linking of macromolecules occurs as a result of the imidization reaction [
92].
- −
-
Radiation cross-linking of powdered polyacrylamides by gamma or beta radiation [
93,
94,
95,
96,
97].
The gel fraction is a particle with a three-dimensional cross-linking; it is capable of swelling up to 1000 times. Such systems containing dispersion of gel particles are able to significantly reduce the permeability of water-conducting, highly permeable porous and fractured interlayers, and the presence of a linear polymer bearing the gel fraction ensures a better filterability, provides viscoelastic properties, and increases the penetration depth of the flow diversion composition into the formation [
98,
99,
100].
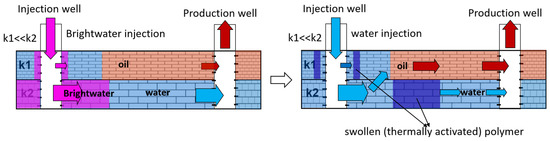
The injection of microparticles of cross-linked polyacrylamide obtained by emulsion polymerization after thermal activation in the formation allows for the formation of flow-diversion gel screens at a particular distance from wells. Polymer microparticles of ≈0.5 microns in size are injected as a dispersion in an organic solvent and forced through the formation. Due to the reservoir temperature in the water, the particles swell, after which they lose the ability to filter. This technology was named BRIGHT WATER and was developed by a consortium of SHEVRON TEXACO, BP, and Nalco Company in 1977 [
101,
102]. A schematic diagram of oil displacement in an inhomogeneous reservoir by intra-reservoir flows is shown in
Figure 24.
Figure 24. Schematic diagram of oil displacement using Bright Water injection.
2. Gel-Dispersed and Sedimentary-Gel-Forming Compositions
The transformation into sedimentary-gel-forming and gel-dispersed materials by introducing dispersions (clay, chalk, marl, and wood flour) has been one of the remarkable trends in the development of conformance control technologies using gels in Russia over the past 10 years, which enhances the structural and mechanical properties of the system, allowing for the reduction of the volume of its injection [
23,
103,
104]. Sedimentation and gel-forming systems are obtained by the interaction of aluminum oxychloride with modified polyacrylamide [
105]. Such gel additives also have selectivity in permeability, i.e., with increasing permeability, the residual resistance factor also increases (
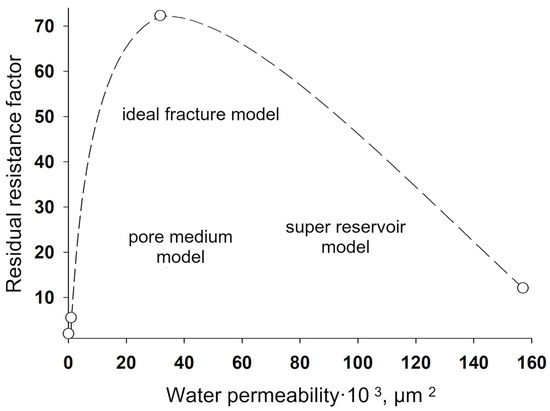
Figure 25).
Figure 25. Dependence of the residual resistance factor for modified polyacrylamide cross-linked with aluminum oxychloride on the initial (before treatment) permeability of the medium to water.
Figure 25 shows the values of the residual resistance factor during filtration of the sedimentary-gel-forming reagent in three media differing in permeability and structure of the pore space: pore medium, super reservoir, and fracture. It can be seen that in the pore medium, the residual resistance factor is minimal compared to the super reservoir and the fracture model.
3. Gels Based on Inorganic Compounds
Methods of regulating intra-layer filtration flows using inorganic gels allow for the creation of strong barriers to water filtration, which leads to a change in the direction of movement of the displacing agent and to the connection of oil-saturated, poorly drained, untreated interlayers to development [
3]. Gels based on silicon compounds, such as silicates and aluminosilicates, have become the most widespread. Silicate gels are formed by the acidification of alkaline solutions of sodium silicate with acids to neutral pH values. The silicate gel, being a pseudoplastic material, “breaks” with the formation of microgel agglomerates ranging in size from 2 to 27 microns when injected into the injection well and advancing through the formation. Such a microgel dispersion in sodium silicate solutions has both viscoplastic and viscoelastic properties, which allows for the redirection of water flows from the developed high-permeable intervals to low-permeable, oil-saturated ones [
107,
108].
Polysilicic acid gels, for leveling the conformance control, are also obtained from aluminosilicate, a natural mineral of nepheline [
109]. The principle of the technology is that the aluminosilicate, when dissolved in inorganic acids, forms a composition that is able to coagulate, turning into a gel. Dissolution occurs with an excess of acid. Subsequent gelling occurs by aggregation with the formation of three-dimensional polymer meshes. The initial particles condense together with an increase in the concentration of the solution, forming a “ringing” gel. As a result of the interaction of aluminosilicate with hydrochloric acid, an aluminosilicate sol is formed, followed by a monosilicic acid, its low-molecular-weight oligomers, and then its silicic acid sol, which turns into a gel [
110].
Solutions of other acids, such as phosphoric and sulfamic acids, can also be used in gel-forming compositions [
111,
112,
113]. In addition, synthetic zeolites are used as aluminosilicates [
114].
The use of bicalcium silicate allows for the accumulation of acidic gels with an adjustable gelling time, which makes it possible to place the flow diversion material at a particular distance from the well [
115,
116].
It is proposed to add partially hydrolyzed polyacrylamide to the composition to provide gels based on sodium silicate with viscoelastic properties [
117]. A similar technique for obtaining a silicate–polymer gel is described in [
118]. Only the spent catalyst Zeokar-10 was used as a source of aluminosilicate after dissolving it in a weak alkali. Water-soluble cellulose derivatives were also used as a polymer, along with polyacrylamide.
L. K. Altunina and her co-researchers experimentally substantiated and implemented the technological process of using both inorganic and organic gels for well conformance control for deposits characterized by high layer-by-layer heterogeneity and temperature. These are gel-forming systems that are low-viscosity solutions in surface conditions and turn into gels in reservoir conditions. The factor that causes gelling is the thermal energy of the reservoir or the injected coolant. Gel-forming compositions with different gelling times—from several minutes to several days—in the temperature range of 40–120 °C were proposed. These include inorganic gel-forming compositions GALKA, GALKA–PAV, and GALKA–U based on the system “aluminum salt—urea—water,” as well as compositions METKA based on thermally reversible polymers of methylcellulose [
4,
119,
120,
121].
GALKA and GALKA–PAV surfactants are low-viscosity solutions with pH = 2.5–3. They contain aluminum salt, carbamide, and some additives that improve their process parameters. The urea is hydrolyzed, forming ammonia and CO2 due to the thermal energy of the reservoir or the energy of the injected coolant, which gradually increases the pH of the solution. The aluminum hydroxide gel is formed in the entire volume of the solution at pH = 10. This is manifested in an abrupt increase in the dynamic shear strain of the gel-forming solution.
The gels formed by the METKA compositions are reversible. When the temperature decreases, they can turn back into a liquid. When the temperature rises, they can form a gel again, which makes it possible to “open” and “close” the interlayers by changing the temperature to regulate filtration flows. This property of gels can be used for a cyclic injection of hot water or steam in order to increase the coverage of the reservoir by thermal action. Currently, thermotropic inorganic gel GALKA is used with steam-assisted gravity drainage (SAGD) technology for steam diversion in horizontal wells [
122].
Thus, various gel-forming compositions based on water-soluble and water-swelling polymers, as well as inorganic silicon and aluminum compounds, are used for flow diversion and conformance control. The combination of gels with dispersants and precipitating compositions allows for the enhancement of the structural–mechanical properties of the diversion system, making it possible to apply these compositions in naturally fractured reservoirs. The ability to regulate the gel formation time allows for the placement of the gel screen at a specified distance from the injection wellbore. This approach has firmly entered industrial practice and continues to be improved both in terms of reducing the cost of the compositions used and increasing their effectiveness.
This entry is adapted from the peer-reviewed paper 10.3390/gels9080609