Vitamin D seems to be involved in infections, autoimmune diseases, cardiometabolic diseases, and cancer development. The relationship between vitamin D and insulin resistance has been a topic of growing interest. Low 25-hydroxyvitamin D (25(OH)D) levels appear to be associated with most of the insulin resistance disorders described to date. In fact, vitamin D deficiency may be one of the factors accelerating the development of insulin resistance. Vitamin D deficiency is a common problem in the population and may be associated with the pathogenesis of diseases related to insulin resistance, such as obesity, diabetes, metabolic syndrome (MS) and polycystic ovary syndrome (PCOS). An important question is the identification of 25(OH)D levels capable of generating an effect on insulin resistance, glucose metabolism and to decrease the risk of developing insulin resistance related disorders.
- vitamin D
- 25-hydroxyvitamin D or calcidiol (25(OH)D)
- calcitriol (1,25(OH)2D)
- vitamin D receptor (VDR)
- 25-hydroxyvitamin D-1alpha-hydroxylase (CYP27B1)
- insulin resistance
- homeostasis model assessment of insulin resistance (HOMA-IR) type 2 diabetes
- o
1. Background
2. Vitamin D and Insulin Resistance Physiology
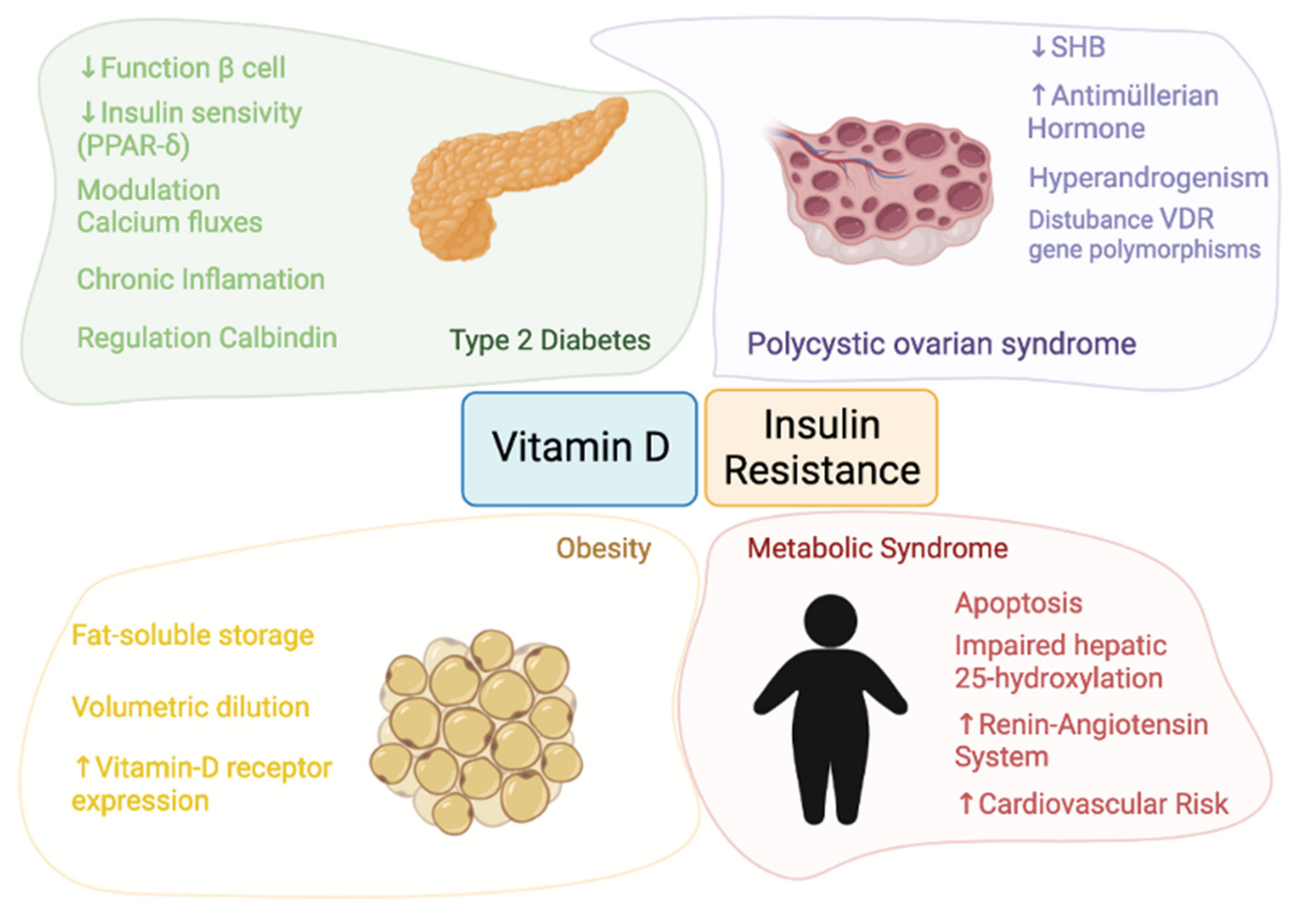
This entry is adapted from the peer-reviewed paper 10.3390/nu13103491
References
- Hilger, J.; Friedel, A.; Herr, R.; Rausch, T.; Roos, F.; Wahl, D.A.; Pierroz, D.D.; Weber, P.; Hoffmann, K. A Systematic Review of Vitamin D Status in Populations Worldwide. Br. J. Nutr. 2014, 111, 23–45.
- Dattola, A.; Silvestri, M.; Bennardo, L.; Passante, M.; Scali, E.; Patruno, C.; Nisticò, S.P. Role of Vitamins in Skin Health: A Systematic Review. Curr. Nutr. Rep. 2020, 9, 226–235.
- Pike, J.W.; Meyer, M.B. The Vitamin D Receptor: New Paradigms for the Regulation of Gene Expression by 1,25-Dihydroxyvitamin D 3. Rheum. Dis. Clin. N. Am. 2012, 38, 13–27.
- Holick, M.F. Vitamin D: Extraskeletal Health. Endocrinol. Metab. Clin. N. Am. 2010, 39, 381–400.
- Mizwicki, M.T.; Norman, A.W. The Vitamin D Sterol-Vitamin D Receptor Ensemble Model Offers Unique Insights into Both Genomic and Rapid-Response Signaling. Sci. Signal. 2009, 2, re4.
- Pittas, A.G.; Lau, J.; Hu, F.B.; Dawson-Hughes, B. The Role of Vitamin D and Calcium in Type 2 Diabetes. A Systematic Review and Meta-Analysis. J. Clin. Endocrinol. Metab. 2007, 92, 2017–2029.
- Maretzke, F.; Bechthold, A.; Egert, S.; Ernst, J.B.; van Lent, D.M.; Pilz, S.; Reichrath, J.; Stangl, G.I.; Stehle, P.; Volkert, D.; et al. Role of Vitamin D in Preventing and Treating Selected Extraskeletal Diseases—An Umbrella Review. Nutrients 2020, 12, 969.
- Grundy, S.M. Obesity, Metabolic Syndrome, and Cardiovascular Disease. J. Clin. Endocrinol. Metab. 2004, 89, 2595–2600.
- Poirier, P.; Giles, T.D.; Bray, G.A.; Hong, Y.; Stern, J.S.; Pi-Sunyer, F.X.; Eckel, R.H. Obesity and Cardiovascular Disease: Pathophysiology, Evaluation, and Effect of Weight Loss: An Update of the 1997 American Heart Association Scientific Statement on Obesity and Heart Disease from the Obesity Committee of the Council on Nutrition, Physica. Circulation 2006, 113, 898–918.
- Goossens, G.H. The Role of Adipose Tissue Dysfunction in the Pathogenesis of Obesity-Related Insulin Resistance. Physiol. Behav. 2008, 94, 206–218.
- Wang, H.; Chen, W.; Li, D.; Yin, X.; Zhang, X.; Olsen, N.; Zheng, S.G. Vitamin D, Aging, and Chronic Diseases. Vitam. Oxidative Stress Immun. Aging 2012, 8, 385–406.
- International Diabetes Federation. IDF Diabetes Atlas, 8th ed.; International Diabetes Federation: Brussels, Belgium, 2017.
- Upreti, V.; Maitri, V.; Dhull, P.; Handa, A.; Prakash, M.S.; Behl, A. Effect of Oral Vitamin D Supplementation on Glycemic Control in Patients with Type 2 Diabetes Mellitus with Coexisting Hypovitaminosis D: A Parellel Group Placebo Controlled Randomized Controlled Pilot Study. Diabetes Metab. Syndr. 2018, 12, 509–512.
- Mirhosseini, N.; Vatanparast, H.; Mazidi, M.; Kimball, S.M. The Effect of Improved Serum 25-Hydroxyvitamin D Status on Glycemic Control in Diabetic Patients: A Meta-Analysis. J. Clin. Endocrinol. Metab. 2017, 102, 3097–3110.
- Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III). JAMA 2001, 285, 2486–2497.
- Swarup, S.; Zeltser, R. Metabolic Syndrome; StatPearls Publishing: Treasure Island, FL, USA, 2020.
- Engin, A. The Definition and Prevalence of Obesity and Metabolic Syndrome. Adv. Exp. Med. Biol. 2017, 960, 1–17.
- Saklayen, M.G. The Global Epidemic of the Metabolic Syndrome. Curr. Hypertens. Rep. 2018, 20, 12.
- Barnard, L.; Ferriday, D.; Guenther, N.; Strauss, B.; Balen, A.H.; Dye, L. Quality of Life and Psychological Well Being in Polycystic Ovary Syndrome. Hum. Reprod. Oxf. Engl. 2007, 22, 2279–2286.
- Revised 2003 Consensus on Diagnostic Criteria and Long-Term Health Risks Related to Polycystic Ovary Syndrome (PCOS). Hum. Reprod. Oxf. Engl. 2004, 19, 41–47.
- McLuskie, I.; Newth, A. New Diagnosis of Polycystic Ovary Syndrome. BMJ 2017, 356, i6456.
- Mayer, S.B.; Evans, W.S.; Nestler, J.E. Polycystic Ovary Syndrome and Insulin: Our Understanding in the Past, Present and Future. Womens Health Lond. Engl. 2015, 11, 137–149.
- Ciampelli, M.; Fulghesu, A.M.; Cucinelli, F.; Pavone, V.; Ronsisvalle, E.; Guido, M.; Caruso, A.; Lanzone, A. Impact of Insulin and Body Mass Index on Metabolic and Endocrine Variables in Polycystic Ovary Syndrome. Metabolism 1999, 48, 167–172.
- Robinson, S.; Henderson, A.D.; Gelding, S.V.; Kiddy, D.; Niththyananthan, R.; Bush, A.; Richmond, W.; Johnston, D.G.; Franks, S. Dyslipidaemia Is Associated with Insulin Resistance in Women with Polycystic Ovaries. Clin. Endocrinol. Oxf. Print 1996, 44, 277–284.
- Holick, M.F. Vitamin D Deficiency. N. Engl. J. Med. 2007, 357, 266–281.
- Sözen, T.; Özışık, L.; Başaran, N.Ç. An Overview and Management of Osteoporosis. Eur. J. Rheumatol. 2017, 4, 46–56.
- Bischoff-Ferrari, H.A.; Giovannucci, E.; Willett, W.C.; Dietrich, T.; Dawson-Hughes, B. Erratum: Estimation of Optimal Serum Concentrations of 25-Hydroxyvitamin D for Multiple Health Outcomes. Am. J. Clin. Nutr. 2006, 84, 18–28.
- Zittermann, A.; Iodice, S.; Pilz, S.; Grant, W.B.; Bagnardi, V.; Gandini, S. Vitamin D Deficiency and Mortality Risk in the General Population: A Meta-Analysis of Prospective Cohort Studies. Am. J. Clin. Nutr. 2012, 95, 91–100.
- Bikle, D.D. Vitamin D Metabolism, Mechanism of Action, and Clinical Applications. Bone 2014, 23, 1–7.
- Hossein-Nezhad, A.; Holick, M.F. Vitamin D for Health: A Global Perspective. Mayo Clin. Proc. 2013, 88, 720–755.
- Pike, J.W.; Meyer, M.B. The Vitamin D Receptor: New Paradigms for the Regulation of Gene Expression by 1,25-Dihydroxyvitamin D(3). Endocrinol. Metab. Clin. N. Am. 2010, 39, 255–269.
- Kato, S. The Function of Vitamin D Receptor in Vitamin D Action. J. Biochem. Tokyo 2000, 127, 717–722.
- Norman, A.W. From Vitamin D to Hormone D: Fundamentals of the Vitamin D Endocrine System Essential for Good Health. Am. J. Clin. Nutr. 2008, 88, 491S–499S.
- Maestro, B.; Dávila, N.; Carranza, M.C.; Calle, C. Identification of a Vitamin D Response Element in the Human Insulin Receptor Gene Promoter. J. Steroid Biochem. Mol. Biol. 2003, 84, 223–230.
- Yamada, S.; Kakuta, H. Retinoid X Receptor Ligands: A Patent Review (2007–2013). Expert Opin. Ther. Pat. 2014, 24, 443–452.
- Mangelsdorf, D.J.; Thummel, C.; Beato, M.; Herrlich, P.; Schütz, G.; Umesono, K.; Blumberg, B.; Kastner, P.; Mark, M.; Chambon, P.; et al. The Nuclear Receptor Superfamily: The Second Decade. Cell 1995, 83, 835–839.
- Haussler, M.R.; Whitfield, G.K.; Haussler, C.A.; Hsieh, J.C.; Thompson, P.D.; Selznick, S.H.; Dominguez, C.E.; Jurutka, P.W. The Nuclear Vitamin D Receptor: Biological and Molecular Regulatory Properties Revealed. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 1998, 13, 325–349.
- Cione, E.; Caroleo, M.C.; Cannataro, R.; Perri, M.; Pingitore, A.; Genchi, G. Vitamin A and Diabesity: New Insight for Drug Discovery. Mini Rev. Med. Chem. 2016, 16, 738–742.
- Shaffer, P.L.; Gewirth, D.T. Vitamin D Receptor-DNA Interactions. Vitam. Horm. 2004, 68, 257–273.
- Parsanathan, R.; Jain, S.K. Glutathione Deficiency Alters the Vitamin D-Metabolizing Enzymes CYP27B1 and CYP24A1 in Human Renal Proximal Tubule Epithelial Cells and Kidney of HFD-Fed Mice. Free Radic. Biol. Med. 2019, 131, 376–381.
- Jain, S.K.; Parsanathan, R.; Achari, A.E.; Kanikarla-Marie, P.; Bocchini, J.A.J. Glutathione Stimulates Vitamin D Regulatory and Glucose-Metabolism Genes, Lowers Oxidative Stress and Inflammation, and Increases 25-Hydroxy-Vitamin D Levels in Blood: A Novel Approach to Treat 25-Hydroxyvitamin D Deficiency. Antioxid. Redox Signal. 2018, 29, 1792–1807.
- Jain, S.K.; Micinski, D. Vitamin D Upregulates Glutamate Cysteine Ligase and Glutathione Reductase, and GSH Formation, and Decreases ROS and MCP-1 and IL-8 Secretion in High-Glucose Exposed U937 Monocytes. Biochem. Biophys. Res. Commun. 2013, 437, 7–11.
- Ballatori, N.; Krance, S.M.; Notenboom, S.; Shi, S.; Tieu, K.; Hammond, C.L. Glutathione Dysregulation and the Etiology and Progression of Human Diseases. Biol. Chem. 2009, 390, 191–214.
- Reusch, J.E.; Begum, N.; Sussman, K.E.; Draznin, B. Regulation of GLUT-4 Phosphorylation by Intracellular Calcium in Adipocytes. Endocrinology 1991, 129, 3269–3273.
- Bland, R.; Markovic, D.; Hills, C.E.; Hughes, S.V.; Chan, S.L.F.; Squires, P.E.; Hewison, M. Expression of 25-Hydroxyvitamin D3-1alpha-Hydroxylase in Pancreatic Islets. J. Steroid Biochem. Mol. Biol. 2004, 89–90, 121–125.
- Leung, P.S. The Potential Protective Action of Vitamin D in Hepatic Insulin Resistance and Pancreatic Islet Dysfunction in Type 2 Diabetes Mellitus. Nutrients 2016, 8, 147.
- Chun, R.F.; Liu, P.T.; Modlin, R.L.; Adams, J.S.; Hewison, M. Impact of Vitamin D on Immune Function: Lessons Learned from Genome-Wide Analysis. Front. Physiol. 2014, 5, 151.
- Christakos, S.; Liu, Y. Biological Actions and Mechanism of Action of Calbindin in the Process of Apoptosis. J. Steroid Biochem. Mol. Biol. 2004, 89–90, 401–404.
- Schmitt, E.B.; Nahas-Neto, J.; Bueloni-Dias, F.; Poloni, P.F.; Orsatti, C.L.; Petri Nahas, E.A. Vitamin D Deficiency Is Associated with Metabolic Syndrome in Postmenopausal Women. Maturitas 2018, 107, 97–102.
- Lin, X.F.; Wu, R.R.; Du, J.; Liao, Y.C.; Du, Y.; Ye, Y.; Wang, Y.; Zhang, X.B.; Wu, C.; Chen, A. Exploring the Significance of Sex Hormone-Binding Globulin Examination in the Treament of Women with Polycystic Ovarian Syndrome (PCOS). Clin. Exp. Obstet. Gynecol. 2015, 42, 315–320.
- Zhu, J.-L.; Chen, Z.; Feng, W.-J.; Long, S.-L.; Mo, Z.-C. Sex Hormone-Binding Globulin and Polycystic Ovary Syndrome. Clin. Chim. Acta Int. J. Clin. Chem. 2019, 499, 142–148.
- Krul-Poel, Y.H.M.; Snackey, C.; Louwers, Y.; Lips, P.; Lambalk, C.B.; Laven, J.S.E.; Simsek, S. The Role of Vitamin D in Metabolic Disturbances in Polycystic Ovary Syndrome: A Systematic Review. Eur. J. Endocrinol. 2013, 169, 853–865.
- Ardabili, H.R.; Gargari, B.P.; Farzadi, L. Vitamin D Supplementation Has No Effect on Insulin Resistance Assessment in Women with Polycystic Ovary Syndrome and Vitamin D Deficiency. Nutr. Res. 2012, 32, 195–201.
