1. Pregnancy-Associated Plasma Protein A2 Mouse Models: What Are We Learning?
While a few controversies exist, many of the findings obtained from studies using knock-out (KO), knock-in (KI) and knock-down (KD) mouse models of
Pappa2 recapitulate the main features described in patients with pregnancy-associated plasma protein (
PAPP-A2) mutations [
58]. In this section, we summarize findings from mouse models of
Pappa2 with a special focus on auxological, hormonal, metabolic and BMD alterations during postnatal growth and adulthood (
Table 1). Finally, we propose studies of experimental treatments using recombinant murine Igf1 (rmIgf1) and recombinant murine Pappa2 (rmPappa2), in mouse models with Pappa2 deficiency, which could help to provide information regarding their ability to palliate growth retardation, as well as the effects on other physiological processes such as pubertal development, metabolism, and bone formation and quality, and possible long-term complications.
Table 1. Summary of the main effects of Pappa2 deficiency on auxological, hormonal, metabolic and bone mineral properties.
| |
Constitutive Pappa2 KO |
Constitutive Pappa2 KO |
Constitutive Induction of Pappa2 KO |
Conditional Pappa2 KO in Osteoblasts |
Constitutive Human PAPP-A2 KI |
| Main references |
Conover et al. 2011 |
Christians et al. 2013, 2015a, 2019; Rogowska et al. 2021; Rubio et al. 2021 |
Christians et al. 2015a |
Amiri and Christians, 2015 |
Fujimoto et al. 2019 |
| Auxological parameters |
| Body weight (BW) |
Ns 1 in males
Reduction in females |
Reduction |
ns |
Reduction |
Reduction |
| Body length |
Ns in males
Reduction in females |
Reduction |
-- |
Reduction in tail |
Reduction |
| Organ size |
Ns in males
Increase in females |
ns |
-- |
-- |
Increase in liver |
| Body composition |
-- |
-- |
-- |
-- |
Higher fat mass. Lower lean mass |
| Hormonal parameters |
| Free Igf1 |
Decrease |
Decrease |
-- |
-- |
Decrease |
| Total Igf1 |
Increase |
Increase |
-- |
-- |
Increase |
| Igfbp5 |
-- |
Increase in plasma
Ns in liver and kidney
Increase in ovaries
Ns in tibia |
ns |
ns |
-- |
| Igfbp3 |
-- |
Decrease in serum
Ns in liver and kidney
Increase in tibia |
Decrease |
-- |
Increase |
| Igfals |
-- |
Ns in tibia |
-- |
-- |
Increase |
| Energy metabolism |
| Glucose tolerance |
-- |
ns |
-- |
-- |
Intolerant |
| Insulin sensitivity |
-- |
ns |
-- |
-- |
Resistant |
| Adiposity |
|
ns |
-- |
-- |
-- |
| BW loss |
-- |
Increase in fasting |
-- |
-- |
-- |
| Caloric intake |
-- |
Increase in HCHD 2 |
-- |
-- |
-- |
| Allometric parameters |
| Femur length |
ns |
Reduction |
-- |
Reduction |
Reduction |
| Femur weight |
-- |
Reduction |
-- |
-- |
-- |
| Other bone dimensions |
-- |
Defects |
-- |
Defects |
-- |
| Bone shape |
-- |
Defects (pelvic girdle and mandible) |
-- |
Defects (pelvic girdle and mandible) |
-- |
| Bone mineral properties |
| Bone mineral content |
Decreases in trabecular and cortical femur |
Decrease in trabecular femur
Increase in cortical femur |
--
-- |
--
-- |
Decrease in trabecular femur
Ns in cortical femur |
| Bone mineral composition |
-- |
Alterations in male femur |
-- |
-- |
-- |
| Collagen maturity |
|
Decrease in female femur |
-- |
-- |
-- |
| Bone remodeling |
| Bone formation and resorption |
-- |
Decreases in female serum
Increases in female tibia |
-- |
-- |
-- |
2. Expression Pattern of Pappa2 in Mice
In mammals [
59,
60,
61], Pappa2 is highly expressed in the feto-maternal interface of the placenta at all gestational stages, in addition to being expressed in specific locations in other tissues throughout the body of the embryo and adult mouse [
26,
62,
63]. Christians et al. [
62] reported high expression of Pappa2 in the stomach and skin of adult mice, as well as in murine embryos, and lower expression in the kidney, brain, heart, lung, testis, pancreas and prostate gland. Later, Conover et al. [
26] indicated that, in addition to the elevated expression of Pappa2 in mouse placenta and embryo, it is also highly expressed in the prostate, colon, lung, ovaries, tibia, brain, spinal cord, testes and kidneys, while being undetectable in the spleen, soleus, adipose tissue, thymus, uterus, liver, skin and heart [
26]. The importance of Pappa2 in bone and joint development is emphasized by the demonstration of its expression in the epiphyseal cartilage of the acetabulum and femoral head of the hip joints of rats at different perinatal stages [
64], as well as in the epiphysis and metaphysis, including osteoblasts, of the femur and tibia of 19-day-old mice [
65]. Analysis of Pappa2 expression has also been performed in zebrafish embryos, larvae and adults, with adult tissues such as bone, muscle, eye, brain, gill, kidney, gastrointestinal tract, ovary and testis shown to have higher expression levels than other tissues [
66]. Together, these findings confirm the physiological importance of PAPP-A2 in the promotion of placental, fetal and postnatal growth, and support the hypothesis that PAPP-A2 may act as a local tissue-specific modulator of IGF1 bioavailability by cleavage of ternary complexes containing IGFBP3 or IGFBP5 and IGFALS.
3. Available Models and Their Differences in Growth and Body Weight
Two knock-out and one knock-in mouse models with targeted disruption of
Pappa2 have been studied to date [
26,
67,
68]. The most striking phenotype of these genetically modified mice is postnatal growth retardation with low levels of free Igf1 and high levels of total Igf1 [
58], similar to that seen in PAPP-A2 deficient patients. The first homozygous mice with a constitutive
Pappa2 deletion were developed and described by Conover and colleagues [
26]. In this study, 16-week-old
Pappa2 KO males weighed 10% less and were modestly smaller on average, but this was not significantly different from male wild-type (WT) littermates. In contrast,
Pappa2 KO females had significantly lower body weight (30%) and length (10%), which was associated with disproportionally larger organs, expressed as wet weight relative to total body weight, including heart, spleen, liver, kidneys and brain. Two years later, the second constitutive
Pappa2 KO mouse model was reported [
68] resulting in further insights into postnatal growth retardation. These authors did not detect any effect of constitutive
Pappa2 deletion on placental or embryonic mass or on the dry weights of stomach, spleen, pancreas, kidneys, liver, lungs or heart of
Pappa2 KO mice at 10 weeks of age. However, they described reductions in both body weight and tail length at 3, 6 and 10 weeks of age [
68]. Subsequently, these authors developed genetically modified mice by disrupting
Pappa2 in SP7-expressing osteoblasts (conditional
Pappa2 deletion) and reported a sex-independent decrease in body weight and tail lengths at all ages [
65]. These results indicated that the effect of whole body
Pappa2 deletion was greater than the effect of osteoblast-specific
Pappa2 deletion. In contrast, when specific
Pappa2 deletion was triggered at 20 weeks of age by tamoxifen-induced
Cre-mediated recombination (resulting in whole body
Pappa2 deletion), no difference between genotypes in body weight was detected [
69]. Recently, using the second mouse model for constitutive
Pappa2 deletion, our research group also reported a sex-independent reduction in body length of
Pappa2 KO mice at 8 months of age [
70]. A constitutive knock-in mouse model employing the human p.Ala1033Val mutation in
PAPP-A2 was generated using the CRISPR/Cas9 system [
67]. These human
PAPP-A2 KI mice, expressing PAPP-A2 proteins that lack protease activity for IGFBP3, confirmed the growth retardation associated with the p.Ala1033Val missense mutation previously described in humans [
48]. These mice showed reductions in body length and weight in both males and females (89.6% and 77.7%, respectively, of mean body weight with respect to WT littermates) when they were measured from 4 to 16 weeks of age. These authors also report a proportionally higher fat mass and lower lean mass in
PAPP-A2 KI mice of both sexes. However, liver weight relative to total body weight was only elevated in female human-
PAPP-A2 KI mice at 16 weeks of age.
4. Differences in Hormonal Alterations in Available Models of Pappa2 Deficiency
In the model presented by Conover and colleagues [
26], no changes in Pappa2 mRNA levels or Igfbp5 proteolysis in fibroblasts of
Pappa2 KO embryos were found. However, at 4 months of age, these
Pappa2 KO mice had a dramatic reduction (95%) in free Igf1 concentrations and elevated concentrations (50%) of total Igf1, supporting functional
PAPP-A2 defects in the release of IGF1 from its ternary complex, as observed in affected patients [
48]. Christians and colleagues [
69] also reported an increase (60%) in circulating levels of total Igf1 at 1.5 months of age in their
Pappa2-KO model. Recently, these authors described a reduction in the circulating levels of a putative free bioactive Igf1 in 2-month-old
Pappa2 KO mice [
71]. Circulating free Igf1 is also decreased and total Igf1 increased in the
Pappa2 KI mice at 4 months of age [
67]. Using the second
Pappa2 KO mouse model developed by Christians et al. [
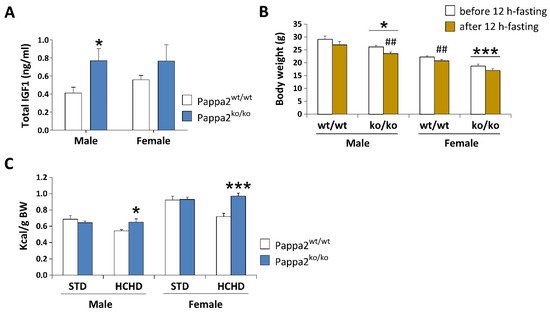
68], we have observed higher circulating levels of total Igf1, specifically in males, but not in
Pappa2 KO females at 8 months of age (
Figure 1A). At 1.5 months of age constitutive
Pappa2 KO mice also had 2-fold higher levels of Igfbp5 and 15-fold lower levels of Igfbp3 [
69]. Increased circulating levels of Igfbp5, recently confirmed in 2-month-old
Pappa2 KO mice, were not influenced by gestation and recovery after lactation [
71], with these variations also continuing to be observed in the plasma of older mice with a mean age of 7 months [
69]. However, at 7 months of age, no effect of
Pappa2 deletion was observed on mRNA levels of
Igfbp3 or
Igfbp5 in the liver or kidney, but Igfbp5 protein levels were increased in ovaries of female
Pappa2 KO mice [
72]. In contrast, osteoblast-specific deletion of
Pappa2 did not alter circulating levels of Igfbp5 in mice at 18–19 days of age [
65], suggesting a local Pappa2-Igfbp5 mechanism for Igf1 function in bone physiology not yet addressed. When whole body
Pappa2 deletion was induced at 5 months of age, no difference in circulating levels of Igfbp5 was found between genotypes, but lower levels of circulating Igfbp3 were detected in a body size and sex-independent manner [
69]. In contrast, increased circulating levels of Igfbp3 and Igfals were found in
Pappa2 KI mice at 4 months of age [
67]. We found a genotype effect on
Igfbp3 mRNA levels in the bone of 8-month-old
Pappa2 KO mice with female
Pappa2 KO mice having higher
Igfbp3 mRNA levels in the tibia than female WT littermates [
70].
Figure 1. Analysis of plasma concentrations of total IGF1 (A), body weight (BW) before and after 12 h-fasting (B), and caloric intake relative to BW after 3 week-feeding of a standard (STD: 2.9 kcal/g) or a high carbohydrate (HCHD: 3.85 kcal/g) diet (C) in constitutive Pappa2 KO mice of both sexes at 8 months old. Data are represented as mean ± S.E.M. (n = 7–8/group). Tukey-corrected tests: */*** p < 0.05/0.001 versus respective Pappa2wt/wt mice; ## p < 0.01 versus mice before 12 h-fasting.
Analysis of a morpholino-mediated
Pappa2 KD zebrafish embryo model (50% and 43% overall sequence identity to human
PAPP-A2 and
PAPP-A, respectively, and conserved proteolytic activity in human IGFBP3 and IGFBP5) suggested that Pappa2 actions in normal growth and development may also be exerted through non-proteolytic modulation of Notch signaling [
66]. While IGFBP-independent mechanisms of PAPP-A2 cannot be excluded, overall results to date indicate that the modulation of IGF1 bioavailability through its release from ternary and binary complexes should be further investigated in a tissue, sex and age-specific manner.
5. Metabolic Disturbance in Available Models of Pappa2 Deficiency
Despite the smaller size and lower weight of
Pappa2 KO mice at 3, 6, 10 and 14 weeks of age in the model described by Christians and colleagues [
69], disruption of circulating Igf1 levels did not affect glucose tolerance, body weight gain or adiposity at 6 weeks of age when constitutive
Pappa2 KO mice were fed a high-fat diet (45% fat) from 17 to 25 weeks of age. Likewise, using the mouse model developed by Christians et al. [
68], we found no modifications in glucose tolerance in constitutive
Pappa2 KO mice at 2 months of age, but we found that these
Pappa2 KO mice lost more body weight than their WT littermates when fasted for 12 h (
Figure 1B). In contrast,
Pappa2 KI mice are reported to be glucose intolerant and insulin resistant [
67]. These discrepancies in glucose handling may be due to differences in the dietary supply of glucose, amino acids and lipids to the liver, in addition to metabolic compensation, such as an increase in insulin concentrations and/or decrease in the insulin-induced autophosphorylation of its receptor and insulin receptor substrate, observed in constitutive
Pappa2 KO mice that may not be produced in human
Pappa2 KI mice due to the presence of a Pappa2 protein, at least in some patients, despite its lack of protease activity. In our preliminary studies, we did not observe differences in body weight gain compared to WT when
Pappa2 KO mice were fed a high-carbohydrate diet (HCHD, 70% sucrose and fructose) for 3 weeks, but they increased their caloric intake (relative to body weight), suggesting higher energy expenditure associated with lower Igf1 availability when exposed to a high carbohydrate diet (
Figure 1C). Because diabetes and obesity induced by hypercaloric diets have been associated with impairments in bone mineral density [
73,
74,
75], future studies should evaluate whether deficiency in Igf1 bioactivity impacts energy homeostasis, regulating bone mass in a sex and diet-specific manner [
76].
This entry is adapted from the peer-reviewed paper 10.3390/cells10123576