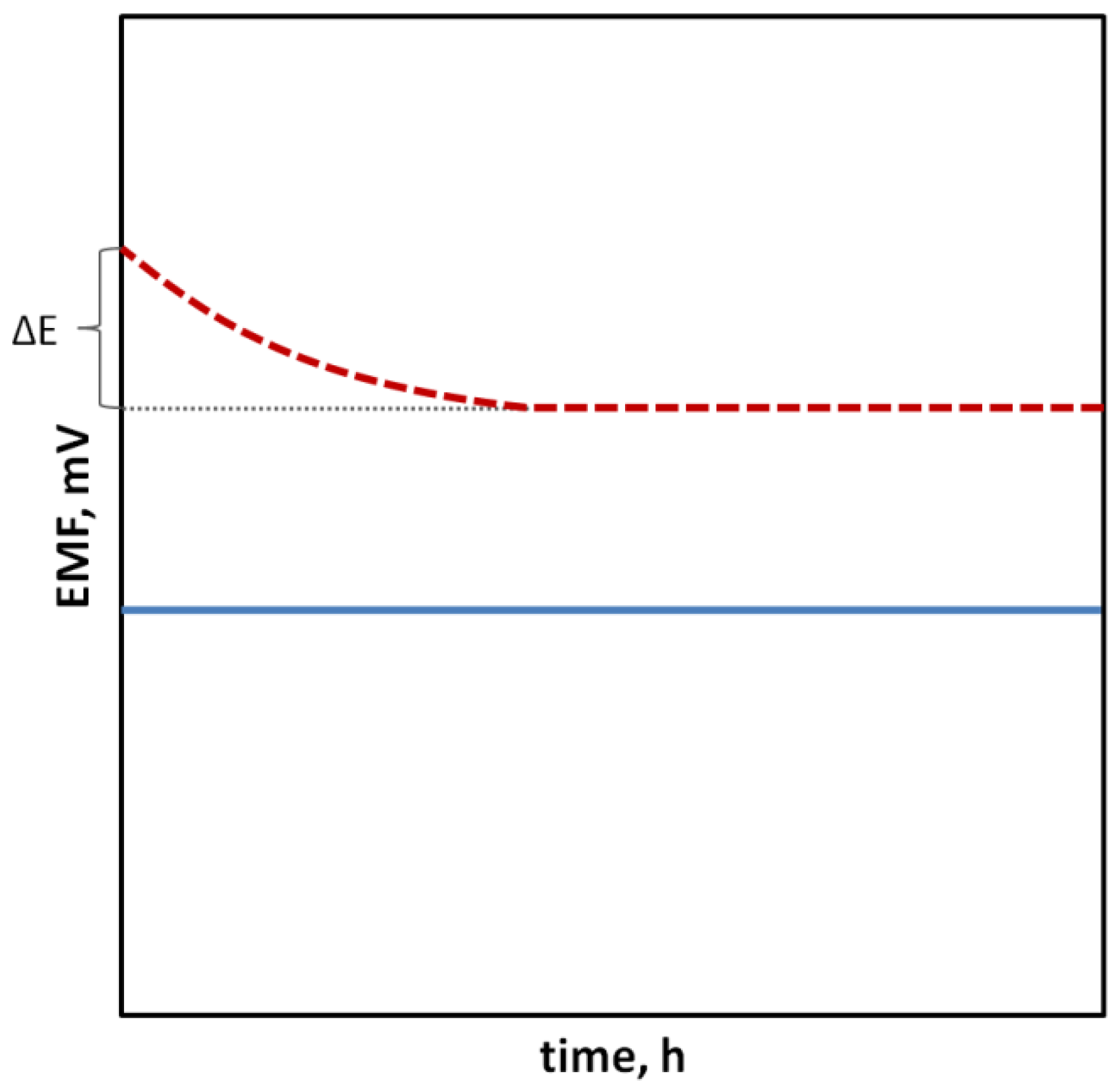
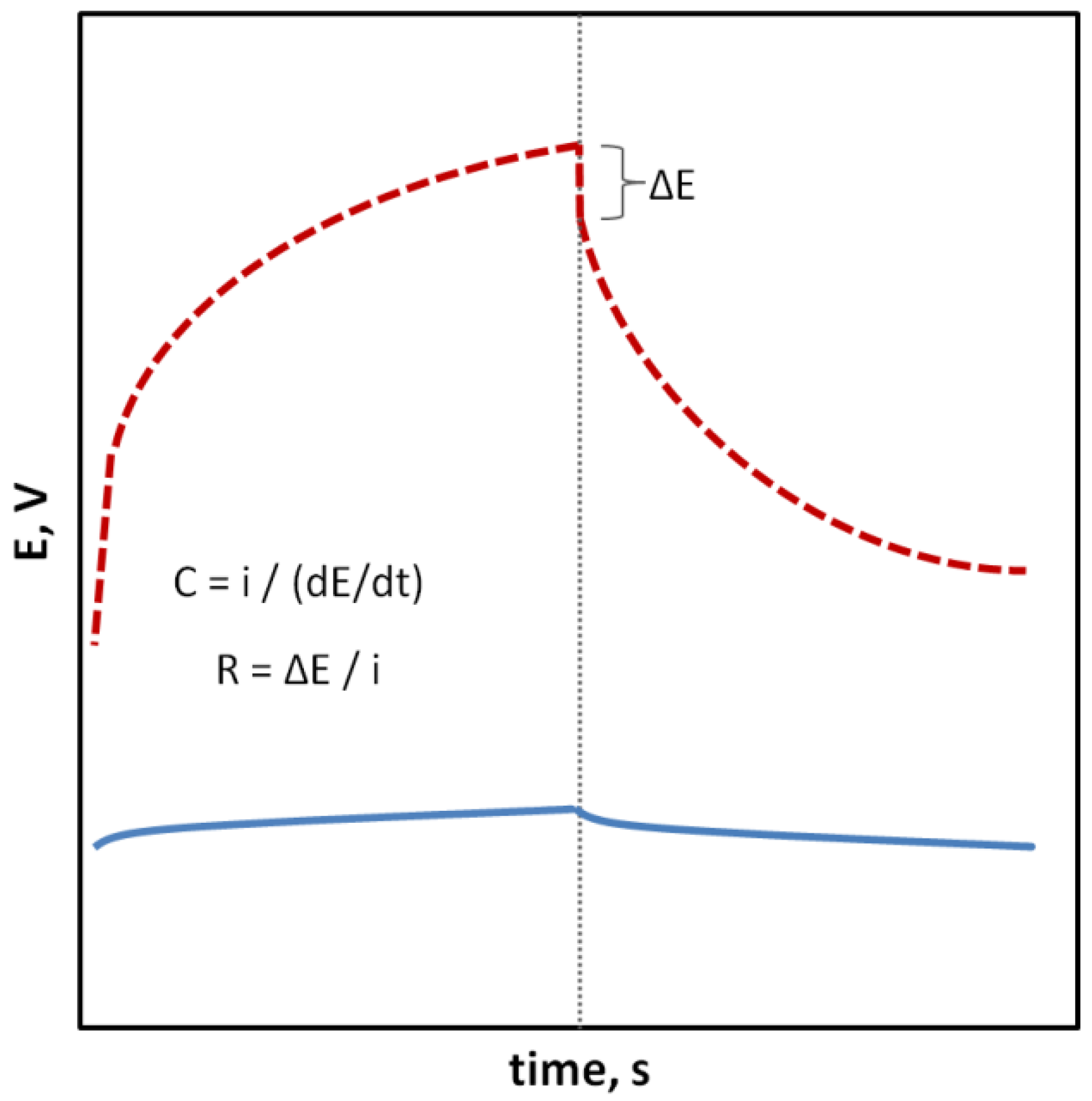
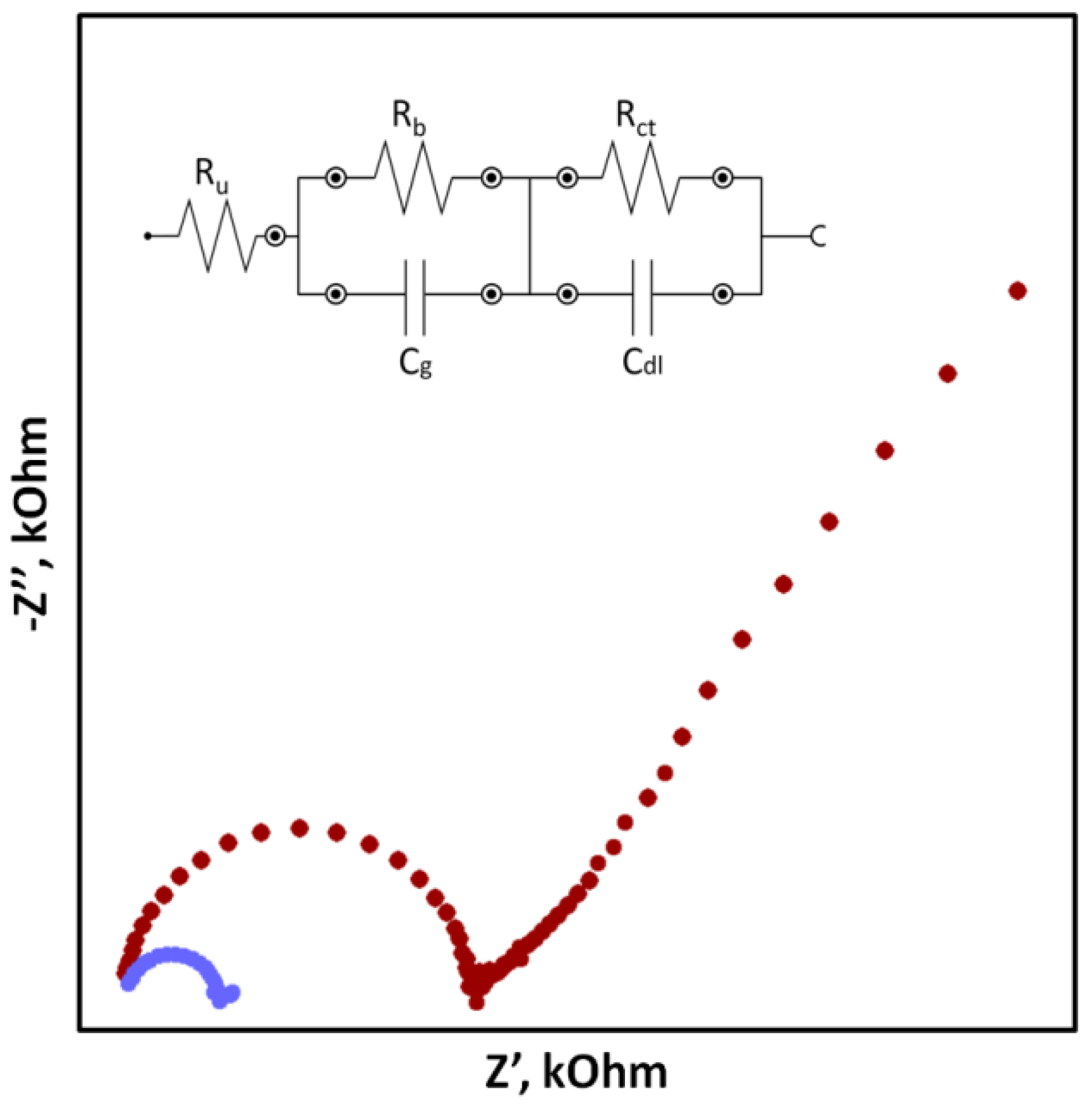
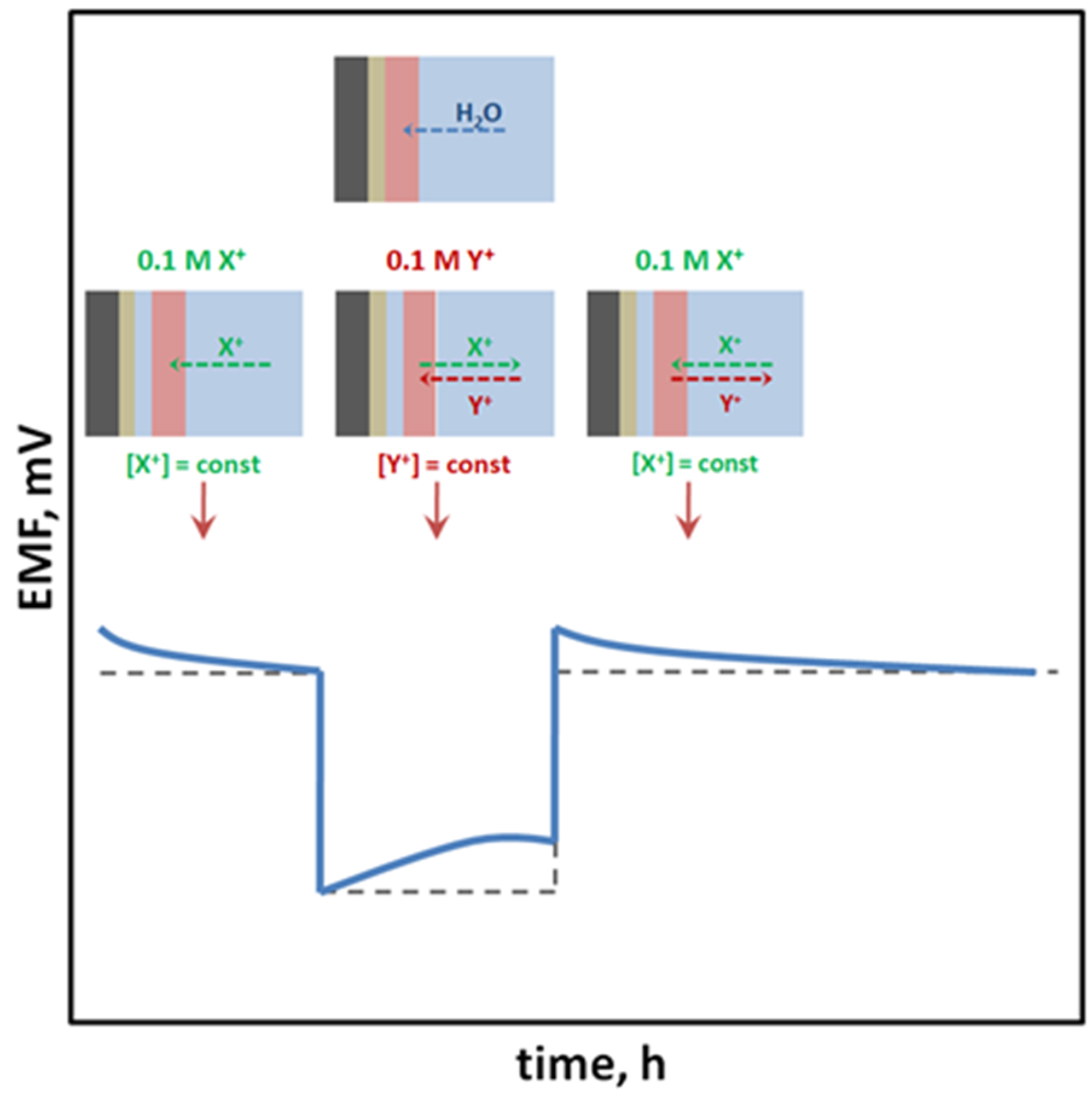
Potentiometry is a highly selective and relatively cheap method that allows achieving low detection limits and a very wide dynamic range of sensors (up to eight orders of units). The principle of the method is to measure the electromotive force (EMF) of a cell made of two types of electrodes: a reference electrode whose potential has to necessarily be constant regardless of the composition and concentration of the sample, and an indicator (working) electrode whose potential changes depending on the activity of the main ion present in the sample solution to which the ion-selective membrane is sensitive.
- ion-selective electrodes
- potentiometry
- solid contact
- composite materials
1. Introduction
2. Potentiometry and Ion-Selective Electrodes
This entry is adapted from the peer-reviewed paper 10.3390/s23135839
References
- Lindner, E.; Gyurcsányi, R.E. Quality Control Criteria for Solid-Contact, Solvent Polymeric Membrane Ion-Selective Electrodes. J. Solid State Electrochem. 2009, 13, 51–68.
- Bieg, C.; Fuchsberger, K.; Stelzle, M. Introduction to Polymer-Based Solid-Contact Ion-Selective Electrodes—Basic Concepts, Practical Considerations, and Current Research Topics. Anal. Bioanal. Chem. 2017, 409, 45–61.
- Roy, S.; David-Pur, M.; Hanein, Y. Carbon Nanotube-Based Ion Selective Sensors for Wearable Applications. ACS Appl. Mater. Interfaces 2017, 9, 35169–35177.
- Parrilla, M.; Cuartero, M.; Crespo, G.A. Wearable Potentiometric Ion Sensors. Trends Anal. Chem. 2019, 110, 303–320.
- Mousavi, Z.; Granholm, K.; Sokalski, T.; Lewenstam, A. All-Solid-State Electrochemical Platform for Potentiometric Measurements. Sens. Actuators B Chem. 2015, 207, 895–899.
- Ozer, T. Low-Cost Pencil-Graphite Multi-Electrodes for Simultaneous Detection of Iron and Copper. J. Turk. Chem. Soc. Sect. A Chem. 2022, 9, 1–12.
- Cuartero, M.; Ruiz, A.; Galián, M.; Ortuño, J.A. Potentiometric Electronic Tongue for Quantitative Ion Analysis in Natural Mineral Waters. Sensors 2022, 22, 6204.
- Chango, G.; Palacio, E.; Cerdà, V. Potentiometric Chip-Based Multipumping Flow System for the Simultaneous Determination of Fluoride, Chloride, PH, and Redox Potential in Water Samples. Talanta 2018, 186, 554–560.
- Zuliani, C.; Diamond, D. Opportunities and Challenges of Using Ion-Selective Electrodes in Environmental Monitoring and Wearable Sensors. Electrochim. Acta 2012, 84, 29–34.
- Chaneam, S.; Taweetong, W.; Kaewyai, K.; Thienwong, P.; Takaew, A.; Chaisuksant, R. Fabrication of a Nitrate Selective Electrode for Determination of Nitrate in Fertilizers by Using Flow Injection Analysis System. Procedia Chem. 2016, 20, 73–75.
- Lin, P.K.T.; Araujo, A.N.; Montenegro, M.C.B.S.M.; Pérez-Olmos, R. New PVC Nitrate-Selective Electrode: Application to Vegetables and Mineral Waters. J. Agric. Food Chem. 2005, 53, 211–215.
- Fan, Y.; Xu, C.; Wang, R.; Hu, G.; Miao, J.; Hai, K.; Lin, C. Determination of Copper(II) Ion in Food Using an Ionic Liquids-Carbon Nanotubes-Based Ion-Selective Electrode. J. Food Compos. Anal. 2017, 62, 63–68.
- Chapman, B.R.; Goldsmith, I.R. Determination of Chloride, Sodium and Potassium in Salted Foodstuffs Using Ion-Selective Electrodes and the Dry Sample Addition Method. Analyst 1982, 107, 1014–1018.
- Abd El-Rahman, M.K.; Salem, M.Y. Ion Selective Electrode (in-Line Analyzer) versus UV-Spectroscopy (at-Line Analyzer); Which Strategy Offers More Opportunities for Real Time Monitoring of the Degradation Kinetics of Pyridostigmine Bromide. Sens. Actuators B Chem. 2015, 220, 255–262.
- Numnuam, A.; Chumbimuni-Torres, K.Y.; Xiang, Y.; Bash, R.; Thavarungkul, P.; Kanatharana, P.; Pretsch, E.; Wang, J.; Bakker, E. Potentiometric Detection of DNA Hybridization. J. Am. Chem. Soc. 2008, 130, 410–411.
- Koncki, R. Recent Developments in Potentiometric Biosensors for Biomedical Analysis. Anal. Chim. Acta 2007, 599, 7–15.
- van de Velde, L.; d’Angremont, E.; Olthuis, W. Solid Contact Potassium Selective Electrodes for Biomedical Applications—A Review. Talanta 2016, 160, 56–65.
- Morris, D.; Coyle, S.; Wu, Y.; Lau, K.T.; Wallace, G.; Diamond, D. Bio-Sensing Textile Based Patch with Integrated Optical Detection System for Sweat Monitoring. Sens. Actuators B Chem. 2009, 139, 231–236.
- Lewenstam, A. Chapter 1 Clinical Analysis of Blood Gases and Electrolytes by Ion-Selective Sensors. Compr. Anal. Chem. 2007, 49, 5–24.
- Cuartero, M.; Bakker, E. Environmental Water Analysis with Membrane Electrodes. Curr. Opin. Electrochem. 2017, 3, 97–105.
- Slaveykova, V.I.; Wilkinson, K.J.; Ceresa, A.; Pretsch, E. Role of Fulvic Acid on Lead Bioaccumulation by Chlorella Kesslerii. Environ. Sci. Technol. 2003, 37, 1114–1121.
- De Marco, R.; Clarke, G.; Pejcic, B. Ion-Selective Electrode Potentiometry in Environmental Analysis. Electroanalysis 2007, 19, 1987–2001.
- Crespo, G.A. Recent Advances in Ion-Selective Membrane Electrodes for in Situ Environmental Water Analysis. Electrochim. Acta 2017, 245, 1023–1034.
- Lindner, E.; Pendley, B.D. A Tutorial on the Application of Ion-Selective Electrode Potentiometry: An Analytical Method with Unique Qualities, Unexplored Opportunities and Potential Pitfalls; Tutorial. Anal. Chim. Acta 2013, 762, 1–13.
- Düzgün, A.; Zelada-Guillén, G.A.; Crespo, G.A.; Macho, S.; Riu, J.; Rius, F.X. Nanostructured Materials in Potentiometry. Anal. Bioanal. Chem. 2011, 399, 171–181.
- Bakker, E.; Bühlmann, P.; Pretsch, E. Polymer Membrane Ion-Selective Electrodes-What Are the Limits? Electroanalysis 1999, 11, 915–933.
- Bobacka, J.; Ivaska, A.; Lewenstam, A. Potentiometric Ion Sensors. Chem. Rev. 2008, 108, 329–351.
- Cuartero, M.; Crespo, G.A. All-Solid-State Potentiometric Sensors: A New Wave for in Situ Aquatic Research. Curr. Opin. Electrochem. 2018, 10, 98–106.
- Bobacka, J. Potential Stability of All-Solid-State Ion-Selective Electrodes Using Conducting Polymers as Ion-to-Electron Transducers. Anal. Chem. 1999, 71, 4932–4937.
- Tóth, K.; Gráf, E.; Horvai, G.; Pungor, E.; Buck, R.P. Plasticized Poly(Vinyl Chloride) Properties and Characteristics of Valinomycin Electrodes. 2. Low-Frequency, Surface-Rate, and Warburg Impedance Characteristics. Anal. Chem. 1986, 58, 2741–2744.
- Suni, I.I. Impedance Methods for Electrochemical Sensors Using Nanomaterials. Trends Anal. Chem. 2008, 27, 604–611.
- Radu, A.; Anastasova-Ivanova, S.; Paczosa-Bator, B.; Danielewski, M.; Bobacka, J.; Lewenstam, A.; Diamond, D. Diagnostic of Functionality of Polymer Membrane—Based Ion Selective Electrodes by Impedance Spectroscopy. Anal. Methods 2010, 2, 1490–1498.
- Fibbioli, M.; Morf, W.E.; Badertscher, M.; De Rooij, N.F.; Pretsch, E. Potential Drifts of Solid-Contacted Ion-Selective Electrodes Due to Zero-Current Ion Fluxes through the Sensor Membrane. Electroanalysis 2000, 12, 1286–1292.
