1. Influence of Heating Mode
Early experiments verified that the cracking reaction process of kerogen is controlled by the reaction temperature and time, and that the time and temperature have complementary effects when the temperature exceeds the critical temperature of the reaction [
8,
9]. Under geological conditions, the temperature of kerogen cracking to generate oil and gas is generally 50–150 °C [
65,
66], but this kerogen cracking process generally lasts for thousands or several millions of years, which is evidently too long for industrial production. In order to ensure that the hydrocarbon generation reaction of kerogen can occur in large quantities in a short period of time, a higher reaction temperature is generally maintained in in situ conversion engineering to reduce the reaction time. Therefore, when a large amount of hydrocarbon is generated from kerogen, it cannot be determined solely via temperature, and the kinetic parameters and heating rate need to be considered. Many semiopen, open, and closed kerogen cracking simulation experiments have proven that higher temperatures can greatly shorten the reaction time of kerogen cracking [
7,
16,
67,
68,
69]. However, too high of a reaction temperature not only unnecessarily increases energy consumption, but also leads to the cracking of large amounts of liquid hydrocarbons into gases, which reduces the economic benefits. Therefore, an appropriate heating temperature or heating mode can be determined to obtain the highest hydrocarbon (HC) yield with the minimal energy consumption.
By obtaining the kinetic parameters of hydrocarbon generation, Hou et al. [
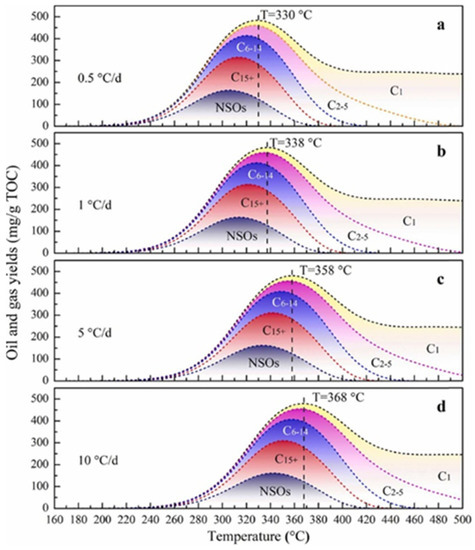
7] simulated the in situ conversion process of organic-rich shale at heating rates of 0.5, 1, 5, and 10 °C/d, and found that the temperatures at which Chang7 kerogen produced the maximal HC yield were 330, 338, 358, and 368 °C, respectively (
Figure 8). Meanwhile, when the temperature exceeds the temperature at maximal HC yield, the HC yield generally exhibits a downward trend, but the gas/oil ratio (GOR) increasingly grows. Song et al. studied the organic-matter evolution of lacustrine shale in the Qingshankou Formation of the Songliao Basin and the Chang 7 Member of the Ordos Basin using a semiclosed heating system under a constant temperature [
70]. Considering the quality and quantity of the produced oil, the conversion degree of kerogen, and the efficiency of hydrocarbon expulsion, the optimal temperature ranges of the Chang7 shale and Qingshankou shale are 400–450 and 350–425 °C during oil shale in situ conversion, respectively [
70]. This indicates that the pyrolytic processes of different shales are distinct. Alhesan et al. conducted isothermal pyrolysis experiments with E
l Lajjun shale at different temperatures and times in a closed system, and good oil production could be achieved at 280 °C for 14 days [
67]. However, when the temperature was lower than 300 °C, the sulfur content of the obtained product was high (12 wt % of oil), but it could be significantly reduced to approximately 7 wt % when the temperature was 300–320 °C [
67]. Ma et al. used experiments to simulate the effects of different heating temperatures, heating rates, and constant temperature times on the quality and quantity of the produced oil. With an increasing heating rate, the total oil and gas yield gradually decreased, and the relative content of the saturated hydrocarbon component decreased [
71]. This may have been because kerogen in organic-rich shale does not have enough time to participate in the reaction under a high heating rate. At a constant temperature of 350 °C with increasing constant temperature time, the fuller the pyrolysis of oil shale organic matter is, the higher the oil and gas yield is, and the saturated hydrocarbon component of the produced oil increases slightly [
69]. Moreover, experiments showed that, with increasing constant temperature time, small pores gradually develop into relatively large pores, and the permeability of the shale increases [
72].
Figure 8. Calculated oil and gas yields during ICP of the Chang 7 shale at different heating rates [
7]. (
a–
d) Calculated oil and gas yields at heating rates of 0.5, 1, 5, and 10 °C/d, respectively.
Therefore, during in situ shale conversion, it is necessary to fully consider the conditions of the target formation and set up a reasonable heating mode to improve the energy consumption ratio. For instance, at the low-temperature stage, the temperature can be rapidly raised to make the kerogen reach the cracking temperature fast, avoiding excessive heat conduction and heat dissipation; at the medium-temperature stage, the temperature should be raised slowly to ensure that the kerogen and NSO compounds can be fully cracked and reduce the content of C6–14 cracking, which can increase the quality and yield of the shale oil. Meanwhile, this heating mode can, to some extent, alleviate the phenomenon of an uneven pyrolysis reaction process caused by the uneven heating of the target layer.
2. Influence of Formation Conditions
Previous simulation experiments found that Types I and IIa (a subtype of Type II) kerogen are more inclined to generate oil, Types IIb (a subtypes of Type II) and III kerogen are more inclined to generate gas, and the main oil generation period of different kerogen types is 0.7–1.35 Easy%Ro [
12,
73]. When the maturity of the kerogen is greater than 1.35 Easy%Ro, its oil generation potential is basically exhausted. Moreover, there are discrepancies in the kinetic parameters between different kerogen types, so the evolutionary process during in situ conversion is also different [
7,
73]. The activation energy of Types Ⅱ and Ⅲ kerogens covers a wide distribution range, while Type Ⅰ a narrow one, and the average activation energy of kerogens follows the order of Type Ⅲ > Type Ⅱ > Type Ⅰ. This shows that the potential of kerogen cracking to generate oil and gas is controlled by the maturity and kerogen type. A hydrocarbon generation simulation experiment found that the presence of some minerals could catalyze the kerogen cracking reaction. For example, carbonate (dolomite) promotes oil generation to a certain extent, but inhibits gas generation, while for silicate (feldspar, quartz, and clay), this is the opposite [
21,
74]. Pyrite is generally beneficial to oil and gas generation [
21], and some findings suggest that the rate of petroleum formation depends critically on the concentration of sulfur radicals generated during the initial stages of thermal maturation [
75]. Moreover, mineralogical composition determines the brittleness index of the shale. The higher the brittleness index of the shale is, the easier fracturing is, which is conducive to the formation of fractures.
The influence of the water content in shale on the energy consumption ratio of in situ conversion mainly has two aspects. On the one hand, water consumes large amounts of heat energy during heating and vaporization due to its large specific heat capacity [
20]. On the other hand, in the presence of water, kerogen in shale can not only undergo thermal cracking reactions, but also chemical reactions with high-temperature water in the gas or liquid phase due to the high temperature [
39,
76]. In hydrothermal cracking reactions, high-temperature water may participate in the reaction as a catalyst, reactant, and/or solvent. In addition, water can provide H
+ and OH
− as reactants that can improve the oil and gas yield of organic-rich shale in situ conversion [
39,
71,
76].
As mentioned above, when the energy consumption of heating shale is less than the energy of producing oil, project and other costs need to be diluted by the scale of exploitation, which renders it necessary to consider its scale when selecting the target formation. Therefore, shale for the commercial development of underground in situ conversion should meet the following conditions: (1) the TOC value of the shale must be more than 6%, mainly consisting of Types I and IIa organic matter; (2) the Ro value must be 0.5%–1.0%; (3) the thickness must be more than 15 m; (4) the burial depth must be less than 3000 m, and the area more than 50 km
2; (5) the roof and floor of the shale section must be well-sealed; (6) the formation water content must be less than 5% [
1,
2,
6].
3. Distribution Pattern of Wells
Fan et al. observed that heating well spacing significantly impacts shale formation temperature through analog computation [
11,
68]. The larger the spacing of the heating wells is, the earlier and sharper the peak of the oil and gas yield is. This may be because the larger heater spacing allows for the reservoir to heat more evenly, which shortens the time interval of kerogen decomposition [
11,
68]. However, exorbitantly heating the well spacing lengthens the heating time too much to perform industrial production, and renders the kerogen conversion rate relatively low. Huang et al. [
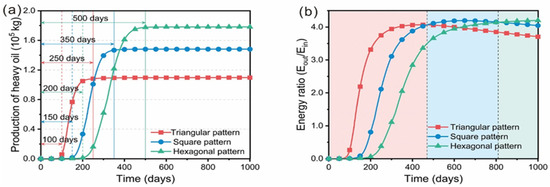
11] considered that, with the same well spacing, if the production period is shorter than 400 days, and a higher heating rate is needed, a more intensive heating well pattern, such as a triangular well pattern, should be selected. If the energy consumption ratio is given priority, and the heating period is longer than 800 days, then the hexagonal well pattern is better (
Figure 9). In the process of in situ shale conversion, the energy consumption ratio of horizontal well heating is significantly higher than that of vertical well heating because horizontal well heating can significantly reduce the consumption of heat energy in the surrounding rock. Lu et al. calculated with a simulation that, when the well spacing of horizontal wells is half the thickness of the shale formation, the energy consumption ratio is the highest, and smaller and larger well spacing leads to a reduction in the energy consumption ratio [
77]. When the well spacing is significantly greater than the thickness of the shale half layer, the surrounding rock also reaches a higher temperature during the heating process, which causes a considerable amount of heat energy to be transferred into the surrounding rock. However, under the condition of small well spacing, when the temperature near the production well reaches the effective cracking temperature, the temperature at the edge of the shale is still low, and the organic matter in the shale does not start to effectively crack, which reduces the oil and gas yield. Therefore, the well spacing should be determined on the basis of the thickness of the target formation. Meanwhile, small well spacing and horizontal wells mean increasing drilling costs and reducing the final benefits after considering the engineering operation costs. Ground conditions are also an important reference indicator for the distribution pattern of wells, and horizontal wells can be considered for areas that are not suitable for construction. In summary, the combination of horizontal well and multiwell methods can slightly reduce the heating cost and significantly reduce the time cost, but this increases the drilling cost. Therefore, it is necessary to comprehensively consider the heating and time costs, ground conditions, and engineering cost in the future, and conduct overall quantitative optimization to determine the distribution pattern of the wells.
Figure 9. (
a) Cumulative oil production and the production period of different well patterns; (
b) energy ratio and predominance period of different well patterns [
11].
4. Catalytic Agent
The simulation calculation results of the kinetic hydrocarbon generation experiment show that, under other conditions being unchanged, with the reduction in activation energy of the kerogen cracking reaction, lower temperature or shorter heating time was required for the effective pyrolysis of kerogen [
8,
9]. In the process of in situ shale conversion, this principle is the same. In industrial production, adding a catalyst is an effective technology to reduce the reaction activation energy. Moreover, the catalyst can accelerate the breaking of long-chain molecules, and convert solid or heavy organic matter into light hydrocarbons, which improves the fluidity and quality of oil [
78,
79,
80,
81,
82]. Therefore, adding a catalyst is one of the efficiency enhancement methods of in situ shale conversion.
Song et al. used natural clay, attapulgite, and 3-mercaptopropyltrimethoxysilane (MPTMS) to optimize the preparation of catalyst SO
3H–APG, which can reduce the pyrolytic temperature and activation energy of shale, improve the hydrocarbon yield, and reduce the content of oxygen- and nitrogen-containing compounds [
78]. Chang et al. used transition metal salts to catalyze the pyrolysis of shale. The results showed that both CoCl
2 and NiCl
2 could promote the pyrolysis of organic-rich shale and the secondary pyrolysis of generated oil [
82]. Yu et al. proposed a clin/SBA-15 catalyst prepared with clinoplite and conducted indepth research on the catalytic behavior of oil shale in situ conversion. The results showed that the saturated hydrocarbon content of the oil produced by the clin/SBA-15 catalyst increased, the H/C ratio increased, and the O/C ratio decreased [
83]. In addition, Clin/SBA-15 did not exhibit activity attenuation in the seven-day shale pyrolysis test [
83]. Sun et al. found that the addition of CrCl
3 reduced the temperature of shale pyrolysis and the activation energy. In addition, the CrCl
3 catalyst reduced the content of oxygen-contained compounds, and increased the content of hydrocarbons and short-chain aliphatic hydrocarbons in shale oil [
84]. Furthermore, many catalysts, such as liquid acid, organometallic complexes, NiMo, CoMo, CoCl
2 6H
2O, dodecane, diatomite, and zeolite molecular sieves, have been proven in the laboratory to change the quality or yield of generated oil [
78,
79,
80,
81,
82]. As mentioned above, water can provide H
+ and OH
- as reactants in the cracking reaction. Therefore, high-temperature water can be used as a catalyst to improve the hydrocarbon yield and prevent coking during the process of in situ conversion [
39,
56].
Generally, the catalyst should come into contact with the reactants, which requires the catalyst to be widely distributed in shale formations. For some solid catalysts, it is difficult to achieve this goal with existing engineering techniques. Evidently, conductive heating technology and radiant heating technology hinder achieving this goal, but convection heating technology can carry the catalyst through the fluid when the catalyst is easily carried by the fluid. Therefore, when studying catalysts for kerogen hydrocarbon generation reactions, it is necessary to consider the applicability of the catalysts. In addition, an experimental study found that the temperature required for shale pyrolysis decreased when the shale was heated by microwaves, which indicates that microwave heating may accelerate the kerogen cracking reaction [
47,
48].
This entry is adapted from the peer-reviewed paper 10.3390/en16114386