Regarding the pathogenetic mechanisms of hypertension in the setting of increased AoStiff, it is currently widely accepted that the increased AoStiff results in a more rapid propagation of the reflected waves from the periphery, which return in phase with cardiac systole, augmenting central SBP (systolic hypertension), increasing the hemodynamic load on the LV (and consequently on the left atrium (LA)), and decreasing the DBP, and consequently the diastolic coronary perfusion (ischemia) [
]. In accordance with the aforementioned points, AoStiff is an independent risk factor for major cardiovascular (CV) events in hypertensive patients.
Hypertension is considered to be the major independent risk factor for AF [
41]. Hypertension, which in the elderly is predominantly due to the increased AoStiff, provokes excessive fibroblast proliferation, increased collagen accumulation, and induces cardiomyocyte apoptosis and inflammation, leading to diffused fibrosis and LV development [
42]. Hypertension, in turn, further increases AoStiff with subsequent systolic and diastolic function loss, resulting in additional heart muscle remodeling. Indeed, a sub-analysis of the MESA (Multiethnic Study of Atherosclerosis) reported that PP is associated with a higher incidence of AF, even after adjusting for LA diameter and LV mass index [
43].
4.3. Ischemic Heart Disease
AoStiff is independently associated with the progression and severity of coronary atherosclerosis [
46]. In a Korean population, a CAVI >8 predicted obstructive IHD [
47], whereas in a recent study that included individuals referred for coronary artery calcium (CAC) scoring and Coronary Computed Tomography Angiography (CCTA), the degree of CAC and severe coronary stenosis demonstrated significant correlation with CAVI [
48]. It is noteworthy that in patients with stable angina and non-obstructive IHD, a higher aortic PWV is associated with stress-induced myocardial ischemia, and assessment of aortic stiffness was recommended for the diagnostic evaluation of this patient population [
49].
The mechanisms underlying the link between AoStiff and IHD have not been delineated. Accepted explanations of the association between increased AoStif and IHD include [
50,
51]: (A) Increased AoStiff disturbs the oxygen demand–supply balance, causing myocardial ischemia. The LV overload due to AoStiff induced an increase in SBP and PP, resulting in LV hypertrophy and, consequently, increased oxygen demand. Further, the AoStiff-induced LV decrease in DBP is closely linked with impaired coronary perfusion. (B) AoStiff increases with age in parallel with the prevalence of traditional risk factors for IHD (e.g., hypertension, diabetes mellitus, dyslipidemia, and smoking) which also increases with age. (C) The systolic hypertension, which results from increased AoStiff, is tightly linked to endothelial dysfunction, one of the first and most important causes of the processes leading to atherosclerosis and IHD [
52]. (D) Atherosclerosis underlying both IHD and increased AoStiff is characterized by the retention of lipids and inflammatory cells such as macrophages, T lymphocytes, and mast cells in the damaged arterial wall [
53].
4.4. Heart Failure
HF has been attributed to the interaction between cardiovascular aging and specific risk factors, comorbidities, and disease modifiers [4,62]. Wave reflections in the rigid aorta of the elderly tend to increase mid-to-late systolic LV load and myocardial wall stress [63]. When the LV pump function is preserved, the reflected wave induces a late systolic pressure peak in the pressure waveform, augmenting aortic pressure in mid-to-late systole. Conversely, when the LV pump function is depressed, wave reflection may exert more pronounced effects to decrease flow, with no apparent alteration in the appearance of the pressure waveform. In patients with severe LV systolic dysfunction, wave reflections truncate flow, reduce stroke volume and shorten the duration of LV ejection. In addition, forward waves may also be altered in patients with severely depressed LV function, as indicated by the decrease in the ratio of the first to second systolic peak, compared to individuals with preserved LV function [64].
5. Chronic Kidney Disease
The kidneys are adversely affected by the increased AoStiff because they are high-flow, low resistance organs receiving approximately 20% of the resting cardiac output. Due to the anatomy of renal microcirculation with small number of branches between the aorta and the glomeruli, the main protective mechanism against increased transmission of pulsatile pressure to the glomerulus is the myogenic response of the afferent arteriole, which can normally autoregulate renal blood flow within a wide range of preglomerular perfusion pressures, in co-ordination with the tubulo-glomerular feedback mechanism [
67]. In a rigid aorta with lost “windkessel effect” a greater percentage of pulsatile power in the arterial pressure and flow waveforms penetrates deeper into the renal microcirculation, where it may cause microvascular damage in the preglomerular vessels [
68]. In people with compromised renal blood flow autoregulation (i.e., in the elderly or patients with diabetes mellitus) [
69,
70], this arterial stiffness-related pressure pulsatility is also transmitted to a greater extent in the glomerular capillary. As such, it can be a major contributor to glomerular hypertension and hyperfiltration, and the development of typical albuminuric chronic kidney disease [
71].
The association between AoStiff and renal dysfunction has been repeatedly demonstrated. Out of the 3666 participants (mean age = 65 years old; 58% women) from the Rotterdam Study, 601 participants with incident CKD were recognized during a median follow-up of 11 years [
72]. In the model adjusted for age, sex, mean arterial pressure, heart rate, and baseline GFR, each standard deviation (SD) higher PP was associated with 0.15-mL/min per 1.73 m
2 steeper annual eGFR decline (95% confidence interval [95% CI], 0.10 to 0.20) and 11% higher risk of incident CKD (95% CI, 1.05 to 1.18), whereas each SD higher PWV was associated with 7% higher risk of incident CKD (95% CI, 1.00 to 1.14). Similar findings of a prospective study [
73] including individuals aged ≥40 years without overt kidney disease reported that PWV, age, and eGFR were independent predictors of renal function decline. In the same study, baseline eGFR did not determine the annual change in PWV, suggesting a unidirectional association between arterial AoStiff and eGFR.
6. Anemia
Anemia is quite commonly diagnosed in older adults and is an important indicator of several reactive and clonal conditions. In a retrospective study of more than 19,000 hospital patients, the incidence of anemia increased from 15% at the ages of 64–69 to 37% in those over aged 90 [
84]. The etiology of anemia in the elderly is multifactorial and ranges from bone marrow failure syndromes to CKD and HF, and from nutritional deficiencies to inflammatory processes, including inflammaging in immunosenescence. In several cases, though, no clear-cut etiology is identified. These patients are referred to as unexplained anemia in the elderly (UAE) [
85].
The mechanisms linking AoStiff with UAE are diverse and complex. There is evidence to suggest that AoStiff is inversely associated with red blood cell volume [
86] and erythropoietin (EPO) levels [
87] in healthy individuals. It is reasonable, therefore, to assume that AoStiff is a contributing factor to the typical low EPO concentration observed in UAE, impairing the endocrine feedback pathways governing the basal regulation of kidney EPO synthesis, as well as renal perfusion. In this regard, it is plausible that an increase in AoStiff may lead to baroreceptor dysfunction, hindering the release of blood volume-regulating hormones that directly stimulate EPO synthesis [
88]. Further, increased AoStiff is strongly associated with a reduction in renal perfusion contributing to impaired hormone delivery to EPO-producing cells. Admittedly, however, the potential impact of AoStiff on UAE etiology deserves further investigation.
7. Ischemic Stroke
The association between AoStiff and ischemic stroke has been attributed to several mechanisms which are not mutually exclusive. Hemodynamic alterations secondary to AoStiff may come into play. Raised PP promotes arterial remodeling, increases arterial wall thickness, and induces the development of atherosclerotic plaques, which may rupture or ulcerate leading to intravascular thrombosis [
94]. Further, the rigid aorta facilitates the penetration of excessive pulsatile energy into the cerebral microcirculation, resulting in its damage [
95]. Undoubtedly, however, the fact that the measured high aortic AoStiff may reflect parallel structural changes in the intracerebral vasculature, such as breaking of elastic fibers, fibrosis, calcifications, medial smooth muscle necrosis, and diffusion of macromolecules into the arterial wall, cannot be excluded [
96]. Finally, the classical cardiovascular risk factors and diseases (e.g., hypertension, atherosclerosis, coronary artery disease, and CKD), which are promoted by AoStiff, are also risk factors for ischemic stroke [
97].
8. Dementia
Neurodegenerative dementias such as Alzheimer’s disease are the most frequent, followed by microangiopathies such as vascular dementia (VaD) and Lewy body dementia (LBD), with mixed pathology often seen.
Dementia is a syndrome defined by the deterioration of cognitive function beyond that expected as a result of biological aging, which presents as a disturbance of multiple higher cortical functions including learning and memory, complex attention, executive function, language, motor perception, and social cognition, affecting the patient’s ability to perform daily activities independently [
98]. Dementia is an umbrella term for a range of conditions characterized by progressive cognitive impairment and behavioral changes that interact with daily functioning [
99,
100].
Different diagnostic criteria have been proposed for VaD, all consisting of the same three core elements: established (1) acquired cognitive impairment, (2) vascular damage in the brain, and (3) a causal association between the two [
101]. Factors characterizing subtypes of VaD encompass the nature and extent of vascular pathologies, the degree of involvement of extra and intracranial vessels, and the anatomical location of tissue changes, as well as the time after the initial vascular event. Cerebral small vessel disease (CSVD), characterized by arteriolosclerosis, lacunar infarcts and cortical and subcortical microinfarcts, and diffuse white matter changes, which involve myelin loss and axonal abnormalities, has gained prominence as an important substrate of cognitive impairment [
102].
The brain, like the kidney, is a high-flow organ receiving approximately 20% of resting cardiac output [
68]. Increased AoStiff exposes the small vessels in the brain to abnormal flow pulsations and, as such, may contribute to the pathogenesis of CSVD and cognitive impairment [
103].
9. Frailty
Frailty is defined as increased vulnerability to poor resolution of homoeostasis after a stressor event (i.e., infection, surgery, or new medical treatment), which increases the risk of adverse outcomes (i.e., death or hospitalization), and it may present as unexplained weight loss, severe fatigue, falls, delirium, or fluctuating disability (i.e., “good and bad days”) [
110]. Its prevalence increases with ageing and has been reported to range between 12–24% among people aged ≥50 across 62 countries [
111]. Frailty and CVD share common risk factors, namely hypertension, diabetes mellitus, and obesity [
112]. The potential common underlying pathophysiologic mechanism is inflammation (“inflammageing”) [
112,
113]. Arterial factors, especially AoStiff, have been suggested to be a potential linking mechanism between frailty and CVD. Frailty has been associated with an increased risk of unfavorable outcomes in patients with peripheral artery disease [
114], valvular disease [
115,
116], as well as in adults with [
117] and without coronary artery disease [
118]. A recent systematic review and meta-analysis of five cross-sectional studies and 7575 participants, aged above 60 years, revealed an association between frailty (defined as a clinical syndrome in which three or more of the following criteria were present: (a) unintentional weight loss (10 lbs in past year), (b) self-reported exhaustion, (c) weakness, (d) slow walking speed, and (e) low physical activity or defined as Simple Frailty score = 2) and prefrailty (defined as Simple Frailty score = 1) and greater AoStiff, compared to non-frail patients [
119].
10. Determinants of Multimorbidity Pattern in the Elderly
1. Sex and ethnicity. Women are more often affected by multimorbidity than men. In the Rotterdam study (n = 6094, median years of follow-up 9.2) two-thirds of people over 45 developed multimorbidity in their remaining lifetime, with women manifesting nearly double the risk of multimorbidity compared to men [126]. MicroRNAs (miRNAs) are small, non-coding endogenous RNA molecules that regulate gene expression either by mRNA cleavage/destabilization or inhibition of translation. miR-181a, which acts as an inflamma-miRNA playing important roles in the aging process, has been identified as multimorbidity-associated miRNA [127].
2. Common pathogenic morbidity routes. In aging populations many patients have multiple diseases characterized by acceleration of the normal aging process. Common signaling pathways and cellular events have been identified in the pathogenesis of increased AoStiff, CVD, CKD, and neurodegenerative disease. These include cellular senescence with telomere shortening, activation of PI3K–AKT–mTOR signaling, impaired autophagy, mitochondrial dysfunction, stem cell exhaustion, epigenetic changes, abnormal microRNA profiles, immunosenescence, and low-grade chronic inflammation (“inflammaging”). Many of these pathways are driven by chronic oxidative stress. There is also a reduction in anti-aging molecules, such as sirtuins and Klotho, which further accelerates the aging process [
130,
131,
132].
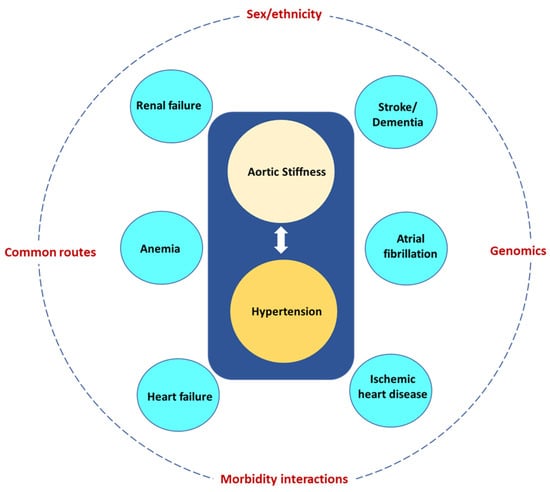
3. Morbidity interactions. Increased AoStiff and the tightly linked hypertension give rise to morbidities, which in turn lead to other morbidities and further increase AoStiff. A common scenario is illustrated in
Figure 6. Increased AoStiff causes hypertension, which in turn further increases AoStiff and may give rise to HF and renal failure, further increasing AoStiff and the severity of hypertension and leading to a vicious cycle. To complicate things further, hypertension is a powerful risk factor for AF, IHD, stroke, and dementia, whereas both renal failure and HF are powerful risk factors for the development of anemia. The above considerations are in accordance with a prospective analysis using the data from the Survey of Health, Ageing and Retirement in Europe in both 2013 and 2015 [
133].
Figure 6. Aortic stiffness and the tightly linked hypertension give rise to several morbidities (blue circles) most of them being powerful risk factors for other morbidities. The eventual multimorbidity pattern depends on modifiers, such as sex and ethnicity, common routes, morbidity interactions, and genomics.
4. Genomics. Genetic data can be used to identify genetic overlaps between multiple diseases, including those that might not be known to have shared genetic pathways [
136]. Using the results of published GWAS (genome wide associated studies), Melzer and colleagues uncovered 22 genetic variants that were linked with multiple age-related diseases, 12 of which were linked and mostly inherited together [
137]. One of these twelve genetic variants was found in apolipoprotein E (APOE) and was linked with Alzheimer’s and coronary artery disease, whereas two other DNA variants were located in the genes LPA (lipoprotein a) and LDLR (low-density lipoprotein receptor) and were linked to altered blood lipids and cardiovascular traits. The remaining nine associations between genetic variants that tended to be inherited together and age-associated diseases included links to three or more of the diseases, including Alzheimer’s disease, stroke, coronary artery disease, type 2 diabetes, kidney disease, osteoarthritis, and several cancers. The phenomenon in which a single genetic variant or gene affects multiple biological pathways or diseases is called “pleiotropy”.
11. Conclusions
AoStiff contributes to the development of pathologies frequently affecting the elderly, such as cardiovascular disorders (hypertension, AF, IHD, HF, CKD, anemia, stroke, and dementia). The coexistence of the aforementioned morbidities in groups of two or more (multimorbidity) is very frequent in the elderly. The appearance of specific multimorbidity patterns depends on several factors, including sex, ethnicity, common morbidity pathogenic routes, morbidity interactions, and genomics. Hypertension is probably the most important morbidity, as it further increases AoStiff, is causally related to most of the other pathologies, and is present in most multimorbidity patterns. Prevention and treatment of increased AoStiff may be one of the most important strategies for the management of multimorbidity in the elderly.