Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Gastroenterology & Hepatology
IBS is a widespread functional GI disorder that determines symptoms such as chronic abdominal pain, flatulence, bloating, and altered bowel habits. Depending on diagnostic standards and the regional area, this pathology has a prevalence between 5% and 20% in adults. IBS can occur among patients of any age, even among children, more precisely 13.5% worldwide, and adolescents, rarely manifesting in older patients. IBS has a slightly higher prevalence among women than males and between 18 and 39 years of age.
- irritable bowel syndrome
- low-FODMAP diet
- oligosaccharides
1. Overview
IBS is a widespread functional GI disorder that determines symptoms such as chronic abdominal pain, flatulence, bloating, and altered bowel habits [16,17]. Depending on diagnostic standards and the regional area, this pathology has a prevalence between 5% and 20% in adults [18,19,20]. IBS can occur among patients of any age, even among children, more precisely 13.5% worldwide [21], and adolescents, rarely manifesting in older patients. IBS has a slightly higher prevalence among women than males and between 18 and 39 years of age [18,19].
Although there is currently no specific biomarker for IBS, the diagnosis was established based on clinical history. Until 2006, diagnosing it seemed difficult for doctors because symptoms could change over time, but with the formulation of diagnostic criteria, the work of physicians became easier. Based on Rome IV diagnostic criteria and their most recent revision in 2016, IBS represents recurrent abdominal pain, which occurred weekly three months prior, coupled with a minimum of two of the subsequent criteria: influenced by bowel movements, associated with changes in the frequency and/or appearance. Following that classification, patients are grouped into three categories according to the pattern of the most frequent bowel movements: IBS with constipation (IBS-C), IBS with diarrhoea (IBS-D), IBS with mixed bowel habits (IBS-M), or IBS unclassified (IBS-U) [22].
2. Pathophysiology
The pathophysiology of IBS is very complex and still incompletely understood, as it involves altered enteric neurotransmitters, intestinal microbiota imbalances, neuroendocrine disorders, visceral hypersensitivity, changes in intestinal barrier function, and changes in motility and the response to maladaptive stress response [23,24]. It has been found to be an alteration of bidirectional communication through the brain–intestinal axis caused by an intricate association of biological, psychological, and social variables that underlie the condition. Communication between the brain and the gut is mediated by the autonomic nervous system. A decrease in parasympathetic activity and an increase in sympathetic nervous system activity are frequently observed in patients with IBS. The decrease in vagal tone, which influences peripheral inflammation and permeability, as well as gastrointestinal motility and sensitivity, can be caused by stress [25]. On the contrary, the vagus nerve can indirectly detect the gut microenvironment and transmit this information to the brain [26,27].
Bacterial overgrowth in the small intestine in the majority of patients provides evidence that gut microbiota is at the forefront of the pathophysiology of IBS [23]. Bloating, constipation, diarrhoea, and flatulence are the main symptoms of intestinal bacterial overgrowth. In approximately 25% of patients, the onset of IBS precedes an enteric infection [28]. As a result, post-infectious IBS is a subtype of enteric pathology dominated by diarrhoea, with a high risk of acquisition in women with severe enteritis or after prolonged antibiotic treatment. The intestinal tract presents an increased number of T cells in the lamina propria, intraepithelial lymphocytes, mast cells at the mucosa level and enteroendocrine cells containing serotonin, thus sustaining the development of a pro-inflammatory environment. In chronic inflammation, juxtaposing mast cell mediators with enteric nerves contributes to the visceral hypersensitivity seen in post-infectious IBS [29].
Food is an additional element that contributes to the pathophysiology of IBS [30,31]. Short-chain carbohydrate fermentation reveals the process through which enteric bacteria and the presence of food allergies, nonimmune food sensitivities, changes in gut hormones, and changes in the gut microbiome produce symptoms of IBS. The use of non-steroidal anti-inflammatory drugs (NSAIDs), antibiotics, infections, and stress are known triggers for IBS symptoms. However, [11] ingesting foods high in FODMAPs and foods high in biogenic amines, which produce histamine [10,30], has been associated with the onset of gastrointestinal symptoms in IBS [11,12,29].
However, it has been found that early life experiences (such as dysfunctional family factors and trauma from psychological and physical abuse) are linked to IBS susceptibility. Anxiety and depression influence pain sensitivity, gut motility, immune function, and QoL [27,29,32,33].
3. Diagnosis
The diagnosis of IBS requires the presence of characteristic symptoms within the last 3 months and the appearance 6 months ago. The Bristol stool form scale can help with the problematic subtyping of IBS because it is based on stool form [2,34,35].
The diagnosis of IBS is made after complete anamnesis based on the characteristic symptoms and results of various preliminary laboratory analyses, including complete blood count (CBC), determination of C-reactive protein (CRP), rapid erythrocyte sedimentation rate (ESR), and serological tests for coeliac disease [26,36,37,38].
Faecal lactoferrin (FL) and faecal calprotectin (fCal) are two biomarkers of intestinal inflammation that are useful for diagnosis. Their analysis is superior to serological tests (e.g., ESR and CRP) for differentiating inflammatory bowel disease (IBD) from IBS. Studies showed that measuring fCal in IBS led to a 67% reduction in the number of adults that require a colonoscopy. The determination of fCal in patients less than 45 years old is necessary to rule out IBD.
Although not widely available, rapid testing is available for both FL and fCal. The combination of CRP and fCal tests provides an even greater discrimination of IBS from IBD [39,40].
However, fCal is not a definitive marker for the diagnosis of IBD and may be elevated in obesity, infection, malignancy, or due to certain drugs (e.g., proton pump inhibitors or non-steroidal anti-inflammatory drugs) [36].
The lysozyme, polymorphonuclear neutrophil elastase, neutrophil lipocalin, and myeloperoxidase are other faecal proteins that have been investigated as biomarkers in IBS. Because these were limited studies, their relevance to the diagnosis of IBS is still uncertain [39].
To rule out other symptoms, a digital abdominal and rectal examination is required. This could confirm stool consistency, including rectal impaction, and it can detect dyssynergic defecation (paradoxical contraction on rectal examination during exertion) or low rectal masses [36].
Endoscopy is the ‘golden investigation’ for diagnosis of diseases of the gastrointestinal tract. It allows direct visualisation and offers the possibility of performing biopsies and establishing a histological diagnosis. However, despite those benefits, it is unpleasant to patients and may cause complications. Therefore, simple, non-invasive, and cheap tests to distinguish between intestinal diseases are beneficial [39].
For IBS, it is important to perform colon cancer screening with the help of colonoscopy. Colonoscopy is a frequent test performed to determine whether a disease, such as IBD, microscopic colitis, or colon cancer, is not the cause of a patient’s digestive symptoms. Polyps, haemorrhoids, and diverticula are just some of the lesions identified in patients with IBS during colonoscopy [26,38].
4. Therapeutic and Nutritional Management
Management of IBS includes three directions (Figure 1): pharmacological therapy (antidepressants, antispasmodics, and laxatives), interventions on hygienic-dietary revitalisation [41,42,43], and psychotherapy (cognitive behavioural psychotherapy, dynamic psychotherapy, hypnotherapy, and biofeedback-assisted stress management intervention) [10,26,44].
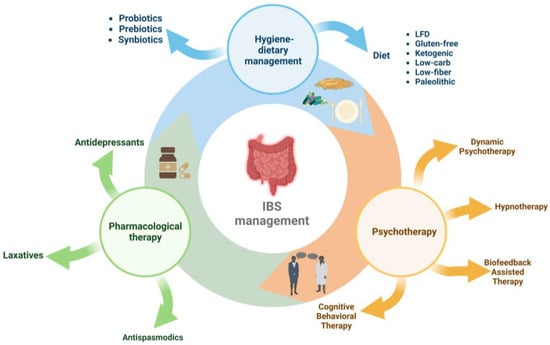
Figure 1. Therapeutic and nutritional management of irritable bowel syndrome.
During the previous ten years, there has been an increase in interest in changing the lifestyle and the hygienic dietary regimen, with patients opting for one of the following diets: LFD, gluten-free diet, low-fibre diet, low-carb diet, ketogenic diet, and palaeolithic diet [22,45,46].
The LFD is a hygienic-dietary intervention option that promotes the intake of foods that include reduced amounts of fermentable oligosaccharides, monosaccharides, and polyols. It improves global symptoms, abdominal pain, bloating, bowel water content, and QoL [47,48,49,50].
This entry is adapted from the peer-reviewed paper 10.3390/nu15102295
This entry is offline, you can click here to edit this entry!
