Ammonia is thought to be a potential alternative for hydrogen storage in the future, allowing for CO2-free energy systems. Ammonia’s beneficial characteristics with regard to hydrogen storage include its high volumetric hydrogen density, low storage pressure, and long-term stability. However, ammonia is characterized by toxicity, flammability, and corrosiveness, making safety a challenge compared to other alternative fuels. In specific circumstances, leakage from ammonia bunkering can cause risks, dispersion, and unsafe areas due to its flammability and toxicity. To avoid dispersion, fire, and explosion hazards on ships, it is crucial to conduct thorough risk analyses
- ammonia
- marine vessels
- risk assessment
1. General Information and Physical Properties of Ammonia
| Properties | Unit | Value |
|---|---|---|
| Energy density | MJ/L | 12.7 |
| Density at standard pressure and temperature | Kg/m3 | 0.769 |
| Latent heat of vaporization | MJ/kg | 188 |
| Vapor pressure at 20 °C | kPa | 858 |
| Heat of vaporization | kJ/kg | 1371 |
| Autoignition temperature | °C | 651 |
| Heat capacity at constant pressure | kJ/mol.°C | 0.037 |
| Minimum ignition energy | mJ | 680 |
| Heat capacity at constant volume | kJ/mol.°C | 0.028 |
| Liquid density | kg/m3 | 600 |
| Adiabatic flame temperature at 1 bar | °C | 1800 |
| Molar mass | g/mol | 17.031 |
| Melting point | °C | −77.7 |
| Condensation pressure at 25 °C | MPa | 0.99 |
| Boiling point at 1 bar | °C | −33.6 |
| Critical temperature | °C | 132.25 |
| Critical pressure | bar | 113 |
| Adiabatic flame temperature | °C | 1800 |
| Flammable range in dry air | % | 15.15 to 27.35 |
| Max. laminar burning | m/s | 0.07 |
| Cetane number | 0 | |
| Octane number | ~130 |
2. Ammonia Bunkering Methods
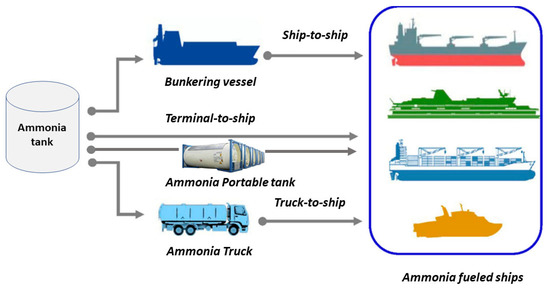
| Bunkering Methods | Advantages | Disadvantages |
|---|---|---|
| STS | Suitable for medium and large ammonia vessels. Quick, saving bunkering time. |
High degree of investment required. Bunkering procedure affected by weather and sea conditions. |
| TTS | Quick ammonia bunkering. Available for all kinds of ammonia vessels, especially large-scale vessels. |
Investment and construction cost of ammonia bunkering terminal is high. Bunkering only available for designated ports. |
| T-TS | Low cost of construction and operation. Suitable for small-scale ammonia bunkering. |
Speed of ammonia bunkering is slow. Limited for large ammonia vessels. |
| APT | Quick ammonia bunkering speed. Flexible bunkering location. Simple process. |
High costs for tank operation and maintenance. Suitable for small-scale ships only. |
3. Hazards of the Ammonia Bunkering Process
3.1. Toxic Effects of Ammonia on Humans
| Ammonia Content (ppm) |
Impacts to Humans |
|---|---|
| 5–10 | Odor-based detection |
| 20–50 | Mild discomfort |
| 50–100 ppm for 2 h | Annoyance and inconvenience |
| 140 ppm for 2 h | Some irritation |
| 200–300 | Effects on throat and eyes |
| 300–500 | Bearable for 20–60 min |
| 2500–5000 | Rapidly fatal (life-threatening around 30 min) |
| 5000–10,000 | Promptly lethal |
3.2. Toxic Effects of Ammonia on the Environment
3.3. Flammability of Ammonia
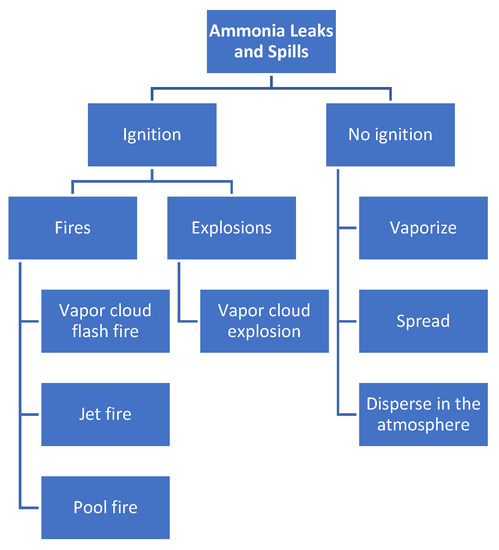
3.4. Corrosiveness
3.5. Other Hazards
3.6. Accidents in Ammonia Bunkering
| Reference | Time | Place | Accident Description | Results |
|---|---|---|---|---|
| Junior et al. [42] | 2007 | Brazil | Ammonia was released from a fish cooperative in Brazil (around 40 kg of AA released). | There were 2 deaths and 18 persons presenting symptoms. |
| Ojha et al. [43] | 1973 | South Africa | An ammonia tank rupture resulted in 38 tons of ammonia leaked. | This accident caused 18 deaths. The ammonia cloud formed by dispersion had a diameter of about 150 m and a depth of 20 m. |
| U.S. CSB [44] | 23 August 2010 | Theodore, Alabama | The catastrophic rupture of a 12-inch pipe caused around 32,000 pounds of ammonia to be released. | The dispersion cloud of ammonia reached about 0.25 miles in size. About 800 contractors at a nearby shipyard were affected. Inside the facility, the ammonia concentration was recorded as about 500 to 600 ppm. |
| Reuters [45] | 31 August 2013 | Shanghai, China | Liquid ammonia leaked from a refrigeration plant. The reason was reported as pipe/valve failure. | There were about 15 deaths and 25 injured. The ammonia dispersion affected residents 15 km downwind from the leakage point. |
| Jain et al. [41] | 7 July 2017 | Elk Grove Village, Illinois | Ammonia leaked from a room (80 × 40 × 20 feet) under a pressure of 150 psi. | About 100 employees were affected. The building was damaged by the explosion. |
This entry is adapted from the peer-reviewed paper 10.3390/en16104019
