] in their study reported that obesity and its associated dysmetabolism, including hyperlipidemia, hyperglycemia and insulin resistance, are strongly correlated with HF [
]. A pro-inflammatory environment characterized by elevated levels of pro-inflammatory cytokines is promoted by obesity and its associated cardiometabolic factors (insulin resistance, dyslipidemia and abdominal adiposity) [
]. The endothelial dysfunction and the nitric oxide unavailability might lead to left ventricular hypertrophy and systolic and diastolic dysfunction in HFpEF [
].
Type II diabetes mellitus (T2DM) is a strongly associated risk factor for HF and other CVD. Patients known to have T2DM present a decreased level of bacterial genera such as
Faecalibacterium,
Bifidobacterium,
Akkermansia,
Bacteroides and
Roseburia. Roseburia,
Bacteroides and
Akkermansia have anti-inflammatory effects.
Bacteroides and
Akkermansia in decreased levels lead to an under expression of tight junctions’ genes, elevated “leaky gut”, and, in consequence, endotoxemia [
89].
5.4. Hypertension
Persistently elevated blood pressure (BP) patients present a higher (up to five-fold)
Bacillota-to-
Bacteroidota ratio in comparison to normotensive controls [
93]. Moreover, the intestinal microbiota is dominated by lactate-producing genera (e.g.,
Turicibacter and
Streptococcus), while SCFA-producing ones appear to be reduced (such as
Clostridiaceae,
Bacteroides and Akkermansia) when hypertension is present [
94,
95]. Some of these associated perturbations in gut microbiota homeostasis are partially related to HF pathogenesis and increase the risk of HF progression.
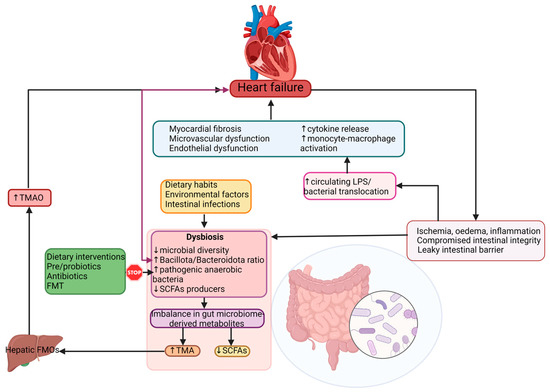
6. Gut-Derived Metabolites as Possible Biomarkers Related to Intestinal Dysbiosis in HF
A biomarker is defined as a biological compound that is easily accessible and measurable in the body. Biomarkers can be classified as molecular, cellular or imaging. Their role is to help in identifying the disease or provide therapeutic guidance. Natriuretic Peptides (NP), brain-type natriuretic peptide (BNP), N-terminal prohormone of BNP and cardiac troponin measurements—classic HF biomarkers—have already been included in the guidelines for HF diagnosis and treatment by the European Society of Cardiology (ESC) [
96] and the American Heart Association (AHA) [
97]. The addition of other diagnostic and prognostic biomarkers that could be associated to such a complex disease would be of benefit for both patients and medical practitioners.
Gut microbial-derived metabolites can also play a significant role in the pathogenesis of HF. It appears that the gut microbiome acts similarly to an endocrine organ. By generating active biometabolites including short-chain fatty acids (SCFAs), trimethylamine (TMA)/trimethylamine N-oxide (TMAO), and bile acids, the gut microbiome influences the host physiology. Several studies described the association of the gut’s microbiome metabolites and different pathologies including hypertension, atherosclerosis, HF, obesity, chronic kidney disease, and T2DM [
2,
7,
98,
99,
100,
101]. These metabolites can be considered as biomarkers of intestinal dysbiosis and can predict inflammation in patients known with HF [
101]. These patients with elevated plasma levels of phenylalanine display increased levels of inflammatory cytokines (IL-8, IL-10), C-reactive protein (CRP) and associate higher mortality [
102], whereas glycine manifest anti-inflammatory effects and seem to offer protection to the cells and heart [
103].
Alterations of gut microbiota composition, especially elevated N-oxidetrimethylamine (TMAO) levels are correlated with the risk of developing HF [
106]. TMAO is a metabolite produced by gut bacteria including
Bacillota and
Pseudomonadota, obtained from choline, phosphatidylcholine, and L-carnitine fermentation [
2]. Chen and colleagues [
106] reported that an elevated level of TMAO resulted from a diet high in saturated fat and sugar can lead to fibrosis, myocardial inflammation and to impaired diastolic function [
106]. Individuals with an increased abundance of
Ruminococcus,
Prevotella and
Clostridium genera and the
Lachnospiraceae family, and decreased levels of
Bacteroidota, revealed higher levels of TMAO in their plasma [
107,
108]. HF–associated dysbiosis is characterized by high levels of circulating TMAO, that are able to stimulate cardiac remodeling through promoting myocardial fibrosis and pro-inflammatory effects [
2,
77,
109,
110].
HF patients display elevated plasma levels of TMAO when compared to healthy individuals. Increased TMAO levels can be used as a prognostic biomarker in both acute and chronic HF, independently of B-type natriuretic peptide (BNP) and traditional risk factors, as TMAO levels are predictive of an augmented risk of mortality in these patients [
80,
81]. Elevated TMAO plasma values correspond with advanced stages of left ventricular diastolic dysfunction [
80]. TMAO can also be considered a prognostic predictor of HFeEF and a marker of risk stratification for this particular category of patients [
112,
113].
The SCFAs are represented by acetate, propionate and butyrate, and they are generated by gut bacteria including
Bacteroides,
Bifidobacterium and
Faecalibacterium spp. [
53]. They are the most important metabolites produced through colon bacteria fermentation of resistant starch and dietary fibers [
101]. Most evidence sustains the fact that SCFAs have a protective role against HF and play a major role in maintaining the integrity of the intestinal barrier: in mucus production and they are active in anti-inflammation protection [
67].
7. Interactions between the Gut Microbiome and Cardiovascular Drugs
Age, sex, nutritional status, disease states, along with genetic and environmental exposures are factors that can explain how individuals will respond to drug therapies [
121]. The human microbiome is known for its involvement in drug metabolism and pharmacological efficacy, but among them there is bidirectional communication, as drugs can also influence microbiota composition.
Drug absorption is an elaborate process, depending on many factors such as their solubility and stability in GI fluids, their pH, GI transit period, permeability through epithelial membranes and the drugs’ interaction with the host and microbial enzymes [
122]. The human gut microbiota is genetically capable of producing enzymes involved in oral drugs’ metabolism, facilitating their absorption across the gut and through the bloodstream [
121]. Dysbiosis of the gut’s bacterial communities can further alter drug pharmacokinetics; the activation of prodrugs can contribute to the production of unwanted toxic metabolites and the inactivation of drugs [
123]. Variation in drug response can also be present in a “healthy” gut, due to inter-individual differences in intestinal bacterial species [
13].
Related to the cardiovascular medication used in HF patients, metagenomic sequencing of stool samples from HF patients revealed that the use of several pharmaceutical agents such as statins, beta-blockers, angiotensin-converting enzyme inhibitors and platelet aggregation inhibitors has an important influence on gut microbial composition [
124]. Despite the fact that specific underlying mechanisms are unknown, partial results of this study were reproduced by another British group of researchers [
125].
7.1. Cardiac Glycosides
Digoxin, a drug frequently recommended in HF is a good example of microbiota influencing drug bioavailability. Some strains of
Eggerthella lenta are responsible for converting digoxin into an inactive microbial metabolite, limiting the quantity of active drug absorbed into the systemic bloodstream in an important 10 percent of patients [
126,
133].
7.2. Blood Thinners and Gut Microbiota
Aspirin is a non-steroidal anti-inflammatory drug (NSAIDS) commonly used to decrease the risk of cerebrovascular and cardiovascular disorders [
133]. Existing evidence demonstrates its ability to disrupt the gut’s microbiota composition. Patients using aspirin present variations of
Ruminococcaceae,
Prevotella,
Barnesiella and
Bacteroides bacterial levels in comparison to individuals not using or using other types of NSAIDs. Furthermore, the gut’s bacterial communities’ composition seems to exert influence on aspirin metabolism. While oral antibiotic administration can decrease the gut microbiota’s metabolic activity by slowing its degradation, increasing its bioavailability and prolonging its anti-thrombotic action, probiotics containing
Bifidobacterium breve Bif195 bacteria can protect against an aspirin intake adverse reaction, such as intestinal wall damage and aspirin-induced gastric ulcers [
135,
136].
7.3. The Effects of Beta-Blockers, ACEi, and ARBs on Gut Microbiota
The effects of antihypertensive medications have been investigated on several occasions, both in animal and human studies. Despite expectations, the association between the use of beta-blockers, angiotensin receptor blockers (ARBs) and angiotensin converting enzyme inhibitors (ACE inhibitors) can modify the composition of gut microbiota.
7.4. Statins and Gut Microbiota
Statins are drugs used for their capacity to decrease low-density-lipoprotein-C (LDL-C) and cholesterol levels. Inter-individual variations in the response to statin treatment are well-known, and are not related to a specific statin agent or dose [
124]. Studies have proven their action on modulating gut bacterial communities’ composition [
121,
128]. Individuals treated with atorvastatin presented an increased level of anti-inflammatory gut bacteria such as
Faecalibacterium prausnitzii and
Akermansia muciniphila, whereas untreated patients known with hypercholesterolemia displayed an increased level of bacterial species with pro-inflammatory effects, such as
Collinsella and
Streptococcus [
140]. LDL-C levels seem to be negatively correlated to the phyla
Bacillota and
Fusobacteria, while
Lentisphaerae and
Cyanobacteria spp. were positively associated with LDL-C [
128].
8. Modulation of Dysbiosis as a Potential Target in Heart Failure
Considering that dysbiosis is a key factor in HF pathogenesis and disease progression, targeting the disrupted gut microbiota could be considered an effective therapeutic objective. The possibility of characterizing each patient’s gut microbiota and his disease-associated dysbiosis allows the initiation of a personalized, targeted therapeutic plan. Although there are various ways to manage and modulate the dysbiotic intestinal microbiota, such as dietary interventions (which also include the use of prebiotics, prebiotics and postbiotics) and fecal transplantation, several reports from the available literature place diet modification and probiotic use as the main interventions for microbiota modulation.
Diet has always been considered a crucial factor in shaping the structure and function of gut’s associated microbiota. A 5-day adjusted diet has been shown to produce beneficial changes in the number and species of the gut microbiota [
145]. Often cited in the medical literature, the Mediterranean diet (MD) consists of elevated levels of polyunsaturated fatty acids, dietary fiber, polyphenols, and a small quantity of red meat [
77]. Among its recognized benefits on human health, an MD provides an increased abundance of probiotics, greater biodiversity, elevated SCFAs, and reduced TMAO [
146,
147]. Adherence to an MD was associated with a decreasing HF incidence up to 74% [
148].
A high-fiber diet has recently been demonstrated to improve gut dysbiosis (described by the
Bacilliota and
Bacteroidota ratio), reduced blood pressure, improved cardiac function and normalized cardiac hypertrophy in a hypertension-induced HF experimental model [
153]. Additionally, fermentation of fiber results in augmented SCFA production, with their beneficial actions on human health [
150].
Related to antibiotic use in modulating the gut microbiota in HF patients, results are controversial. In animal models, oral vancomycin use induced smaller left ventricular infarct size, and improved recovery cardiac function following ischemia/reperfusion experiments in treated, compared to untreated, rats [
166]. Rifamixin, besides its bactericidal and bacteriostatic effect, also has the capacity to reduce translocation of bacteria and toxicity, has an anti-inflammatory effect and can positively regulate the composition of the intestinal microbiota, promoting the growth of
lactobacillus and bifidobacteria [
167,
168]. As for human clinical trials, the results are contradictory. The use of a cocktail of tobramicyn and polymixin B, in HF patients, normalized the level of intestinal Gram-negative bacilli, significantly decreased pro-inflammatory cytokines and improved flow-mediated dilation: evidence of endothelial dysfunction [
169].
Prebiotics are “a selectively fermented ingredient that results in specific changes in the composition and/or activity of the gastrointestinal microbiota, thus conferring benefit(s) upon host health” [
171]. Prebiotics use could increase the amount of
Bifidobacterium and promotes a higher body weight loss, which decreased systolic and diastolic blood pressure [
172].