Antimicrobial resistance (AMR) is one of the top 10 global health threats facing humanity, and the sub-Saharan Africa (SSA) is among the heavily affected regions due to its weak health systems and limited resources. Due to an escalating number of AMR pathogens and the scarcity of new antimicrobials, efforts in the prevention of infections and the search for alternative treatment options are ongoing. The highlighted weapons against AMR include vaccines, education and awareness, infection prevention and control (IPC) using water, sanitation, and hygiene (WASH), alternative treatment options, the One Health (OH) approach, AMR surveillance, operational national action plans (NAPs) on AMR, antimicrobial stewardship (AMS) programs, and good governance and regulations. Despite not being used at a satisfactory level in SSA, advanced techniques in dealing with AMR in SSA include (i) metagenomics, (ii) whole-genome sequencing (WGS) in AMR surveillance to track resistance trends and know when to intervene, and (iii) use of artificial intelligence in AMR prediction based on genomics data.
- antimicrobial resistance
- One Health
- alternative treatment
1. Vaccines
2. Education and Awareness
-
Training healthcare professionals
-
Public awareness/engagement
3. Infection Prevention and Control by Improving WASH
4. Natural Products
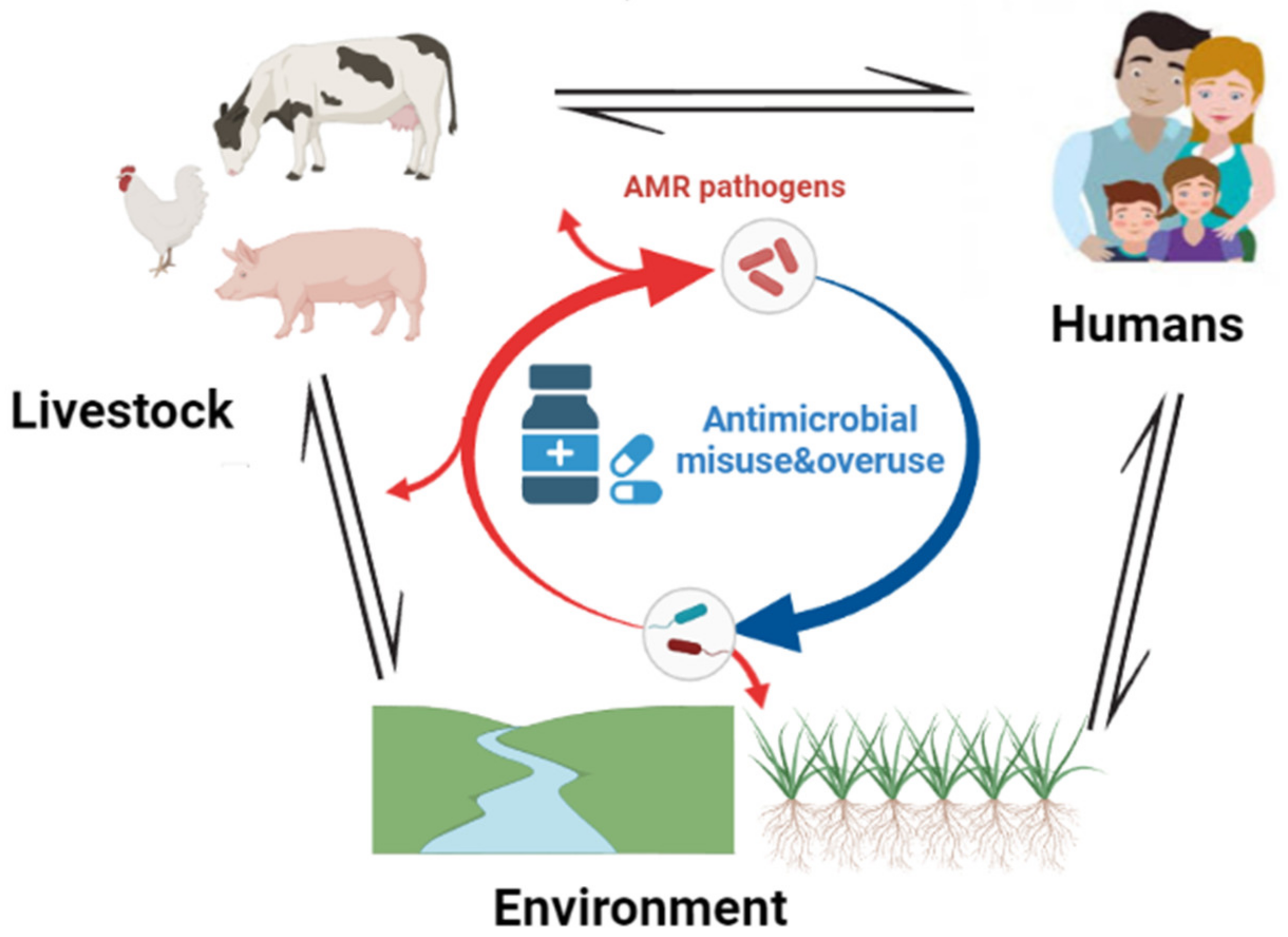
5. One Health Approach
6. AMR Surveillance Program
7. Antimicrobial Stewardship Programs
8. Functional National Action Plans (NAPs) on AMR
9. Political Will/Governance/Regulations
References
- Rosini, R.; Nicchi, S.; Pizza, M.; Rappuoli, R. Vaccines against Antimicrobial Resistance. Front. Immunol. 2020, 11, 1048. Available online: https://www.frontiersin.org/article/10.3389/fimmu.2020.01048 (accessed on 19 June 2022).
- Autran, B.; Carcelain, G.; Combadiere, B.; Debre, P. Therapeutic Vaccines for Chronic Infections. Science 2004, 305, 205–208.
- Riedel, S. Edward Jenner and the History of Smallpox and Vaccination. Bayl. Univ. Med. Cent. Proc. 2005, 18, 21–25.
- Iwasaki, A.; Omer, S.B. Why and How Vaccines Work. Cell 2020, 183, 290–295.
- Bloom, D.E.; Black, S.; Salisbury, D.; Rappuoli, R. Antimicrobial resistance and the role of vaccines. Proc. Natl. Acad. Sci. USA 2018, 115, 12868–12871.
- Micoli, F.; Bagnoli, F.; Rappuoli, R.; Serruto, D. The role of vaccines in combatting antimicrobial resistance. Nat. Rev. Microbiol. 2021, 19, 287–302.
- Lipsitch, M.; Siber, G.R. How Can Vaccines Contribute to Solving the Antimicrobial Resistance Problem? Mbio 2016, 7, e00428-16.
- Jansen, K.U.; Knirsch, C.; Anderson, A.S. The role of vaccines in preventing bacterial antimicrobial resistance. Nat. Med. 2018, 24, 10–19.
- Bloom; Curtis, N. D.E. The Value of Vaccination In Hot Topics in Infection and Immunity in Children VII; Curtis, N., Finn, A., Pollard, A.J., Eds.; Springer: New York, NY, USA, 2011; pp. 1–8.
- Rodrigues, C.; Plotkin, S.A. Impact of Vaccines; Health, Economic and Social Perspectives. Front. Microbiol. 2020, 11, 1526.
- Alghamdi, S. The role of vaccines in combating antimicrobial resistance (AMR) bacteria. Saudi J. Biol. Sci. 2021, 28, 7505–7510.
- WHO. Bacterial Vaccines in Clinical and Preclinical Development. 2021. Available online: https://www.who.int/publications-detail-redirect/9789240052451 (accessed on 15 July 2022).
- Musa, B.M.; Adamu, A.L.; Galadanci, N.A.; Zubayr, B.; Odoh, C.N.; Aliyu, M.H. Trends in prevalence of multi drug resistant tuberculosis in sub-Saharan Africa: A systematic review and meta-analysis. PLoS ONE 2017, 12, e0185105.
- Hlashwayo, D.F.; Sigaúque, B.; Bila, C.G. Epidemiology and antimicrobial resistance of Campylobacter spp. in animals in Sub-Saharan Africa: A systematic review. Heliyon 2020, 6, e03537.
- Ismail, S.; Shahid, F.; Khan, A.; Bhatti, S.; Ahmad, S.; Naz, A.; Almatroudi, A.; Qamar, M.T.U. Pan-vaccinomics approach towards a universal vaccine candidate against WHO priority pathogens to address growing global antibiotic resistance. Comput. Biol. Med. 2021, 136, 104705.
- Madhi, S.A.; Rees, H. Special focus on challenges and opportunities for the development and use of vaccines in Africa. Hum. Vaccines Immunother. 2018, 14, 2335–2339.
- Amponsah-Dacosta, E.; Muloiwa, R.; Wiysonge, C.S.; Gold, M.; Hussey, G.; Kagina, B.M. Developing vaccinology expertise for Africa: Fifteen years and counting. Pan Afr. Med. J. 2021, 38, 313.
- Stoop, N.; Hirvonen, K.; Maystadt, J.-F. Institutional mistrust and child vaccination coverage in Africa. BMJ Glob. Health 2021, 6, e004595.
- Janusz, C.B.; Frye, M.; Mutua, M.K.; Wagner, A.L.; Banerjee, M.; Boulton, M.L. Vaccine Delay and Its Association With Undervaccination in Children in Sub-Saharan Africa. Am. J. Prev. Med. 2021, 60 (Suppl. 1), S53–S64.
- Bobo, F.T.; Asante, A.; Woldie, M.; Dawson, A.; Hayen, A. Child vaccination in sub-Saharan Africa: Increasing coverage addresses inequalities. Vaccine 2022, 40, 141–150.
- Saied, A.A.; Metwally, A.A.; Dhawan, M.; Choudhary, O.P.; Aiash, H. Strengthening vaccines and medicines manufacturing capabilities in Africa: Challenges and perspectives. EMBO Mol. Med. 2022, 14, e16287.
- Haenssgen, M.J.; Xayavong, T.; Charoenboon, N.; Warapikuptanun, P.; Zaw, Y.K. The Consequences of AMR Education and Awareness Raising: Outputs, Outcomes, and Behavioural Impacts of an Antibiotic-Related Educational Activity in Lao PDR. Antibiotics 2018, 7, 95.
- Ajuebor, O.; Shetty, N.; Mah, K.; Cometto, G. Health workers’ education and training to prevent antimicrobial resistance. Bull. World Health Organ. 2019, 97, 791.
- Nand, P.; Wilson, M.; Cohen, S.H.; Brown, J. Curbing antimicrobial resistance: Do physicians receive adequate education about antibiograms? J. Infect. 2016, 72, 127–129.
- Lubwama, M.; Onyuka, J.; Ayazika, K.T.; Ssetaba, L.J.; Siboko, J.; Daniel, O.; Mushi, M.F. Knowledge, attitudes, and perceptions about antibiotic use and antimicrobial resistance among final year undergraduate medical and pharmacy students at three universities in East Africa. PLoS ONE 2021, 16, e0251301.
- Nisabwe, L.; Brice, H.; Umuhire, M.C.; Gwira, O.; Harelimana, J.D.D.; Nzeyimana, Z.; Sebatunzi, O.R.; Rusingiza, E.K.; Hahirwa, I.; Muvunyi, C.M. Knowledge and attitudes towards antibiotic use and resistance among undergraduate healthcare students at University of Rwanda. J. Pharm. Policy Pract. 2020, 13, 7.
- Majumder, A.A.; Singh, K.; Hilaire, M.G.-S.; Rahman, S.; Sa, B.; Haque, M. Tackling Antimicrobial Resistance by promoting Antimicrobial stewardship in Medical and Allied Health Professional Curricula. Expert Rev. Anti-infective Ther. 2020, 18, 1245–1258.
- Pezzani, M.D.; Tornimbene, B.; Pessoa-Silva, C.; de Kraker, M.; Rizzardo, S.; Salerno, N.D.; Harbarth, S.; Tacconelli, E. Methodological quality of studies evaluating the burden of drug-resistant infections in humans due to the WHO Global Antimicrobial Resistance Surveillance System target bacteria. Clin. Microbiol. Infect. 2021, 27, 687–696.
- Catalán-Matamoros, D.; Pariente, A.; Elías-Pérez, C. What we know about media communication on antibiotics and antimicrobial resistance: A systematic review of the scientific literature. Patient Educ. Couns. 2019, 102, 1427–1438.
- Venant, E.; Stanley, B.K.; Mosha, M.J.; Msovela, K.E.; Mushi, D.J.; Masanja, P.; Wiedenmayer, K.; Ombaka, E. P23 Assessment of knowledge, attitude and practice towards antimicrobial use and resistance among students in three secondary schools in Dodoma City. JAC-Antimicrobial Resist. 2022, 4 (Suppl. 1), dlac004.022.
- Mitchell, J.; Cooke, P.; Ahorlu, C.; Arjyal, A.; Baral, S.; Carter, L.; Dasgupta, R.; Fieroze, F.; Fonseca-Braga, M.; Huque, R.; et al. Community engagement: The key to tackling Antimicrobial Resistance (AMR) across a One Health context? Glob. Public Health 2021, 17, 2647–2664.
- Willis, L.D.; Chandler, C. Quick fix for care, productivity, hygiene and inequality: Reframing the entrenched problem of antibiotic overuse. BMJ Glob. Health 2019, 4, e001590.
- Godman, B.; Egwuenu, A.; Wesangula, E.; Schellack, N.; Kalungia, A.C.; Tiroyakgosi, C.; Kgatlwane, J.; Mwita, J.C.; Patrick, O.; Niba, L.L.; et al. Tackling Antimicrobial Resistance across Sub-Saharan Africa: Current Challenges and Implications for the Future. Expert Opin. Drug Saf. 2022, 21, 1089–1111.
- Michaelidou, M.; Karageorgos, S.; Tsioutis, C. Antibiotic Use and Antibiotic Resistance: Public Awareness Survey in the Republic of Cyprus. Antibiotics 2020, 9, 759.
- Pinder, R.J.; Berry, D.; Sallis, A.; Chadborn, T. Antibiotic Prescribing and Behaviour Change in Healthcare Settings: Literature Review and Behavioural Analysis. In Department of Health & Public Health England, Report, Feb. 2015; Department of Health & Public Health England: London, UK.
- Ayukekbong, J.A.; Ntemgwa, M.; Atabe, A.N. The threat of antimicrobial resistance in developing countries: Causes and control strategies. Antimicrob. Resist. Infect. Control. 2017, 6, 47.
- World Bank. Drug-Resistant Infections: A Threat to Our Economic Future. Available online: https://documents.worldbank.org/en/publication/documents-reports/documentdetail/323311493396993758/final-report (accessed on 9 October 2022).
- Kimera, Z.I.; Frumence, G.; Mboera, L.E.G.; Rweyemamu, M.; Mshana, S.E.; Matee, M.I.N. Assessment of Drivers of Antimicrobial Use and Resistance in Poultry and Domestic Pig Farming in the Msimbazi River Basin in Tanzania. Antibiotics 2020, 9, 838.
- Gahamanyi, N.; Mboera, L.E.G.; Matee, M.I.; Mutangana, D.; Komba, E.V.G. Prevalence, Risk Factors, and Antimicrobial Resistance Profiles of Thermophilic Campylobacter Species in Humans and Animals in Sub-Saharan Africa: A Systematic Review. Int. J. Microbiol. 2020, 2020, 2092478. Available online: https://www.hindawi.com/journals/ijmicro/2020/2092478/ (accessed on 23 November 2020).
- Williams, M.R.; Stedtfeld, R.D.; Guo, X.; Hashsham, S.A. Antimicrobial Resistance in the Environment. Water Environ. Res. 2016, 88, 1951–1967.
- WHO. Progress on WASH in Health Care Facilities 2000–2021: Special Focus on WASH and Infection Prevention and Control (IPC). Available online: https://www.who.int/publications-detail-redirect/progress-on-wash-in-health-care-facilities-2000-2021--special-focus-on-wash-and-infection-prevention-and-control-(ipc) (accessed on 1 March 2023).
- Sano, D.; Wester, A.L.; Schmitt, H.; Amarasiri, M.; Kirby, A.; Medlicott, K.; Husman, A.M.D.R. Updated research agenda for water, sanitation and antimicrobial resistance. J. Water Health 2020, 18, 858–866.
- Bain, R.; Johnston, R.; Mitis, F.; Chatterley, C.; Slaymaker, T. Establishing Sustainable Development Goal Baselines for Household Drinking Water, Sanitation and Hygiene Services. Water 2018, 10, 1711.
- Ikhimiukor, O.O.; Odih, E.E.; Donado-Godoy, P.; Okeke, I.N. A bottom-up view of antimicrobial resistance transmission in developing countries. Nat. Microbiol. 2022, 7, 757–765.
- WHO. Technical Brief on Water, Sanitation, Hygiene and Wastewater Management to Prevent Infections and Reduce the Spread of Antimicrobial Resistance. Available online: https://www.who.int/publications/i/item/9789240006416 (accessed on 20 October 2022).
- Prendergast, A.J.; Gharpure, R.; Mor, S.; Viney, M.; Dube, K.; Lello, J.; Berger, C.; Siwila, J.; Joyeux, M.; Hodobo, T.; et al. Putting the “A” into WaSH: A call for integrated management of water, animals, sanitation, and hygiene. Lancet Planet. Heal. 2019, 3, e336–e337.
- Kraemer, S.A.; Ramachandran, A.; Perron, G.G. Antibiotic Pollution in the Environment: From Microbial Ecology to Public Policy. Microorganisms 2019, 7, 180.
- Essack, S. Water, sanitation and hygiene in national action plans for antimicrobial resistance. Bull. World Health Organ. 2021, 99, 606–608.
- Ramay, B.M.; Caudell, M.A.; Cordón-Rosales, C.; Archila, L.D.; Palmer, G.H.; Jarquin, C.; Moreno, P.; McCracken, J.P.; Rosenkrantz, L.; Amram, O.; et al. Antibiotic use and hygiene interact to influence the distribution of antimicrobial-resistant bacteria in low-income communities in Guatemala. Sci. Rep. 2020, 10, 13767.
- Chandra, H.; Bishnoi, P.; Yadav, A.; Patni, B.; Mishra, A.P.; Nautiyal, A.R. Antimicrobial Resistance and the Alternative Resources with Special Emphasis on Plant-Based Antimicrobials—A Review. Plants 2017, 6, 16.
- Borges, A.; Abreu, A.C.; Dias, C.; Saavedra, M.J.; Borges, F.; Simões, M. New Perspectives on the Use of Phytochemicals as an Emergent Strategy to Control Bacterial Infections Including Biofilms. Molecules 2016, 21, 877.
- Gahamanyi, N.; Munyaneza, E.; Dukuzimana, E.; Tuyiringire, N.; Pan, C.-H.; Komba, E.V.G. Ethnobotany, Ethnopharmacology, and Phytochemistry of Medicinal Plants Used for Treating Human Diarrheal Cases in Rwanda: A Review. Antibiotics 2021, 10, 1231.
- Bartlett, J.G.; Gilbert, D.N.; Spellberg, B. Seven Ways to Preserve the Miracle of Antibiotics. Clin. Infect. Dis. 2018, 56, 1445–1450.
- Rawat, P.; Singh, P.K.; Kumar, V. Evidence based traditional anti-diarrheal medicinal plants and their phytocompounds. Biomed. Pharmacother. 2017, 96, 1453–1464.
- Atlas, R.M. One Health: Its Origins and Future In One Health: The Human-Animal-Environment Interfaces in Emerging Infectious Diseases: The Concept and Examples of a One Health Approach; Mackenzie, J.S., Jeggo, M., Daszak, P., Richt, J.A., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 1–13.
- One Health High-Level Expert Panel (OHHLEP); Adisasmito, W.B.; Almuhairi, S.; Behravesh, C.B.; Bilivogui, P.; Bukachi, S.A.; Casas, N.; Becerra, N.C.; Charron, D.F.; Chaudhary, A.; et al. One Health: A new definition for a sustainable and healthy future. PLoS Pathog. 2022, 18, e1010537.
- Evans, B.; Leighton, F. A history of One Health. Rev. Sci. Tech. 2014, 33, 413–420.
- McEwen, S.A.; Collignon, P.J. Antimicrobial Resistance: A One Health Perspective. Microbiol. Spectr. 2018, 6, 521–547.
- Woolhouse, M.; Ward, M.; Van Bunnik, B.; Farrar, J. Antimicrobial resistance in humans, livestock and the wider environment. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140083.
- Jassim, S.A.A.; Limoges, R.G. Bacteriophage and Antimicrobial Resistance In Bacteriophages: Practical Applications for Nature’s Biocontrol; Jassim, S.A.A., Limoges, R.G., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 19–57.
- The Global Leaders Group on Antimicrobial Resistance. Available online: https://www.amrleaders.org/resources (accessed on 10 August 2022).
- Cornejo, J.; Asenjo, G.; Zavala, S.; Venegas, L.; Galarce, N.; Hormazábal, J.C.; Vergara-E, C.; Lapierre, L. Advances in Integrated Antimicrobial Resistance Surveillance and Control Strategies in Asia-Pacific Economic Cooperation Economies: Assessment of a Multiyear Building Capacity Project. Antibiotics 2022, 11, 1022.
- Pariente, N.; PLoS Biology Staff Editors. The antimicrobial resistance crisis needs action now. PLoS Biol. 2022, 20, e3001918.
- Wu, D.; Walsh, T.R.; Wu, Y. World Antimicrobial Awareness Week 2021—Spread Awareness, Stop Resistance. China CDC Wkly. 2021, 3, 987–993.
- Langford, B.J.; Matson, K.L.; Eljaaly, K.; Apisarnthanarak, A.; Bailey, P.L.; MacMurray, L.; Marra, A.R.; Simonsen, K.A.; Sreeramoju, P.; Nori, P.; et al. Ten ways to make the most of World Antimicrobial Awareness Week. Antimicrob. Steward. Health Epidemiology 2022, 2, e187.
- Groseclose, S.L.; Buckeridge, D.L. Public Health Surveillance Systems: Recent Advances in Their Use and Evaluation. Annu. Rev. Public Health 2017, 38, 57–79.
- WHO. Global Antimicrobial Resistance and Use Surveillance System (GLASS) Report: 2021. Available online: https://www.who.int/publications-detail-redirect/9789240027336 (accessed on 21 August 2022).
- Njukeng, P.A.; Ako-Arrey, D.E.; Amin, E.T.; Njumkeng, C.; Wirsiy, F.S. Antimicrobial Resistance in the Central African Region: A Review. J. Environ. Sci. Public Health 2019, 3, 358–378.
- Fuller, W.F.; Hamzat, O.T.; Aboderin, A.O.; Gahimbare, L.; Kapona, O.; Yahaya, A.A.; Kasambara, W.; Nikiema, J.-B.; Ilboudo, D.W.; Mpundu, M.M. National action plan on antimicrobial resistance: An evaluation of implementation in the World Health Organization Africa region. J. Public Health Afr. 2022, 13, 2000.
- Kariuki, S.; Kering, K.; Wairimu, C.; Onsare, R.; Mbae, C. Antimicrobial Resistance Rates and Surveillance in Sub-Saharan Africa: Where Are We Now? Infect. Drug Resist. 2022, 15, 3589–3609.
- Department Health. South Africa Antimicrobial Resistance National Action Plan 2018–2024. Available online: https://www.who.int/publications/m/item/south-africa-south-african-antimicrobial-resistance-national-strategy-framework-a-one-health-approach (accessed on 15 August 2022).
- MCC. National Antimicrobial Resistance Surveillance Framework Tanzania. Available online: https://www.mifugouvuvi.go.tz/uploads/publications/sw1602246063-NATIONAL%20ANTIMICROBIAL%20RESISTANCE%20SURVEILLANCE%20FRAMEWORK.pdf (accessed on 16 August 2022).
- Hein, W.; Aglanu, L.M.; Mensah-Sekyere, M.; Harant, A.; Brinkel, J.; Lamshöft, M.; Lorenz, E.; Eibach, D.; Amuasi, J. Fighting Antimicrobial Resistance: Development and Implementation of the Ghanaian National Action Plan (2017–2021). Antibiotics 2022, 11, 613.
- Kariuki, S.; Keddy, K.H.; Antonio, M.; Okeke, I.N. Antimicrobial resistance surveillance in Africa: Successes, gaps and a roadmap for the future. Afr. J. Lab. Med. 2018, 7, 924.
- Akpan, M.R.; Isemin, N.U.; Udoh, A.E.; Ashiru-Oredope, D. Implementation of antimicrobial stewardship programmes in African countries: A systematic literature review. J. Glob. Antimicrob. Resist. 2020, 22, 317–324.
- Malani, A.N.; Richards, P.G.; Kapila, S.; Otto, M.H.; Czerwinski, J.; Singal, B. Clinical and economic outcomes from a community hospital’s antimicrobial stewardship program. Am. J. Infect. Control. 2013, 41, 145–148.
- Kaki, R.; Elligsen, M.; Walker, S.; Simor, A.; Palmay, L.; Daneman, N. Impact of antimicrobial stewardship in critical care: A systematic review. J. Antimicrob. Chemother. 2011, 66, 1223–1230.
- Stach, L.M.; Hedican, E.B.; Herigon, J.C.; Jackson, M.A.; Newland, J.G. Clinicians’ Attitudes Towards an Antimicrobial Stewardship Program at a Children’s Hospital. J. Pediatr. Infect. Dis. Soc. 2012, 1, 190–197.
- World Health Organization. Antimicrobial Stewardship Programmes in Health-Care Facilities in Low- and Middle-Income Countries: A WHO Practical Toolkit. Available online: https://apps.who.int/iris/handle/10665/329404 (accessed on 10 September 2022).
- Davey, P.; Marwick, C.A.; Scott, C.L.; Charani, E.; McNeil, K.; Brown, E.; Ramsay, C.R.; Wiffen, P.J.; Wilcox, M. Interventions to improve antibiotic prescribing practices for hospital inpatients. Cochrane Database Syst. Rev. 2013, 4, CD003543.
- Brink, A.J.; Messina, A.P.; Feldman, C.; Richards, G.A.; Becker, P.J.; Goff, D.A.; Bauer, K.A.; Nathwani, D.; van den Bergh, D.; on behalf of theNetcare Antimicrobial Stewardship Study Alliance. Antimicrobial stewardship across 47 South African hospitals: An implementation study. Lancet Infect. Dis. 2016, 16, 1017–1025.
- Howard, P.; Pulcini, C.; Levy, H.G.; West, R.M.; Gould, I.M.; Harbarth, S.; Nathwani, D.; ESCMID Study Group for Antimicrobial Policies (ESGAP); ISC Group on Antimicrobial Stewardship. An international cross-sectional survey of antimicrobial stewardship programmes in hospitals. J. Antimicrob. Chemother. 2015, 70, 1245–1255.
- Boyles, T.H.; Whitelaw, A.; Bamford, C.; Moodley, M.; Bonorchis, K.; Morris, V.; Rawoot, N.; Naicker, V.; Lusakiewicz, I.; Black, J.; et al. Antibiotic Stewardship Ward Rounds and a Dedicated Prescription Chart Reduce Antibiotic Consumption and Pharmacy Costs without Affecting Inpatient Mortality or Re-Admission Rates. PLoS ONE 2013, 8, e79747.
- World Health Organization. Global Action Plan on Antimicrobial Resistance. Available online: https://www.who.int/publications/i/item/9789241509763 (accessed on 10 September 2022).
- Munkholm, L.; Rubin, O. The global governance of antimicrobial resistance: A cross-country study of alignment between the global action plan and national action plans. Glob. Health 2020, 16, 109.
- World Health Organization. Supporting Countries with National Action Plan Implementation. Available online: https://www.who.int/activities/supporting-countries-with-national-action-plan-implementation (accessed on 10 September 2022).
- Kakkar, M.; Chatterjee, P.; Chauhan, A.S.; Grace, D.; Lindahl, J.; Beeche, A.; Jing, F.; Chotinan, S. Antimicrobial resistance in South East Asia: Time to ask the right questions. Glob. Health Action 2018, 11, 1483637.
- Wernli, D.; Haustein, T.; Conly, J.; Carmeli, Y.; Kickbusch, I.; Harbarth, S. A Call for Action: The Application of the International Health Regulations to the Global Threat of Antimicrobial Resistance. PLoS Med. 2011, 8, e1001022.
- Bretaudeau, L.; Tremblais, K.; Aubrit, F.; Meichenin, M.; Arnaud, I. Good Manufacturing Practice (GMP) Compliance for Phage Therapy Medicinal Products. Front. Microbiol. 2020, 11, 1161.
