Chickens can acquire bacteria at different stages, and bacterial diversity can occur due to production practices, diet, and environment. The changes in consumer trends have led to increased animal production, and chicken meat is one of the most consumed meats. To ensure high levels of production, antimicrobials have been used in livestock for therapeutic purposes, disease prevention, and growth promotion, contributing to the development of antimicrobial resistance across the resident microbiota. Enterococcus spp. and Escherichia coli are normal inhabitants of the gastrointestinal microbiota of chickens that can develop strains capable of causing a wide range of diseases, i.e., opportunistic pathogens. Enterococcus spp. isolated from broilers have shown resistance to at least seven classes of antibiotics, while E. coli have shown resistance to at least four.
- antibiotic resistance
- broilers
- food animals
- gastrointestinal microbiota
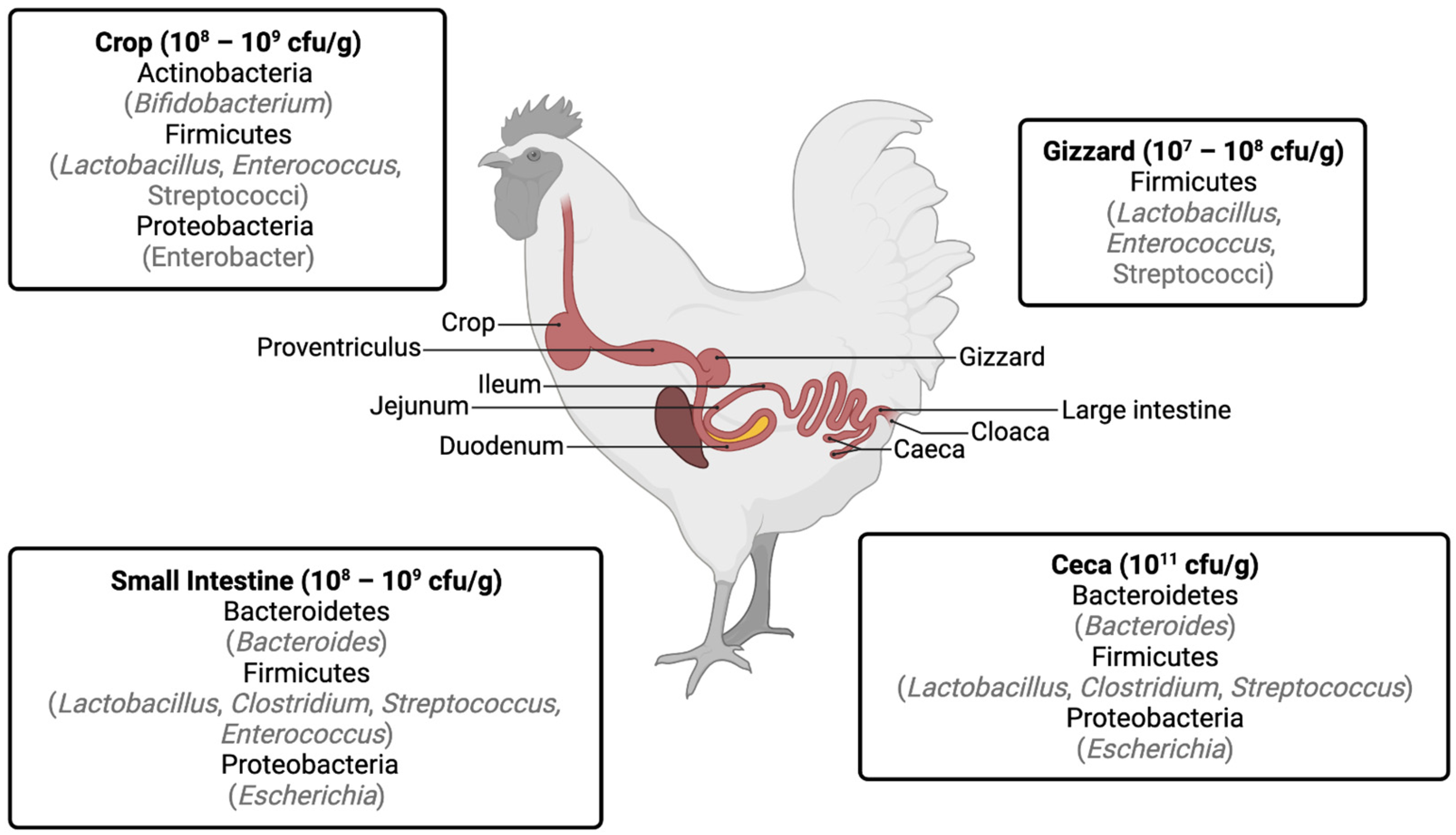
1. Gastrointestinal Bacteria in Chickens
2. Development of Antibiotic Resistance by Gastrointestinal Bacteria in Broilers
The growth of the human population, the increase in incomes, and the changes in consumer trends (more protein in the diet) have increased the consumption of animal products. This high demand for animal products led to high levels of animal production [15]. A large sector of the food-producing animal industry is represented by poultry, and over the last three decades, it has expanded with an annual growth rate of over 5%, while the bovine and swine industries grew 1.5% and 3%, respectively [16][17]. The poultry meat production varies, but in Europe, over 80% is chicken [18]. Commercial meat-type chicken production is highly specialized and includes two types of farms: breeding hens to obtain fertile eggs and broilers to obtain meat [19]. According to Statistics Portugal (INE), the gross production of chicken meat in Portugal in 2021 recorded a level similar to the previous year (+0.8%), having reached 313 thousand tons [20]. To ensure high levels of production, antimicrobials have been used in animal production for therapeutic purposes, disease prevention, and growth promotion [21]. In 2020, in Portugal, approximately 179.1 tons of antimicrobials were sold to food animal producers, and the antimicrobial classes for veterinary use with higher sales were tetracyclines (34.4%), penicillins (22.1%), macrolides (11.4%), pleuromutilins (7.2%), polymyxins (6.7%), quinolones (4.2%), sulfonamides (4.1%), lincosamides (3.5%), aminoglycosides (2.8%), amphenicols (2.5%), and trimethoprim (1%) [22]. The use of antimicrobials as growth promoters involves administering subtherapeutic concentrations of antibiotics orally or mixed into feed or water with the final goal of increasing the rate of weight gain and the efficiency of the feed [21]. In poultry, antibiotics are generally administered to the entire flock, and antibiotic usage for disease prevention is allowed in all large poultry-producing countries [23][24]. Antimicrobial use in livestock is linked to the development of antimicrobial resistance, and antibiotic resistance mechanisms can be easily spread within microbial communities [25]. The development of resistant strains has raised some public health concerns, and to overcome this issue, the EU and South Korea have banned the use of antibiotics as feed additives [26][27]. Other countries, such as Denmark, Japan, and Canada, formally monitor antibiotic use and the development of antimicrobial resistance [28][29][30]. In the US, in early 2017, the Food and Drug Administration (FDA) banned antimicrobials used as growth promoters, but they continue to be legally administered via animal feed for disease prevention, often at lower dosages and for longer periods, similarly to production uses now prohibited [21][31]. However, in some European countries, a substantial decline in the sales of antimicrobials for food-producing animals has been observed [32]. Antibiotic-free poultry production has also become popular in many developed countries, particularly within the US poultry industry, mainly due to a consumer perception that antibiotic-free produced poultry is superior to conventionally raised poultry [33]. However, broilers raised with no antibiotics are more susceptible to enteric diseases that can negatively impact their intestinal health and general welfare [34]. Improving poultry production, increasing poultry immunity, and reducing the spread of disease are vital, and studies have reported that these can be achieved by adding diverse, potentially valuable ingredients to the feed or drinking water of poultry flocks [35]. A significant increase in the use of antibiotics is caused by broiler farming, and their permanent use disrupts the gastrointestinal metabolism of chickens [9][36]. The gastrointestinal tract is considered the main intervening part of productivity, pathogen entrance, and disease prevention [37]. Any disturbance might lead to poor digestion or absorption of nutrients or the inability to fight pathogens and the development of a disease [5]. Since antibiotics have been largely used as feed additives in animal production for therapeutic or growth promotion purposes, it is crucial to pay attention to the health of the broiler’s gastrointestinal tract and the development of drug-resistant bacteria [36]. Vancomycin is one of the “last-line” antibiotics used to treat life-threatening infections caused by Gram-positive bacteria [38]. Avoparcin, a glycopeptide antibiotic analog of vancomycin, was introduced as a feed additive in chicken feed in 1987, and, even though it was banned from the EU in 1997, the development of vancomycin-resistant Enterococci might have been potentiated by its use as a feed additive in livestock [38][39][40][41]. Furthermore, β-lactams are among the most used classes of antibiotics both in humans and animals, and an increasing trend of gastrointestinal colonization by extended-spectrum β-lactamase (ESBL) producing bacteria has been observed in commercial poultry farms and humans [42][43]. ESBL-producing bacteria are often reported among broilers, and the acquisition of ESBL genes among gastrointestinal microflora may play an important role in the spread of multidrug-resistant bacteria among humans, animals, and the environment via the food chain [44][45][46]. Antibiotic-resistant bacteria and antibiotic-resistant genes can be found in many hosts and environments, including wild animals, organically produced food animals, and even in newborn babies never exposed to antibiotics [44][47][48][49]. In addition, both pathogenic and non-pathogenic bacteria can harbor antimicrobial resistance genes [50]. Antimicrobial resistance studies have mainly focused on clinical pathogens, but recently, due to the emergence of zoonotic diseases, the impact of antimicrobial resistance on animals, agricultural practices, wildlife, and the environment has gained new attention and led to cooperation from various sectors [51]. Thus, Commission Implementing Decision 2020/1729 of 17 November 2020 determined that to monitor and report antimicrobial resistance, commensal Enterococcus faecalis, Enterococcus faecium, and E. coli, as well as food-producing animals, such as broilers, must be considered [52].3. Enterococcus spp.
Enterococcus spp. are Gram-positive, catalase-negative, non-spore-forming, facultative anaerobic lactic acid bacteria that can be found in the gastrointestinal microbiota of humans and other animals [53]. These bacteria can tolerate many adverse conditions, surviving for several months in hostile environments, including extreme pH and temperature conditions (between 10 ℃ and 45 ℃) and high NaCl concentrations [54]. Since they prefer intestinal habitats and are widespread, robust, and easy to cultivate, they are often used as indicators of fecal contamination and integrated hygiene criteria for water and food products [55]. They are also appropriate for veterinary and human resistance surveillance systems [56][57]. These naturally gut-oriented bacteria were considered harmless commensal bacteria, but when the commensal relationship with the host is disturbed, enterococci can cause aggressive infections [58]. They are currently recognized as one of the main nosocomial pathogens and are progressively becoming more resistant to antimicrobial agents. These species have also been associated with an increasing number of hospital-acquired infections in both human and veterinary medicine [59][60][61]. In poultry, Enterococcus spp. can cause several diseases such as osteomyelitis, femoral head necrosis, spondylitis, skeletal disease, and arthritis. Furthermore, these organisms have been linked to musculoskeletal disease in broiler breeders and broilers [56][62].3.1. Enterococci Species Diversity
More than 50 different species of enterococci have been described, and E. faecalis, E. faecium, Enterococcus hirae, and Enterococcus durans are the most common species in the gastrointestinal tract of chickens [60][63][64]. E. faecalis and E. faecium are almost entirely responsible for nosocomial enterococcal infections [65]. In poultry, E. faecalis is responsible for increased first-week mortality, amyloid arthropathy in layers, and valvular endocarditis, salpingitis, peritonitis and arthritis in broilers, while E. faecium has been linked to septicemic disease in white Peking ducklings [66][67]. In 1-day-old chicks, E. faecalis and E. faecium can be found mainly in the intestines, while E. durans can be found in the crop. Later, at an age of 3–4 weeks, E. durans can be found in the intestines [68]. A study that included meat samples from turkeys and organic and conventional chickens revealed that E. durans was the most common species isolated from conventional chickens [63]. Moreover, a European study performed with commensal enterococci from healthy cattle, pigs, and chickens revealed that E. durans was among the most prevalent enterococci [69]. E. hirae is the fourth most common Enterococcus species identified in poultry, and it has been demonstrated that these bacteria can colonize the small intestines of 3-week-old chickens, and, even though less frequently, 12-week-old chickens [68][70]. In the past few years, E. hirae has been among the most common species of opportunistic pathogenic bacteria in poultry and can often be isolated from broiler chickens with endocarditis [71][72][73]. Different species can also be identified in particular age groups, such as Enterococcus cecorum in older poultry [68]. In fact, an age-dependent succession of enterococcal species colonization seems to occur in chickens. Chickens are initially colonized by E. faecalis, but this population is then replaced, mostly by E. faecium. This replacement may occur due to the use of tylosin, to which E. faecium is frequently resistant, as a growth promoter. When the chickens mature, these species seem to be replaced by E. cecorum [68][74]. The earliest existence of commensal E. cecorum in the digestive tract of chickens was recognized at the age of 3 to 4 weeks, and by 12 weeks of age, this species was the most prevalent enterococcal component in the crop and intestines of chickens [68][70]. E. cecorum was isolated from the cecal flora of chickens and described as a gastrointestinal commensal of various mammals and birds [75]. However, E. cecorum is an opportunistic pathogen that may also play a role as an etiological agent of diseases in humans (nosocomial infections), chickens, and racing pigeons [72][76][77][78]. Several studies have described this bacterium as an emerging pathogen in the poultry industry [79][80][81][82]. Borst et al. (2017) identified E. cecorum with pathogenic genotypes in one-week-old naturally infected broilers. These autscholars also reported that the ability to colonize the gastrointestinal tract early in life may offer a competitive advantage to pathogenic E. cecorum strains and potentiate dissemination through a flock [83]. E. cecorum has been recognized as a cause of inflammatory musculoskeletal lesions, such as enterococcal spondylitis in chickens, broiler breeders, and broilers in Europe, Canada, and the US [62][83][84][85].3.2. Antimicrobial Resistance in Enterococci
Enterococci have been described as intrinsically resistant to β-lactam antibiotics, such as penicillins (ampicillin, amoxicillin/clavulanic acid, penicillin G, methicillin, piperacillin), carbapenems (imipenem), and cephalosporins (cefoperazone, ceftriaxone) [86]. In addition, they can also be extrinsically resistant due to the accumulation of mutations or the acquisition of exogenous genes. The acquisition of resistance genes frequently occurs by conjugation using pheromone-responsive plasmids, conjugative plasmids with a broad host range, or conjugative transposons with the potential to carry multiple antibiotic resistance genes [87][88]. Enterococci express low-affinity penicillin-binding proteins (PBPs) that are responsible for their weak binding to β-lactam antibiotics [54]. E. faecium isolates from healthy poultry in Portugal have revealed a 30% rate of resistance to ampicillin [89]. Increased production of PBP5 has been associated with acquired resistance to penicillins (penicillin or ampicillin) among clinical E. faecium isolates [54][90]. In Portugal, E. faecium isolated from fecal samples of healthy broilers presented pbp5 genes [91]. In E. faecalis, acquired ampicillin resistance is unusual but is mostly mediated by mutations in the pbp4 gene [92]. Hasan et al. (2018) reported high rates of pbp4 genes in E. faecalis isolated from poultry environments (poultry feces, air, and feed) [59]. Low-level resistance to aminoglycosides, such as streptomycin or gentamicin, is the result of enterococci intrinsic resistance. However, the acquisition of aminoglycoside-modifying enzymes can lead to high-level resistance [87]. Enterococci isolated from broilers with vertebral osteomyelitis have shown high-level aminoglycoside resistance [93]. Kanamycin-resistant E. faecalis isolated from healthy broilers and E. gallinarum isolated from chicken meat contained the aac(6’)–aph(2”) gene [94][95]. This gene is also frequently responsible for gentamicin resistance in enterococci [96]. Gentamicin resistance has been found among enterococci isolated from humans, retail food, and healthy farm animals from six US states [97]. High levels of kanamycin resistance were also identified in E. faecium isolates from healthy poultry in Portugal [89]. Amphenicols are broad-spectrum antibiotics, and due to their toxicity and adverse effects in humans, chloramphenicol and its derivates were banned in 1994 from use in food-producing animals in the EU [98]. Accordingly, a low frequency of chloramphenicol resistance was observed among E. faecium and E. faecalis isolates from healthy broilers in Denmark [95][99]. More recently, a Turkish study performed with broilers from a slaughterhouse revealed a 33.1% rate of Enterococcus species resistant to chloramphenicol [100]. Chloramphenicol-resistant strains usually contain the catpIP501 gene, and horizontal dissemination of phenicol resistance genes among enterococcal isolates may also contribute to the increase in chloramphenicol resistance [54][74]. Accordingly, E. faecium and E. faecalis isolates collected from healthy broilers in Denmark contained the catpIP501 gene [95]. Linezolid, the first clinically available oxazolidinone, is globally used in human medicine as a last-resort antimicrobial agent to treat infections caused by multidrug-resistant Gram-positive pathogens, such as VRE [101][102]. This drug class is not approved for food animals in the USA and EU and, as expected, Tyson et al. (2018) reported very low levels of linezolid-resistant Enterococcus spp. (LRE) from food animal cecal content in the USA, and De Jong et al. (2019) revealed that commensal enterococci from healthy cattle, pigs, and chickens across Europe and broiler breeder farms in Korea were rarely resistant to linezolid [69][103][104]. In the United Arab Emirates, Habib et al. (2022) reported as well low levels of LRE from retail broiler meat [101]. Besides linezolid, daptomycin, which is a cyclic lipopeptide antibiotic, is also used for the treatment of complicated infections caused by Gram-positive organisms. Although there are no daptomycin formulations approved for animal use in the EU, Diarra et al. (2010) were able to isolate two daptomycin-resistant Enterococcus spp. (DRE) from broiler chickens [64][105]. The absence or very low levels of clinical resistance to several antibiotics that are highly valuable for human medicine, such as linezolid and daptomycin, is encouraging [69]. However, these findings add to the importance of monitoring the emergence of LRE and DRE at retail and farm levels. Cross-resistance to linezolid is attributed to different groups of acquired resistance genes [88]. Among them are the cfr gene, which confers transferable resistance to oxazolidinones, phenicols, lincosamides, pleuromutilins, and streptogramin A, and the optrA gene, which confers resistance to linezolid, tedizolid, chloramphenicol, and florfenicol [106][107]. Although not common among food animals, the cfr gene has been detected in E. faecalis isolated from retail chicken meat [108][109]. The optrA gene as well as the fexA gene were found among E. faecalis isolated from fecal samples of broilers [103]. A Chinese study reported that the optrA gene was more frequently detected in enterococci from food-producing animals (15.9%) than in humans (2.0%), which might suggest an animal reservoir or that the optrA gene has disseminated more quickly in enterococci of animal origin due to the selective pressure imposed by the use of florfenicol [106]. A high level of resistance against macrolide-lincosamide-streptogramin B (MLSB) has been shown by enterococci from the internal organs of healthy and diseased poultry. In addition, enterococci that express the ermB gene can also exhibit resistance to tetracycline [72][110]. Tetracycline-resistant Enterococcus isolates harboring tetL, tetM, tetO, or tetS in association with the ermB gene encoding resistance to MLSB have been isolated from fecal and cloacal samples from broilers [64][110]. Vancomycin and teicoplanin are important members of the glycopeptides class, and resistance to vancomycin has been recently detected among 11% of the enterococci collected from cecal samples of healthy broilers at a Swedish slaughterhouse [54][111]. Glycopeptide resistance determinants have been detected in all farm species, and the mechanism of resistance usually involves altering the peptidoglycan synthesis pathway [54][112]. E. faecium, E. faecalis, and E. hirae isolated from broilers carried the vanA gene, as did VRE isolated from a Norwegian broiler production facility [49][113][114]. The vanC gene has been detected in E. gallinarum isolated from fecal and cecal samples of broilers [64].3.3. Emergence and Dissemination of Vancomycin-Resistant Enterococci (VRE)
Enterococci were the first pathogens to show acquired resistance to vancomycin, and they emerged in the late 1970s as leading hospital-associated pathogens likely due to the extensive use of vancomycin to treat enterococcal infections [87][115][116]. The use of avoparcin as a growth promoter in farm animals may have also contributed to the emergence of vancomycin-resistant Enterococcus spp. (VRE) [39]. Moreover, in Europe, the VRE problem was initially confined to livestock, and VRE was observed in animals regularly exposed to antibiotics [95][114]. In the late 1990s, several food-producing animals, healthy humans, food products, and environmental samples, all over Europe and other countries, were colonized by VRE [49][89][94][114][117][118]. When avoparcin was banned as a growth promoter in the European Union, a decrease in VRE fecal carriage in animal meat products and human fecal flora was observed in a German study [119]. However, many reports suggested that VRE persisted in food animals. However, many reports suggested that VRE persisted in food animals. A Norwegian study documented a high prevalence of VRE in broiler and turkey carcasses three years after avoparcin was banned in Norway [49]. Denmark also presented similar findings [118]. In Sweden, the proportion of VRE-positive samples from healthy broilers increased from less than 1% in 2000 to over 40% in 2005 [113]. More recently, Leinweber et al. (2018) reported a 7.5% prevalence of vancomycin-resistant E. faecium in retail chicken meat [120]. Currently, VRE represents a serious threat to global health [121]. Resistance to vancomycin in enterococci has been mainly associated with the vanA and vanB gene clusters that allow the synthesis of different cell wall precursors with little affinity to vancomycin [105][122]. VRE containing the vanA gene are considered endemic, and they have been previously reported in human and animal samples, as well as in food and water [48][123][124][125]. Moreover, some enterococcal species, such as E. gallinarum and E. casseliflavus, have shown a different vancomycin resistance mechanism, related to a chromosomally encoded VanC operon [64][126]. All of these vast resistance characteristics limit therapeutic options, particularly the antibiotic treatment of nosocomial infections in humans and multiple diseases in poultry [56].3.4. Molecular Characteristics of Enterococcus Clones
Stępień-Pyśniak et al. (2021) carried out a study that included 35 Polish E. faecalis and 41 Danish E. faecalis strains collected during post-mortem examination from broiler chicks showing lesions characteristic of yolk sac infection. The most prevalent clonal lineage among the Polish isolates was ST59, followed by ST282 and ST16. Regarding the Danish isolates, the most prevalent clonal lineages were ST116 and ST16. Only two Danish isolates were identified as VRE, and one belonged to the ST387 clonal lineage, while the other belonged to ST838 [127]. A Brazilian study that analyzed 12 E. faecalis strains isolated from natural cases of vertebral osteomyelitis in broilers revealed that almost half of these belonged to ST49. In addition, ST202 was represented by one strain that was vancomycin-resistant [93]. A study performed with 45 E. faecalis strains isolated from the cloaca of healthy broilers in Saudi Arabia reported that most of those strains belonged to ST16, ST302, and ST179, respectively. Two isolates were VRE, and these also belonged to ST16 [128]. In China, 61 strains of E. faecalis isolated from the cecal tissue of broiler chickens with swollen cecal lesions belonged to 34 sequence types, and the most prevalent was ST631 [129]. Kim et al. (2018) studied the molecular characteristics of 85 E. faecalis strains isolated from chicken meat samples, and ST256 was observed in over 50% of the isolates [130]. E. faecalis strains isolated from retail chicken carcasses in the Emirate of Abu Dhabi were assigned to five different sequence types, and half of them belonged to the clonal lineage ST476 [101]. ST314, followed by ST16, were the most prevalent clonal lineages reported among broilers across Australia [60]. Overall, the most frequent and wide-ranging clonal lineage that has been identified among E. faecalis isolated from broilers or broiler meat since 2018 is ST16. This sequence type has already been identified in Poland, the Netherlands, Saudi Arabia, China, and Australia, and in both vancomycin-resistant and vancomycin-susceptible E. faecalis.
Leinweber et al. (2018) isolated three vancomycin-resistant E. faecium (VREfm) strains from Danish chicken meat, and all the strains belonged to ST32 [120]. VREfm strains were also isolated from cecal samples from healthy broilers in Sweden, but all of these belonged to ST310 [111]. In Turkey, a study that included vancomycin-susceptible E. faecium and VREfm isolated from broiler cloaca reported that ST1346 was the most prevalent clonal lineage among vancomycin-susceptible E. faecium, while all VREfm presented different and novel STs (ST1341, ST1342, ST1343, ST1244, and ST1345) [131]. A study that included 30 E. faecium strains isolated from the cloaca of healthy broilers in Saudi Arabia reported that most of those strains belonged to ST194, ST82, and ST157, respectively [128]. Kim et al. (2018) isolated one E. faecium strain from chicken meat samples that was revealed to belong to ST451 [130]. E. faecium isolated from retail chicken carcasses in Abu Dhabi Emirate has been assigned to four different sequence types: one known ST (ST195) and three novel STs (ST2236, ST2238, and ST2239) [101]. ST492, followed by ST195 and ST241, were the most prevalent clonal lineages reported among broilers across Australia [60]. Overall, E. faecium isolates from broilers or broiler meat since 2018 do not share many clonal lineages. However, ST194 and ST195 were already identified in two different sources (broilers and broiler meat) on at least two different continents.
4. Escherichia coli
E. coli are facultative, anaerobic Gram-negative rods that can be found in the intestinal tract of food-production animals and humans [132][133][134]. They are commonly acknowledged as antimicrobial resistance indicators in Gram-negative bacterial populations and are a model for antimicrobial resistance surveillance studies [23][134]. This bacterium has a special place in the microbiological world since it represents a substantial part of the endemic microbiota of different hosts and can also cause severe infections in humans and animals [135]. E. coli can be classified into different pathotypes capable of causing various diseases. Intestinal pathogenic E. coli (IPEC) are responsible for disorders in the gastrointestinal tract ranging from mild diarrhea to severe colitis [136][137][138]. In contrast, extraintestinal pathogenic E. coli (ExPEC) are mainly asymptomatic inhabitants of the intestinal tract that can cause extra-intestinal diseases after migrating to other body parts, such as the urinary tract or the bloodstream [139][140]. ExPEC has already been isolated from healthy production chickens in Canada, from diseased broiler chickens in Egypt, and from meat chickens in Australia [141][142][143]. Avian pathogenic E. coli (APEC), a subset of ExPEC, is mainly responsible for respiratory or systemic infections in poultry [144][145]. Additionally, in poultry production, it is a major cause of colibacillosis, which is considered the main cause of decreased productivity and increased mortality, leading to major economic losses [146][147][148]. Colibacillosis is characterized by acute fatal septicemia or sub-acute fibrinous pericarditis, airsacculitis, salpingitis, and peritonitis [149]. Good husbandry, strict biosecurity, and vaccination are essential to prevent colibacillosis. Vaccination against colibacillosis is generating interest, and Śmiałek et al. (2020) have already reported that vaccination decreased the number of E. coli isolates from broilers of commercial farms and that these isolates were more susceptible to the antimicrobials [150]. Ebrahimi-Nik et al. (2018) also showed an efficient vaccine against colibacillosis in poultry [151]. However, since different strains can cause outbreaks, it is challenging to develop a vaccine that is effective against multiple strains [152].4.1. Antimicrobial Resistance in E. coli
In recent decades, a growing number of resistance genes have been identified in E. coli isolates, and many of these were acquired by horizontal gene transfer. E. coli can act as a donor or a recipient of resistance genes, so resistance genes can be passed on or acquired by E. coli [135]. Ampicillin is a β-lactam antibiotic, and resistance to β-lactams in Gram-negative bacteria is primarily mediated by β-lactamase enzymes that hydrolyze the β-lactam ring, thereby inactivating the drug [145]. A study performed with cloacal swab samples from apparently healthy broilers revealed that the E. coli isolates were 100% resistant to ampicillin [153]. Ampicillin-resistant E. coli were screened for several genes, and the most frequently found were blaTEM, blaSHV, blaOXA, blaCMY, and blaCTX-M [154][155]. Al Azad et al. (2019) and Sarker et al. (2019) both revealed a high prevalence of blaTEM in E. coli isolated from cloacal swabs of broiler chickens [153][156]. The genes blaCTX, blaCMY, and blaSHV have also been identified in E. coli isolated from broilers [155][157]. Tetracyclines are among the most common therapeutic agents used in animals. A. M. Ahmed et al. (2013), reported that 91.8% of the APEC isolates from septicemic broilers in Egypt harbored tetracycline resistance determinants [145]. Tetracycline resistance in broilers is possibly due to the acquisition of the tetA gene [158]. A study that included cecal samples from healthy broilers and broiler meat revealed a high prevalence of antimicrobial resistance, particularly quinolone resistance [159]. Regarding the quinolone-resistant genes, qnrA and qnrS play an important role in broiler chickens [160][161]. De Koster et al. (2021) reported resistance to ciprofloxacin in E. coli isolated from Belgian and Dutch broiler farms [162]. Mutations in the gyrA and gyrB genes could be the molecular mechanisms responsible for the acquisition of ciprofloxacin resistance [158]. Resistance of E. coli from broiler breeding animals, that had just arrived in Sweden, to nalidixic acid was identified by Börjesson et al. (2016), suggesting that the importation of birds can be a source of the occurrence of these bacteria in Swedish broiler production [163]. Sulfonamides are listed for use in poultry in all countries and, according to Roth et al. (2019), the resistance rates in E. coli of broiler origin to sulfamethoxazole are higher than 40% in all countries [23]. One Portuguese research study focused on the resistance of E. coli isolated from carcasses and internal organs of healthy chickens from intensive farms detected sul1 as the most common gene of the sulfonamide class [164]. On the other hand, the sul2 gene was the most prevalent gene detected in isolates from broilers, Danish broiler meat, and imported broiler meat [165]. However, when analyzing the genes involved in sulfamethoxazole-trimethoprim resistance, it is necessary to consider the genes sul and dfr, since they act synergistically to confer resistance [166]. Genes dfrA1, dfrA12, dfrA14, and dfrA17 were the most commonly identified genes in trimethoprim-resistant strains of avian fecal E. coli recovered from clinically healthy chickens [167].4.2. ESBL-Producing E. coli
As a member of the Enterobacteriaceae family, E. coli can produce extended-spectrum β-lactamases (ESBLs) [168][169]. ESBLs are enzymes that can degrade extended-spectrum β-lactam antibiotics, such as third-generation cephalosporins, commonly used to treat numerous systemic infections [170]. Organisms capable of producing ESBLs were first reported in Europe in the early 1980s, and since then, their prevalence rates increased [171][172]. ESBL-producing E. coli are becoming the most challenging multidrug-resistant pathogens worldwide, and they have been extensively described among broilers [133][155][173][174][175]. In particular, Rousham et al. (2021) reported a high prevalence of ESBL-producing E. coli in broiler ceca and feces in households, farms, and live poultry. Furthermore, this study also revealed that the majority of the isolates were resistant to fluoroquinolones, cefepime, sulfonamides, and aminoglycosides [42]. Interestingly, a study performed by Van Hoek et al. (2018) showed that almost 30% of one-day-old broilers were already ESBL-positive [44]. ESBLs can be categorized into three main subtypes: TEM, SHV, and CTX-M β-lactamases. The TEM and SHV subtypes are large and widespread groups that differ from their parental enzymes by one or two amino acids [176]. However, these minor alterations in their amino acid sequences are sufficient to extend the spectrum of their enzymatic activity, which allows them to hydrolyze cephalosporins that have an oxyimino side chain, such as third-generation cephalosporins and aztreonam [177]. Both TEM and SHV subtypes were reported in the United States and France in the late 1980s and 1990s [169][178]. Unlike other ESBLs, the CTX-M family is a heterogenous and complex group of enzymes that possibly resulted from the relocation of chromosomal Kluyvera genes and that can confer resistance to cefotaxime and ceftazidime [179]. TEM and SHV types were the prevailing ESBL enzymes worldwide for a long time. Now, CTX-M enzymes may represent the most prevalent subtype of ESBLs [180][181][182]. Worryingly, a significant proportion of ESBL-producing isolates are represented by E. coli-expressing CTX-M β-lactamases that have quickly spread not only among healthcare settings but also in the community [42][139]. The spread of CTX-M variants in animals and humans is responsible for the high frequency of ESBLs [132][183][184]. An Indonesian study revealed a prevalence of almost 97.8% of CTX-M-producing E. coli among broilers’ cloacal swabs [174]. Currently, over 123 types of CTX-M have been identified [139]. CTX-M-14 and CTX-M-15 are extensively disseminated among chickens [185][186][187]. Still, the CTX-M-1 gene has also been reported as one of the common CTX-M types that have been recovered from poultry in many European countries [140][155][188][189]. Liu et al. (2020) revealed that CTX-M-14 was the most predominant CTX-M subtype identified among apparently healthy broiler chickens, and CTX-M-14 has also been detected in broiler meat in Portugal [190][191]. Subramanya et al. (2020) collected samples from healthy poultry from backyard farms and commercial broiler farms, and their data indicated that CTX-M-15 was the most prevalent ESBL enzyme [192]. CTX-M-15-producing E. coli is strongly linked to sequence type 131 (ST131) clones, which are related to fluoroquinolone resistance [168][193]. Many European countries use fluoroquinolones in farm animals that could be related to the fluoroquinolone-resistant E. coli strains [194]. Moreover, fluoroquinolones are approved for use in poultry in the largest poultry-producing countries, with the exception of the US [23][195]. However, a study from Awawdeh et al. (2022) reported fluoroquinolone-resistant E. coli from meat chickens in Australia, a country that does not use fluoroquinolones in poultry, which suggests that this resistance is likely due to horizontal transmission of antibiotic-resistant genes [143][196]. The CTX-M-1 gene was described in commensal isolates from French layers and healthy poultry [188][197]. Moreover, a Dutch study that collected samples from an organic broiler farm revealed that all E. coli isolates carried CTX-M-1 genes [44].4.3. Molecular Characteristics of E. coli Clones
Päivärinta et al. (2020) collected broiler cecal samples from a high-capacity slaughterhouse and from vacuum-packed raw broiler meat without marinade intended for consumer use, all from the same high-capacity slaughterhouse. In total, three ESBL-producing E. coli strains were isolated: two from the ceca that belonged to ST1594, and one from the meat that belonged to ST351 [175]. Retail chicken meat was also studied in Egypt, and ST1196 was the most prevalent sequence type among ESBL-producing E. coli, while ST156 and ST189 were identified among non-ESBL-producing E.coli [198]. Broilers infected with colibacillosis were studied in Norway, Croatia, Tunisia, and Pakistan [144][147][199][200]. In the Norwegian study, ST429 accounted for over 60% of the clonal lineages identified in E. coli isolates [144]. However, in Croatia, ST429 was reported at a much lower rate (0.65%). The most prevalent sequence types in Croatia were ST95 and ST117 [199]. ST117 was also predominant among the Pakistani E. coli isolates from broilers with colibacillosis [201]. The Tunisian study reported four different sequence types in ESBL-producing E. coli strains, with the majority belonging to ST4187 [147]. Two different Pakistani studies that included cecal and fecal samples from broilers reported ST131 between the most prevalent sequence types in ESBL-producing E. coli strains [202][203]. A study carried out by Aslantaş (2020) in Turkey detected 19 sequence types in 28 ESBL-producing E. coli isolates, and the most prevalent were ST114 and ST354 [200]. In Australia, ESBL-producing E. coli isolated from healthy broilers belonged to different clonal lineages, while E. coli from chickens with colibacillosis belonged mainly to ST354 [143]. Overall, according to the studies mentioned in Table 5, the most frequent and wide-ranging clonal lineage that was identified in both ESBL-producing and non-ESBL-producing E. coli isolated from broilers or broiler meat since 2020 was ST117.
5. Impact of Antibiotic Usage and Antibiotic-Resistant Bacteria in the Gastrointestinal Tract of Broilers: A One Health Approach
The use and abuse of antibiotics select and enrich antibiotic-resistant bacteria in the gastrointestinal microbiota of food animals, particularly broilers [204]. Therefore, antibiotic-resistant bacteria may be carried in the large intestine of adult laying hens and shed in their feces, leading to contamination of the eggshell surface [3]. Specific foodborne and poultry pathogens found on the eggshell surface might infect the hatchlings and consequently affect the health of the growing broiler and their derived meat products [205]. The antimicrobial-resistant bacteria that have emerged and live in the animal production environment can spread to humans through human–animal contact or the consumption of or contact with animal products [120][121][206]. Furthermore, during food processing, when an animal is slaughtered, the muscles are exposed and can be cross-contaminated if the gastrointestinal tract ruptures or if contaminated instruments and materials are used [207]. An American study reported that 95% of retail chicken meat samples were contaminated with enterococci, mainly with E. faecium, followed by E. faecalis [57]. Adeyanju and Ishola (2014) revealed that almost 44% of retail chicken samples from Nigeria presented E. coli [208]. Antibiotic usage is the most important factor that provides the selection pressure that enables the dissemination of antimicrobial resistance genes, and unfortunately, antibiotic exposure is not only caused by antibiotic consumption [209][210]. Almost 90% of the administered doses of antibiotics are excreted unmodified or partly metabolized through urine and feces [25]. Animal manure is acknowledged as a rich reservoir of antibiotic residues, and its use as crop fertilizer exposes the environment to antibiotic determinants [13]. Antibiotic-resistance genes can persist in soils for several weeks, and their transmission to crops, and therefore, to animals or humans when consumed, represents a health risk [211]. Discharges from the wastewater treatment process also represent a way for resistant bacteria to enter the environment [212]. Once in the environment, the bacterial resistance can be transferred to wild animals, such as wild birds, particularly migratory raptors, who travel long distances through different ecological niches and prey on synanthropic rodents and small birds in urban and rural environments (Figure 2) [121][213].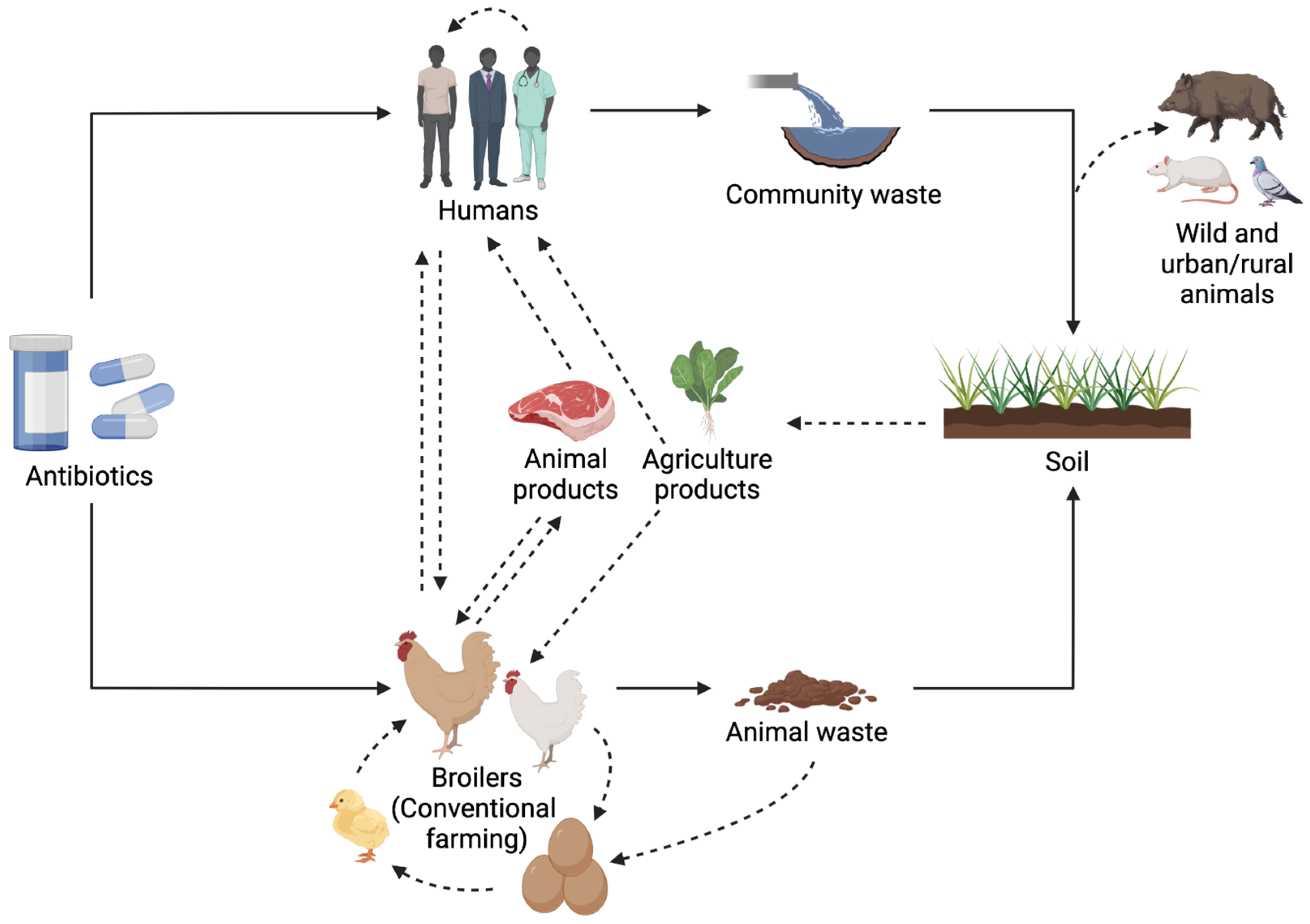
Recently, broilers have increased significantly as a meat source, and the largest broiler meat producers worldwide include the United States, China, and Brazil, respectively. Within the European Union, Poland, Spain, Germany, France, and Italy present the higher gross domestic production of broilers [217]. Broiler meat produced by some of these countries is exported globally [23]. For example, Brazil is the world’s largest poultry exporter; about a third of Brazil’s chicken production is exported—4.6 million out of 14.3 million metric tons in 2020—to over 150 countries worldwide [218]. Therefore, ongoing surveillance systems for antimicrobial resistance in broiler production are mandatory to avoid the spread of antimicrobial resistance among broiler meat or other foods derived from these animals.
5.1. Transmission of Enterococcus spp. and E. coli
5.2. Clonal Relationship from a One Health Perspective
E. faecalis ST16 isolated from yolk sac infections was previously characterized as an epidemic clone in hospitals in Poland and other European countries [243][244]. Its presence has also been detected in many animals, such as poultry, pigs, and cattle [245]. vanA E. faecalis ST116 isolates were isolated from turkey meat, non-hospitalized humans, and patients [122]. E. faecalis sequence types ST4, ST59, ST82, ST116, and ST245 have been found in hospitalized patients [245]. ST49 was detected more frequently in hospitalized human patients than in non-hospitalized human patients [246]. Furthermore, ST16, ST21, ST179, and ST480 have been reported among E. faecalis hospital isolates in Saudi Arabia [247]. In China, human ST631 derived from E. faecalis primarily manifests in diseases of the abdominal cavity, gastrointestinal tract, and other related sites [129]. ST256 has been isolated from chickens, pigs, and humans and has shown a high prevalence of multidrug resistance [130][248][249]. Furthermore, E. faecalis ST256 carries the optrA gene, which is related to oxazolidinone and phenicol resistance [106][248]. Freitas et al. (2020) reported genetic relatedness between optrA-positive E. faecalis of ST476 in animal and clinical (human) hosts worldwide over several decades [250]. Alzahrani et al. (2022), described E. faecium isolates from eight different sequence types in cloacal swabs from healthy broilers. Five of these belonged to CC9 (ST9, ST157, ST82, ST194, and ST12) and three to CC17 (ST16, ST18, and ST360) [128]. CC17 was considered a nosocomial clonal complex, but several studies have reported the dissemination of E. faecium CC17 in animals [122][219]. Chickens can possibly acquire the CC17 E. faecium isolates from contaminated environments or humans visiting the farm. This idea is reinforced by a study that showed the transmission of E. faecium of human origin to chickens [251]. E. faecium ST492 isolates found in broilers were clustered with the human isolates, which may also indicate reverse zoonotic transmission from humans to chickens along the production chain [60]. Two cecal E. coli ST1594 isolates that came from two different flocks from the same farm carried blaCTX-M-1 and sul2 resistance genes. These findings indicate that clonal transfer of blaCTX-M-1 and sul2 genes may occur between different E. coli ST1594 strains [175]. MLST findings by Ramadan et al. (2020) showed overlapping E. coli STs from different sources: ST1011, ST156, ST48, and ST224 in chicken and beef isolates; ST10 in human and chicken; and ST226 in human and beef isolates. This suggests the adaptability of some STs to distinctive hosts with a potential for inter-species transmission [198]. Furthermore, ST10 and ST48 belonged to CC10, which is linked to diarrheagenic E. coli infections in humans worldwide [252]. E. coli ST429 has been identified as APEC [253]. ST95 has also been previously associated with APEC. In addition, uropathogenic E. coli ST95 has been isolated from humans [254]. In the study carried out by Kravik et al. (2018), both ST429 and ST95 were analyzed to deduce their phylogenetic relationship, and ST429 revealed a high sequence resemblance between isolates from the same flock, while ST95 isolates from a single flock were more diverse [144]. ST69, ST23, and ST131 are also frequently responsible for extraintestinal infections in humans and poultry [255][256][257]. As a carrier of many resistance and virulence-associated genes, ST131 has been often described as the accountable agent for human urinary infections and bacteremia [258]. ST131 is considered a well-established pandemic clone, and it was isolated from poultry samples from different European countries [132]. Jouini et al. (2021) isolated the pandemic high-risk human lineage CTX-M-15-B2-O25b-ST131 E. coli from diseased chickens in Tunisia [136]. ST117 was already reported in several Nordic countries, as well as in Canada [186][259]. The ST4187 lineage has been considered relevant regarding the spread of mcr-1-mediated colistin resistance and ESBL-encoding genes in E. coli isolates from broilers with colibacillosis [147]. ST4187 was also described in E. coli isolated from hospitalization units in Angola and birds from Chile [260][261].6. Conclusions
Antimicrobials have played an essential role in diminishing mortality and morbidity rates in animal production. However, their misuse is considered one of the major threats to public health. The inappropriate application of antibiotics contributed to the selection and enrichment of antibiotic-resistant bacteria in the gastrointestinal microbiota of animals, and the consumption of contaminated animal-source food, direct contact with animals, or environmental exposure can lead to the transmission of antimicrobial-resistant bacteria to humans. In addition, food-animal waste may contain antimicrobial residues that will lead to the contamination of the environment, and consequently, to the spread of antimicrobial resistance to other sources. The results presented in this review cannot exclude the possibility that pathotypes of Enterococcus spp. and E. coli isolated from broilers might represent transmission to or from humans. Infections by antibiotic-resistant bacteria are an increasing problem, and antimicrobial resistance can be responsible for treatment failures for both animal and human diseases, which have significant economic and public health consequences, such as prolonged treatment and extended hospital stays, which might further promote the transmission of resistant pathogens in hospitals and represent a financial burden. Furthermore, results concerning foodborne strains suggest that the food chain also represents a possible means of bacterial infection in humans. Therefore, the inappropriate use of antimicrobials in broiler chicken production is a primary concern, and it is imperative to restrict the use of critically important antibiotics for humans in food animals and explore antibiotic alternatives for animal production. Practices to prevent bacterial cross-contamination and manure treatment options that avoid the dissemination of antibiotic resistance into the environment are also important. In addition, to prevent and control the spread of antibiotic resistance, individuals should only use antibiotics when and as prescribed by a certified health professional, never share or use leftover antibiotics, prevent infections by regularly washing hands, avoiding close contact with sick people, and keeping vaccinations up to date, prepare food hygienically, and choose foods that have been produced without the use of antibiotics for growth promotion or disease prevention in healthy animals. To better understand the potential of antimicrobial resistance transmission, more studies regarding human and veterinary epidemiology are needed.References
- Stanley, D.; Hughes, R.J.; Moore, R.J. Microbiota of the chicken gastrointestinal tract: Influence on health, productivity and disease. Appl. Microbiol. Biotechnol. 2014, 98, 4301–4310.
- Chen, C.Y.; Chen, C.K.; Chen, Y.Y.; Fang, A.; Shaw, G.T.W.; Hung, C.M.; Wang, D. Maternal gut microbes shape the early-life assembly of gut microbiota in passerine chicks via nests. Microbiome 2020, 8, 129.
- Olsen, R.; Kudirkiene, E.; Thøfner, I.; Pors, S.; Karlskov-Mortensen, P.; Li, L.; Papasolomontos, S.; Angastiniotou, C.; Christensen, J. Impact of egg disinfection of hatching eggs on the eggshell microbiome and bacterial load. Poult. Sci. 2017, 96, 3901–3911.
- Fathima, S.; Shanmugasundaram, R.; Adams, D.; Selvaraj, R.K. Gastrointestinal Microbiota and Their Manipulation for Improved Growth and Performance in Chickens. Foods 2022, 11, 1401.
- Yadav, S.; Jha, R. Strategies to modulate the intestinal microbiota and their effects on nutrient utilization, performance, and health of poultry. J. Anim. Sci. Biotechnol. 2019, 10, 2.
- Shang, Y.; Kumar, S.; Oakley, B.; Kim, W.K. Chicken gut microbiota: Importance and detection technology. Front. Vet. Sci. 2018, 5.
- Hinton, A.; Buhr, R.J.; Ingram, K.D. Physical, Chemical, and Microbiological Changes in the Crop of Broiler Chickens Subjected to Incremental Feed Withdrawal. Poult. Sci. 2000, 79, 212–218.
- Gong, J.; Si, W.; Forster, R.J.; Huang, R.; Yu, H.; Yin, Y.; Yang, C.; Han, Y. 16S rRNA gene-based analysis of mucosa-associated bacterial community and phylogeny in the chicken gastrointestinal tracts: From crops to ceca. FEMS Microbiol. Ecol. 2006, 59, 147–157.
- Videnska, P.; Faldynova, M.; Juricova, H.; Babak, V.; Sisak, F.; Havlickova, H.; Rychlik, I. Chicken faecal microbiota and disturbances induced by single or repeated therapy with tetracycline and streptomycin. BMC Vet. Res. 2013, 9, 6–8.
- Takahashi, M.; Kametaka, M.; Mitsuoka, T. Influence of Diets Low in Protein or Lysine on the Intestinal Flora of Chicks with Reference to Cecal Contents. J. Nutr. Sci. Vitaminol. 1982, 28, 501–510.
- Lu, J.; Idris, U.; Harmon, B.; Hofacre, C.; Maurer, J.J.; Lee, M.D. Diversity and Succession of the Intestinal Bacterial Community of the Maturing Broiler Chicken. Appl. Environ. Microbiol. 2003, 69, 6816–6824.
- Gong, J.; Forster, R.J.; Yu, H.; Chambers, J.R.; Sabour, P.M.; Wheatcroft, R.; Chen, S. Diversity and phylogenetic analysis of bacteria in the mucosa of chicken ceca and comparison with bacteria in the cecal lumen. FEMS Microbiol. Lett. 2002, 208, 1–7.
- Subirats, J.; Murray, R.; Scott, A.; Lau, C.H.F.; Topp, E. Composting of chicken litter from commercial broiler farms reduces the abundance of viable enteric bacteria, Firmicutes, and selected antibiotic resistance genes. Sci. Total Environ. 2020, 746, 141113.
- Rychlik, I. Composition and function of chicken gut microbiota. Animals 2020, 10, 103.
- Dhama, K.; Rajagunalan, S.; Chakraborty, S.; Verma, A.K.; Kumar, A.; Tiwari, R.; Kapoor, S. Food-borne Pathogens of Animal Origin-Diagnosis, Prevention, Control and Their Zoonotic Significance: A Review. Pakistan J. Biol. Sci. 2013, 16, 1076–1085.
- Edwards, P.; Zhang, W.; Belton, B.; Little, D.C. Misunderstandings, myths and mantras in aquaculture: Its contribution to world food supplies has been systematically over reported. Mar. Policy 2019, 106, 103547.
- FAO. World Agriculture: Towards 2030/2050 Prospects for Food, Nutrition, Agriculture and Major Commodity Groups; FAO: Yokohama, Japan, 2006.
- GPP/GOV Análise Setorial Carne de Aves; Gabinete de Planeamento, Políticas e Administração Geral: Lisboa, Portugal, 2020.
- Pereira, J.L.S.; Ferreira, S.; Pinheiro, V.; Trindade, H. Ammonia, Nitrous Oxide, Carbon Dioxide and Methane Emissions from Commercial Broiler Houses in Mediterranean Portugal. Water. Air. Soil Pollut. 2018, 229, 377.
- INE Estatísticas Agrícolas 2021; Instituto Nacional De Estatística: Lisboa, Portugal, 2022.
- Patel, S.J.; Wellington, M.; Shah, R.M.; Ferreira, M.J. Antibiotic Stewardship in Food-producing Animals: Challenges, Progress, and Opportunities. Clin. Ther. 2020, 42, 1649–1658.
- ESVAC. Sales of Veterinary Antimicrobial Agents in 31 European Countries in 2019 and 2020. Trends from 2010 to 2020 (EMA/294674/2019); European Medicines Agency: Zuidas, Amsterdam, The Netherlands, 2019; ISBN 9789291550685.
- Roth, N.; Käsbohrer, A.; Mayrhofer, S.; Zitz, U.; Hofacre, C.; Domig, K.J. The application of antibiotics in broiler production and the resulting antibiotic resistance in Escherichia coli: A global overview. Poult. Sci. 2019, 98, 1791–1804.
- Poole, T.; Sheffield, C. Use and Misuse of Antimicrobial Drugs in Poultry and Livestock: Mechanisms of Antimicrobial Resistance. Pak. Vet. J. 2013, 33, 85–92.
- Diarra, M.S.; Malouin, F. Antibiotics in Canadian poultry productions and anticipated alternatives. Front. Microbiol. 2014, 5, 282.
- European Commission. Ban on Antibiotics as Growth Promoters in Animal Feed Enters Into Effect; European Commission: Brussels, Belgium, 2005.
- Lee, J.H.; Cho, S.; Paik, H.D.; Choi, C.W.; Nam, K.T.; Hwang, S.G.; Kim, S.K. Investigation on antibacterial and antioxidant activities, phenolic and flavonoid contents of some thai edible plants as an alternative for antibiotics. Asian-Australas. J. Anim. Sci. 2014, 27, 1461–1468.
- Bager, F. DANMAP: Monitoring antimicrobial resistance in Denmark. Int. J. Antimicrob. Agents 2000, 14, 271–274.
- PHAC. Canadian Integrated Program for Antimicrobial Resistance Surveillance (CIPARS) 2017: Design and Methods; PHAC: Ottawa, ON, Canada, 2017.
- Shimazaki, Y.; Ozawa, M.; Matsuda, M.; Akama, R.; Shirakawa, T.; Furuya, Y.; Harada, S. Contributors: Report on the Japanese Veterinary Antimicrobial Resistance Monitoring System; National Veterinary Assay Laboratory Ministry of Agriculture, Forestry and Fisheries: Tokyo, Japan, 2020.
- Wallinga, D.; Smit, L.A.M.; Davis, M.F.; Casey, J.A.; Nachman, K.E. A Review of the Effectiveness of Current US Policies on Antimicrobial Use in Meat and Poultry Production. Curr. Environ. Health Rep. 2022, 9, 339–354.
- ESVAC. Sales of Veterinary Antimicrobial Agents in 30 European Countries in 2015. Trends from 2010 to 2015 (EMA/184855/2017); European Medicines Agency: Zuidas, Amsterdam, The Netherlands, 2017.
- Singer, R.S.; Porter, L.J.; Thomson, D.U.; Gage, M.; Beaudoin, A.; Wishnie, J.K. Raising Animals Without Antibiotics: U.S. Producer and Veterinarian Experiences and Opinions. Front. Vet. Sci. 2019, 6, 452.
- McKeith, A.; Loper, M.; Tarrant, K.J. Research Note: Stocking density effects on production qualities of broilers raised without the use of antibiotics. Poult. Sci. 2019, 99, 698–701.
- Seidavi, A.; Tavakoli, M.; Slozhenkina, M.; Gorlov, I.; Hashem, N.M.; Asroosh, F.; Taha, A.E.; Abd El-Hack, M.E.; Swelum, A.A. The use of some plant-derived products as effective alternatives to antibiotic growth promoters in organic poultry production: A review. Environ. Sci. Pollut. Res. 2021, 28, 47856–47868.
- Magnusson, U.; Lewerin, S.S.; Eklund, G.; Rozstalnyy, A. Prudent and Efficient Use of Antimicrobials in Pigs and Poultry; FAO: Rome, Italy, 2019.
- Rajput, D.S.; Zeng, D.; Khalique, A.; Rajput, S.S.; Wang, H.; Zhao, Y.; Sun, N.; Ni, X. Pretreatment with probiotics ameliorate gut health and necrotic enteritis in broiler chickens, a substitute to antibiotics. AMB Express 2020, 10, 220.
- Wijesekara, P.N.K.; Kumbukgolla, W.W.; Jayaweera, J.A.A.S.; Rawat, D. Review on usage of vancomycin in livestock and humans: Maintaining its efficacy, prevention of resistance and alternative therapy. Vet. Sci. 2017, 4, 6.
- Simm, R.; Slettemeås, J.S.; Norström, M.; Dean, K.R.; Kaldhusdal, M.; Urdahl, A.M. Significant reduction of vancomycin resistant E. faecium in the Norwegian broiler population coincided with measures taken by the broiler industry to reduce antimicrobial resistant bacteria. PLoS ONE 2019, 14, e0226101.
- Sabença, C.; de Sousa, T.; Oliveira, S.; Viala, D.; Théron, L.; Chambon, C.; Hébraud, M.; Beyrouthy, R.; Bonnet, R.; Caniça, M.; et al. Next-Generation Sequencing and MALDI Mass Spectrometry in the Study of Multiresistant Processed Meat Vancomycin-Resistant Enterococci (VRE). Biology 2020, 9, 89.
- Bacanlı, M.; Başaran, N. Importance of antibiotic residues in animal food. Food Chem. Toxicol. 2019, 125, 462–466.
- Rousham, E.K.; Asaduzzaman, M.; Amin Uddin Mozmader, T.I.M.; Amin, M.B.; Rahman, M.; Hossain, M.I.; Islam, M.R.; Mahmud, Z.H.; Unicomb, L.; Islam, M.A. Human colonization with extended-spectrum beta-lactamase-producing E. coli in relation to animal and environmental exposures in bangladesh: An observational one health study. Environ. Health Perspect. 2021, 129, 037001.
- Ferreira, M.; Leão, C.; Clemente, L.; Albuquerque, T.; Amaro, A. Antibiotic Susceptibility Profiles and Resistance Mechanisms to β-Lactams and Polymyxins of Escherichia coli from Broilers Raised under Intensive and Extensive Production Systems. Microorganisms 2022, 10, 2044.
- Van Hoek, A.H.A.M.; Veenman, C.; Florijn, A.; Huijbers, P.M.C.; Graat, E.A.M.; De Greeff, S.; Dierikx, C.M.; Van Duijkeren, E. Longitudinal study of ESBL Escherichia coli carriage on an organic broiler farm. J. Antimicrob. Chemother. 2018, 73, 3298–3304.
- Subramanya, S.H.; Bairy, I.; Metok, Y.; Baral, B.P.; Gautam, D.; Nayak, N. Detection and characterization of ESBL-producing Enterobacteriaceae from the gut of subsistence farmers, their livestock, and the surrounding environment in rural Nepal. Sci. Rep. 2021, 11, 2091.
- Dierikx, C.M.; van der Goot, J.; van Essen-Zandbergen, A.; Mevius, D.J. Dynamics of cefotaxime resistant Escherichia coli in broilers in the first week of life. Vet. Microbiol. 2018, 222, 64–68.
- Zhang, L.; Kinkelaar, D.; Huang, Y.; Li, Y.; Li, X.; Wang, H.H. Acquired antibiotic resistance: Are we born with it? Appl. Environ. Microbiol. 2011, 77, 7134–7141.
- Silva, V.; Igrejas, G.; Carvalho, I.; Peixoto, F.; Cardoso, L.; Pereira, J.E.; Del Campo, R.; Poeta, P. Genetic Characterization of vanA-Enterococcus faecium Isolates from Wild Red-Legged Partridges in Portugal. Microb. Drug Resist. 2017, 24, 89–94.
- Borgen, K.; Sorum, M.; Wasteson, Y.; Kruse, H. VanA-type vancomycin-resistant enterococci (VRE) remain prevalent in poultry carcasses 3 years after avoparcin was banned. Int. J. Food Microbiol. 2001, 64, 89–94.
- Haley, B.J.; Van Kessel, J.A.S. The resistome of the bovine gastrointestinal tract. Curr. Opin. Biotechnol. 2022, 73, 213–219.
- Silva, V.; Caniça, M.; Capelo, J.L.; Igrejas, G.; Poeta, P. Diversity and genetic lineages of environmental staphylococci: A surface water overview. FEMS Microbiol. Ecol. 2020, 96, fiaa191.
- European Commission. Commission Implementing Decision (EU) 2020/1729; European Commission: Brussels, Belgium, 2020.
- Comerlato, C.B.; Ritter, A.C.; Miyamoto, K.N.; Brandelli, A. Proteomic study of Enterococcus durans LAB18S growing on prebiotic oligosaccharides. Food Microbiol. 2020, 89, 103430.
- Torres, C.; Alonso, C.A.; Ruiz-Ripa, L.; León-Sampedro, R.; Del Campo, R.; Coque, T.M. Antimicrobial Resistance in Enterococcus spp. of animal origin. Microbiol. Spectr. 2018, 6, 185–227.
- Ramos, S.; Silva, V.; Dapkevicius, M.d.L.E.; Igrejas, G.; Poeta, P. Enterococci, from harmless bacteria to a pathogen. Microorganisms 2020, 8, 1118.
- Roy, K.; Islam, M.S.; Paul, A.; Ievy, S.; Talukder, M.; Sobur, M.A.; Ballah, F.M.; Khan, M.S.R.; Rahman, M.T. Molecular detection and antibiotyping of multi-drug resistant Enterococcus faecium from healthy broiler chickens in Bangladesh. Vet. Med. Sci. 2022, 8, 200–210.
- Tyson, G.H.; Nyirabahizi, E.; Crarey, E.; Kabera, C.; Lam, C.; Rice-Trujillo, C.; McDermott, P.F.; Tate, H. Prevalence and antimicrobial resistance of enterococci isolated from retail meats in the United States, 2002 to 2014. Appl. Environ. Microbiol. 2017, 84, e01902-17.
- Lengfelder, I.; Sava, I.G.; Hansen, J.J.; Kleigrewe, K.; Herzog, J.; Neuhaus, K.; Hofmann, T.; Sartor, R.B.; Haller, D. Complex bacterial consortia reprogram the colitogenic activity of Enterococcus faecalis in a gnotobiotic mouse model of chronic, immune-mediated colitis. Front. Immunol. 2019, 10, 1420.
- Hasan, K.A.; Ali, S.A.; Rehman, M.; Bin-Asif, H.; Zahid, S. The unravelled Enterococcus faecalis zoonotic superbugs: Emerging multiple resistant and virulent lineages isolated from poultry environment. Zoonoses Public Health 2018, 65, 921–935.
- O’Dea, M.; Sahibzada, S.; Jordan, D.; Laird, T.; Lee, T.; Hewson, K.; Pang, S.; Abraham, R.; Coombs, G.W.; Harris, T.; et al. Genomic, Antimicrobial Resistance, and Public Health Insights into Enterococcus spp. from Australian Chickens. J. Clin. Microbiol. 2019, 57, e00319-19.
- Daniel, D.S.; Lee, S.M.; Gan, H.M.; Dykes, G.A.; Rahman, S. Genetic diversity of Enterococcus faecalis isolated from environmental, animal and clinical sources in Malaysia. J. Infect. Public Health 2017, 10, 617–623.
- Robbins, K.M.; Suyemoto, M.M.; Lyman, R.L.; Martin, M.P.; Barnes, H.J.; Borst, L.B. An outbreak and source investigation of enterococcal spondylitis in broilers caused by Enterococcus cecorum. Avian Dis. 2012, 56, 768–773.
- Miranda, J.M.; Guarddon, M.; Vázquez, B.I.; Fente, C.A.; Barros-Velázquez, J.; Cepeda, A.; Franco, C.M. Antimicrobial resistance in Enterobacteriaceae strains isolated from organic chicken, conventional chicken and conventional turkey meat: A comparative survey. J. Food Prot. 2007, 70, 412–416.
- Diarra, M.S.; Rempel, H.; Champagne, J.; Masson, L.; Pritchard, J.; Topp, E. Distribution of antimicrobial resistance and virulence genes in Enterococcus spp. and characterization of isolates from broiler chickens. Appl. Environ. Microbiol. 2010, 76, 8033–8043.
- Michaux, C.; Hansen, E.E.; Jenniches, L.; Gerovac, M.; Barquist, L.; Vogel, J. Single-Nucleotide RNA Maps for the Two Major Nosocomial Pathogens Enterococcus faecalis and Enterococcus faecium. Front. Cell Infect. Microbiol. 2020, 10, 600325.
- Gregersen, R.H.; Petersen, A.; Christensen, H.; Bisgaard, M. Multilocus sequence typing of Enterococcus faecalis isolates demonstrating different lesion types in broiler breeders. Avian Pathol. 2010, 39, 435–440.
- Sandhu, T.S. Fecal streptococcal infection of commercial white Pekin ducklings. Avian Dis. 1988, 32, 570–573.
- Devriese, L.A.; Hommez, J.; Wijfels, R.; Haesebrouck, F. Composition of the enterococcal and streptococcal intestinal flora of poultry. J. Appl. Bacteriol. 1991, 71, 46–50.
- De Jong, A.; Simjee, S.; Rose, M.; Moyaert, H.; El Garch, F.; Youala, M.; Butty, P.; Haag-Diergarten, S.; Klein, U.; Pellet, T.; et al. Antimicrobial resistance monitoring in commensal enterococci from healthy cattle, pigs and chickens across Europe during 2004-14 (EASSA Study). J. Antimicrob. Chemother. 2019, 74, 921–930.
- Dolka, B.; Gołȩbiewska-Kosakowska, M.; Krajewski, K.; Kwieciński, P.; Nowak, T.; Szubstarski, J.; Wilczyński, J.; Szeleszczuk, P. Occurrence of Enterococcus spp. in poultry in Poland based on 2014-2015 data. Med. Weter. 2017, 73, 220–224.
- Dolka, B.; Cisek, A.A.; Szeleszczuk, P. The application of the loop-mediated isothermal amplification (LAMP) method for diagnosing Enterococcus hirae-associated endocarditis outbreaks in chickens. BMC Microbiol. 2019, 19, 48.
- Stȩpień-Pyśniak, D.; Marek, A.; Banach, T.; Adaszek, Ł.; Pyzik, E.; Wilczyński, J.; Winiarczyk, S. Prevalence and antibiotic resistance of Enterococcus strains isolated from poultry. Acta Vet. Hung. 2016, 64, 148–163.
- Talarmin, J.P.; Pineau, S.; Guillouzouic, A.; Boutoille, D.; Giraudeau, C.; Reynaud, A.; Lepelletier, D.; Corvec, S. Relapse of Enterococcus hirae prosthetic valve endocarditis. J. Clin. Microbiol. 2011, 49, 1182–1184.
- Kim, M.H.; Moon, D.C.; Kim, S.-J.; Mechesso, A.F.; Song, H.-J.; Kang, H.Y.; Choi, J.-H.; Yoon, S.-S.; Lim, S.-K. Nationwide Surveillance on Antimicrobial Resistance Profiles of Enterococcus faecium and Enterococcus faecalis Isolated from Healthy Food Animals in South Korea, 2010–2019. Microorganisms 2021, 9, 925.
- Dolka, B.; Chrobak-Chmiel, D.; Makrai, L.; Szeleszczuk, P. Phenotypic and genotypic characterization of Enterococcus cecorum strains associated with infections in poultry. BMC Vet. Res. 2016, 12, 129.
- Greub, G.; Devriese, L.A.; Pot, B.; Dominguez, J.; Bille, J. Enterococcus cecorum Septicemia in a Malnourished Adult Patient. Eur. J. Clin. Microbiol. Infect Dis. 1997, 16, 594–598.
- Warnke, P.; Köller, T.; Stoll, P.; Podbielski, A. Nosocomial infection due to Enterococcus cecorum identified by MALDI-TOF MS and Vitek 2 from a blood culture of a septic patient. Eur. J. Microbiol. Immunol. 2015, 5, 177–179.
- Jung, A.; Teske, L.; Rautenschlein, S. Enterococcus cecorum infection in a racing pigeon. Avian Dis. 2014, 58, 654–658.
- Jung, A.; Rautenschlein, S. Comprehensive report of an Enterococcus cecorum infection in a broiler flock in Northern Germany. BMC Vet. Res. 2014, 10, 311.
- Aitchison, H.; Poolman, P.; Coetzer, M.; Griffiths, C.; Jacobs, J.; Meyer, M.; Bisschop, S. Enterococcal-related vertebral osteoarthritis in South African broiler breeders: A case report. J. S. Afr. Vet. Assoc. 2014, 85, 5–9.
- Schreier, J.; Rautenschlein, S.; Jung, A. Different virulence levels of Enterococcus cecorum strains in experimentally infected meat-type chickens. PLoS ONE 2021, 16, e0259904.
- Schreier, J.; Karasova, D.; Crhanova, M.; Rychlik, I.; Rautenschlein, S.; Jung, A. Influence of lincomycin-spectinomycin treatment on the outcome of Enterococcus cecorum infection and on the cecal microbiota in broilers. Gut Pathog. 2022, 14, 3.
- Borst, L.B.; Suyemoto, M.M.; Sarsour, A.H.; Harris, M.C.; Martin, M.P.; Strickland, J.D.; Oviedo, E.O.; Barnes, H.J. Pathogenesis of Enterococcal Spondylitis Caused by Enterococcus cecorum in Broiler Chickens. Vet. Pathol. 2017, 54, 61–73.
- Kense, M.J.; Landman, W.J.M. Enterococcus cecorum infections in broiler breeders and their offspring: Molecular epidemiology. Avian Pathol. 2011, 40, 603–612.
- Stalker, M.J.; Brash, M.L.; Weisz, A.; Ouckama, R.M.; Slavic, D. Arthritis and osteomyelitis associated with Enterococcus cecorum infection in broiler and broiler breeder chickens in Ontario, Canada. J. Vet. Diagnostic Investig. 2010, 22, 643–645.
- Sanlibaba, P.; Senturk, E. Prevalence, characterization, and antibiotic resistance of enterococci from traditional cheeses in turkey. Int. J. Food Prop. 2018, 21, 1955–1963.
- Arias, C.A.; Murray, B.E. The rise of the Enterococcus: Beyond vancomycin resistance. Nat. Rev. Microbiol. 2012, 10, 266–278.
- Shang, Y.; Li, D.; Shan, X.; Schwarz, S.; Zhang, S.M.; Chen, Y.X.; Ouyang, W.; Du, X.D. Analysis of two pheromone-responsive conjugative multiresistance plasmids carrying the novel mobile optra locus from Enterococcus faecalis. Infect. Drug Resist. 2019, 12, 2355–2362.
- Poeta, P.; Costa, D.; Rodrigues, J.; Torres, C. Antimicrobial resistance and the mechanisms implicated in faecal enterococci from healthy humans, poultry and pets in Portugal. Int. J. Antimicrob. Agents 2006, 27, 131–137.
- Miller, W.R.; Munita, J.M.; Arias, C.A. Mechanisms of antibiotic resistance in enterococci. Expert Rev. Anti. Infect. Ther. 2014, 12, 1221–1236.
- Semedo-Lemsaddek, T.; Bettencourt Cota, J.; Ribeiro, T.; Pimentel, A.; Tavares, L.; Bernando, F.; Oliveira, M. Resistance and virulence distribution in enterococci isolated from broilers reared in two farming systems. Ir. Vet. J. 2021, 74, 22.
- Ono, S.; Muratani, T.; Matsumoto, T. Mechanisms of resistance to imipenem and ampicillin in Enterococcus faecalis. Antimicrob. Agents Chemother. 2005, 49, 2954–2958.
- Braga, J.F.V.; Leal, C.A.G.; Silva, C.C.; Fernandes, A.A.; Martins, N.R.d.S.; Ecco, R. Genetic diversity and antimicrobial resistance profile of Enterococcus faecalis isolated from broilers with vertebral osteomyelitis in Southeast Brazil. Avian Pathol. 2018, 47, 14–22.
- Harada, T.; Mito, Y.; Otsuki, K.; Murase, T. Resistance to gentamicin and vancomycin in enterococcal strains isolated from retail broiler chickens in Japan. J. Food Prot. 2004, 67, 2292–2295.
- Aarestrup, F.M.; Agerso, Y.; Gerner-Smidt, P.; Madsen, M.; Jensen, L.B. Comparison of antimicrobial resistance phenotypes and resistance genes in Enterococcus faecalis and Enterococcus faecium from humans in the community, broilers, and pigs in Denmark. Diagn. Microbiol. Infect. Dis. 2000, 37, 127–137.
- Weinbren, M.J.; Johnson, A.P.; Woodford, N. Defining high-level gentamicin resistance in enterococci. J. Antimicrob. Chemother. 2000, 45, 404–405.
- Donabedian, S.M.; Thal, L.A.; Hershberger, E.; Perri, M.B.; Chow, J.W.; Bartlett, P.; Jones, R.; Joyce, K.; Rossiter, S.; Gay, K.; et al. Molecular characterization of gentamicin-resistant Enterococci in the United States: Evidence of spread from animals to humans through food. J. Clin. Microbiol. 2003, 41, 1109–1113.
- Schwarz, S.; Kehrenberg, C.; Doublet, B.; Cloeckaert, A. Molecular basis of bacterial resistance to chloramphenicol and florfenicol. FEMS Microbiol. Rev. 2004, 28, 519–542.
- Baynes, R.E.; Dedonder, K.; Kissell, L.; Mzyk, D.; Marmulak, T.; Smith, G.; Tell, L.; Gehring, R.; Davis, J.; Riviere, J.E. Health concerns and management of select veterinary drug residues. Food Chem. Toxicol. 2016, 88, 112–122.
- Ünal, N.; Aşkar, Ş.; Yildirim, M. Antibiotic resistance profile of Enterococcus faecium and Enterococcus faecalis isolated from broiler cloacal samples. Turkish J. Vet. Anim. Sci. 2017, 41, 199–203.
- Habib, I.; Ghazawi, A.; Lakshmi, G.B.; Mohamed, M.I.; Li, D.; Khan, M.; Sahibzada, S. Emergence and Genomic Characterization of the First Reported optrA-Carrying Linezolid-Resistant Enterococci Isolated from Retail Broiler Meat in the United Arab Emirates. Foods 2022, 11, 3190.
- Hui, L.A.; Bodolea, C.; Vlase, L.; Hiriscau, E.I.; Popa, A. Linezolid Administration to Critically Ill Patients: Intermittent or Continuous Infusion? A Systematic Literature Search and Review. Antibiotics 2022, 11, 436.
- Yoon, S.; Kim, Y.B.; Seo, K.W.; Ha, J.S.; Noh, E.B.; Lee, Y.J. Characteristics of linezolid-resistant Enterococcus faecalis isolates from broiler breeder farms. Poult. Sci. 2020, 99, 6055–6061.
- Tyson, G.H.; Sabo, J.L.; Hoffmann, M.; Hsu, C.H.; Mukherjee, S.; Hernandez, J.; Tillman, G.; Wasilenko, J.L.; Haro, J.; Simmons, M.; et al. Novel linezolid resistance plasmids in Enterococcus from food animals in the USA. J. Antimicrob. Chemother. 2018, 73, 3254–3258.
- Gião, J.; Leão, C.; Albuquerque, T.; Clemente, L.; Amaro, A. Antimicrobial Susceptibility of Enterococcus Isolates from Cattle and Pigs in Portugal: Linezolid Resistance Genes optrA and poxtA. Antibiotics 2022, 11, 615.
- Wang, Y.; Lv, Y.; Cai, J.; Schwarz, S.; Cui, L.; Hu, Z.; Zhang, R.; Li, J.; Zhao, Q.; He, T.; et al. A novel gene, optrA, that confers transferable resistance to oxazolidinones and phenicols and its presence in Enterococcus faecalis and Enterococcus faecium of human and animal origin. J. Antimicrob. Chemother. 2015, 70, 2182–2190.
- Long, K.S.; Poehlsgaard, J.; Kehrenberg, C.; Schwarz, S.; Vester, B. The cfr rRNA methyltransferase confers resistance to phenicols, lincosamides, oxazolidinones, pleuromutilins, and streptogramin A antibiotics. Antimicrob. Agents Chemother. 2006, 50, 2500–2505.
- Kim, Y.B.; Seo, K.W.; Son, S.H.; Noh, E.B.; Lee, Y.J. Genetic characterization of high-level aminoglycoside-resistant Enterococcus faecalis and Enterococcus faecium isolated from retail chicken meat. Poult. Sci. 2019, 98, 5981–5988.
- Tamang, M.D.; Moon, D.C.; Kim, S.R.; Kang, H.Y.; Lee, K.; Nam, H.M.; Jang, G.C.; Lee, H.S.; Jung, S.C.; Lim, S.K. Detection of novel oxazolidinone and phenicol resistance gene optrA in enterococcal isolates from food animals and animal carcasses. Vet. Microbiol. 2017, 201, 252–256.
- Cauwerts, K.; Decostere, A.; De Graef, E.M.; Haesebrouck, F.; Pasmans, F. High prevalence of tetracycline resistance in Enterococcus isolates from broilers carrying the erm(B) gene. Avian Pathol. 2007, 36, 395–399.
- Nilsson, O.; Alm, E.; Greko, C.; Bengtsson, B. The rise and fall of a vancomycin-resistant clone of Enterococcus faecium among broilers in Sweden. J. Glob. Antimicrob. Resist. 2019, 17, 233–235.
- Skarzynska, M.; Leekitcharoenphon, P.; Hendriksen, R.S.; Aarestrup, F.M.; Wasyl, D. A metagenomic glimpse into the gut of wild and domestic animals: Quantification of antimicrobial resistance and more. PLoS ONE 2020, 15, e0242987.
- Nilsson, O.; Greko, C.; Top, J.; Franklin, A.; Bengtsson, B. Spread without known selective pressure of a vancomycin-resistant clone of Enterococcus faecium among broilers. J. Antimicrob. Chemother. 2009, 63, 868–872.
- Robredo, B.; Singh, K.V.; Baquero, F.; Murray, B.E.; Torres, C. From vanA Enterococcus hirae to vanA Enterococcus faecium: A study of feed supplementation with avoparcin and tylosin in young chickens. Antimicrob. Agents Chemother. 1999, 43, 1137–1143.
- Zhu, Y.; Huang, W.E.; Yang, Q. Clinical Perspective of Antimicrobial Resistance in Bacteria. Infect. Drug Resist. 2022, 15, 735–746.
- Dubin, K.; Pamer, E.G. Enterococci and Their Interactions with the Intestinal Microbiome. Microbiol. Spectr. 2017, 5, 5–6.
- Manson, J.M.; Keis, S.; Smith, J.M.B.; Cook, G.M. A clonal lineage of VanA-type Enterococcus faecalis predominates in vancomycin-resistant enterococci isolated in New Zealand. Antimicrob. Agents Chemother. 2003, 47, 204–210.
- Heuer, O.E.; Pedersen, K.; Jensen, L.B.; Madsen, M.; Olsen, J.E. Persistence of vancomycin-resistant enterococci (VRE) in broiler houses after the avoparcin ban. Microb. Drug Resist. 2002, 8, 355–361.
- Klare, I.; Badstübner, D.; Konstabel, C.; Böhme, G.; Claus, H.; Witte, W. Decreased incidence of VanA-type vancomycin-resistant enterococci isolated from poultry meat and from fecal samples of humans in the community after discontinuation of avoparcin usage in animal husbandry. Microb. Drug Resist. 1999, 5, 45–52.
- Leinweber, H.; Alotaibi, S.M.I.; Overballe-Petersen, S.; Hansen, F.; Hasman, H.; Bortolaia, V.; Hammerum, A.M.; Ingmer, H. Vancomycin resistance in Enterococcus faecium isolated from Danish chicken meat is located on a pVEF4-like plasmid persisting in poultry for 18 years. Int. J. Antimicrob. Agents 2018, 52, 283–286.
- Aun, E.; Kisand, V.; Laht, M.; Telling, K.; Kalmus, P.; Väli, Ü.; Brauer, A.; Remm, M.; Tenson, T. Molecular Characterization of Enterococcus Isolates From Different Sources in Estonia Reveals Potential Transmission of Resistance Genes Among Different Reservoirs. Front. Microbiol. 2021, 12, 601490.
- Ahmed, M.O.; Baptiste, K.E. Vancomycin-Resistant Enterococci: A Review of Antimicrobial Resistance Mechanisms and Perspectives of Human and Animal Health. Microb. Drug Resist. 2018, 24, 590–606.
- Morris, D.; Galvin, S.; Boyle, F.; Hickey, P.; Mulligan, M.; Cormican, M. Enterococcus faecium of the vanA genotype in rural drinking water, effluent, and the aqueous environment. Appl. Environ. Microbiol. 2011, 78, 596–598.
- Poeta, P.; Costa, D.; Rodrigues, J.; Torres, C. Study of faecal colonization by vanA-containing Enterococcus strains in healthy humans, pets, poultry and wild animals in Portugal. J. Antimicrob. Chemother. 2005, 55, 278–280.
- Wilson, I.G.; McAfee, G.G. Vancomycin-resistant enterococci in shellfish, unchlorinated waters, and chicken. Int. J. Food Microbiol. 2002, 79, 143–151.
- Dutta, I.; Reynolds, P.E. Biochemical and genetic characterization of the vanC-2 vancomycin resistance gene cluster of Enterococcus casseliflavus ATCC 25788. Antimicrob. Agents Chemother. 2002, 46, 3125–3132.
- Stępień-Pyśniak, D.; Hauschild, T.; Dec, M.; Marek, A.; Brzeski, M.; Kosikowska, U. Antimicrobial resistance and genetic diversity of Enterococcus faecalis from yolk sac infections in broiler chicks. Poult. Sci. 2021, 100, 101491.
- Alzahrani, O.M.; Fayez, M.; Alswat, A.S.; Alkafafy, M.; Mahmoud, S.F.; Al-Marri, T.; Almuslem, A.; Ashfaq, H.; Yusuf, S. Antimicrobial Resistance, Biofilm Formation, and Virulence Genes in Enterococcus Species from Small Backyard Chicken Flocks. Antibiotics 2022, 11, 380.
- Yu, L.; Liu, Y.; Liu, M.; Li, Z.; Li, L.; Wang, F. Research Note: Molecular characterization of antimicrobial resistance and virulence gene analysis of Enterococcus faecalis in poultry in Tai’an, China. Poult. Sci. 2022, 101, 101763.
- Kim, Y.B.; Seo, H.J.; Seo, K.W.; Jeon, H.Y.; Kim, D.K.; Kim, S.W.; Lim, S.K.; Lee, Y.J. Characteristics of high-Level ciprofloxacin-Resistant Enterococcus faecalis and Enterococcus faecium from retail chicken meat in Korea. J. Food Prot. 2018, 81, 1357–1363.
- Aslanta, Ö. Molecular and phenotypic characterization of enterococci isolated from broiler flocks in Turkey. Trop. Anim. Health Prod. 2019, 51, 1073–1082.
- Day, M.J.; Rodríguez, I.; van Essen-Zandbergen, A.; Dierikx, C.; Kadlec, K.; Schink, A.K.; Wu, G.; Chattaway, M.A.; DoNascimento, V.; Wain, J.; et al. Diversity of STs, plasmids and ESBL genes among Escherichia coli from humans, animals and food in Germany, the Netherlands and the UK. J. Antimicrob. Chemother. 2016, 71, 1178–1182.
- Badr, H.; Reda, R.M.; Hagag, N.M.; Kamel, E.; Elnomrosy, S.M.; Mansour, A.I.; Shahein, M.A.; Ali, S.F.; Ali, H.R. Multidrug-Resistant and Genetic Characterization of Recovered from Chickens and Humans in Egypt. Animals 2022, 12, 346.
- Ramos, S.; Silva, V.; Dapkevicius, M.d.L.E.; Caniça, M.; Tejedor-Junco, M.T.; Igrejas, G.; Poeta, P. Escherichia coli as Commensal and Pathogenic Bacteria among Food-Producing Animals: Health Implications of Extended Spectrum β-Lactamase (ESBL) Production. Animals 2020, 10, 2239.
- Poirel, L.; Madec, J.-Y.; Lupo, A.; Schink, A.-K.; Kieffer, N.; Nordmann, P.; Schwarz, S. Antimicrobial Resistance in Escherichia coli. Microbiol. Spectr. 2018, 6, 4–6.
- Jouini, A.; Klibi, A.; Elarbi, I.; Chaabene, M.B.; Hamrouni, S.; Souiai, O.; Hanachi, M.; Ghram, A.; Maaroufi, A. First detection of human ST131-CTX-M-15-o25-b2 clone and high-risk clonal lineages of ESBL/pAmpC-producing E. coli isolates from diarrheic poultry in Tunisia. Antibiotics 2021, 10, 70.
- Lindstedt, B.A.; Finton, M.D.; Porcellato, D.; Brandal, L.T. High frequency of hybrid Escherichia coli strains with combined Intestinal Pathogenic Escherichia coli (IPEC) and Extraintestinal Pathogenic Escherichia coli (ExPEC) virulence factors isolated from human faecal samples. BMC Infect. Dis. 2018, 18, 544.
- Burke, D.A.; Axon, A.T.R. Ulcerative colitis and Escherichia coli with adhesive Properties. J. Clin. Pathol. 1987, 40, 782–786.
- Akya, A.; Ahmadi, M.; Khodamoradi, S.; Rezaei, M.R.; Karani, N.; Elahi, A.; Lorestani, R.C.; Rezaei, M. Prevalence of blaCTX-M, blaCTX-M-2, blaCTX-M-8, blaCTX-M-25 and blaCTX-M-3 Genes in Escherichia coli Isolated from Urinary Tract Infection in Kermanshah City, Iran. J. Clin. Diagnostic Res. 2019, 13, 13–16.
- Leverstein-van Hall, M.A.; Dierikx, C.M.; Cohen Stuart, J.; Voets, G.M.; van den Munckhof, M.P.; van Essen-Zandbergen, A.; Platteel, T.; Fluit, A.C.; van de Sande-Bruinsma, N.; Scharinga, J.; et al. Dutch patients, retail chicken meat and poultry share the same ESBL genes, plasmids and strains. Clin. Microbiol. Infect. 2011, 17, 873–880.
- Stromberg, Z.R.; Johnson, J.R.; Fairbrother, J.M.; Kilbourne, J.; Van Goor, A.; Curtiss, R.; Mellata, M. Evaluation of Escherichia coli isolates from healthy chickens to determine their potential risk to poultry and human health. PLoS ONE 2017, 12, e0180599.
- Mohamed, M.A.; Shehata, M.A.; Rafeek, E. Virulence genes content and antimicrobial resistance in Escherichia coli from broiler chickens. Vet. Med. Int. 2014, 2014, 195189.
- Awawdeh, L.; Turni, C.; Mollinger, J.L.; Henning, J.; Cobbold, R.N.; Trott, D.J.; Wakeham, D.L.; Gibson, J.S. Antimicrobial susceptibility, plasmid replicon typing, phylogenetic grouping, and virulence potential of avian pathogenic and faecal Escherichia coli isolated from meat chickens in Australia. Avian Pathol. 2022, 51, 349–360.
- Kravik, I.H.; Kaspersen, H.; Sjurseth, S.K.; Jonsson, M.; David, B.; Aspholm, M.; Sekse, C. High sequence similarity between avian pathogenic E. coli isolates from individual birds and within broiler chicken flocks during colibacillosis outbreaks. Vet. Microbiol. 2022, 267, 109378.
- Ahmed, A.M.; Shimamoto, T.; Shimamoto, T. Molecular characterization of multidrug-resistant avian pathogenic Escherichia coli isolated from septicemic broilers. Int. J. Med. Microbiol. 2013, 303, 475–483.
- Solà-Ginés, M.; Cameron-Veas, K.; Badiola, I.; Dolz, R.; Majó, N.; Dahbi, G.; Viso, S.; Mora, A.; Blanco, J.; Piedra-Carrasco, N.; et al. Diversity of multi-drug resistant avian pathogenic Escherichia coli (APEC) Causing outbreaks of colibacillosis in broilers during 2012 in Spain. PLoS ONE 2015, 10, e0143191.
- Dhaouadi, S.; Soufi, L.; Hamza, A.; Fedida, D.; Zied, C.; Awadhi, E.; Mtibaa, M.; Hassen, B.; Cherif, A.; Torres, C.; et al. Co-occurrence of mcr-1 mediated colistin resistance and β-lactamase-encoding genes in multidrug-resistant Escherichia coli from broiler chickens with colibacillosis in Tunisia. J. Glob. Antimicrob. Resist. 2020, 22, 538–545.
- Kim, Y.B.; Yoon, M.Y.; Ha, J.S.; Seo, K.W.; Noh, E.B.; Son, S.H.; Lee, Y.J. Molecular characterization of avian pathogenic Escherichia coli from broiler chickens with colibacillosis. Poult. Sci. 2020, 99, 1088–1095.
- Saif, Y.M.; Fadly, A.M.; Glisson, J.R.; McDougald, L.R.; Nolan, L.K.; Swayne, D.E. Diseases of Poultry, 12th ed.; Blackwell Publishing: Hoboken, NJ, USA, 2008; ISBN 9781119350927.
- Śmiałek, M.; Kowalczyk, J.; Koncicki, A. Influence of vaccination of broiler chickens against Escherichia coli with live attenuated vaccine on general properties of E. coli population, IBV vaccination efficiency, and production parameters—A field experiment. Poult. Sci. 2020, 99, 5452–5460.
- Ebrahimi-Nik, H.; Bassami, M.R.; Mohri, M.; Rad, M.; Khan, M.I. Bacterial ghost of avian pathogenic E. coli (APEC) serotype O78:K80 as a homologous vaccine against avian colibacillosis. PLoS ONE 2018, 13, e0194888.
- Oliveira, J.M.; Cardoso, M.F.; Moreira, F.A.; Müller, A. Phenotypic antimicrobial resistance (AMR) of avian pathogenic Escherichia coli (APEC) from broiler breeder flocks between 2009 and 2018. Avian Pathol. 2022, 51, 388–394.
- Sarker, M.S.; Mannan, M.S.; Ali, M.Y.; Bayzid, M.; Ahad, A.; Bupasha, Z.B. Antibiotic resistance of Escherichia coli isolated from broilers sold at live bird markets in Chattogram, Bangladesh. J. Adv. Vet. Anim. Res. 2019, 6, 272–277.
- Borges, C.A.; Tarlton, N.J.; Riley, L.W. Escherichia coli from Commercial Broiler and Backyard Chickens Share Sequence Types, Antimicrobial Resistance Profiles, and Resistance Genes with Human Extraintestinal Pathogenic Escherichia coli. Foodborne Pathog. Dis. 2019, 16, 813–822.
- Apostolakos, I.; Mughini-Gras, L.; Fasolato, L.; Piccirillo, A. Assessing the occurrence and transfer dynamics of ESBL/pAmpC-producing Escherichia coli across the broiler production pyramid. PLoS ONE 2019, 14, e0217174.
- Al Azad, M.A.R.; Rahman, M.M.; Amin, R.; Begum, M.I.A.; Fries, R.; Husna, A.; Khairalla, A.S.; Badruzzaman, A.T.M.; El Zowalaty, M.E.; Lampang, K.N.; et al. Susceptibility and multidrug resistance patterns of Escherichia coli isolated from cloacal swabs of live broiler chickens in Bangladesh. Pathogens 2019, 8, 118.
- Hojabri, Z.; Darabi, N.; Arab, M.; Saffari, F.; Pajand, O. Clonal diversity, virulence genes content and subclone status of Escherichia coli sequence type 131: Comparative analysis of E. coli ST131 and non-ST131 isolates from Iran. BMC Microbiol. 2019, 19, 117.
- Das, A.; Dhar, P.K.; Dutta, A.; Jalal, M.S.; Ghosh, P.; Das, T.; Barua, H.; Biswas, P.K. Circulation of oxytetracycline- and ciprofloxacin-resistant commensal Escherichia coli strains in broiler chickens and farm environments, Bangladesh. Vet. World 2020, 13, 2395–2400.
- Thorsteinsdottir, T.R.; Haraldsson, G.; Fridriksdottir, V.; Kristinsson, K.G.; Gunnarsson, E. Prevalence and genetic relatedness of antimicrobial-resistant Escherichia coli isolated from animals, foods and humans in Iceland. Zoonoses Public Health 2010, 57, 189–196.
- Yoon, M.Y.; Kim, Y.B.; Ha, J.S.; Seo, K.W.; Noh, E.B.; Son, S.H.; Lee, Y.J. Molecular characteristics of fluoroquinolone-resistant avian pathogenic Escherichia coli isolated from broiler chickens. Poult. Sci. 2020, 99, 3628–3636.
- Mahmud, S.; Nazir, K.H.M.N.H.; Rahman, M.T. Prevalence and molecular detection of fluoroquinolone-resistant genes (qnrA and qnrS) in Escherichia coli isolated from healthy broiler chickens. Vet. World 2018, 11, 1720–1724.
- De Koster, S.; Ringenier, M.; Lammens, C.; Stegeman, A.; Tobias, T.; Velkers, F.; Vernooij, H.; Kluytmans-Van Den Bergh, M.; Kluytmans, J.; Dewulf, J.; et al. ESBL-producing, carbapenem-and ciprofloxacin-resistant Escherichia coli in Belgian and Dutch broiler and pig farms: A cross-sectional and cross-border study. Antibiotics 2021, 10, 945.
- Börjesson, S.; Guillard, T.; Landén, A.; Bengtsson, B.; Nilsson, O. Introduction of quinolone resistant Escherichia coli to Swedish broiler population by imported breeding animals. Vet. Microbiol. 2016, 194, 74–78.
- Mendonça, N.; Figueiredo, R.; Mendes, C.; Card, R.M.; Anjum, M.F.; da Silva, G.J. Microarray evaluation of antimicrobial resistance and virulence of Escherichia coli isolates from Portuguese poultry. Antibiotics 2016, 5, 4.
- Trobos, M.; Jakobsen, L.; Olsen, K.E.P.; Frimodt-Møller, N.; Hammerum, A.M. Prevalence of sulphonamide resistance and class 1 integron genes in Escherichia coli isolates obtained from broilers, broiler meat, healthy humans and urinary infections in Denmark. Int. J. Antimicrob. Agents 2008, 32, 366–367.
- Amador, P.; Fernandes, R.; Prudêncio, C.; Duarte, I. Prevalence of antibiotic resistance genes in multidrug-resistant Enterobacteriaceae on portuguese livestock manure. Antibiotics 2019, 8, 23.
- Messaili, C.; Messai, Y.; Bakour, R. Virulence gene profiles, antimicrobial resistance and phylogenetic groups of fecal Escherichia coli strains isolated from broiler chickens in Algeria. Vet. Ital. 2019, 55, 35–46.
- Castanheira, M.; Simner, P.J.; Bradford, P.A. Extended-spectrum β-lactamases: An update on their characteristics, epidemiology and detection. JAC-Antimicrobial Resist. 2021, 3, dlab092.
- De Champs, C.; Sirot, D.; Chanal, C.; Bonnet, R.; Sirot, J.; Avril, J.L.; Cattoen, C.; Chardon, H.; Croix, J.L.; Dabernat, H.; et al. A 1998 survey of extended-spectrum β-lactamases in Enterobacteriaceae in France. Antimicrob. Agents Chemother. 2000, 44, 3177–3179.
- Subramanya, S.H.; Bairy, I.; Nayak, N.; Padukone, S.; Sathian, B.; Gokhale, S. Low rate of gut colonization by extended-spectrum β-lactamase producing Enterobacteriaceae in HIV infected persons as compared to healthy individuals in Nepal. PLoS ONE 2019, 14, e0212042.
- Cantón, R.; Novais, A.; Valverde, A.; Machado, E.; Peixe, L.; Baquero, F.; Coque, T.M. Prevalence and spread of extended-spectrum β-lactamase-producing Enterobacteriaceae in Europe. Clin. Microbiol. Infect. 2008, 14, 144–153.
- Knothe, H.; Shah, P.; Krcmery, V.; Antal, M.; Mitsuhashi, S. Transferable resistance to cefotaxime, cefoxitin, cefamandole and cefuroxime in clinical isolates of Klebsiella pneumoniae and Serratia marcescens. Infection 1983, 11, 315–317.
- Carvalho, I.; Silva, N.; Carrola, J.; Silva, V.; Currie, C.; Igrejas, G.; Poeta, P. Antibiotic resistance: Immunity-acquired resistance: Evolution of antimicrobial resistance among extended-spectrum β-lactamases and carbapenemases in Klebsiella pneumoniae and Escherichia coli. In Antibiotic Drug Resistance; CDC: Atlanta, GA, USA, 2019.
- Wibisono, F.J.; Sumiarto, B.; Untari, T.; Effendi, M.H.; Permatasari, D.A.; Witaningrum, A.M. CTX Gene of Extended Spectrum Beta-Lactamase (ESBL) Producing Escherichia coli on Broilers in Blitar, Indonesia. Syst. Rev. Pharm. 2020, 11, 396–403.
- Päivärinta, M.; Latvio, S.; Fredriksson-Ahomaa, M.; Heikinheimo, A. Whole genome sequence analysis of antimicrobial resistance genes, multilocus sequence types and plasmid sequences in ESBL/AmpC Escherichia coli isolated from broiler caecum and meat. Int. J. Food Microbiol. 2020, 315, 108361.
- Smet, A.; Martel, A.; Persoons, D.; Dewulf, J.; Heyndrickx, M.; Herman, L.; Haesebrouck, F.; Butaye, P. Broad-spectrum β-lactamases among Enterobacteriaceae of animal origin: Molecular aspects, mobility and impact on public health. FEMS Microbiol. Rev. 2010, 34, 295–316.
- Rawat, D.; Nair, D. Extended-spectrum ß-lactamases in gram negative bacteria. J. Glob. Infect. Dis. 2010, 2, 263.
- Sader, H.S.; Pfaller, M.A.; Jones, R.N. Prevalence of important pathogens and the antimicrobial activity of parenteral drugs at numerous medical centers in the United States. Diagn. Microbiol. Infect. Dis. 1994, 20, 203–208.
- Cantón, R.; González-Alba, J.M.; Galán, J.C. CTX-M enzymes: Origin and diffusion. Front. Microbiol. 2012, 3, 110.
- Seyedjavadi, S.S.; Goudarzi, M.; Sabzehali, F. Relation between blaTEM, blaSHV and blaCTX-M genes and acute urinary tract infections. J. Acute Dis. 2016, 5, 71–76.
- Nandagopal, B.; Sankar, S.; Sagadevan, K.; Arumugam, H.; Jesudason, M.V.; Aswathaman, K.; Nair, A. Frequency of extended spectrum β-lactamase producing urinary isolates of Gram-negative bacilli among patients seen in a multispecialty hospital in Vellore district, India. Indian J. Med. Microbiol. 2015, 33, 282–285.
- El Bouamri, M.C.; Arsalane, L.; Zerouali, K.; Katfy, K.; El Kamouni, Y.; Zouhair, S. Molecular characterization of extended spectrum β-lactamase-producing Escherichia coli in a university hospital in Morocco, North Africa. African J. Urol. 2015, 21, 161–166.
- Carvalho, I.; Tejedor-Junco, M.T.; González-Martín, M.; Corbera, J.A.; Silva, V.; Igrejas, G.; Torres, C.; Poeta, P. Escherichia coli producing extended-spectrum β-lactamases (ESBL) from domestic camels in the Canary Islands: A one health approach. Animals 2020, 10, 1295.
- O’Keefe, A.; Hutton, T.A.; Schifferli, D.M.; Rankin, S.C. First detection of CTX-M and SHV extended-spectrum β-lactamases in Escherichia coli urinary tract isolates from dogs and cats in the United States. Antimicrob. Agents Chemother. 2010, 54, 3489–3492.
- Seo, K.W.; Lee, Y.J. The occurrence of CTX-M–producing E. coli in the broiler parent stock in Korea. Poult. Sci. 2021, 100, 1008–1015.
- Cormier, A.; Zhang, P.L.C.; Chalmers, G.; Weese, J.S.; Deckert, A.; Mulvey, M.; McAllister, T.; Boerlin, P. Diversity of CTX-M-positive Escherichia coli recovered from animals in Canada. Vet. Microbiol. 2019, 231, 71–75.
- Maciuca, I.E.; Williams, N.J.; Tuchilus, C.; Dorneanu, O.; Guguianu, E.; Carp-Carare, C.; Rimbu, C.; Timofte, D. High prevalence of Escherichia coli-producing CTX-M-15 extended-spectrum beta-lactamases in poultry and human clinical isolates in Romania. Microb. Drug Resist. 2015, 21, 651–662.
- Girlich, D.; Poirel, L.; Carattoli, A.; Kempf, I.; Lartigue, M.F.; Bertini, A.; Nordmann, P. Extended-spectrum β-lactamase CTX-M-1 in Escherichia coli isolates from healthy poultry in France. Appl. Environ. Microbiol. 2007, 73, 4681–4685.
- Kola, A.; Kohler, C.; Pfeifer, Y.; Schwab, F.; Kühn, K.; Schulz, K.; Balau, V.; Breitbach, K.; Bast, A.; Witte, W.; et al. High prevalence of extended-spectrum-β-lactamase-producing Enterobacteriaceae in organic and conventional retail chicken meat, Germany. J. Antimicrob. Chemother. 2012, 67, 2631–2634.
- Liu, X.; Wei, X.; Liu, L.; Feng, X.; Shao, Z.; Han, Z.; Li, Y. Prevalence and characteristics of extended-spectrum β-lactamases-producing Escherichia coli from broiler chickens at different day-age. Poult. Sci. 2020, 99, 3688–3696.
- Clemente, L.; Leão, C.; Moura, L.; Albuquerque, T.; Amaro, A. Prevalence and characterization of ESBL/AmpC producing Escherichia coli from fresh meat in Portugal. Antibiotics 2021, 10, 1333.
- Subramanya, S.H.; Bairy, I.; Nayak, N.; Amberpet, R.; Padukone, S.; Metok, Y.; Bhatta, D.R.; Sathian, B. Detection and characterization of ESBLproducing Enterobacteriaceae from the gut of healthy chickens, Gallus gallus domesticus in rural Nepal: Dominance of CTX-M-15-non-ST131 Escherichia coli clones. PLoS ONE 2020, 15, e0227725.
- Hussain, A.; Shaik, S.; Ranjan, A.; Suresh, A.; Sarker, N.; Semmler, T.; Wieler, L.H.; Alam, M.; Watanabe, H.; Chakravortty, D.; et al. Genomic and Functional Characterization of Poultry Escherichia coli From India Revealed Diverse Extended-Spectrum β-Lactamase-Producing Lineages With Shared Virulence Profiles. Front. Microbiol. 2019, 10, 2766.
- ESVAC. Sales of Veterinary Antimicrobial Agents in 31 European Countries in 2021 (EMA/294674/2019); European Medicines Agency: Zuidas, Amsterdam, The Netherlands, 2021.
- FDA Withdrawal of Enrofloxacin for Poultry. Available online: https://www.fda.gov/animal-veterinary/recalls-withdrawals/withdrawal-enrofloxacin-poultry (accessed on 25 March 2023).
- Cheng, A.C.; Turnidge, J.; Collignon, P.; Looke, D.; Barton, M.; Gottlieb, T. Control of fluoroquinolone resistance through successful regulation, Australia. Emerg. Infect. Dis. 2012, 18, 1453–1460.
- Chauvin, C.; Le Devendec, L.; Jouy, E.; Le Cornec, M.; Francart, S.; Marois-Créhan, C.; Kempf, I. National prevalence of resistance to third-generation cephalosporins in Escherichia coli isolates from layer flocks in France. Antimicrob. Agents Chemother. 2013, 57, 6351–6353.
- Ramadan, H.; Jackson, C.R.; Frye, J.G.; Hiott, L.M.; Samir, M.; Awad, A.; Woodley, T.A. Antimicrobial resistance, genetic diversity and multilocus sequence typing of Escherichia coli from humans, retail chicken and ground beef in Egypt. Pathogens 2020, 9, 357.
- Lozica, L.; Repar, J.; Gottstein, Ž. Longitudinal study on the effect of autogenous vaccine application on the sequence type and virulence profiles of Escherichia coli in broiler breeder flocks. Vet. Microbiol. 2021, 259, 109159.
- Aslantaş, Ö. High occurrence of CMY-2-type beta-lactamase-producing Escherichia coli among broiler flocks in Turkey. Trop. Anim. Health Prod. 2020, 52, 1681–1689.
- Azam, M.; Mohsin, M.; Johnson, T.J.; Smith, E.A.; Johnson, A.; Umair, M.; Saleemi, M.K. Sajjad-ur-Rahman Genomic landscape of multi-drug resistant avian pathogenic Escherichia coli recovered from broilers. Vet. Microbiol. 2020, 247, 108766.
- Ilyas, S.; Rasool, M.H.; Arshed, M.J.; Qamar, M.U.; Aslam, B.; Almatroudi, A.; Khurshid, M. The Escherichia coli sequence type 131 harboring extended-spectrum beta-lactamases and carbapenemases genes from poultry birds. Infect. Drug Resist. 2021, 14, 805–813.
- Jamil, A.; Zahoor, M.A.; Nawaz, Z.; Siddique, A.B.; Khurshid, M. Genetic Diversity of Escherichia coli Coharboring mcr-1 and Extended Spectrum Beta-Lactamases from Poultry. Biomed Res. Int. 2022, 2022, 8224883.
- Bengtsson-Palme, J. Antibiotic resistance in the food supply chain: Where can sequencing and metagenomics aid risk assessment? Curr. Opin. Food Sci. 2017, 14, 66–71.
- Gantois, I.; Ducatelle, R.; Pasmans, F.; Haesebrouck, F.; Gast, R.; Humphrey, T.J.; Van Immerseel, F. Mechanisms of egg contamination by Salmonella enteritidis. FEMS Microbiol. Rev. 2009, 33, 718–738.
- Bertelloni, F.; Salvadori, C.; Moni, A.; Cerri, D.; Mani, P.; Ebani, V.V. Antimicrobial resistance in Enterococcus spp. Isolated from laying hens of backyard poultry flocks. Ann. Agric. Environ. Med. 2015, 22, 665–669.
- Rouger, A.; Tresse, O.; Zagorec, M. Bacterial contaminants of poultry meat: Sources, species, and dynamics. Microorganisms 2017, 5, 50.
- Adeyanju, G.T.; Ishola, O. Salmonella and Escherichia coli contamination of poultry meat from a processing plant and retail markets in Ibadan, Oyo state, Nigeria. Springerplus 2014, 3, 139.
- Almeida, A.; Duarte, S.; Nunes, R.; Rocha, H.; Pena, A.; Meisel, L. Human and veterinary antibiotics used in Portugal-A ranking for ecosurveillance. Toxics 2014, 2, 188–225.
- Hu, Y.; Gao, G.F.; Zhu, B. The antibiotic resistome: Gene flow in environments, animals and human beings. Front. Med. 2017, 11, 161–168.
- Scott, A.; Tien, Y.C.; Drury, C.F.; Reynolds, W.D.; Topp, E. Enrichment of antibiotic resistance genes in soil receiving composts derived from swine manure, yard wastes, or food wastes, and evidence for multiyear persistence of swine Clostridium spp. Can. J. Microbiol. 2018, 64, 201–208.
- Silva, V.; Ribeiro, J.; Rocha, J.; Manaia, C.M.; Silva, A.; Pereira, J.E.; Maltez, L.; Capelo, J.L.; Igrejas, G.; Poeta, P. High Frequency of the EMRSA-15 Clone (ST22-MRSA-IV) in Hospital Wastewater. Microorganisms 2022, 10, 147.
- Stępień-Pyśniak, D.; Hauschild, T.; Kosikowska, U.; Dec, M.; Urban-Chmiel, R. Biofilm formation capacity and presence of virulence factors among commensal Enterococcus spp. from wild birds. Sci. Rep. 2019, 9, 11204.
- Aslam, B.; Khurshid, M.; Arshad, M.I.; Muzammil, S.; Rasool, M.; Yasmeen, N.; Shah, T.; Chaudhry, T.H.; Rasool, M.H.; Shahid, A.; et al. Antibiotic Resistance: One Health One World Outlook. Front. Cell Infect. Microbiol. 2021, 11, 1153.
- Park, S.Y.; Woodward, C.L.; Kubena, L.F.; Nisbet, D.J.; Birkhold, S.G.; Ricke, S.C. Environmental dissemination of foodborne Salmonella in preharvest poultry production: Reservoirs, critical factors, and research strategies. Crit. Rev. Environ. Sci. Technol. 2008, 38, 73–111.
- Trudeau, S.; Thibodeau, A.; Côté, J.C.; Gaucher, M.L.; Fravalo, P. Contribution of the Broiler Breeders’ Fecal Microbiota to the Establishment of the Eggshell Microbiota. Front. Microbiol. 2020, 11, 666.
- AVEC Annual Report 2022; AVEC: Brussels, Belgium, 2022.
- Poultry. Brazilian Farmers Poultry. 2022. Available online: brazilianfarmers.com/discover/poultry–2/ (accessed on 27 March 2023).
- Getachew, Y.; Hassan, L.; Zakaria, Z.; Abdul Aziz, S. Genetic variability of vancomycin-resistant Enterococcus faecium and Enterococcus faecalis isolates from humans, chickens, and pigs in Malaysia. Appl. Environ. Microbiol. 2013, 79, 4528–4533.
- Brinkwirth, S.; Ayobami, O.; Eckmanns, T.; Markwart, R. Hospital-acquired infections caused by enterococci: A systematic review and meta-analysis, WHO European region, 1 January 2010 to 4 February 2020. Eurosurveillance 2021, 26, 2001628.
- Hammerum, A.M. Enterococci of animal origin and their significance for public health. Clin. Microbiol. Infect. 2012, 18, 619–625.
- Anghinah, R.; Watanabe, R.G.S.; Simabukuro, M.M.; Guariglia, C.; Pinto, L.F.; de Menezes e Gonçalves, D.C. Native Valve Endocarditis due to Enterococcus hirae Presenting as a Neurological Deficit. Case Rep. Neurol. Med. 2013, 2013, 636070.
- Poyart, C.; Lambert, T.; Morand, P.; Abassade, P.; Quesne, G.; Baudouy, Y.; Trieu-Cuot, P. Native valve endocarditis due to Enterococcus hirae. J. Clin. Microbiol. 2002, 40, 2689–2690.
- Ahmed, F.Z.; Baig, M.W.; Gascoyne-Binzi, D.; Sandoe, J.A.T. Enterococcus cecorum aortic valve endocarditis. Diagn. Microbiol. Infect. Dis. 2011, 70, 525–527.
- Woo, P.C.Y.; Tam, D.M.W.; Lau, S.K.P.; Fung, A.M.Y.; Yuen, K.Y. Enterococcus cecorum Empyema Thoracis Successfully Treated with Cefotaxime. J. Clin. Microbiol. 2004, 42, 919–922.
- Hsueh, P.R.; Teng, L.J.; Chen, Y.C.; Yang, P.C.; Ho, S.W.; Luh, K.T. Recurrent bacteremic peritonitis caused by Enterococcus cecorum in a patient with liver cirrhosis. J. Clin. Microbiol. 2000, 38, 2450–2452.
- De Baere, T.; Claeys, G.; Verschraegen, G.; Devriese, L.A.; Baele, M.; Van Vlem, B.; Vanholder, R.; Dequidt, C.; Vaneechoutte, M. Continuous ambulatory peritoneal dialysis peritonitis due to Enterococcus cecorum. J. Clin. Microbiol. 2000, 38, 3511–3512.
- Sørensen, T.L.; Blom, M.; Monnet, D.L.; Frimodt-Møller, N.; Poulsen, R.L.; Espersen, F. Transient intestinal carriage after ingestion of Antibiotic-resistant Enterococcus faecium from chicken and pork. N. Engl. J. Med. 2001, 345, 1161–1166.
- Tzavaras, I.; Siarkou, V.I.; Zdragas, A.; Kotzamanidis, C.; Vafeas, G.; Bourtzi-Hatzopoulou, E.; Pournaras, S.; Sofianou, D. Diversity of vanA-type vancomycin-resistant Enterococcus faecium isolated from broilers, poultry slaughterers and hospitalized humans in Greece. J. Antimicrob. Chemother. 2012, 67, 1811–1818.
- Nowakiewicz, A.; Ziółkowska, G.; Trościańczyk, A.; Zięba, P.; Gnat, S. Determination of resistance and virulence genes in Enterococcus faecalis and E. faecium strains isolated from poultry and their genotypic characterization by ADSRRS-fingerprinting. Poult. Sci. 2017, 96, 986–996.
- Pormohammad, A.; Nasiri, M.J.; Azimi, T. Prevalence of antibiotic resistance in Escherichia coli strains simultaneously isolated from humans, animals, food, and the environment: A systematic review and meta-analysis. Infect. Drug Resist. 2019, 12, 1181–1197.
- Winokur, P.L.; Vonstein, D.L.; Hoffman, L.J.; Uhlenhopp, E.K.; Doern, G.V. Evidence for transfer of CMY-2 AmpC β-lactamase plasmids between Escherichia coli and Salmonella isolates from food animals and humans. Antimicrob. Agents Chemother. 2001, 45, 2716–2722.
- Fei, X.; Yin, K.; Yin, C.; Hu, Y.; Li, J.; Zhou, Z.; Tian, Y.; Geng, S.; Chen, X.; Pan, Z.; et al. Analyses of prevalence and molecular typing reveal the spread of antimicrobial-resistant Salmonella infection across two breeder chicken farms. Poult. Sci. 2018, 97, 4374–4383.
- Noh, E.B.; Kim, Y.B.; Seo, K.W.; Son, S.H.; Ha, J.S.; Lee, Y.J. Antimicrobial resistance monitoring of commensal Enterococcus faecalis in broiler breeders. Poult. Sci. 2020, 99, 2675–2683.
- Ferreira, J.C.; Penha Filho, R.A.C.; Kuaye, A.P.Y.; Andrade, L.N.; Chang, Y.F.; Darini, A.L.C. Virulence potential of commensal multidrug resistant Escherichia coli isolated from poultry in Brazil. Infect. Genet. Evol. 2018, 65, 251–256.
- Díaz-Jiménez, D.; García-Meniño, I.; Fernández, J.; García, V.; Mora, A. Chicken and turkey meat: Consumer exposure to multidrug-resistant Enterobacteriaceae including mcr-carriers, uropathogenic E. coli and high-risk lineages such as ST131. Int. J. Food Microbiol. 2020, 331, 108750.
- Johnson, J.R.; Porter, S.B.; Johnston, B.; Thuras, P.; Clock, S.; Crupain, M.; Ranganb, U. Extraintestinal Pathogenic and Antimicrobial-Resistant Escherichia coli, Including Sequence Type 131 (ST131), from Retail Chicken Breasts in the United States in 2013. Appl. Enviromental Microbiol. 2017, 83, e02956-16.
- Falgenhauer, L.; Imirzalioglu, C.; Oppong, K.; Akenten, C.W.; Hogan, B.; Krumkamp, R.; Poppert, S.; Levermann, V.; Schwengers, O.; Sarpong, N.; et al. Detection and characterization of ESBL-producing Escherichia coli from humans and poultry in Ghana. Front. Microbiol. 2019, 10, 3358.
- Mughini-Gras, L.; Dorado-García, A.; van Duijkeren, E.; van den Bunt, G.; Dierikx, C.M.; Bonten, M.J.M.; Bootsma, M.C.J.; Schmitt, H.; Hald, T.; Evers, E.G.; et al. Attributable sources of community-acquired carriage of Escherichia coli containing β-lactam antibiotic resistance genes: A population-based modelling study. Lancet Planet. Health 2019, 3, e357–e369.
- Wada, Y.; Harun, A.B.; Yean, C.Y.; Zaidah, A.R. Vancomycin-Resistant Enterococcus: Issues in Human Health, Animal Health, Resistant Mechanisms and the Malaysian Paradox. Adv. Anim. Vet. Sci. 2019, 7, 1021–1034.
- Mollenkopf, D.F.; Cenera, J.K.; Bryant, E.M.; King, C.A.; Kashoma, I.; Kumar, A.; Funk, J.A.; Rajashekara, G.; Wittum, T.E. Organic or antibiotic-free labeling does not impact the recovery of enteric pathogens and antimicrobial-resistant Escherichia coli from fresh retail chicken. Foodborne Pathog. Dis. 2014, 11, 920–929.
- Musa, L.; Proietti, P.C.; Marenzoni, M.L.; Stefanetti, V.; Kika, T.S.; Blasi, F.; Magistrali, C.F.; Toppi, V.; Ranucci, D.; Branciari, R.; et al. Susceptibility of commensal E. coli isolated from conventional, antibiotic-free, and organic meat chickens on farms and at slaughter toward antimicrobials with public health relevance. Antibiotics 2021, 10, 1321.
- Kawalec, M.; Pietras, Z.; Daniłowicz, E.; Jakubczak, A.; Gniadkowski, M.; Hryniewicz, W.; Willems, R.J.L. Clonal structure of Enterococcus faecalis isolated from Polish hospitals: Characterization of epidemic clones. J. Clin. Microbiol. 2007, 45, 147–153.
- Kuch, A.; Willems, R.J.L.; Werner, G.; Coque, T.M.; Hammerum, A.M.; Sundsfjord, A.; Klare, I.; Ruiz-Garbajosa, P.; Simonsen, G.S.; van Luit-Asbroek, M.; et al. Insight into antimicrobial susceptibility and population structure of contemporary human Enterococcus faecalis isolates from Europe. J. Antimicrob. Chemother. 2012, 67, 551–558.
- Jolley, K.A.; Bray, J.E.; Maiden, M.C.J. Open-access bacterial population genomics: BIGSdb software, the PubMLST.org website and their applications. Wellcome Open Res. 2018, 3, 124.
- Tedim, A.P.; Ruiz-Garbajosa, P.; Corander, J.; Rodríguez, C.M.; Cantón, R.; Willems, R.J.; Baquero, F.; Coque, T.M. Population biology of intestinal Enterococcus Isolates from hospitalized and nonhospitalized individuals in different age groups. Appl. Environ. Microbiol. 2015, 81, 1820–1831.
- Farman, M.; Yasir, M.; Al-Hindi, R.R.; Farraj, S.A.; Jiman-Fatani, A.A.; Alawi, M.; Azhar, E.I. Genomic analysis of multidrug-resistant clinical Enterococcus faecalis isolates for antimicrobial resistance genes and virulence factors from the western region of Saudi Arabia. Antimicrob. Resist. Infect. Control 2019, 8, 55.
- He, T.; Shen, Y.; Schwarz, S.; Cai, J.; Lv, Y.; Li, J.; Feßler, A.T.; Zhang, R.; Wu, C.; Shen, J.; et al. Genetic environment of the transferable oxazolidinone/phenicol resistance gene optrA in Enterococcus faecalis isolates of human and animal origin. J. Antimicrob. Chemother. 2016, 71, 1466–1473.
- Lee, J.H.; Shin, D.; Lee, B.; Lee, H.; Lee, I.; Jeong, D.W. Genetic diversity and antibiotic resistance of Enterococcus faecalis isolates from traditional Korean fermented soybean foods. J. Microbiol. Biotechnol. 2017, 27, 916–924.
- Freitas, A.R.; Tedim, A.P.; Novais, C.; Lanza, V.F.; Peixe, L. Comparative genomics of global optrA-carrying Enterococcus faecalis uncovers a common chromosomal hotspot for optrA acquisition within a diversity of core and accessory genomes. Microb. Genomics 2020, 6, e000350.
- Sakai, Y.; Tsukahara, T.; Ushida, K. Possibility of vancomycin-resistant enterococci transmission from human to broilers, and possibility of using the vancomycin-resistant gram-positive cocci as a model in a screening study of vancomycin-resistant enterococci infection in the broiler chick. Anim. Sci. J. 2006, 77, 538–544.
- Yu, F.; Chen, X.; Zheng, S.; Han, D.; Wang, Y.; Wang, R.; Wang, B.; Chen, Y. Prevalence and genetic diversity of human diarrheagenic Escherichia coli isolates by multilocus sequence typing. Int. J. Infect. Dis. 2018, 67, 7–13.
- Papouskova, A.; Papouskova, A.; Masarikova, M.; Masarikova, M.; Valcek, A.; Valcek, A.; Senk, D.; Cejkova, D.; Jahodarova, E.; Cizek, A.; et al. Genomic analysis of Escherichia coli strains isolated from diseased chicken in the Czech Republic. BMC Vet. Res. 2020, 16, 189.
- Jørgensen, S.L.; Stegger, M.; Kudirkiene, E.; Lilje, B.; Poulsen, L.L.; Ronco, T.; Pires Dos Santos, T.; Kiil, K.; Bisgaard, M.; Pedersen, K.; et al. Diversity and Population Overlap between Avian and Human Escherichia coli Belonging to Sequence Type 95. mSphere 2019, 4, e00333-18.
- Danzeisen, J.L.; Wannemuehler, Y.; Nolan, L.K.; Johnson, T.J. Comparison of multilocus sequence analysis and virulence genotyping of Escherichia coli from live birds, retail poultry meat, and human extraintestinal infection. Avian Dis. 2013, 57, 104–108.
- Bert, F.; Johnson, J.R.; Ouattara, B.; Leflon-Guibout, V.; Johnston, B.; Marcon, E.; Valla, D.; Moreau, R.; Nicolas-Chanoine, M.H. Genetic diversity and virulence profiles of Escherichia coli isolates causing spontaneous bacterial peritonitis and bacteremia in patients with cirrhosis. J. Clin. Microbiol. 2010, 48, 2709–2714.
- Pires-dos-Santos, T.; Bisgaard, M.; Christensen, H. Genetic diversity and virulence profiles of Escherichia coli causing salpingitis and peritonitis in broiler breeders. Vet. Microbiol. 2013, 162, 873–880.
- Nicolas-Chanoine, M.H.; Bertrand, X.; Madec, J.Y. Escherichia coli ST131, an intriguing clonal group. Clin. Microbiol. Rev. 2014, 27, 543–574.
- Ronco, T.; Stegger, M.; Olsen, R.H.; Sekse, C.; Nordstoga, A.B.; Pohjanvirta, T.; Lilje, B.; Lyhs, U.; Andersen, P.S.; Pedersen, K. Spread of avian pathogenic Escherichia coli ST117 O78: H4 in Nordic broiler production. BMC Genom. 2017, 18, 13.
- Kieffer, N.; Nordmann, P.; Aires-De-Sousa, M.; Poirel, L. High prevalence of carbapenemase-producing Enterobacteriaceae among hospitalized children in Luanda, Angola. Antimicrob. Agents Chemother. 2016, 60, 6189–6192.
- Báez, J.; Hernández-García, M.; Guamparito, C.; Díaz, S.; Olave, A.; Guerrero, K.; Cantón, R.; Baquero, F.; Gahona, J.; Valenzuela, N.; et al. Molecular characterization and genetic diversity of ESBL-producing Escherichia coli colonizing the migratory Franklin’s Gulls (Leucophaeus pipixcan) in Antofagasta, North of Chile. Microb. Drug Resist. 2015, 21, 111–116.
