The widespread use of peptide receptor radionuclide therapy (PRRT) represents a major therapeutic breakthrough in nuclear medicine, particularly since the introduction of 177Lu-radiolabeled somatostatin analogs. These radiopharmaceuticals have especially improved progression-free survival and quality of life in patients with inoperable metastatic gastroenteropancreatic neuroendocrine tumors expressing somatostatin receptors. In the case of aggressive or resistant disease, the use of somatostatin derivatives radiolabeled with an alpha-emitter could provide a promising alternative. Among the currently available alpha-emitting radioelements, actinium-225 has emerged as the most suitable candidate, especially regarding its physical and radiochemical properties.
- actinium-225
- radionuclide production
- radiolabeling
- targeted radionuclide therapy
- targeted alpha-therapy
- radiobiology
- neuroendocrine tumors
- 225Ac–DOTATATE
- radiopharmaceuticals
- 225Ac–DOTATATE
1. Introduction
1.1. About Neuroendocrine Tumors
1.2. Somatostatin Receptors and Octreotide Analogs
Although NETs are heterogeneous diseases in their pathophysiology and clinical expression, they usually share the characteristic of overexpressing somatostatin receptors (SSTRs) [13][4]. Five SSTR subtypes are described (SSTR1 to SSTR5), SSTR2 being the most frequently encountered in differentiated NETs [14][5]. However, several subtypes can be expressed concomitantly on tumor cells in various combinations and proportions [15,16][6][7]. NETs overexpressing SSTRs most often have a gastrointestinal, pancreatic, bronchial, pulmonary, or even thymic or breast origin. SSTRs belong to the G-protein-coupled receptor family and are localized at the cell membrane. Their natural peptide ligand, somatostatin, is found in humans under two different forms: one of 14 amino acids (SS-14) and one of 28 amino acids (SS-28) (Figure 1) [17,18][8][9]. Natural somatostatin has been shown to be unsuitable for in vivo use due to its short plasma half-life (about 3 min) [19][10]. Analogues of this hormone, more resistant to enzymatic degradation, have therefore been developed by making various modifications to the natural molecule [20,21][11][12]. The introduction of D-series amino acids to improve in vivo stability, the retention of the minimum chain length to maintain biological activity, the use of the hexapeptide motif Cys-Phe-D-Trp-Lys-Thr-Cys and the elongation of the N- and C-terminal ends allowed the characterization, in 1982, of the most stable active somatostatin analog known as octreotide (Figure 1) [22][13].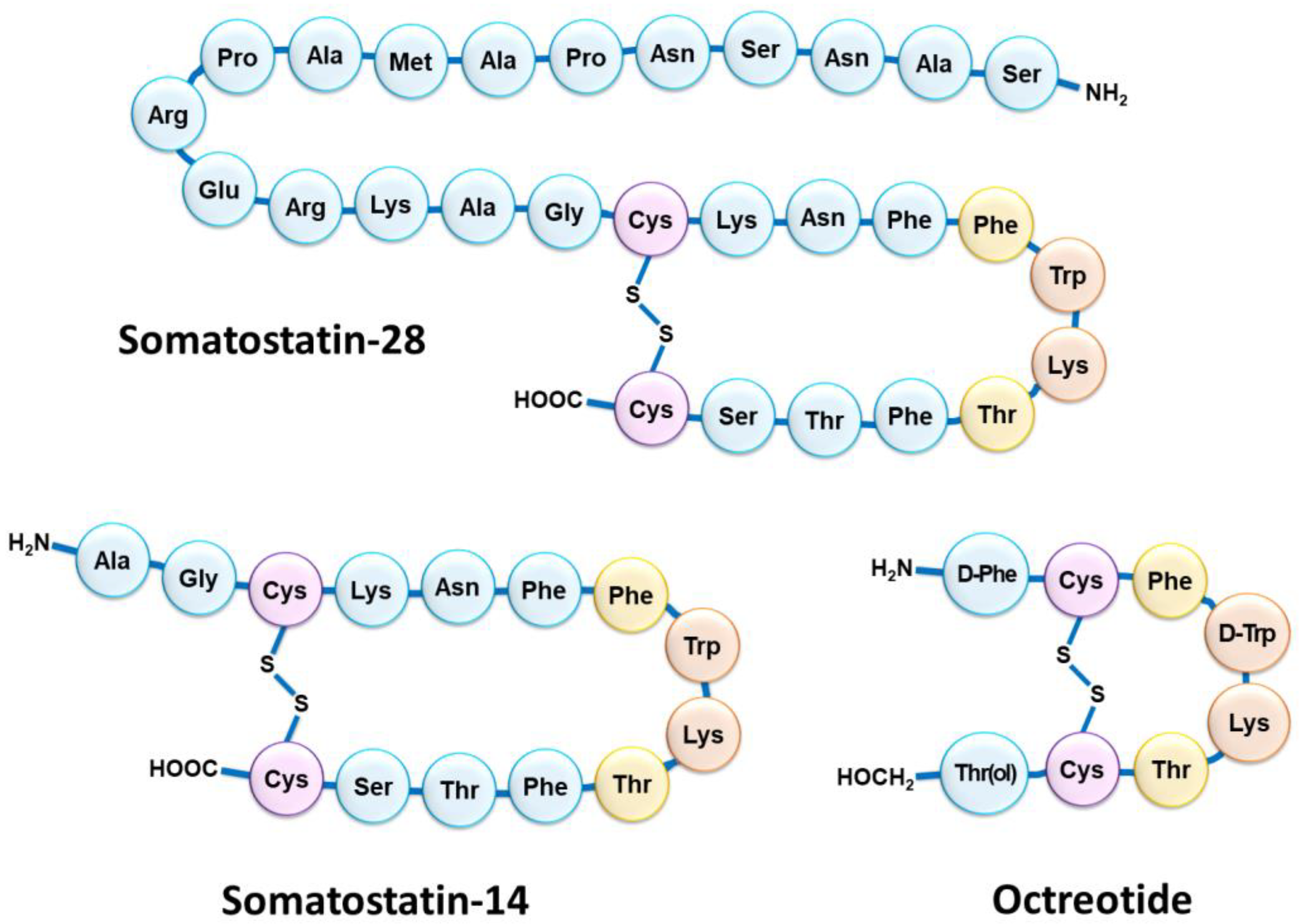
1.3. SSTR Targeting for Peptide Receptor Radionuclide Therapy
1.4. PRRT Using Somatostatin Analogs Radiolabeled with Alpha-Emitters
Although β-PRRT remains an approved treatment for unresectable metastatic NETs, some tumors show resistance to β-emissions despite somatostatin receptor expression [69][25]. Furthermore, not all treated patients achieve partial or complete response following SSTR-targeting 177Lu-PRRT, and relapse is often observed in the years post-treatment [70][26]. Thus, among the strategies considered in an effort to overcome these drawbacks, octreotide derivatives radiolabeled with alpha-emitting radionuclides have received particular attention [71][27]. Within this group of radioisotopes, radium-223 (alkaline earth metal, group 2) has been extensively studied both in vitro and in vivo, and has paved the way for the use of alpha-emitting radioelements in patients [72][28]. To date, radium-223 is used in its dichloride form for the treatment of symptomatic bone metastases in patients with castration-resistant prostate cancer, without known visceral metastatic disease. However, due to its particular chemistry, 223Ra is not suitable for DOTA-peptide radiolabeling. Thus, a special interest has emerged for several α-emitting lanthanides (e.g., 149Tb) and actinides (e.g., 227Th and 225Ac), as well as some radioelements from their decay chain (e.g., 213Bi) to achieve a convenient complex formation with DOTA [73][29]. An initial preclinical evaluation of 213Bi–DOTATOC showed its potential value in NETs resistant to 177Lu-PRRT [74,75,76[30][31][32][33],77], these properties being promptly confirmed in the clinic [78][34].2. Actinium-225: Decay Characteristics, Radiobiological and Dosimetry Considerations
2.1. Physical Properties of Actinium-225
Actinium-225 is a relatively long-lived pure alpha-emitter, with a half-life of 9.9 days that is well-suited for radionuclide therapy applications and for centralized industrial production, distant from the (pre)clinical user sites. It is formed from the 229Th decay product 225Ra and decays via a cascade of six short-lived daughter radionuclides to the nearly stable bismuth-209 (Figure 32) [81][35]. These intermediates include francium-221 (t1/2 = 4.8 min, 6.3 MeV α-particle and 218 keV γ-emission), astatine-217 (t1/2 = 33 ms, 7.1 MeV α-particle), bismuth-213 (t1/2 = 45.6 min, 5.9 MeV α-particle, 1.4 MeV β-particle and 440 keV γ-emission), polonium-213 (t1/2 = 4.3 μs, 8.5 MeV α-particle), thallium-209 (t1/2 = 2.2 min, 3.9 MeV β-particle) and lead-209 (t1/2 = 3.2 h, 0.6 MeV β-particle) before reaching 209Bi. Overall, the predominant decay pathway of 225Ac produces four alpha-particles with energies ranging from 5.8 to 8.5 MeV and associated tissue ranges of 47 to 85 µm. In addition, the cascade includes two main beta-disintegrations of 1.4 and 0.6 MeV maximum energy. Therefore, 225Ac is considered as an in vivo radionuclide generator or a “nanogenerator” with regard to its decay chain.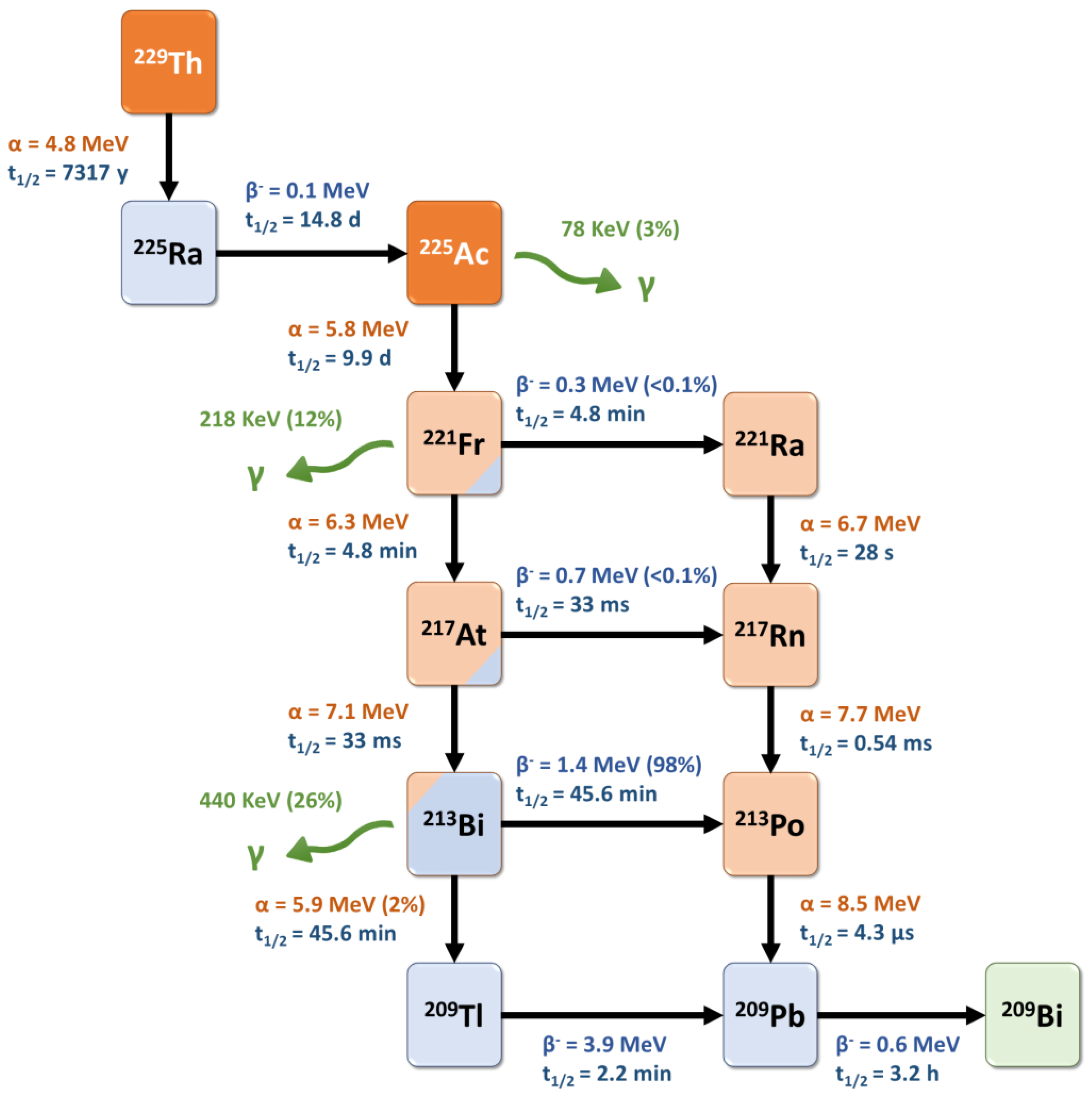
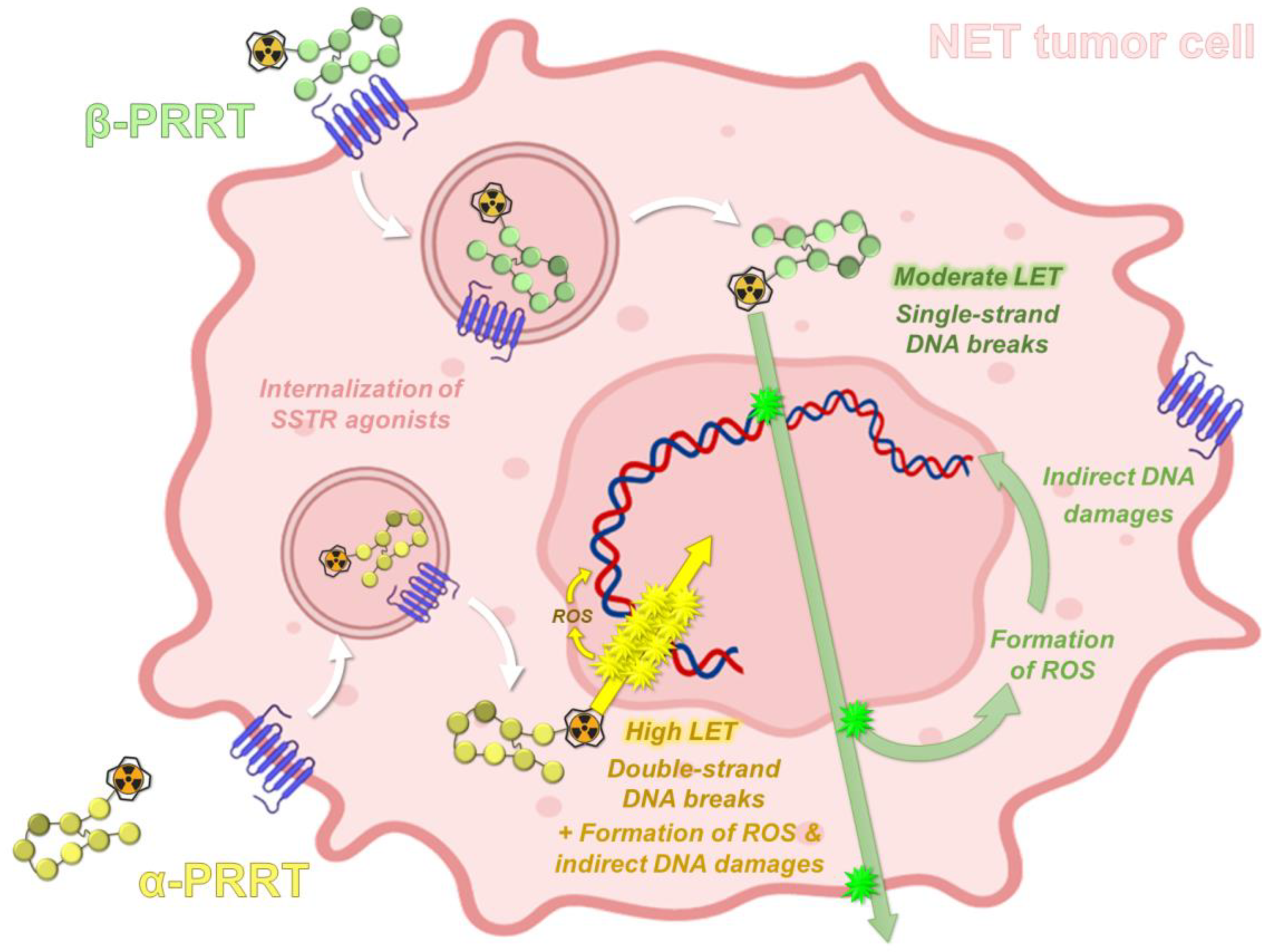
2.2. Radiobiological Properties of Actinium-225
2.3. Dosimetry for Targeted Alpha-Therapy with
225
Ac
3. Radiochemical and Preclinical Development of [225Ac]Ac–DOTATATE
3.1. Production of Actinium-225
Two isotopes of actinium, 227Ac and 228Ac, exist in nature within the natural decay chain of uranium-235 and thorium-232, respectively [94][44]. However, neither of these two isotopes is used in the clinic, with 228Ac representing a minimal part of natural actinium and 227Ac having a very long half-life (t1/2 = 21.77 y). Therefore, 225Ac is the only one of the more than 30 known actinium isotopes to be used in preclinical and clinical studies to date. The main method for generating 225Ac for clinical use is through radiochemical extraction following the decay of 229Th (t1/2 = 7397 y), which originates from reactor-bred 233U [95,96][45][46]. The main sources of 229Th in the world for 225Ac used in preclinical and clinical studies are Oak Ridge National Laboratory (ORNL, Oak Ridge, TN, USA) [95][45], the Institute of Physics and Power Engineering (IPPE, Obninsk, Russia) [97][47], and the Directorate for Nuclear Safety and Security of the Joint Research Center of the European commission (JRC, Halstenbek, Germany), formerly the Institute for Transuranium Elements (ITE, Karlsruhe, Germany) [98][48]. More recently, the Canadian Nuclear Laboratories set up a 225Ac production chain that could supply up to 3.7 GBq of this radioisotope annually [99][49]. Consequently, accelerator-based production techniques to obtain 225Ac have been developed. The most promising approach to obtain 225Ac at a large scale may be the cyclotron proton irradiation of a 226Ra target, involving the 226Ra(p,2n)225Ac transformation [111,112][50][51]. With a high cross-section peak (710 mb) at 16.8 MeV, this convenient method can be performed on low-energy cyclotrons.3.2. Chemistry of Actinium
3.2.1. Actinium in Aqueous Solution
Actinium is the chemical element with atomic number 89 and the first element of the actinide group, to which it gives its name. Nevertheless, actinium has rather similar chemical properties to lanthanum and other lanthanides. Actinium exists essentially in the +3 oxidation state in aqueous solution; additionally, Ac3+ is the largest +3 cation in the periodic table. It is also the most basic +3 ion due to its low charge density, directly related to its large size. Although the +3 state is the most stable in aqueous solution, the +2 oxidation state may also be encountered [120][52]. This second species is assumed because a reduction half-wave potential in a 225Ac3+ aqueous solution can be observed. The progressive negative shift of this potential in the presence of increasing 18-crown-6 concentrations has been attributed to the formation of a complex between crown ether and divalent actinium [121][53]. However, without the effect of 18-crown-6 on the reduction of 225Ac3+, the existence of stable 225Ac2+ ions in aqueous solution remains unlikely regarding the low extraction yields of actinium using sodium amalgam in aqueous sodium acetate, an extraction technique usually efficient for lanthanides at a stable +2 oxidation state [122][54].3.2.2. Coordination Chemistry of Actinium
The usefulness of actinium-225 as a radionuclide for therapeutic purposes has been limited for a long time by the unavailability of chelating agents that are both capable of being compatible with this bulky radionuclide and of controlling the fate of the resulting daughter emitters, particularly with regard to their alpha-recoil, which is related to the conservation of momentum laws that occurs upon release of an alpha-particle [137][55]. Nevertheless, the coordination chemistry of such a clinically relevant alpha-emitter has recently gained more and more interest [138][56]. Considering its low polarizability and despite its large ionic radius, the Ac3+ ion is considered a hard Lewis acid [139][57], showing a medium absolute chemical hardness value of 14.4 eV [140][58]. As such, it will complex more easily with hard ligands, such as anionic oxygen donors. The complexation reaction will preferentially occur under charge control and the acid–base bond will be essentially ionic. Indeed, Ac3+ displays an electrostatic interaction constant (EA) value of 2.84 and a covalent interaction constant (CA) value of 0.28 [138][56]. This predominance of charge interactions can be predicted from the character of the frontier molecular orbitals, which are centered on the nuclei of the donor and acceptor atoms; when these atoms are close together in space, the overlaps of the orbitals are negligible while the charge interactions are strong. This is mainly attributed to the density of the charge, which is very significant in ions of hard consistency. The high ionic radius of the Ac3+ cation suggests that the most suitable chelators would be polydentate agents, with a high denticity between 8 and 12. Initial works investigated the suitability of linear polyaminocarboxylate chelators, such as CHX-A″-DTPA, for the chelation of the 225Ac3+ cation [141,142][59][60]. These efforts were motivated by the advantageous radiolabeling kinetic properties of these ligands; however, the complexes obtained did not show sufficient in vivo stability. Subsequently, large macrocyclic chelators were considered and the 18-membered polyaminocarboxylic acid core HEHA was rapidly identified as particularly suitable for actinium complexation [142,143][60][61].3.2.3. Relevance of DOTA in Actinium Radiopharmaceuticals
The in vivo fate of the 225Ac–DOTA complex alone was initially shown to be safe, with only low activity amounts in liver and bone of BALB/c mice [142][60]. Subsequently, DOTA-bioconjugated constructs (either antibodies or peptides) also showed the sufficient stability of the complex, both in vitro [162,166,167,173][62][63][64][65] and in vivo [162,166][62][63]. Nevertheless, early studies raised some concerns about the compatibility of DOTA with actinium [142,146][60][66]. Indeed, the large ionic radius of the Ac3+ ion is not in favor of the good thermodynamic stability of the DOTA complex, which may also be subject to transmetalation with other cations. In order to minimize adverse in vivo effects associated with the loss of 225Ac and its daughter radionuclides (especially 213Bi, significantly increasing the kidney-absorbed dose [194][67]) from DOTA, several approaches have been considered, such as the co-administration of chelating agents or concomitant diuresis [195,196][68][69].3.3. Somatostatin Analogs Radiolabeled with
225
Ac: Preclinical Studies
Only a few studies have reported preclinical efficacy results of 225Ac-radiolabeled somatostatin analogues, due to this group of vector molecules having already been widely studied with beta-emitters such as 90Y or 177Lu [197][70].
Activities between 10 and 60 kBq were well-tolerated by the mice; however, activities over 30 kBq induced pathologic changes in the renal cortex, suggesting radiation-induced acute tubular necrosis in both the distal and proximal tubules. Similar results were obtained in another study on Sprague Dawley rats that received 111 or 370 kBq [225Ac]Ac–DOTATOC and developed renal tubular nephrosis or renal glomerulopathy [199][71]. Only a slight accumulation in the liver was objectified, probably due to the release of free 225Ac. After a single administration of the highest non-toxic activity (20 kBq), tumor weights 14 days after treatment were lower with [225Ac]Ac–DOTATOC than with [177Lu]Lu–DOTATOC (1 MBq), in accordance with previous studies investigating [213Bi]Bi–DOTATOC [74,75][30][31].
4. Clinical Use of 225Ac–DOTATATE
To date, [177Lu]Lu–DOTATATE is considered as the standard PRRT treatment for GEP NETs. In this regard, the phase 3 randomized control trial NETTER-1 specifically demonstrated that [177Lu]Lu–DOTATATE therapy plus long-acting octreotide was associated with a significantly longer PFS (28.4 vs. 8.5 months) than high-dose long-acting octreotide in advanced midgut GEP NET patients, although the OS endpoint of thise study did not reach statistical significance (48 vs. 36.3 months, p = 0.3) [62,205][72][73]. Thus, this therapy offers a promising option as an early-line treatment for advanced NET [60,62][72][74]. Nevertheless, this type of pathology is known to frequently relapse, which may lead to patient retreatment. In this context, several studies have investigated the value of a renewed treatment with β-PRRT; furthermore, TAT protocols using somatostatin analogs, especially 225Ac-based approaches, were also rapidly proposed as an alternative for patients that did not respond to β-PRRT. Although it was used several years earlier, the first literature report of an alpha-PRRT with [225Ac]Ac–DOTATOC in human dates from October 2018 [206][75]. Ten patients with metastatic NETs progressing after 90Y– and/or [177Lu]Lu–DOTATOC therapy were treated with intra-arterial [225Ac]Ac–DOTATOC (~8 MBq). Overall, the treatment was well-tolerated and effective, demonstrating its potential as a possible therapeutic alternative in advanced NETs resistant to β-PRRT. Then, two major studies involving 225Ac-labeled octreotide analogs were reported in patients with advanced-stage SSTR-expressing metastatic GEP NETs. These works primarily focused on the hematologic and renal toxicity of [225Ac]Ac–DOTATOC, and on the long-term outcomes of this therapeutic, respectively.5. Conclusions
Ahead of other α-emitters, TAT using 225Ac-labeled somatostatin analogs seems to be a promising therapeutic approach for metastatic or inoperable NETs, especially considering its preliminary efficacy and safety results. Efforts to achieve the sufficient production of 225Ac and extensive radiochemistry works aimed at optimizing the chelation of this radioelement reflect the high expectations for its clinical use, including in other pathologies such as prostate cancer with 225Ac-labeled PSMA ligands [88,233,234,235,236,237][39][76][77][78][79][80], or even in hematological cancers such as acute myeloid leukemia [238][81]. However, the role of TAT versus β-PRRT in terms of OS, PFS and long-term toxicity is still difficult to define without formal comparative studies. Beforehand, the further investigation into the therapeutic use modalities of 225Ac-radiolabeled somatostatin analogs will be required. Some of these questions may be answered by the ACTION-1 clinical trial (NCT05477576) [239][82], which is designed to determine the safety, pharmacokinetics, and recommended phase 3 dose of [225Ac]Ac–DOTATATE and its efficacy compared to the investigator-selected standard of care therapy in patients with inoperable GEP NETs that progressed following 177Lu–somatostatin analogues. Similarly, preliminary data on the efficacy of 225Ac-labeled somatostatin analogs in other cancers such as paragangliomas [211][83] or pheochromocytomas [240][84] will need to be further consolidated. From a radiopharmaceutical perspective, the importance of developing a reliable method for measuring the radiochemical purity of 225Ac conjugates produced in-house appears to be crucial and would certainly be a key requirement to obtain approval for clinical use from regulatory agencies. In addition, it will be interesting to develop a standardized dosimetric tool for the accurate estimation of adsorbed doses in target and non-target organs. For the time being, TATs constitute an emerging therapeutic alternative for patients with either highly resistant or late-stage disease, particularly in the context of compassionate access, depending on the country.References
- Basu, B.; Sirohi, B.; Corrie, P. Systemic Therapy for Neuroendocrine Tumours of Gastroenteropancreatic Origin. Endocr. Relat. Cancer 2010, 17, R75–R90.
- Scalettar, B.A.; Jacobs, C.; Fulwiler, A.; Prahl, L.; Simon, A.; Hilken, L.; Lochner, J.E. Hindered Submicron Mobility and Long-Term Storage of Presynaptic Dense-Core Granules Revealed by Single-Particle Tracking. Dev. Neurobiol. 2012, 72, 1181–1195.
- Kaltsas, G.A.; Besser, G.M.; Grossman, A.B. The Diagnosis and Medical Management of Advanced Neuroendocrine Tumors. Endocr. Rev. 2004, 25, 458–511.
- Hankus, J.; Tomaszewska, R. Neuroendocrine Neoplasms and Somatostatin Receptor Subtypes Expression. Nucl. Med. Rev. 2016, 19, 111–117.
- Reubi, J.C.; Waser, B.; Schaer, J.C.; Laissue, J.A. Somatostatin Receptor Sst1-Sst5 Expression in Normal and Neoplastic Human Tissues Using Receptor Autoradiography with Subtype-Selective Ligands. Eur. J. Nucl. Med. 2001, 28, 836–846.
- Schaer, J.C.; Waser, B.; Mengod, G.; Reubi, J.C. Somatostatin Receptor Subtypes Sst1, Sst2, Sst3 and Sst5 Expression in Human Pituitary, Gastroentero-Pancreatic and Mammary Tumors: Comparison of MRNA Analysis with Receptor Autoradiography. Int. J. Cancer 1997, 70, 530–537.
- Reubi, J.C. Peptide Receptors as Molecular Targets for Cancer Diagnosis and Therapy. Endocr. Rev. 2003, 24, 389–427.
- Böhlen, P.; Brazeau, P.; Benoit, R.; Ling, N.; Esch, F.; Guillemin, R. Isolation and Amino Acid Composition of Two Somatostatin-like Peptides from Ovine Hypothalamus: Somatostatin-28 and Somatostatin-25. Biochem. Biophys. Res. Commun. 1980, 96, 725–734.
- Shen, L.P.; Pictet, R.L.; Rutter, W.J. Human Somatostatin I: Sequence of the CDNA. Proc. Natl. Acad. Sci. USA 1982, 79, 4575–4579.
- Rai, U.; Thrimawithana, T.R.; Valery, C.; Young, S.A. Therapeutic Uses of Somatostatin and Its Analogues: Current View and Potential Applications. Pharmacol. Ther. 2015, 152, 98–110.
- Veber, D.F.; Holly, F.W.; Nutt, R.F.; Bergstrand, S.J.; Brady, S.F.; Hirschmann, R.; Glitzer, M.S.; Saperstein, R. Highly Active Cyclic and Bicyclic Somatostatin Analogues of Reduced Ring Size. Nature 1979, 280, 512–514.
- Veber, D.F.; Freidlinger, R.M.; Perlow, D.S.; Paleveda, W.J.; Holly, F.W.; Strachan, R.G.; Nutt, R.F.; Arison, B.H.; Homnick, C.; Randall, W.C.; et al. A Potent Cyclic Hexapeptide Analogue of Somatostatin. Nature 1981, 292, 55–58.
- Bauer, W.; Briner, U.; Doepfner, W.; Haller, R.; Huguenin, R.; Marbach, P.; Petcher, T.J.; Pless, J. SMS 201-995: A Very Potent and Selective Octapeptide Analogue of Somatostatin with Prolonged Action. Life Sci. 1982, 31, 1133–1140.
- Al-Toubah, T.; Strosberg, J. Peptide Receptor Radiotherapy Comes of Age. Endocrinol. Metab. Clin. N. Am. 2018, 47, 615–625.
- Krenning, E.P.; Kooij, P.P.; Bakker, W.H.; Breeman, W.A.; Postema, P.T.; Kwekkeboom, D.J.; Oei, H.Y.; de Jong, M.; Visser, T.J.; Reijs, A.E. Radiotherapy with a Radiolabeled Somatostatin Analogue, -Octreotide. A Case History. Ann. N. Y. Acad. Sci. 1994, 733, 496–506.
- Krenning, E.P.; Kooij, P.P.M.; Pauwels, S.; Breeman, W.A.P.; Postema, P.T.E.; DeHerder, W.W.; Valkema, R.; Kwekkeboom, D.J. Somatostatin Receptor: Scintigraphy and Radionuclide Therapy. Digestion 1996, 57, 57–61.
- Krenning, E.P.; de Jong, M.; Kooij, P.P.M.; Breeman, W.A.P.; Bakker, W.H.; de Herder, W.W.; van Eijck, C.H.J.; Kwekkeboom, D.J.; Jamar, F.; Pauwels, S.; et al. Radiolabelled Somatostatin Analogue(s) for Peptide Receptor Scintigraphy and Radionuclide Therapy. Ann. Oncol. 1999, 10, S23–S30.
- Valkema, R.; De Jong, M.; Bakker, W.H.; Breeman, W.A.P.; Kooij, P.P.M.; Lugtenburg, P.J.; De Jong, F.H.; Christiansen, A.; Kam, B.L.R.; De Herder, W.W.; et al. Phase I Study of Peptide Receptor Radionuclide Therapy with Octreotide: The Rotterdam Experience. Semin. Nucl. Med. 2002, 32, 110–122.
- Cybulla, M.; Weiner, S.; Otte, A. End-Stage Renal Disease after Treatment with 90Y-DOTATOC. Eur. J. Nucl. Med. Mol. Imaging 2001, 28, 1552–1554.
- Valkema, R.; Pauwels, S.; Kvols, L.K.; Barone, R.; Jamar, F.; Bakker, W.H.; Kwekkeboom, D.J.; Bouterfa, H.; Krenning, E.P. Survival and Response after Peptide Receptor Radionuclide Therapy with Octreotide in Patients with Advanced Gastroenteropancreatic Neuroendocrine Tumors. Semin. Nucl. Med. 2006, 36, 147–156.
- Bushnell, D.L.; O’Dorisio, T.M.; O’Dorisio, M.S.; Menda, Y.; Hicks, R.J.; Van Cutsem, E.; Baulieu, J.-L.; Borson-Chazot, F.; Anthony, L.; Benson, A.B.; et al. 90Y-Edotreotide for Metastatic Carcinoid Refractory to Octreotide. J. Clin. Oncol. 2010, 28, 1652–1659.
- Imhof, A.; Brunner, P.; Marincek, N.; Briel, M.; Schindler, C.; Rasch, H.; Mäcke, H.R.; Rochlitz, C.; Müller-Brand, J.; Walter, M.A. Response, Survival, and Long-Term Toxicity after Therapy with the Radiolabeled Somatostatin Analogue -TOC in Metastasized Neuroendocrine Cancers. J. Clin. Oncol. 2011, 29, 2416–2423.
- Kwekkeboom, D.J.; Bakker, W.H.; Kooij, P.P.; Konijnenberg, M.W.; Srinivasan, A.; Erion, J.L.; Schmidt, M.A.; Bugaj, J.L.; de Jong, M.; Krenning, E.P. Octreotate: Comparison with Octreotide in Patients. Eur. J. Nucl. Med. 2001, 28, 1319–1325.
- Kam, B.L.R.; Teunissen, J.J.M.; Krenning, E.P.; de Herder, W.W.; Khan, S.; van Vliet, E.I.; Kwekkeboom, D.J. Lutetium-Labelled Peptides for Therapy of Neuroendocrine Tumours. Eur. J. Nucl. Med. Mol. Imaging 2012, 39, 103–112.
- McClellan, K.; Chen, E.Y.; Kardosh, A.; Lopez, C.D.; Del Rivero, J.; Mallak, N.; Rocha, F.G.; Koethe, Y.; Pommier, R.; Mittra, E.; et al. Therapy Resistant Gastroenteropancreatic Neuroendocrine Tumors. Cancers 2022, 14, 4769.
- Krenning, E.P.; Kwekkeboom, D.J.; Valkema, R.; Pauwels, S.; Kvols, L.K.; Jong, M. Peptide Receptor Radionuclide Therapy. Ann. N. Y. Acad. Sci. 2004, 1014, 234–245.
- Navalkissoor, S.; Grossman, A. Targeted Alpha Particle Therapy for Neuroendocrine Tumours: The Next Generation of Peptide Receptor Radionuclide Therapy. Neuroendocrinology 2019, 108, 256–264.
- Parker, C.; Nilsson, S.; Heinrich, D.; Helle, S.I.; O’Sullivan, J.M.; Fosså, S.D.; Chodacki, A.; Wiechno, P.; Logue, J.; Seke, M.; et al. Alpha Emitter Radium-223 and Survival in Metastatic Prostate Cancer. N. Engl. J. Med. 2013, 369, 213–223.
- Eychenne, R.; Chérel, M.; Haddad, F.; Guérard, F.; Gestin, J.-F. Overview of the Most Promising Radionuclides for Targeted Alpha Therapy: The “Hopeful Eight”. Pharmaceutics 2021, 13, 906.
- Norenberg, J.P.; Krenning, B.J.; Konings, I.R.H.M.; Kusewitt, D.F.; Nayak, T.K.; Anderson, T.L.; de Jong, M.; Garmestani, K.; Brechbiel, M.W.; Kvols, L.K. 213Bi-Octreotide Peptide Receptor Radionuclide Therapy of Pancreatic Tumors in a Preclinical Animal Model. Clin. Cancer Res. 2006, 12, 897–903.
- Nayak, T.K.; Norenberg, J.P.; Anderson, T.L.; Prossnitz, E.R.; Stabin, M.G.; Atcher, R.W. Somatostatin-Receptor-Targeted Alpha-Emitting 213Bi Is Therapeutically More Effective than Beta(-)-Emitting 177Lu in Human Pancreatic Adenocarcinoma Cells. Nucl. Med. Biol. 2007, 34, 185–193.
- Chan, H.S.; Konijnenberg, M.W.; de Blois, E.; Koelewijn, S.; Baum, R.P.; Morgenstern, A.; Bruchertseifer, F.; Breeman, W.A.; de Jong, M. Influence of Tumour Size on the Efficacy of Targeted Alpha Therapy with 213Bi--Octreotate. EJNMMI Res. 2016, 6, 6.
- Chan, H.S.; Konijnenberg, M.W.; Daniels, T.; Nysus, M.; Makvandi, M.; de Blois, E.; Breeman, W.A.; Atcher, R.W.; de Jong, M.; Norenberg, J.P. Improved Safety and Efficacy of 213Bi-DOTATATE-Targeted Alpha Therapy of Somatostatin Receptor-Expressing Neuroendocrine Tumors in Mice Pre-Treated with l-Lysine. EJNMMI Res. 2016, 6, 83.
- Kratochwil, C.; Giesel, F.L.; Bruchertseifer, F.; Mier, W.; Apostolidis, C.; Boll, R.; Murphy, K.; Haberkorn, U.; Morgenstern, A. 213Bi-DOTATOC Receptor-Targeted Alpha-Radionuclide Therapy Induces Remission in Neuroendocrine Tumours Refractory to Beta Radiation: A First-in-Human Experience. Eur. J. Nucl. Med. Mol. Imaging 2014, 41, 2106–2119.
- Morgenstern, A.; Apostolidis, C.; Kratochwil, C.; Sathekge, M.; Krolicki, L.; Bruchertseifer, F. An Overview of Targeted Alpha Therapy with 225Actinium and 213Bismuth. CRP 2018, 11, 200–208.
- Pouget, J.-P.; Lozza, C.; Deshayes, E.; Boudousq, V.; Navarro-Teulon, I. Introduction to Radiobiology of Targeted Radionuclide Therapy. Front. Med. 2015, 2, 12.
- Robertson, A.K.H.; Ramogida, C.F.; Rodríguez-Rodríguez, C.; Blinder, S.; Kunz, P.; Sossi, V.; Schaffer, P. Multi-Isotope SPECT Imaging of the 225Ac Decay Chain: Feasibility Studies. Phys. Med. Biol. 2017, 62, 4406–4420.
- Ocak, M.; Toklu, T.; Demirci, E.; Selçuk, N.; Kabasakal, L. Post-Therapy Imaging of 225Ac-DOTATATE Treatment in a Patient with Recurrent Neuroendocrine Tumor. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 2711–2712.
- Kratochwil, C.; Bruchertseifer, F.; Rathke, H.; Bronzel, M.; Apostolidis, C.; Weichert, W.; Haberkorn, U.; Giesel, F.L.; Morgenstern, A. Targeted α-Therapy of Metastatic Castration-Resistant Prostate Cancer with 225Ac-PSMA-617: Dosimetry Estimate and Empiric Dose Finding. J. Nucl. Med. 2017, 58, 1624–1631.
- Belli, M.L.; Sarnelli, A.; Mezzenga, E.; Cesarini, F.; Caroli, P.; Di Iorio, V.; Strigari, L.; Cremonesi, M.; Romeo, A.; Nicolini, S.; et al. Targeted Alpha Therapy in MCRPC (Metastatic Castration-Resistant Prostate Cancer) Patients: Predictive Dosimetry and Toxicity Modeling of 225Ac-PSMA (Prostate-Specific Membrane Antigen). Front. Oncol. 2020, 10, 531660.
- Sgouros, G.; He, B.; Ray, N.; Ludwig, D.L.; Frey, E.C. Dosimetric Impact of Ac-227 in Accelerator-Produced Ac-225 for Alpha-Emitter Radiopharmaceutical Therapy of Patients with Hematological Malignancies: A Pharmacokinetic Modeling Analysis. EJNMMI Phys. 2021, 8, 60.
- Tranel, J.; Feng, F.Y.; James, S.S.; Hope, T.A. Effect of Microdistribution of Alpha and Beta-Emitters in Targeted Radionuclide Therapies on Delivered Absorbed Dose in a GATE Model of Bone Marrow. Phys. Med. Biol. 2021, 66, 035016.
- Sato, T.; Furuta, T.; Liu, Y.; Naka, S.; Nagamori, S.; Kanai, Y.; Watabe, T. Individual Dosimetry System for Targeted Alpha Therapy Based on PHITS Coupled with Microdosimetric Kinetic Model. EJNMMI Phys. 2021, 8, 4.
- Fry, C.; Thoennessen, M. Discovery of Actinium, Thorium, Protactinium, and Uranium Isotopes. At. Data Nucl. Data Tables 2013, 99, 345–364.
- Boll, R.A.; Malkemus, D.; Mirzadeh, S. Production of Actinium-225for Alpha Particle Mediated Radioimmunotherapy. Appl. Radiat. Isot. 2005, 62, 667–679.
- Apostolidis, C.; Molinet, R.; Rasmussen, G.; Morgenstern, A. Production of Ac-225from Th-229 for Targeted α Therapy. Anal. Chem. 2005, 77, 6288–6291.
- Kotovskii, A.A.; Nerozin, N.A.; Prokof’ev, I.V.; Shapovalov, V.V.; Yakovshchits, Y.A.; Bolonkin, A.S.; Dunin, A.V. Isolation of Actinium-225for Medical Purposes. Radiochemistry 2015, 57, 285–291.
- Zielinska, B.; Apostolidis, C.; Bruchertseifer, F.; Morgenstern, A. An Improved Method for the Production of Ac-225/Bi-213 from Th-229 for Targeted Alpha Therapy. Solvent Extr. Ion Exch. 2007, 25, 339–349.
- Perron, R.; Gendron, D.; Causey, P.W. Construction of a Thorium/Actinium Generator at the Canadian Nuclear Laboratories. Appl. Radiat. Isot. 2020, 164, 109262.
- Apostolidis, C.; Molinet, R.; McGinley, J.; Abbas, K.; Möllenbeck, J.; Morgenstern, A. Cyclotron Production of Ac-225for Targeted Alpha Therapy. Appl. Radiat. Isot. 2005, 62, 383–387.
- Nagatsu, K.; Suzuki, H.; Fukada, M.; Ito, T.; Ichinose, J.; Honda, Y.; Minegishi, K.; Higashi, T.; Zhang, M.-R. Cyclotron Production of 225Ac from an Electroplated 226Ra Target. Eur. J. Nucl. Med. Mol. Imaging 2021, 49, 279–289.
- Morss, L.R.; Edelstein, N.M.; Fuger, J. The Chemistry of the Actinide and Transactinide Elements, 3rd ed.; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2007; ISBN 978-1-4020-3598-2.
- Yamana, H.; Mitsugashira, T.; Shiokawa, Y.; Sato, A.; Suzuki, S. Possibility of the Existence of Divalent Actinium in Aqueous Solution. J. Radioanal. Chem. 1983, 76, 19–26.
- Malý, J. The Amalgamation Behavior of Heavy Elements—III Extraction of Radium, Lead and the Actinides by Sodium Amalgam from Acetate Solutions. J. Inorg. Nucl. Chem. 1969, 31, 1007–1017.
- Kozempel, J.; Mokhodoeva, O.; Vlk, M. Progress in Targeted Alpha-Particle Therapy. What We Learned about Recoils Release from In Vivo Generators. Molecules 2018, 23, 581.
- Thiele, N.A.; Wilson, J.J. Actinium-225for Targeted α Therapy: Coordination Chemistry and Current Chelation Approaches. Cancer Biother. Radiopharm. 2018, 33, 336–348.
- Pearson, R.G. Hard and Soft Acids and Bases. J. Am. Chem. Soc. 1963, 85, 3533–3539.
- Cotton, S.A. Lanthanide and Actinide Chemistry; Inorganic Chemistry; Wiley: Chichester, UK; Hoboken, NJ, USA, 2006; ISBN 978-0-470-01005-1.
- Davis, I.A.; Glowienka, K.A.; Boll, R.A.; Deal, K.A.; Brechbiel, M.W.; Stabin, M.; Bochsler, P.N.; Mirzadeh, S.; Kennel, S.J. Comparison of 225actinium Chelates: Tissue Distribution and Radiotoxicity. Nucl. Med. Biol. 1999, 26, 581–589.
- Deal, K.A.; Davis, I.A.; Mirzadeh, S.; Kennel, S.J.; Brechbiel, M.W. Improved in Vivo Stability of Actinium-225Macrocyclic Complexes. J. Med. Chem. 1999, 42, 2988–2992.
- Kennel, S.J.; Brechbiel, M.W.; Milenic, D.E.; Schlom, J.; Mirzadeh, S. Actinium-225Conjugates of Mab CC49 and Humanized ΔCH2 CC49. Cancer Biother. Radiopharm. 2002, 17, 219–231.
- Maguire, W.F.; McDevitt, M.R.; Smith-Jones, P.M.; Scheinberg, D.A. Efficient 1-Step Radiolabeling of Monoclonal Antibodies to High Specific Activity with 225Ac for α-Particle Radioimmunotherapy of Cancer. J. Nucl. Med. 2014, 55, 1492–1498.
- Pandya, D.N.; Hantgan, R.; Budzevich, M.M.; Kock, N.D.; Morse, D.L.; Batista, I.; Mintz, A.; Li, K.C.; Wadas, T.J. Preliminary Therapy Evaluation of 225Ac-DOTA-c(RGDyK) Demonstrates That Cerenkov Radiation Derived from 225Ac Daughter Decay Can Be Detected by Optical Imaging for In Vivo Tumor Visualization. Theranostics 2016, 6, 698–709.
- Pruszynski, M.; D’Huyvetter, M.; Bruchertseifer, F.; Morgenstern, A.; Lahoutte, T. Evaluation of an Anti-HER2 Nanobody Labeled with 225Ac for Targeted α-Particle Therapy of Cancer. Mol. Pharm. 2018, 15, 1457–1466.
- Majkowska-Pilip, A.; Rius, M.; Bruchertseifer, F.; Apostolidis, C.; Weis, M.; Bonelli, M.; Laurenza, M.; Królicki, L.; Morgenstern, A. In Vitro Evaluation of 225Ac-DOTA-Substance P for Targeted Alpha Therapy of Glioblastoma Multiforme. Chem. Biol. Drug Des. 2018, 92, 1344–1356.
- McDevitt, M.R.; Ma, D.; Simon, J.; Frank, R.K.; Scheinberg, D.A. Design and Synthesis of 225Ac Radioimmunopharmaceuticals. Appl. Radiat. Isot. 2002, 57, 841–847.
- Schwartz, J.; Jaggi, J.S.; O’Donoghue, J.A.; Ruan, S.; McDevitt, M.; Larson, S.M.; Scheinberg, D.A.; Humm, J.L. Renal Uptake of Bismuth-213 and Its Contribution to Kidney Radiation Dose Following Administration of Actinium-225-Labeled Antibody. Phys. Med. Biol. 2011, 56, 721–733.
- Singh Jaggi, J.; Kappel, B.J.; McDevitt, M.R.; Sgouros, G.; Flombaum, C.D.; Cabassa, C.; Scheinberg, D.A. Efforts to Control the Errant Products of a Targeted In Vivo Generator. Cancer Res. 2005, 65, 4888–4895.
- Yoshimoto, M.; Yoshii, Y.; Matsumoto, H.; Shinada, M.; Takahashi, M.; Igarashi, C.; Hihara, F.; Tachibana, T.; Doi, A.; Higashi, T.; et al. Evaluation of Aminopolycarboxylate Chelators for Whole-Body Clearance of Free 225Ac: A Feasibility Study to Reduce Unexpected Radiation Exposure during Targeted Alpha Therapy. Pharmaceutics 2021, 13, 1706.
- Uccelli, L.; Boschi, A.; Cittanti, C.; Martini, P.; Panareo, S.; Tonini, E.; Nieri, A.; Urso, L.; Caracciolo, M.; Lodi, L.; et al. 90Y/177Lu-DOTATOC: From Preclinical Studies to Application in Humans. Pharmaceutics 2021, 13, 1463.
- Norenberg, J.; Daniels, T.; Kevin, J.; Heloisa, S.; Kusewitt, D.; Hesterman, J.; Orcutt, K.; Nysus, M.; Goff, C.; Jacquez, Q.; et al. Pre-Clinical Evaluation of 225Ac-DOTATOC Pharmacokinetics, Dosimetry, and Istopathology to Enable Phase-1 Clinical Trial in Patients with Neuroendocrine Tumors. J. Med. Imaging Radiat. Sci. 2019, 50, S105.
- Strosberg, J.R.; Caplin, M.E.; Kunz, P.L.; Ruszniewski, P.B.; Bodei, L.; Hendifar, A.; Mittra, E.; Wolin, E.M.; Yao, J.C.; Pavel, M.E.; et al. 177Lu-Dotatate plus Long-Acting Octreotide versus High-dose Long-Acting Octreotide in Patients with Midgut Neuroendocrine Tumours (NETTER-1): Final Overall Survival and Long-Term Safety Results from an Open-Label, Randomised, Controlled, Phase 3 Trial. Lancet Oncol. 2021, 22, 1752–1763.
- Strosberg, J.R.; Caplin, M.E.; Kunz, P.L.; Ruszniewski, P.B.; Bodei, L.; Hendifar, A.E.; Mittra, E.; Wolin, E.M.; Yao, J.C.; Pavel, M.E.; et al. Final Overall Survival in the Phase 3 NETTER-1 Study of Lutetium-177-DOTATATE in Patients with Midgut Neuroendocrine Tumors. JCO 2021, 39, 4112.
- Strosberg, J.; El-Haddad, G.; Wolin, E.; Hendifar, A.; Yao, J.; Chasen, B.; Mittra, E.; Kunz, P.L.; Kulke, M.H.; Jacene, H.; et al. Phase 3 Trial of 177Lu-Dotatate for Midgut Neuroendocrine Tumors. N. Engl. J. Med. 2017, 376, 125–135.
- Zhang, J.; Singh, A.; Kulkarni, H.R.; Schuchardt, C.; Müller, D.; Wester, H.-J.; Maina, T.; Rösch, F.; van der Meulen, N.P.; Müller, C.; et al. From Bench to Bedside—The Bad Berka Experience with First-in-Human Studies. Semin. Nucl. Med. 2019, 49, 422–437.
- Kratochwil, C.; Bruchertseifer, F.; Rathke, H.; Hohenfellner, M.; Giesel, F.L.; Haberkorn, U.; Morgenstern, A. Targeted α-Therapy of Metastatic Castration-Resistant Prostate Cancer with 225Ac-PSMA-617: Swimmer-Plot Analysis Suggests Efficacy Regarding Duration of Tumor Control. J. Nucl. Med. 2018, 59, 795–802.
- Sathekge, M.; Bruchertseifer, F.; Knoesen, O.; Reyneke, F.; Lawal, I.; Lengana, T.; Davis, C.; Mahapane, J.; Corbett, C.; Vorster, M.; et al. 225Ac-PSMA-617 in Chemotherapy-Naive Patients with Advanced Prostate Cancer: A Pilot Study. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 129–138.
- Sathekge, M.; Bruchertseifer, F.; Vorster, M.; Lawal, I.O.; Knoesen, O.; Mahapane, J.; Davis, C.; Reyneke, F.; Maes, A.; Kratochwil, C.; et al. Predictors of Overall and Disease-Free Survival in Metastatic Castration-Resistant Prostate Cancer Patients Receiving 225Ac-PSMA-617 Radioligand Therapy. J. Nucl. Med. 2020, 61, 62–69.
- Yadav, M.P.; Ballal, S.; Sahoo, R.K.; Tripathi, M.; Seth, A.; Bal, C. Efficacy and Safety of 225Ac-PSMA-617 Targeted Alpha Therapy in Metastatic Castration-Resistant Prostate Cancer Patients. Theranostics 2020, 10, 9364–9377.
- Khreish, F.; Ebert, N.; Ries, M.; Maus, S.; Rosar, F.; Bohnenberger, H.; Stemler, T.; Saar, M.; Bartholomä, M.; Ezziddin, S. 225Ac-PSMA-617/177Lu-PSMA-617 Tandem Therapy of Metastatic Castration-Resistant Prostate Cancer: Pilot Experience. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 721–728.
- Rosenblat, T.L.; McDevitt, M.R.; Carrasquillo, J.A.; Pandit-Taskar, N.; Frattini, M.G.; Maslak, P.G.; Park, J.H.; Douer, D.; Cicic, D.; Larson, S.M.; et al. Treatment of Patients with Acute Myeloid Leukemia with the Targeted Alpha-Particle Nanogenerator Actinium-225-Lintuzumab. Clin. Cancer Res. 2022, 28, 2030–2037.
- Hope, T.; Mehr, S.; Morris, M.; Li, D.; Halperin, M.D.; Strosberg, J.; Soares, H.; Jacene, H.; Pavel, M.; Pamela, L.; et al. ACTION-1: A Randomized Phase Ib/3 Trial of RYZ101 Compared with SoC in SSTR2+ Well-Differentiated GEP-NET with Progression Following Lu-177 SSA. EJEA 2023.
- Yadav, M.P.; Ballal, S.; Sahoo, R.K.; Bal, C. Efficacy and Safety of 225Ac-DOTATATE Targeted Alpha Therapy in Metastatic Paragangliomas: A Pilot Study. Eur. J. Nucl. Med. Mol. Imaging 2021, 49, 1595–1606.
- Peng, D.; Liu, H.; Huang, L.; Cao, J.; Chen, Y. 225Ac-DOTATATE Therapy in a Case of Metastatic Pheochromocytoma. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 3596–3597.
