2. Peritendinous Injection
The dermo-epidermal complex, subcutaneous tissue, and forearm fascia wrap the lateral elbow compartment and, using a high-frequency linear probe, they can all easily be identified as superficial to the CET
[8][9][8,9]. At this level, the recurrent radial artery, posterior branch of the radial collateral artery, and the interosseous recurrent artery anastomose—forming a superficial vascular plexus which covers the lateral epicondyle and the CET
[10]. Interestingly, in patients with signs and symptoms of LE, a hypertrophic neurovascular network was histologically identified within the aforementioned superficial soft tissues which envelops and penetrates the superficial fibers of the CET
[11]. This condition is known as superficial-to-deep vascular invasion of the tendon tissue (
Figure 1)—with larger vascular elements being in the subcutaneous tissue (i.e., the donator vessels) and thinner vascular signals residing within the CET (i.e., the penetrating vessels)
[1].
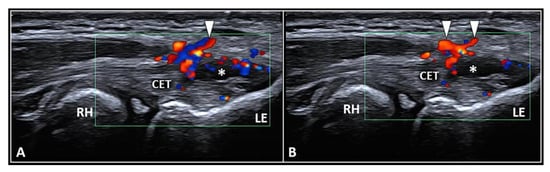
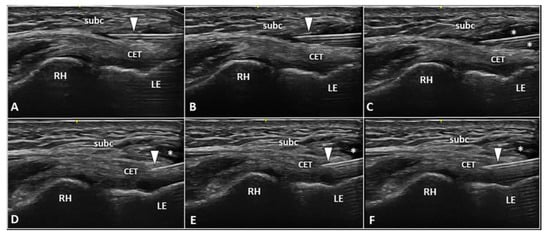
Figure 1. Ultrasound examination with color Doppler (A,B) clearly shows how the aberrant neovessels (white arrowheads) originating from the superficial vascular plexus of the subcutis pierce the forearm fascia invading the hypoechoic zone (white asterisk) of the CET related to insertional tendinosis. RH: radial head, LE: lateral epicondyle, CET: common extensor tendon.
To optimize the sonographic visibility of the aforementioned vascular infiltration of the CET, the authors suggest a few technical tips and tricks. At first, a large amount of gel should be used, i.e., the suspension technique—to avoid/minimize the compression of skin with the ultrasound probe as well as the unintentional squeeze of the small-sized vascular elements
[8][9][8,9]. Second, by adjusting the frame rate and positioning the superior edge of the color/power Doppler box at the level of the subcutaneous tissue, correct assessment of the superficial microvasculature can be performed—avoiding fake vascular signals related to acoustic artifacts
[12]. Lastly, a large region of interest should be avoided to preserve the Doppler sensitivity
[12].
Based on the anatomical and histopathological findings quoted above, USG injection of the interface between the forearm fascia and the underlying CET can be performed to disrupt the bridging neovessels and neonerves originating from the superficial tissues and infiltrating the tendon fibers
[1]. An in-plane technique is recommended to guarantee real-time visualization of the needle for its entire course during the intervention and a proximal-to-distal or distal-to-proximal approach can be used according to the physician’s experience
[13][14].
Especially for beginners, the main pitfall of the USG peritendinous injection of the CET would be the risk of advancing the needle’s tip within the most superficial fibers of the tendon and not at the extra-tendinous fascial layers. In this aspect, the authors suggest a few tips/tricks to avoid the aforementioned complication
[14][15].
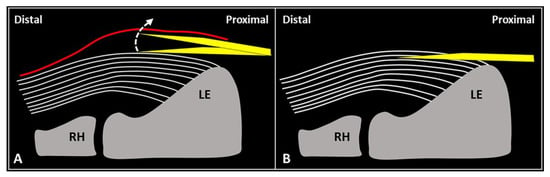
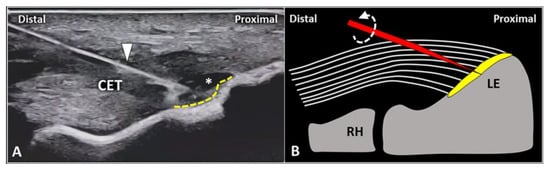
First, gentle inclination of the needle pointing the tip toward the skin can be performed. The movement can be easily performed when the needle is located inside the soft superficial tissues, while a stiff “block” can be encountered as the needle is inserted within the tendon (
Figure 2). Second, slight pressure can be applied on the syringe plunger to observe if the mixture can be easily injected or not. However, hard resistance can be felt if the needle is located within the tendon fibers and very low resistance (coupled with smooth flow of the mixture covering the tendon surface) can be observed when the needle tip is positioned inside the fascial layers.
Figure 2. When the needle’s tip (yellow) is correctly positioned just over the superficial edge of the CET, by gently tilting (curved dotted arrow) the needle, it is possible to “separate” the forearm fascia (red) from the underlying tendon without hard resistance (A). Conversely, if the needle’s tip (yellow) is located inside the superficial portion of the CET, a “block” can be felt during this maneuver due to the intratendinous entrapment of the needle between the tendon fibers (white lines) (B). RH: radial head, LE: lateral epicondyle.
3. Intratendinous Injection
The CET is an anchor tendon, mainly composed of longitudinally oriented collagen fibers attaching to the lateral epicondyle of the humerus. Normally, it presents a hyperechoic fibrillar pattern quite similar to the underlying RCL
[1]. The extensor carpi radialis brevis tendon represents its deepest layer whereas the extensor digitorum communis tendon constitutes the most superficial portion of the common tendinous mass
[15][18]. Interestingly, extensor carpi radialis longus, extensor carpi ulnaris, and the CET are not fused with each other—presenting as separate anatomical structures
[16][19]. The vascular network is not uniformly distributed within the CET and unlike the superficial layers perfused by a rich subcutaneous plexus, the deep portion is almost avascular
[10]. Among several pathological conditions potentially involved in the clinical scenario of LE, focal tendinosis, partial tear, and intratendinous calcific deposition are the most commonly encountered in daily practice (
Figure 3)
[1].
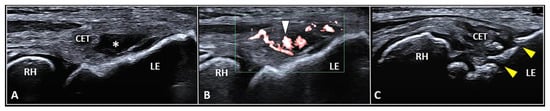
Figure 3. Partial tear (white asterisk) of the insertion zone of CET (A), focal tendinosis with inner hypervascularization (white arrowhead) (B), and enthesitis with cortical irregularities (yellow arrowhead) (C) of the lateral epicondyle (LE) should be considered among the most common sonographic findings related to the clinical picture of lateral epicondylitis. RH: radial head.
For all the aforementioned disorders, color/power Doppler assessment should be coupled with the B-mode to visualize the presence of aberrant neovessels within the pathological tendon tissue
[1][3][4][1,3,4]. Zeising et al. demonstrated that USG injection of the intratendinous microvasculature with local anesthetic efficiently reduced lateral elbow pain, suggesting that only the pathological (neuro)vascular tangle could represent the main pain generator in patients with LE
[4]. Interestingly, using sclerosing polidocanol instead of local anesthetic, they described several cases characterized by a significant pain reduction despite the persistence of intratendinous hypervascularization on color/power Doppler
[17][20]. In this sense, it seems that the neonerves more than the neovessels play a pivotal role in painful tendinopathy of the LE. Based on speculations from the aforementioned findings, the authors used the vascular signals of color/power Doppler as an indirect sonographic sign to localize the real target of procedure, i.e., the intratendinous neural tangle
[17][20]. More specifically, Substance P and calcitonin gene-regulated peptide seem to be the main neuropeptides involved in (i) neurogenic inflammation of tendon sensory fibers and (ii) microvascular leakage with local edema
[18][21].
Using B-mode (tendon thickness, bony spur) and color Doppler (vascular signals), Krogh et al.
[19][22] published a cross-sectional study of 264 participants with healthy elbows and 60 patients with chronic LE. Interestingly, they reported that color doppler activity was a strong indicator of ongoing tendinopathy (although not pathognomonic) and that the absence of vascular signals in patients with suspected lateral tendinopathy should raise the suspicion of an alternative diagnosis
[19][22]. Likewise, in a between-group cross-sectional study of 25 patients and 19 asymptomatic participants, du Toit et al.
[20][23] concluded that neovascularity identified with power Doppler ultrasonography (vs. B-mode imaging) was diagnostically superior in identifying chronic tennis elbow. Moreover, the lack of both neovascularity and grey scale changes in ultrasound examinations substantially increased the probability that the condition is not present and should prompt the clinician to consider other causes for lateral elbow pain
[20][23].
Recently, dynamic imaging protocols of the elbow augmented the diagnostic potential of ultrasound examination in detecting soft tissue injuries which could hardly be identified. For instance, in patients with signs/symptoms suggestive of LE, dynamic assessment can be performed with active/passive movements of the elbow—to use the intraarticular synovial fluid as a natural contrast agent, pushing it within a partial tear of the CET on its articular side
[21][24]. Interestingly, in some patients, a painful pinching of the radio-humeral synovial plica can be observed between the bony surfaces, which can otherwise mimic the painful/clinical scenario of LE
[21][24].
Considering the superficial location of the CET, a 25 to 38 mm needle is considered long enough to accurately reach the tendon tissue in the literature
[22][25]. Likewise, 18–25 G needles can be used depending on the technical features of the procedure. Of note, a thinner needle can be used to inject an orthobiologic agent within a partial tear, a larger needle is necessary to perform USG fenestrations in focal tendinosis (
Figure 3). As previously mentioned for peritendinous injections, an in-plane technique is also recommended to visualize the needle’s shaft and tip during intratendinous procedures
[13][14]. Again, a proximal-to-distal or distal-to-proximal approach can be used. Needless to say, to plan for an accurate injection, the authors suggest matching the longitudinal and transverse scans to exactly “pinpoint” the pathological segment of the common tendinous mass
[23][24][26,27].
The researchers suggest using a specific approach depending on the main purpose of the intervention:
USG injection of the CET: a distal-to-proximal approach (
Figure 4) allows for reaching a very specific zone of the CET, especially if located in close proximity to the attachment zone, e.g., a focal disruption of the fibers near the fibrocartilaginous plate of the enthesis. Of note, several authors in the pertinent literature have clearly demonstrated that the volume injected within the CET partially migrates outside, reaching the surrounding tissues of the lateral elbow compartment. Park et al.
[25][28] performed a USG intratendinous injection of 1.5 mL of platelet-rich plasma into the CET of 25 patients with chronic LE (duration of symptoms > 6 months). They evaluated the spread of the mixture immediately after the procedure. Interestingly, the mixture was sonographically identified as fluids and/or gas microbubbles both inside the subcutaneous tissue and within the radio-humeral joint
[25][28]. In this sense, the CET should not be considered as a sealed chamber but rather as a multilayer structure in which fluid can slip between the interlaminar zones and leak out from the tendon structure.
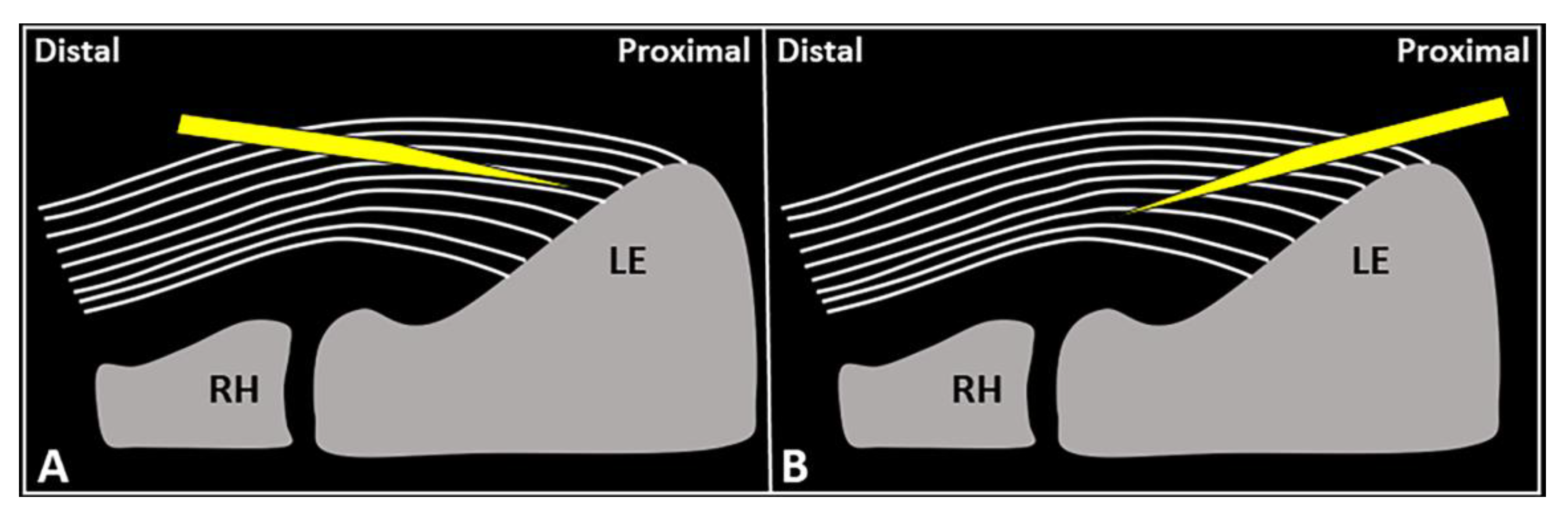
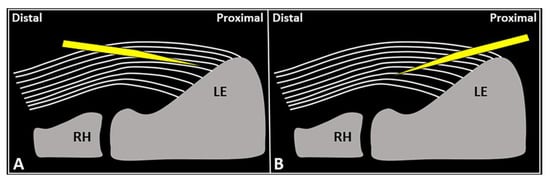
Schematic drawing shows the distal-to-proximal (
) and proximal-to-distal (
) approaches for performing USG fenestration of the CET. While the needle tip (
) can slip in between the tendon fibers (
) in the former technique (
); crossing in an orthogonal fashion, it efficiently disrupts them in the latter (
). RH: radial head, LE: lateral epicondyle.
USG fenestration of the CET: a proximal-to-distal approach might guarantee needle movements orthogonal to the spatial orientation of the CET fibers (
Figure 4). The bevel of the needle can efficiently disrupt the tendon tissue, i.e., the main goal of USG fenestration. Instead, if a distal-to-proximal approach is used, the needle tends to slip in between the different layers of the CET—traveling inside the interlaminar zones without a satisfactory transection of the fibers (
Figure 4). The exact number of back-and-forward passages of the needle within the CET is not well defined in the literature, whereby the feeling of local softness is usually considered as practical clinical feedback for a successful procedure. In addition, a small amount of blood might commonly flow back to the syringe—due to mechanical disruption of the aforementioned abundant microvasculature of the CET in pathological conditions.
Based on the currently available evidence, it seems that the mechanical effects of the USG procedure—rather than the chemical effects of the injectate—may play a more pivotal role in the clinical/functional outcomes. Indeed, after repetitive perforation of a tendon, transcription factors involved in the proliferation and differentiation of mesenchymal cells, genes involved in inflammatory responses, and angiogenesis pathways are regulated, modulating the healing response
[26][27][28][31,32,33]. Histologically, the back-and-forward movements of the needle within the pathological tendon tissue induce micro-injuries and local bleeding with progressive deposition of granulation tissue that mechanically reinforces/stabilizes the tendon microarchitecture
[27][29][32,34].
Notably, different USG techniques can be combined if clinically indicated. For instance, in a patient with sonographic findings of a superficial-to-deep vascular invasion of pre-insertional focal tendinosis of CET, a two-step procedure can be planned. First, a high-volume USG hydrodissection of the tendon-fascia interface can be performed to release adhesions and to disrupt the perforating neovessels/neonerves. Second, a USG fenestration of the degenerated tendon tissue can be performed to transect the disorganized fibers with multiple back-and-forward movements of the needle (
Figure 5).
Figure 5. Two-step USG procedure of the lateral elbow using in-plane proximal-to-distal approach. The needle (white arrowhead) is advanced in between the subcutaneous tissue (subc) and the CET. High volume (white asterisks) is injected to efficiently “open” the pathological interface (A–C). The needle (white arrowhead) is redirected more deeply to perform the fenestration of the pre-insertional segment of CET (D–F). RH: radial head, LE: lateral epicondyle.
Recently, Pringels et al.
[30][35] theorized that the remodeling of tendon tissue into fibrocartilage-like tissue can cause increased intratendinous resting pressure, mainly due to excessive water-binding glycosaminoglycans and proteoglycans. Moreover, this increased pressure might explain the hypoxic state and the formation of leaky (neo)vessels in tendon pathology
[30][35].
4. Enthesis Injection
The fibers of the CET anchor to the lateral epicondyle through a transitional plate known as enthesis. Histologically, it is a double-layer structure made of an uncalcified fibrocartilaginous stratum in continuum with the tendon fibers and a calcified fibrocartilage attached to the subchondral bone
[31][32][36,37]. In physiological conditions, the sonographic appearance of lateral enthesis is similar to a smooth hypoechoic band located between the hyperechoic cortical surface of the lateral epicondyle and the regular fibrillar pattern of the CET.
[1][33][34][1,38,39] Likewise, in patients with enthesopathies, focal interruptions of the cortical bone, pitting of the trabecular bone, lamellar calcifications of the fibrocartilaginous plate, and vascular signals at color/power Doppler are the most common sonographic findings (
Figure 3)
[1][33][34][1,38,39].
In the authors’ experience, the USG injection of the enthesis is less commonly performed when compared to peri/intratendinous injections in the daily management of patients with LE. Likewise, focal detachment (i.e., avulsion) of the CET fibers from the fibrocartilaginous plate of the lateral elbow can be considered a non-rare pathological condition requiring this intervention. Technically, using the in-plane distal-to-proximal approach, suggest a two-step procedure (
Figure 6) with an intralesional injection of local anesthetic followed by abrasion of the fibrocartilage with rotations of the needle bevel causing a “drilling effect”.
Figure 6. In a patient with focal avulsion of deep fibers of CET from the lateral epicondyle (LE), a two-step USG procedure can be performed. Using an in-plane distal-to-proximal approach, the needle (white arrowhead) is advanced within the injury (white asterisk) and local anesthetic is injected to “uncover” the underlying enthesis (yellow dotted line) (A). Schematic drawing shows multiple rotations (curved dotted arrow) of the needle’s tip (red) for mechanical drilling of the fibrocartilage (yellow) to induce a local micro-bleeding (B). RH: radial head.
Of note, not rarely in daily practice, radio-humeral synovitis can mimic the clinical scenario of lateral enthesopathy
[35][43]. Intra-articular effusion in the annular recess and vascular signals within/around the radio-humeral synovial plica are the most common sonographic findings suggestive of articular pain
[1][12][1,12]. Therefore, a USG intra-articular rather than an extra-articular procedure should be planned for prompt management
[36][44]. With the elbow flexed at 90 degrees and the forearm pronated, an out-of-plane technique—by advancing the needle through the CET—is usually preferred to release the mixture inside the synovial space of the elbow
[37][45]. A hyperechoic flash within the hypo/anechoic radio-humeral cleft during the USG procedure can be considered as a common “confirming” sign for correct intra-articular placement.
Chen et al.
[38][46] have described a two-step USG procedure to simultaneously inject the radio-humeral joint and the tendon-bone interface of the CET. Using an out-of-plane technique with a lateral approach, the white dot representing the cross-section of the needle is first within the articular cleft and subsequently inside the enthesis of the lateral epicondyle. As such, the authors have considered the lateral elbow pain (refractory to conservative treatments) as cross-talking between two different pain generators—the fibrocartilaginous plate of the CET and the synovial tissue of the radio-humeral joint.
Lastly, among the different types of USG techniques described in the pertinent literature to manage chronic pain in patients with recalcitrant LE, muscular injections have also been reported. Indeed, considering the excessive traction of the muscle-tendon unit, a well-established pathophysiological element for progression to the chronic phase of lateral epicondylopathy, botulinum toxin injections into the forearm extensor muscles were performed
[39][47]. Of note, very careful selection of the muscle(s) to be injected and the dosage of toxin for each muscle belly have to be planned before the intervention to avoid adverse events such as muscular weakness
[40][48].