Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Robert Zymliński and Version 2 by Rita Xu.
Heart failure (HF) constitutes a significant clinical problem and is associated with a sizeable burden for the healthcare system. Numerous novel techniques, including device interventions, are investigated to improve clinical outcome. Interventions regarding autonomic nervous system imbalance, i.e., baroreflex activation therapy; vagus, splanchnic and cardiopulmonary nerves modulation; respiratory disturbances, i.e., phrenic nerve stimulation and synchronized diaphragmatic therapy; decongestion management, i.e., the Reprieve system, transcatheter renal venous decongestion system, Doraya, preCardia, WhiteSwell and Aquapass, are presented.
- heart failure
- cardiorenal syndrome
- autonomic dysregulation
1. Introduction
Heart failure (HF) is a clinical syndrome resulting from structural and/or functional abnormality of the heart, leading to elevated intracardiac pressures and/or insufficient cardiac output. Increased cardiac filling pressures and neuro-hormonal disturbances resulting in fluid retention and redistribution are major factors responsible for congestion development and acute decompensation in heart failure [1].
As the HF pathophysiology is multidimensional, device interventions allow direct or indirect targeting of biological HF pathways, e.g. methods to manipulate sympathetic nervous system (SNS) imbalance, respiratory dysregulation or volume overload have been developed (Table 1). To preserve the research'ticle’s coherence and compactness, researcherswe decided not to describe all promising techniques, but theywe focused on selected pathophysiological processes crucial in HF (Figure 1).
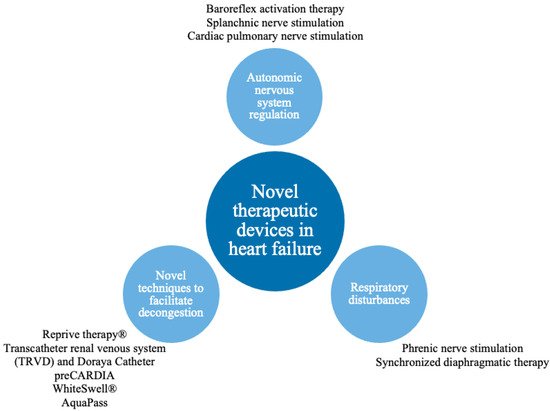
Figure 1. Pathophysiological pathways addressed by novel therapeutic devices.
Table 1. Summary of the proposed novel methods.
| Method | Pathophysiological Mechanism | Solution | Trial Design and Size | Primary Outcomes | Evidence | Adverse Events | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Baroreflex activation therapy | Overactivity of SNS (increased heart rate, arterial pressure, RAAS activity and negative cardiac remodeling). | Stimulation of carotid bodies to restore autonomic system balance. | Multicenter, prospective, controlled trial | n | = 408 | Rate of cardiovascular and HF morbidity, MANCE, Change in: NT-proBNP, 6 MHW, MLWHF QOL |
BeAT-HF showed improvements of quality of life, exercise capacity, functional status and decrease of NT-proBNP [2] | MANCE event-free rate: 97%. A system or procedure-related serious adverse event occurred in seven patients. | |||
| Single-center, open-label | n | = 11 | Not reported | Dell’Oro et al. demonstrated significant improvement of EF and reduction in hospitalization [3] | No adverse effects were reported. | ||||||
| Vagus nerve stimulation | Overactivity of SNS (increased heart rate, arterial pressure, RAAS activity and negative cardiac remodeling). | Increase of PNS activity. | Multicenter, prospective, randomized, controlled trial | n | = 95 | Change in LVESD, Percentage of surviving patients. |
NECTAR-HF presented significant improvement in quality of life, NYHA class and functional status [4] | There were no significant differences in serious adverse events between control and therapy groups. The overall rate of implantation-related infections was 7.4% |
|||
| Multicenter, open-label, uncontrolled trial | n | = 60 | Change in: LVESV EF, Adverse events. |
ANTHEM-HF showed positive, durable improvement of cardiac function [5] | Serious adverse events occurred in 16 patients. There was one death related to system implantation due to an embolic stroke that occurred 3 days after surgery. |
||||||
| Splanchnic nerve stimulation | Excessive cardiac filling pressure due to overactivity of SNS resulting in visceral vasoconstriction and rapid volume shift from visceral to central compartment during exercise. | GSN modulation preventing exercise provoked visceral vasoconstriction and subsequent fluid shift from the visceral compartment to the central venous system. | Single-center, prospective, open-label, uncontrolled trials | n | = 11, | n | = 15 | Change in CVPPAMP PCWP |
Splanchnic-HF 1, and Splanchnic-HF 2 showed a reduction in PCPW and improvement of the cardiac index during exercise [6][7] | Splanchnic-HF 1, and Splanchnic-HF 2 showed a reduction in PCPW and improvement of the cardiac index during exercise [6,7] | No adverse events were reported. |
| Multicenter, prospective, uncontrolled, pilot study |
Change in: mean PCPW at rest and exercise (20 W). Adverse events. |
REBALANCE-HF confirmed the reduction in exercise PCPW in HFpEF and NYHA class improvement [8] | There were three non-serious device-related adverse events reported in this study: HF decompensation due to periprocedural fluid overload, transient hypertension and back pain following ablation. | ||||||||
| Cardiopulmonary nerve stimulation | Impaired LV contractility and relaxation. | Stimulation of the autonomic system area responsible for LV contractility resulting in positive lusitropic and inotropic effects. | Single-center, first-in-human, proof-of-concept study | n | = 15 | Adverse events. | A proof-of-concept study showed improvement of LV contractility and an increase in mean arterial pressure without affecting the heart rate [9] | No device-related serious adverse events were reported. | |||
| Phrenic nerve stimulation | Central apnea due to periodic drop in CO | 2 | partial pressure to below the threshold for triggering the action potential in the respiratory center caused by greater sensitivity to carbon dioxide leading to potent stimulus of rhythmic breathing. | Transvenous stimulation of phrenic nerve during apneas. | Multicenter, randomized, open-label study | n | = 151 | Reduction in AHI and freedom from serious adverse events | The remedē System Pivotal Trial showed significant reduction in AHI, arousal index, desaturation and apnea episodes. It also revealed improvement in quality of life, sleep structure and EF [10][11] | The remedē System Pivotal Trial showed significant reduction in AHI, arousal index, desaturation and apnea episodes. It also revealed improvement in quality of life, sleep structure and EF [10,11] | Cumulatively, 21 (14%) serious adverse events were observed in 5-year follow-ups (15; (10%) in the first 12 months). It predominantly included electrode dysfunction, electrode dislocation and infection of the implantation site [10] |
| Asymptomatic diaphragmatic stimulation | High left ventricle pre-load and after-load pressures increase remodeling and HF progression. | Stimulation of diaphragm muscle fibers synchronized with cardiac cycle to decrease intrathoracic pressures. | Single-center, randomized, open-label study | n | = 33 | LVEF improvement | EPIPHRENIC II Study showed significant improvement of LVEF, maximal power on effort, reduction in NYHA class, without differences in 6-min walking test or BNP concentration [12][13] | EPIPHRENIC II Study showed significant improvement of LVEF, maximal power on effort, reduction in NYHA class, without differences in 6-min walking test or BNP concentration [12,13] | Three patients were excluded due to dysfunctional diaphragmatic electrode. No adverse events were observed [12] | ||
| Multicenter, non-randomized, open-label study | n | = 15 | Freedom from serious adverse events during procedural recovery or acute therapy | VisONE study showed improvement in LVEF and life quality (evaluated in SF-36); extended walking distance during the 6 MWT was observed at a 1-year follow-up. [13] | No adverse events were observed during procedural recovery, acute therapy (primary outcome) and in 12month follow-up (secondary outcome) [13] | ||||||
| Reprieve system | Problems with controlling decongestive therapy to avoid too rapid diuretic response and hypovolemia and, on the other hand, providing too much fluid, which worsens volume overload. | Sustaining the accurate fluid balance by measuring the urine output and providing the exact amount of replacement solution to achieve preset fluid balance. | Non-randomized, single-center, prospective, open-label, studies, both | n | = 19 | Device and procedure-related adverse events and decongestive efficacy | Higher urine output and decrease in CVP in comparison to the baseline. Actual fluid loss did not exceed target fluid loss at the end of therapy in every patient [14] | No serious adverse events were observed. One case of hypokalemia occurred. | |||
| Transcatheter renal venous decongestion system | Congestion in renal veins. | Transfemoral inserted flow pump, which reduces renal vein pressure to the desired level. | No results have been published so far. | Device and procedure-related adverse events, technical and procedural feasibility | The trial to evaluate TRVD was terminated prematurely, no results have been published so far. | No results have been published so far. | |||||
| Doraya Catheter | Congestion in renal veins. | Partial obstruction of the flow in the inferior vena cava below the level of the renal veins reduces renal vein pressure | First in-human, single-arm, open-label study | n | = 9 | Serious adverse events. | The catheter was successfully deployed in all patients. Clinical symptoms, as well as diuresis and natriuresis, improved [15] | No device-related or embolic events were reported. One serious procedure-related adverse event: bleeding hematoma from the injection site, resolved without sequelae. | |||
| preCARDIA | Increased right ventricle preload. | Obstruction of the superior vena cava leading to an intermittent decrease in preload. | Multicenter, prospective, single-arm exploratory safety and feasibility, open-label, trial | n | = 30 | Freedom from device or procedure-related serious adverse events | Successful decrease in right atrial pressure and PCWP, increase in net fluid balance and urine output [16] | No device or procedure-related serious adverse events were observed. | |||
| WhiteSwell | Increased preload causes lymphatic congestion, which impairs interstitial drainage and exacerbates oedema. | Reduction in the pressure in the area of lymphatic duct outflow into venous vessels. | The animal model study, | n | = 7 sheep, used in 1 human, | n | = 1 | Serious adverse events. | Examined in a ovine model. Trend toward improved oxygenation an diuresis was noticed [17] | No adverse events were reported in in-human application. | |
| AquaPass | Insufficient urine volume removal. | Enhancing the sweat rate to remove fluid directly from interstitial space. | Feasibility and short-term performance, single-arm, open-label study, | n | = 16 | Serious adverse events, treatment tolerance, ability to control skin temperature between 33 and 38 Celsius degrees). | The procedure was safe in HF patients, successful weight loss was observed. Increased skin temperature without elevating core temperature above average was achieved in each patients [18] | No adverse event occurred. |
HF remains a major medical problem and is associated with a high occurrence of rehospitalization and deaths, which constitute a huge problem for patients as well as healthcare systems worldwide [19]. Given that, numerous methods to improve outcome in HF have arisen, some including device-based treatment techniques.
