Neurons are the basic building blocks of the human body’s neurological system. Atrophy is defined by the disintegration of the connections between cells that enable them to communicate. Peripheral neuropathy and demyelinating disorders, as well as cerebrovascular illnesses and central nervous system (CNS) inflammatory diseases, have all been linked to brain damage, including Parkinson’s disease (PD). It turns out that these diseases have a direct impact on brain atrophy. However, it may take some time after the onset of one of these diseases for this atrophy to be clearly diagnosed. With the emergence of the Coronavirus disease 2019 (COVID-19) pandemic, there were several clinical observations of COVID-19 patients.
- COVID-19
- brain atrophy
- cerebrovascular diseases
1. Introduction
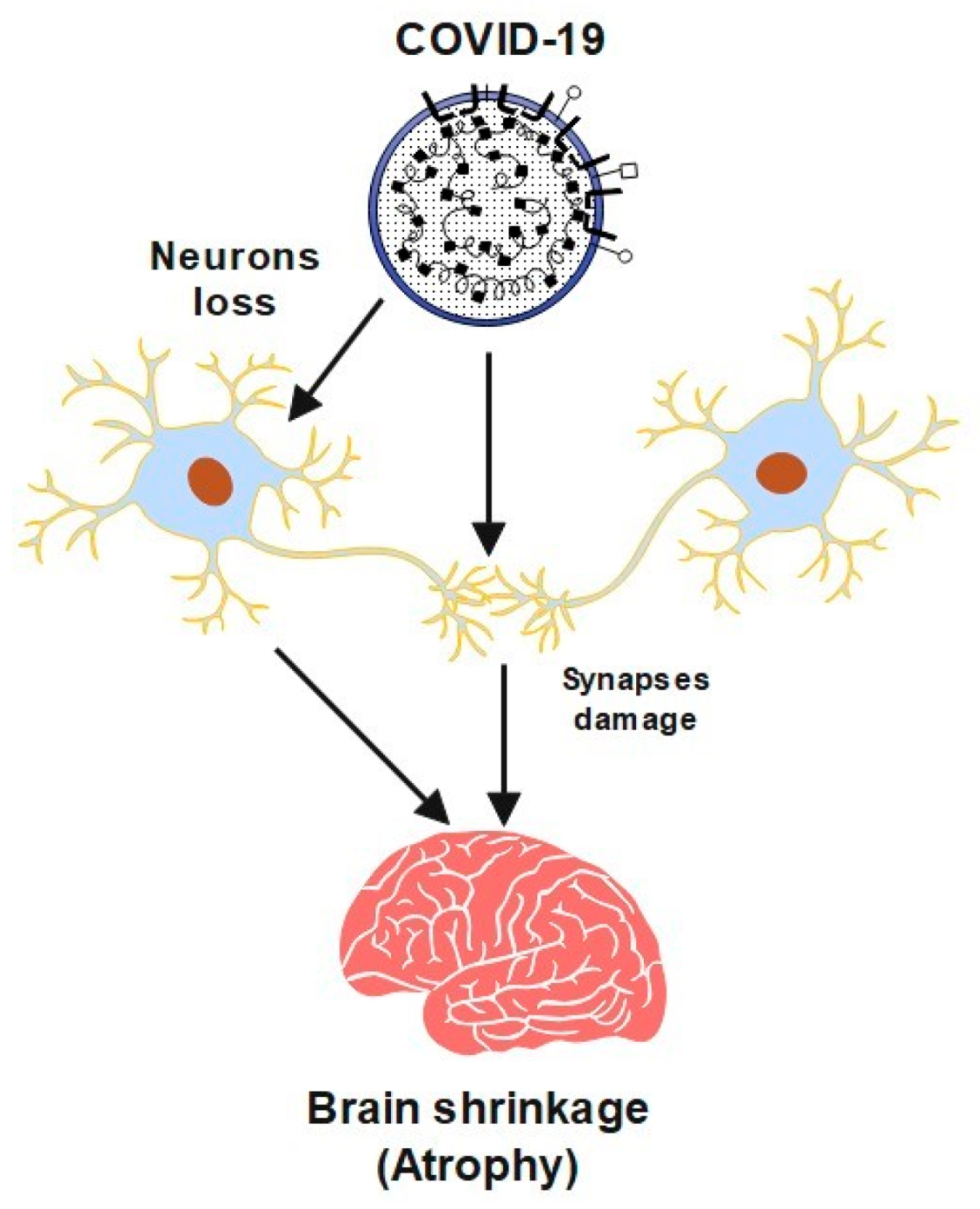
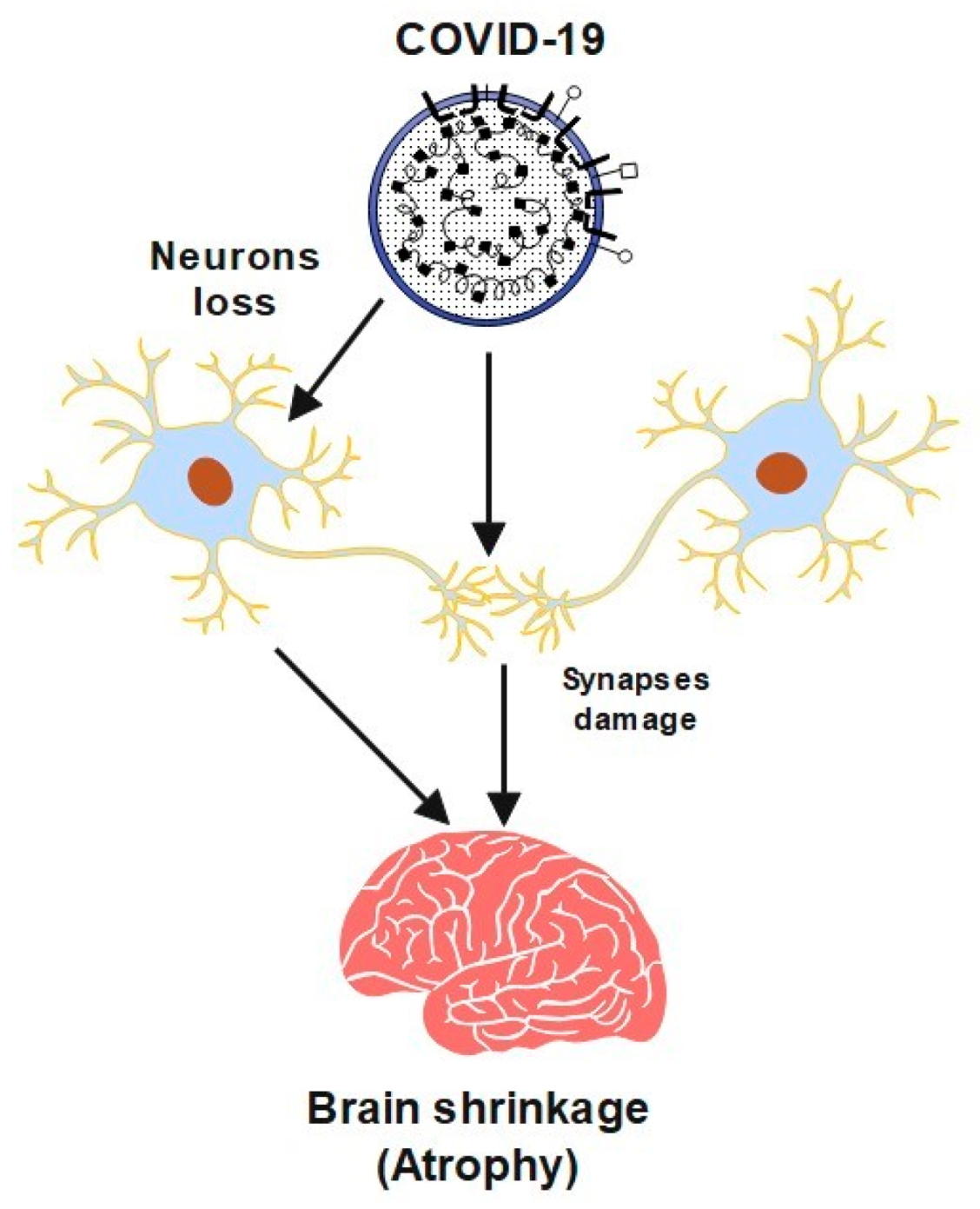
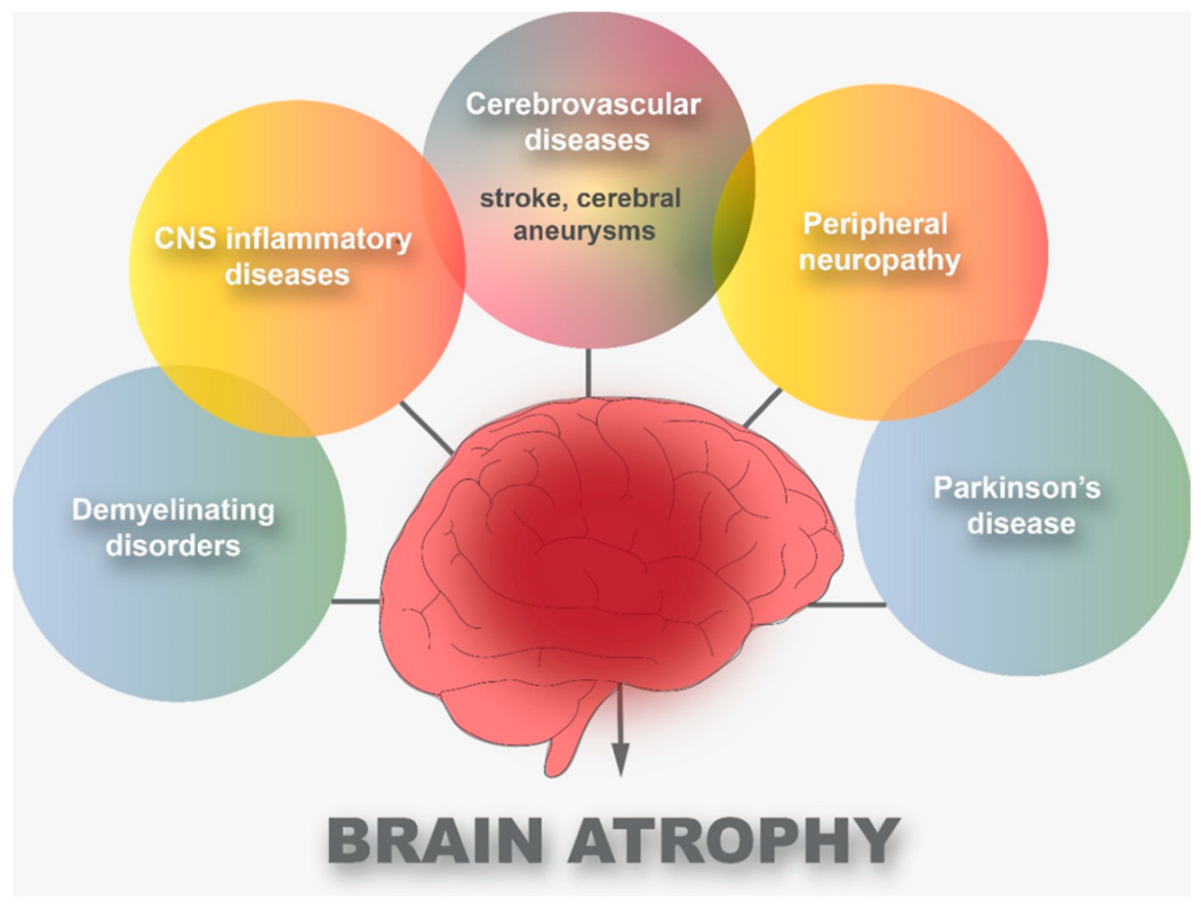
2. Cerebrovascular Diseases and COVID-19
References
- Verma, K.; Amitabh Prasad, D.N.; Kumar, B.; Kohli, E. Brain and COVID-19 Crosstalk: Pathophysiological and Psychological Manifestations. ACS Chem. Neurosci. 2020, 11, 3194–3203.
- Li, Z.; Liu, T.; Yang, N.; Han, D.; Mi, X.; Li, Y.; Liu, K.; Vuylsteke, A.; Xiang, H.; Guo, X. Neurological manifestations of patients with COVID-19: Potential routes of SARS-CoV-2 neuroinvasion from the periphery to the brain. Front. Med. 2020, 14, 533–541.
- Dixon, L.; Varley, J.A.; Gontsarova, A.; Mallon, D.; Tona, F.; Muir, D.A.; Luqmani, A.; Jenkins, I.H.; Nicholas, R.S.; Jones, B.; et al. COVID-19-related acute necrotizing encephalopathy with brain stem involvement in a patient with aplastic anemia. Neurol. Neuroimmunol. Neuroinflammation 2020, 7, e789.
- Coolen, T.; Lolli, V.; Sadeghi, N.; Rovai, A.; Trotta, N.; Taccone, F.S.; Creteur, J.; Henrard, S.; Goffard, J.; Dewitte, O.; et al. Early postmortem brain MRI findings in COVID-19 non-survivors. Neurology 2020, 95, e2016-27.
- Gandhi, S.; Srivastava, A.K.; Ray, U.; Tripathi, P.P. Is the Collapse of the Respiratory Center in the Brain Responsible for Respiratory Breakdown in COVID-19 Patients? ACS Chem. Neurosci. 2020, 11, 1379–1381.
- Espinosa, P.S.; Rizvi, Z.; Sharma, P.; Hindi, F.; Filatov, A. Neurological Complications of Coronavirus Disease (COVID-19): Encephalopathy, MRI Brain and Cerebrospinal Fluid Findings: Case 2. Cureus 2020, 12, e7930.
- Hagerty, S.L.; Williams, L.M. The impact of COVID-19 on mental health: The interactive roles of brain biotypes and human connection. Brain Behav. Immun. Health 2020, 5, 100078.
- Yin, X.; Zheng, X.; Peng, W.; Wu, M.; Mao, X. Vascular Endothelial Growth Factor (VEGF) as a Vital Target for Brain Inflammation during the COVID-19 Outbreak. ACS Chem. Neurosci. 2020, 11, 1704–1705.
- Lin, E.; Lantos, J.; Strauss, S.; Phillips, C.; Campion, T.R.; Navi, B.B.; Parikh, N.S.; Merkler, A.E.; Mir, S.; Zhang, C.; et al. Brain Imaging of Patients with COVID-19: Findings at an Academic Institution during the Height of the Outbreak in New York City. Am. J. Neuroradiol. 2020, 41, 2001–2008.
- Chigr, F.; Merzouki, M.; Najimi, M. Autonomic Brain Centers and Pathophysiology of COVID-19. ACS Chem. Neurosci. 2020, 11, 1520–1522.
- Saavedra, J.M. COVID-19, Angiotensin Receptor Blockers, and the Brain. Cell. Mol. Neurobiol. 2020, 40, 667–674.
- Fischer, D.; Threlkeld, Z.D.; Bodien, Y.G.; Kirsch, J.E.; Huang, S.; Schaefer, P.W.; Rapalino, O.; Hochberg, L.R.; Rosen, B.R.; Edlow, B.L. Intact Brain Network Function in an Unresponsive Patient with COVID-19. Ann. Neurol. 2020, 88, 851–854.
- Crunfli, F.; Carregari, V.C.; Veras, F.P.; Vendramini, P.H.; Valença, A.G.; Antunes, A.S.; Brandão-Teles, C.; Zuccoli, G.D.; Reis-de-Oliveira, G.; Silva-Costa, L.C.; et al. SARS-CoV-2 infects brain astrocytes of COVID-19 patients and impairs neuronal viability. medRxiv 2021.
- Achar, A.; Ghosh, C. COVID-19-Associated Neurological Disorders: The Potential Route of CNS Invasion and Blood-Brain Barrier Relevance. Cells 2020, 9, 2360.
- Yang, A.C.; Kern, F.; Losada, P.M.; Maat, C.A.; Schmartz, G.P.; Fehlmann, T.; Schaum, N.; Lee, D.P.; Calcuttawala, K.; Vest, R.T.; et al. Broad transcriptional dysregulation of brain and choroid plexus cell types with COVID-19. BioRxiv 2020.
- Fabbri, V.P.; Foschini, M.P.; Lazzarotto, T.; Gabrielli, L.; Cenacchi, G.; Gallo, C.; Aspide, R.; Frascaroli, G.; Cortelli, P.; Riefolo, M.; et al. Brain ischemic injury in COVID-19-infected patients: A series of 10 post-mortem cases. Brain Pathol. 2020, 31, 205–210.
- Pinggera, D.; Klein, B.E.; Thomė, C.; Grassner, L. The influence of the COVID-19 pandemic on traumatic brain injuries in Tyrol: Experiences from a state under lockdown. Eur. J. Trauma Emerg. Surg. 2020, 47, 653–658.
- Malentacchi, M.; Gned, D.; Angelino, V.; Demichelis, S.; Perboni, A.; Veltri, A.; Bertolotto, A.; Capobianco, M. Concomitant brain arterial and venous thrombosis in a COVID-19 patient. Eur. J. Neurol. 2020, 27, e38–e39.
- Haider, A.; Siddiqa, A.; Ali, N.; Dhallu, M. COVID-19 and the Brain: Acute Encephalitis as a Clinical Manifestation. Cureus 2020, 12, e10784.
- Mao, X.; Jin, W. The COVID-19 Pandemic: Consideration for Brain Infection. Neuroscience 2020, 437, 130–131.
- Choi, J.; Lee, H.; Park, J.; Cho, S.; Kwon, M.; Jo, C.; Koh, Y.H. Altered COVID-19 receptor ACE2 expression in a higher risk group for cerebrovascular disease and ischemic stroke. Biochem. Biophys. Res. Commun. 2020, 528, 413–419.
- Altable, M.; De la Serna, J. Cerebrovascular disease in COVID-19: Is there a higher risk of stroke? Brain Behav. Immun. Health 2020, 6, 100092.
- Escalard, S.; Chalumeau, V.; Escalard, C.; Redjem, H.; Delvoye, F.; Hébert, S.; Smajda, S.; Ciccio, G.; Desilles, J.P.; Mazighi, M.; et al. Early Brain Imaging Shows Increased Severity of Acute Ischemic Strokes with Large Vessel Occlusion in COVID-19 Patients. Stroke 2020, 51, 3366–3370.
- Voznyuk, I.A.; Ilyina, O.; Kolomentsev, S.V. Ischemic Stroke as a Clinical Form and Pathogenetic Model in the Structure of Central Nervous System Lesions in COVID-19. Bull. Rehabil. Med. 2020, 4, 90–98.
- Bekelis, K.; Missios, S.; Ahmad, J.; Labropoulos, N.; Schirmer, C.M.; Calnan, D.R.; Skinner, J.; Mackenzie, T. Ischemic Stroke Occurs Less Frequently in Patients with COVID-19. Stroke 2020, 51, 3570–3576.
- Landa, N.A.; Oficialdegui, C.V.; Fernández, K.I.; Larrabe, I.G.; Onaindia, S.R.; Benguria, S.T. Ischemic-hemorrhagic stroke in patients with Covid-19. Rev. Espanñola Anestesiol. Y Reanim. 2020, 67, 516–520.
- Zhou, Y.; Lu, J.; Cheng, Y.; Xin, N. Nervous system complications of COVID-19 with a focus on stroke. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 13044–13048.
- Nunes, N.D.; Nascimento, J.F.; Nascimento, J.K.; Castro, R.R.; Azizi, M.A.; Junior, G.C.; Catharino, A.M.; Silveira, V.; Neves, M.A. Brain and Covid-19: An Integrative Review. E-Cronicon Neurol. 2020, 12, 101–107.
- Pennisi, M.; Lanza, G.; Falzone, L.; Fisicaro, F.; Ferri, R.; Bella, R. SARS-CoV-2 and the Nervous System: From Clinical Features to Molecular Mechanisms. Int. J. Mol. Sci. 2020, 21, 5475.
- Goldberg, M.F.; Goldberg, M.F.; Cerejo, R.; Tayal, A. Cerebrovascular Disease in COVID-19. Am. J. Neuroradiol. 2020, 41, 1170–1172.
- Hernández-Fernández, F.; Sandoval Valencia, H.; Barbella-Aponte, R.A.; Collado-Jiménez, R.M.; Ayo-Martin, O.; Barrena, C.; Molina-Nuevo, J.D.; García-García, J.; Lozano-Setién, E.; Alcahut-Rodríguez, C.; et al. Cerebrovascular disease in patients with COVID-19: Neuroimaging, histological and clinical description. Brain 2020, 143, 3089–3103.
- Rea, G.; Lassandro, F.; Lieto, R.; Bocchini, G.; Romano, F.Y.; Sica, G.; Valente, T.; Muto, E.; Murino, P.; Pinto, A.; et al. Lesson by SARS-CoV-2 disease (COVID-19): Whole-body CT angiography detection of “relevant” and “other/incidental” systemic vascular findings. Eur. Radiol. 2021, 31, 7363–7370.
- Bittmann, S. COVID-19: Expression of ACE2-receptors in the Brain Suggest Neurotropic Damage. J. Regen. Biol. Med. 2020, 2, 1–3.
- Jaunmuktane, Z.; Mahadeva, U.; Green, A.C.; Sekhawat, V.; Barrett, N.A.; Childs, L.; Shankar-Hari, M.; Thom, M.; Jäger, H.R.; Brandner, S. Microvascular injury and hypoxic damage: Emerging neuropathological signatures in COVID-19. Acta Neuropathol. 2020, 140, 397–400.
- Boyko, A.N.; Sivertseva, S.; Spirin, N.N. Nervous system damage in COVID-19 with an emphasis on the management of patients with multiple sclerosis. Neurol. Neuropsychiatry Psychosom. 2020, 12, 44–47.
- Mehta, S.; Bhandari, S.; Mehta, S. Brain autopsies in fatal COVID-19 and postulated pathophysiology: More puzzling than a Rubik’s cube. J. Clin. Pathol. 2020, 74, 612–613.
- Babkina, A.S.; Golubev, A.; Ostrova, I.V.; Volkov, A.V.; Kuzovlev, A.N. Brain Morphological Changes in COVID-19. Gen. Reanimatol. 2021, 17, 4–15.
- Haque, E.; Akther, M.; Azam, S.; Kim, I.; Choi, D. COVID-19 and The Brain. J. Microb. Biochem. Technol. 2020, 12, 1–9.
- Bulfamante, G.; Bocci, T.; Falleni, M.; Campiglio, L.; Coppola, S.; Tosi, D.; Chiumello, D.; Priori, A. Brainstem neuropathology in two cases of COVID-19: SARS-CoV-2 trafficking between brain and lung. J. Neurol. 2021, 268, 4486–4491.
