Pollination is essential to maintain ecosystem balance and agricultural production. Domesticated bee pollination, which is easy to feed and manage, and mechanized pollination, which is not restricted by the environment, are considered the main technical means to alleviate the “pollinating insect crisis”. The selection of pollination method should be based on the physiological characteristics of crops and the actual environmental conditions of natural pollination. The pollination ability of bees is closely related to the status of the bees. Maintaining the pollination ability of bees in a reasonable range is the goal of bee pollination services. Colony state control technology needs to develop in two directions. First, there is a need to develop colony state monitoring technology based on multi-feature information fusion and to explore the self-regulation mechanisms of the colony in response to various factors. Second, based on these self-regulation mechanisms, there is a need to develop a low-cost and non-invasive bee colony state and pollination capacity estimation model, monitoring technology, and equipment based on single feature information. The goals of mechanized pollination are “efficiency” and “precision”. Mechanized pollination technology needs to be developed in two directions. First, the mechanisms of pollen abscission, transport, and sedimentation in different crops and mechanized pollination conditions should be explored. Second, research and development of efficient and accurate pollination equipment and technology based on the integration of multiple technologies such as pneumatic assistance, auxiliaries, static electricity, target, variables, and navigation, are needed.
- bee pollination
- mechanized pollination
- crops
- efficient pollination technology
1. Introduction
2. Pollination Methods for Different Crops
2.1. Basic Structural Features of Flowers
As shown in Figure 1, a flower comprises six parts: flower stalk, receptacle, calyx, corolla, stamen, and pistil. The stamens comprise filaments and anthers that contain a large amount of pollen; the pistil comprises the stigma, style, and ovary. The stigma is at the top of the pistil and is used to receive pollen grains. A flower with a calyx, corolla, stamen, and pistil is called a complete flower, whereas those lacking one or more parts are called incomplete flowers. Flowers with pistils and stamens are called hermaphroditic flowers. A flower lacking a stamen is called a unisexual flower; a flower with only stamens is a male flower, and a one with only pistils is a female flower. A plant with female and male flowers is called a hermaphrodite, whereas the separation of female and male flowers in different plants is called dioecism [10].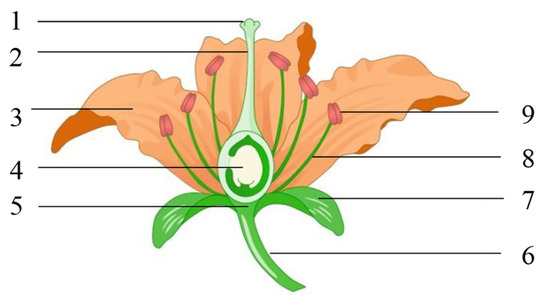
2.2. Self-Pollination and Cross-Pollination
2.3. Differences in Pollination Methods of Different Crops
3. Status of Efficient Bee Pollination Technology
3.1. Principle of Efficient Pollination of Bees
Bee pollination is a “bonus” of bees’ out-foraging behavior. In the long-term coevolution of bees and plants, bees have formed numerous characteristics suitable for plant pollination, including being easily attracted by bright colored entomophilous flowers that emit an aroma and secrete nectar; in addition, bees have identifying memory and pollination activity focused on specific species over a long period, high-frequency vibration generated by the wings during foraging that causes pollen to fall off, morphological structures such as villi and pollen baskets that easily adhere to pollen, social group structure, and numerous groups [21].
3.2. Breeding of High-Quality Pollinating Bee Varieties
There are 2 Apis species, and 9 Bombus species, and 8 solidary bee species that provide pollination services for crops. In addition, more bee varieties are waiting to be selected, including 6 bumble bees, 15 stingless bees, and 14 solitary bees. Europe, Asia, North America, and South America have more species that can be more easily used to breed high-quality pollinating bee varieties [25][22]. Fertility, population growth rate, bee sorting, collecting ability, pollination habit, disease resistance, and stress resistance are the biological characteristics that need to be closely investigated in pollinating bee breeding. Compared with bees, Bombus spp. have longer beaks, greater ability for nectar collection, and adaptability to low light density. Because bumblebees lack a developed information exchange system similar to bees, they are more suitable for pollination of crops in greenhouses where they are widely used [26,27][23][24].3.3. Climate and Environment Control Technology Outside the Hive
Climate and environmental factors (e.g., temperature, humidity, light, rain and wind) outside the hive directly affect the physiological function of bees, the secretion of nectar, and pollen germination of nectar crops, thus affecting the foraging behavior of bees and their pollination effects. In a subtropical climate, the initial temperature at which Apis Spp. commence foraging behavior on apple blossoms is 16 °C. As the temperature increases, the number of bees out-foraging continuously increases and reaches a relatively stable high level when the temperature exceedes 20 °C. Temperatures below 20 °C, wind speeds of over 15 mph, rain, and lower light levels limit bees’ out-foraging behavior [28][25]. In the regional climate of Lithuania, Apis mellifera almost stopped its foraging behavior on rape crops when the temperature reached 43 °C [29][26]. A light intensity of 600–1700 lx and solar radiation of 9–20 mW/cm2 are the minimum climatic and environmental conditions required for the four types of bees (Apis dorsata F; A. mellifera L; A. cerana F; and A. florea F) to go out for food [30][27]. Under high humidity conditions, the nectar secretion rate of crops is higher and the evaporation rate is lower; therefore, the foraging rate of bees is positively correlated with environmental humidity. However, considering both pollen collection and transportation of bees, researchers believe that a lower humidity environment is more conducive to bee foraging [31][28]. Fields and orchards are open-air spaces and bees require favorable natural climate conditions to go out for food. Bees can regulate their body temperature to ensure physiological functions, such as food absorption, respiration, metabolism, and energy supply [37][29]. As shown in Figure 3, foraging bees use solar energy to increase body temperature and save energy in a low-temperature environment. At the same time, they choose between “investment-oriented” and “energy-saving” thermoregulation strategies based on whether increasing body temperature can maximize the efficiency of food absorption [38][30]. In a high-temperature environment, foraging bees can dissipate heat and cool through the evaporation of honey sacs and oral water droplets [39,40][31][32].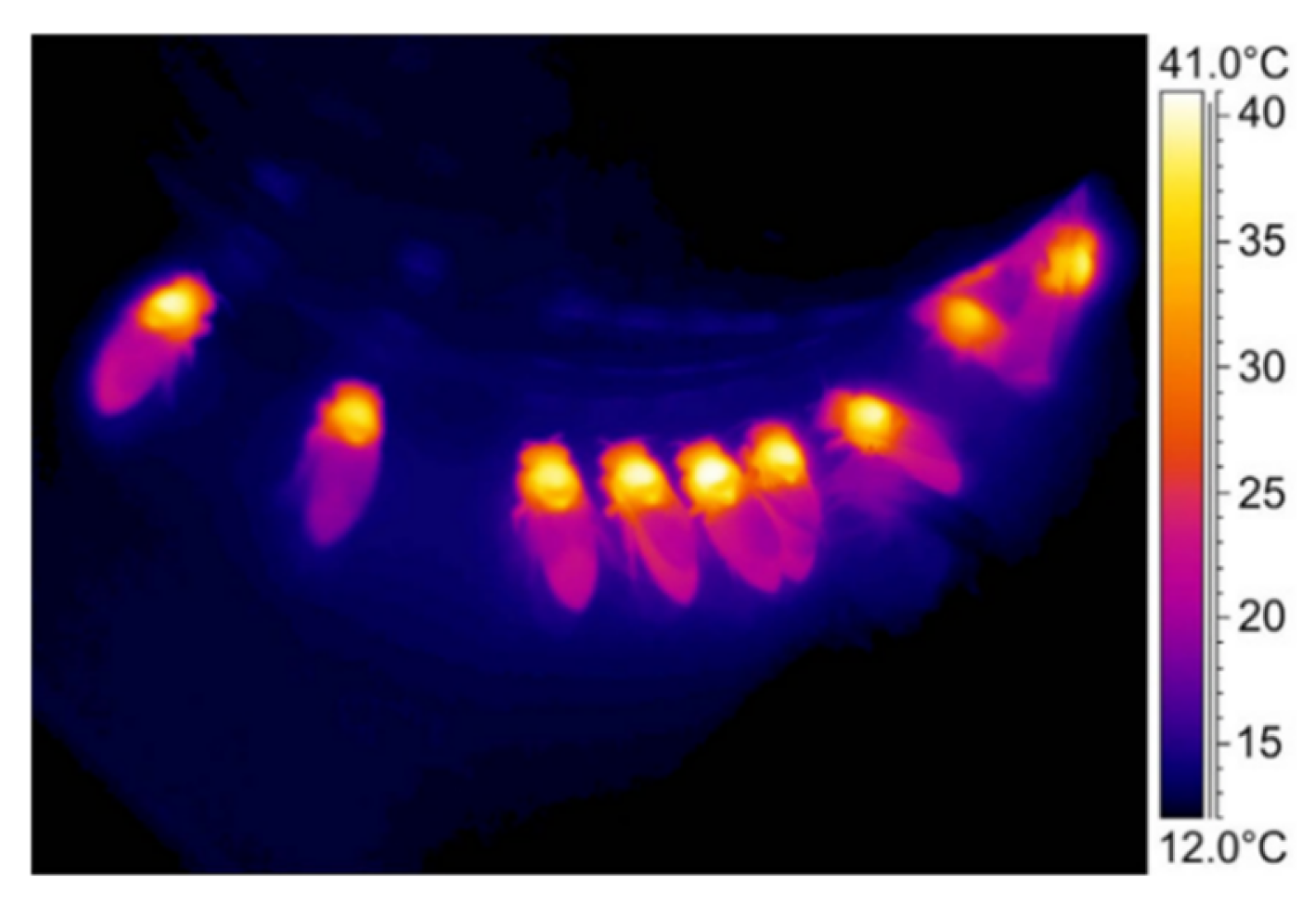
3.4. Temperature and Humidity Control Technology in the Beehive
Healthy breeding of queen bees, drones, and worker bees is the basis for the health, strength, and stability of the colony. The beehive provides a breeding and living place that shields the bee colony from light, rain, and wind. Temperature and humidity in the beehive are the most critical factors that affect colony reproduction and are directly affected by the climate outside the beehive.
3.4.1. Influence of Temperature and Humidity on Beehive Colony Reproduction
The optimum temperature for the development of the fertilized eggs of Apis mellifera and the queen bee pupae in the capping platform is 35–36 °C; between 32 °C and 36 °C, the fertilized eggs and queen bee pupae develop normally. When the temperature reaches 37 °C, the queen bee pupae cannot develop normally [46][36]. The temperate ranges 29–37 °C and 31–37 °C are the development temperatures of Apis mellifera and Apis cerana, respectively. With a decrease in temperature, the development period of worker bee capping is significantly prolonged, and this affects developmental and morphological indicators such as birth weight, snout length, and forewing area. Beyond this temperature zone, bees are deformed or die [47][37]. The temperature of pupa development can significantly affect the behavioral performance of honeybee adults, thus affecting their foraging tasks. The learning and memory behavior of honeybee adults under a pupa development temperature of 36 °C is better than that under 32 °C and 34.5 °C [48][38]. The synaptic organization of the adult honeybee brain is mediated by the temperature experienced during pupal development, which affects its communication and learning behavior. In the olfactory input area of the mushroom body of the brain, the number of microglomers was highest in bees incubated at a temperature of 34.5 °C that is normally maintained by brood cells, and the number of microglomers was significantly reduced in bees incubated at 1 °C below or above this standard [49][39]. Pupal developmental temperature affected JH metabolism and octopamine levels in bee brains; bees developing at higher temperatures showed an earlier tendency to go out for food [50][40]. The optimum humidity range for normal hatching of bee eggs is 90% to 95%; outside this humidity range, the number of larvae that hatch normally decreases significantly [51][41]. In environments with high relative humidity, bees cannot excrete metabolized and food-dissolved water, which shortens their lifespan [52][42].3.4.2. Colony Self-Regulation Mechanism of Beehive Temperature and Humidity
Honeybee antennae are multimodal sensory organs that can sense various information, such as temperature, humidity, smell, taste, and mechanical stimuli, among others. Their temperature sensitivity is 0.25 °C. Honeybees receive real-time information on temperature, humidity, and spleen temperature in the hive through their antennae [53,54,55][43][44][45]. When the temperature, humidity, and temperature of the spleen in the hive are beyond the reasonable range for breeding and life, the bee colony takes measures to modify it. When the temperature in the hive or spleen is low, thermogenic bees raise the temperature of their thoracic cavity and attach the breast to the wax cover of the sealing lid, transferring this heat to maintain the temperature in the hive and spleen. Non-thermogenic bees gather and squeeze the cell comb to increase the sealing performance of the hive and reduce heat loss [56,57][46][47]. When the temperature in the beehive or spleen is high, the bee colony increases airflow between the inside of the beehive and the outside world through ordered directional fans to discharge the overheated air in the beehive; furthermore, the bee colony is also scattered in the beehive, further improving ventilation and heat dissipation [58][48].
3.4.3. Temperature and Humidity Monitoring Technology in Beehives
The existing temperature and humidity control in the beehive mainly relies on the self-regulating ability of the bee colony. A healthy, strong, and stable bee colony can maintain the temperature and humidity in the beehive within a reasonable range to ensure the breeding and life of the bee colony. A typical winter colony of 17,500 bees can survive for over 300 h even when the temperature outside the hive is as low as −25 °C [61][49]. However, when the state of the bee colony changes because of, for example, the occurrence of disease and insect pests, separation of bees, escape, or changes in the temperature and humidity inside the beehive caused by external changes that exceed the ability of the existing bee colony to adjust, the survival of the entire bee colony is seriously threatened.
3.5. Bee Colony State Management and Control Technology
Colony state is a broad concept that includes multi-characteristic information about the queen bee, drones, and worker bees (e.g., breeding status, population structure, number, nest, spleen status, degree of diseases, and insect pests). In actual production, beekeepers still need to manually estimate the status of the bee colony regularly before implementation of strategic management; this approach is labor intensive, low efficiency, extremely dependent on professional experience, and interferes with the life of the bee colony and pollination operations. With the development of modern information technologies such as artificial intelligence and the internet of things, researchers have attempted to use non-invasive intelligent equipment to obtain real-time bee colony information using images, sound, vibration, weight, and incoming and outgoing bee activity.3.5.1. Image Monitoring Technology in Beehives
Image-based information from the beehive is the most intuitive technical means to assess the state of the bee colony. As honey bees have phototaxis and cannot recognize red light, image monitoring technology in beehives often uses red light irradiation or infrared thermal imaging to obtain image information.3.5.2. Sound and Vibration Monitoring Technology in Beehives
The sound and vibration emitted by bees are an essential part of the communication mechanism of the bee colony. The temperature and humidity regulation of the colony, group activities such as going out for food, separating bees, escaping, losing the king, and disease can all be reflected by sound and vibration information.
3.5.3. Bee Colony Weight Monitoring Technology
The colony weight includes the main colony elements such as the adult bee colony, larvae, honey, and pollen, and its changes can accurately reflect the productivity, health, robustness, and stability of the colony in the beehive. To obtain bee colony weight information, professional electronic balances or electronic scales are often used to weigh beehives, comb spleens, adult bees, honey, and pollen.3.5.4. Bee Colony Monitoring Technology in the Hive Gate Area
Monitoring bee colony activities in the hive gate area can not only directly provide information on the foraging enthusiasm of bee colonies but also indirectly estimate bee colony status. Research on bee colony monitoring technology outside the hive focuses mainly on the number of bees and the number entering and leaving the hive gate area. Four main technical solutions are currently used: radio frequency identification tags, capacitive sensors, photoelectric sensors, and machine vision [95,96,97,98,99][50][51][52][53][54]. However, radio frequency identification tags, capacitive sensors, and photoelectric sensors suffer from regular beehive modifications, high costs, and complex maintenance. The bee colony monitoring system based on machine vision is a non-invasive solution that is easy to install and maintain, low cost, easy to popularize and apply, and has attracted increasing research attention.3.5.5. Bee Colony Monitoring Technology Based on Multi-Feature Information Fusion
The climate outside the hive and the temperature and humidity inside the hive affect bees’ behaviors such as foraging and breeding, thus affecting the colony status. Human activities, pesticides, bee habits, and other factors also significantly affect the state of bee colonies; moreover, these factors are interdependent. Currently, there is no effective model to reasonably estimate the influence of these factors and the self-adjustment mechanism of the colony on the colony state. Furthermore, it is inaccurate to estimate the state of a bee colony only based on single feature such as the climate outside the beehive or the temperature, humidity, sound, or weight inside the beehive.
4. Status of Mechanized Pollination Technology
4.1. Principle of Mechanized Pollination
When there is an unfavorable natural pollination environment, or crops cannot attract pollinating insects, or with staggered flowering periods of male and female flowers, natural wind pollination and bee pollination cannot occur, and artificial assisted pollination is required. Therefore, labor-saving and efficient mechanized pollination has attracted increasing attention. Mechanized pollination refers to using mechanical equipment to shed crop pollen and transport it to the pistil stigma. For crops with staggered flowering periods of male and female flowers, a common method is to collect the pollen first and then spray or atomize the target pistil. Various methods are available, including mechanical collision, ultrasonic, and pneumatic assistance.4.2. Mechanized Pollination Technology and Equipment for Field Crops
Mechanized pollination of field crops usually involves pneumatic-assisted pollination to create pollination conditions similar to natural wind pollination. Wang et al. [116][55] studied the effect of airflow speed on the pollination of hybrid rice; they suggested that pneumatic-assisted pollination mainly relies on two processes (air flow directly blowing out the pollen and airflow making the plant vibrate) that cause pollen to shed and be transported. Their results showed that airflow speed significantly affects pollen distribution and that horizontal distribution, vertical distribution, and total pollen increase with airflow speed. The horizontal distribution has a unique bimodal image, and the later peak moves away from the pollen source with increasing flow velocity. The vertical distribution moves closer to the airflow center with increasing flow velocity.4.3. Mechanized Pollination Technology and Equipment for Orchards
Compared with field crops, the canopy of orchard crops is large and dense, and the planting row spacing is wide. As such, it is unreasonable to rely on rotorcraft to create wind-pollinated pollination conditions; moreover, different types of fruit trees are planted in the same orchard to ensure the fruit setting rate, but the flowering period of various fruit tree varieties is not synchronized, which increases the difficulty of pollination [127][56]. Cross-pollinated orchard crops represented by apples, pears, and kiwi fruit have a large amount of pollen that can be collected and stored in advance, and therefore, air-blown or atomized pollination is now commonly used. This also overcomes the problem that the flowering periods of various fruit tree varieties are not synchronized [128,129,130][57][58][59]. For cross-pollinated orchard crops, the efficient collection of high-quality pollen is the basis of their mechanized pollination. The quality varies with species, climatic conditions during flowering, flower maturity, and agronomic practices [131,132,133,134][60][61][62][63]. The pollen germination rate is an important index for measuring the quality of pollen, which determines the amount of pollen sprayed by mechanized pollination [135][64]. The germination rate of some commercial kiwi pollen exceeds 80%. Some researchers tried setting a device at the hive entrance to obtain pollen collected by bees, but the quality of the pollen processed and collected by some varieties of bees was not ideal, which affected the success rate of crop pollination [136][65]. Mechanized pollen collection is suitable for cross-pollinated crops with a large amount of pollen. A backpack type pollen vacuum cleaner can collect over 500 cc/h [137][66]. For crops in which it is difficult to obtain pollen mechanically, manual pollen collection is still the main method [138][67]. In 2018, the cost of kiwi pollen in New Zealand was USD 3452/kg, and the recommended application rate is 300–400 g/hm2. The lower storage temperature and ideal drying conditions affect the pollen life [139][68]. Pollen cells with a high water content cannot survive in frozen storage, which may be due to the formation of lethal intracellular ice and subsequent membrane rupture [140][69].4.4. Mechanized Pollination Technology and Equipment for Greenhouses
Compared with fields and orchards, the space in greenhouses is confined and narrow; as such, pollination equipment requires miniaturization. For greenhouse strawberries, Hu et al. [151][70] proposed first covering strawberry flowers with pollen and then using a rotating brush to gently shake the flowers to form a breeze, causing the flower to flutter in the pollen cover to complete the pollination work. Wu et al. [152][71] proposed a method for pollination of greenhouse crops with pneumatic tilting, which involves an automatic cross-pollination mobile operation consisting of the staggered blowing of double-row crops with pneumatic tilting, adaptive humidifying of the first row powder, and then heating and blowing powder.5. Conclusions
The pollination ability of bees is closely related to the status of bees. Maintaining the pollination ability of bees in a reasonable range is the goal of bee pollination services. Currently, there is no effective model that can reasonably incorporate the influence of various factors (including climate outside the hive, temperature, and humidity inside the hive, and the self-regulation mechanism of the hive) to accurately estimate the state of the hive. As such, colony state control technology needs to develop in two directions. First, there is a need to develop colony state monitoring technology based on multi-feature information fusion and to explore the self-regulation mechanisms of the colony in response to various factors. Second, based on the self-regulation mechanisms of bee colonies in response to various factors, there is a need to develop low-cost and non-invasive bee colony state and pollination capacity estimation models, monitoring technology, and equipment based on single feature information. The goals of mechanized pollination are “efficiency” and “precision”. Different operation scenarios and crops rely on different mobile carriers, execution devices, and technologies for mechanized pollination. Mechanized pollination technology must be developed in two directions. First, the mechanisms of abscission, transport, and sedimentation of pollen should be explored in different crops and mechanized pollination conditions. Second, the research and development of efficient and accurate pollination equipment and technology based on the integration of multiple technologies such as pneumatic assistance, auxiliaries, static electricity, target, variables, and navigation are needed. Efficient pollination of agricultural crops should not be regarded as an isolated subject that requires substantial investment. Instead, pollination is an essential link in crop production and management, and it should be included in the concept and application systems of common “smart agriculture” methods. Efficient crop pollination requires the acquisition of information from the massive data generated by “smart agriculture” to guide efficient and accurate operation. Moreover, understanding the feedback of the pollination effect will provide important dimensional information for “smart agriculture”.References
- Ollerton, J.; Winfree, R.; Tarrant, S. How many flowering plants are pollinated by animals? Oikos 2011, 120, 321–326.
- Klein, A.-M.; Vaissiere, B.E.; Cane, J.H.; Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Tscharntke, T. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. B-Biol. Sci. 2007, 274, 303–313.
- Gallai, N.; Salles, J.-M.; Settele, J.; Vaissiere, B.E. Economic valuation of the vulnerability of world agriculture confronted with pollinator decline. Ecol. Econ. 2009, 68, 810–821.
- Kjøhl, M.; Nielsen, A.; Stenseth, N.C. Potential Effects of Climate Change on Crop Pollination; Food and Agriculture Organization of the United Nations: Rome, Italy, 2011.
- Connolly, C. The risk of insecticides to pollinating insects. Commun. Integr. Biol. 2013, 6, e25074.
- Maes, J.; Hauck, J.; Paracchini, M.L.; Ratamaki, O.; Hutchins, M.; Termansen, M.; Furman, E.; Perez-Soba, M.; Braat, L.; Bidoglio, G. Mainstreaming ecosystem services into EU policy. Curr. Opin. Environ. Sustain. 2013, 5, 128–134.
- FitzPatrick, Ú.; Stout, J.; Bertrand, C.; Bradley, K.; Clabby, G.; Keena, C.; Walsh, J. All-Ireland Pollinator Plan 2015–2020; National Biodiversity Data Centre: Waterford, Ireland, 2015.
- Hung, K.-L.J.; Kingston, J.M.; Albrecht, M.; Holway, D.A.; Kohn, J.R. The worldwide importance of honey bees as pollinators in natural habitats. Proc. R. Soc. B-Biol. Sci. 2018, 285, 20172140.
- Binns, C. Robotic Insects Could Pollinate Flowers and Find Disaster Victims Popular Science USA. 2009. Available online: https://www.popsci.com/technology/article/2009-12/flight-robobee/ (accessed on 10 November 2022).
- Leins, P.; Erbar, C. Flower and fruit. Hoppea 2010, 71, 354–355.
- Brown, A.H.D.; Zohary, D.; Nevo, E. Outcrossing rates and heterozygosity in natural populations of Hordeum spontaneum Koch in Israel. Heredity 1978, 41, 49–62.
- Vuletin Selak, G.; Perica, S.; Ban Goreta, S.; Radunic, M.; Poljak, M. Reproductive Success after Self-pollination and Cross-pollination of Olive Cultivars in Croatia. HortScience 2011, 46, 186–191.
- Chavez, J.D.; Lyrene, M.P. Effects of Self-pollination and Cross-pollination of Vaccinium darrowii (Ericaceae) and Other Low-chill Blueberries. HortScience 2009, 44, 1538–1541.
- Lloyd, D.G.; Webb, C.J. The avoidance of interference between the presentation of pollen and stigmas in angiosperms I. Dichogamy. N. Z. J. Bot. 2011, 24, 135–162.
- Webb, C.J.; Lloyd, D.G. The avoidance of interference between the presentation of pollen and stigmas in angiosperms II. Herkogamy. N. Z. J. Bot. 1986, 24, 163–178.
- Trelease, W. The Heterogony of Oxalis violacea. Am. Nat. 1882, 16, 13–19.
- East, E.M. The distribution of self-sterility in the flowering plants. Proc. Am. Philos. Soc. 1940, 82, 449–518.
- Lord, E.M. Cleistogamy: A Tool for the Study of Floral Morphogenesis, Function and Evolution. Bot. Rev. 1981, 47, 421–449.
- Kumar, S.; Rao, M.; Gupta, N.C. Breeding Strategies of Self Pollinated Crop with Special Emphasis on Hybrid Rice: Present and Future Perspectives. Res. Rev. J. Agric. Sci. Technol. 2014, 3, 2349–3682.
- Dicenta, F.; Ortega, E.; Canovas, J.A.; Egea, J. Self-pollination vs. cross-pollination in almond: Pollen tube growth, fruit set and fruit characteristics. Plant Breed. 2002, 121, 163–167.
- Winston, M.L. The Biology of the Honey Bee; Harvard University Press: Cambridge, MA, USA, 1991.
- Osterman, J.; Aizen, M.A.; Biesmeijer, J.C.; Bosch, J.; Howlett, B.G.; Inouye, D.W.; Jung, C.; Martins, D.J.; Medel, R.; Pauw, A. Global trends in the number and diversity of managed pollinator species. Agric. Ecosyst. Environ. 2021, 322, 107653.
- Zhigang, L.; Hongsong, R.; Guangyu, F.; Aihemaiti, M.; Hu Xidan, M.; Kurban, A.; Ruihua, W.; Hongmei, G.; Jian, W.; Haifeng, L. Effect of Bumblebee Pollination on Yield and Quality of Greenhouse Tomato; Jiangsu Academy of Agricultural Sciences: Nanjing, China, 2021.
- Hongdong, W.; Shuang, H.; Bing, H.; Mingfei, W.; Guanxiong, Z.; Donggang, L. Research progress of bumblebee pollination technology in protected agriculture. Yangtze River Veg. 2022, 8, 34–37.
- Joshi, N.C.; Joshi, P.C. Foraging Behaviour of Apis spp. on Apple Flowers in a Subtropical Environment. N. Y. Sci. J. 2010, 3, 71–76.
- Blazyte-Cereskiene, L.; Vaitkeviciene, G.; Venskutonyte, S.; Buda, V. Honey bee foraging in spring oilseed rape crops under high ambient temperature conditions. Zemdirb. Agric. 2010, 97, 61–70.
- Abrol, D.P. Diversity of pollinating insects visiting litchi flowers (Litchi chinensis Sonn.) and path analysis of environmental factors influencing foraging behaviour of four honeybee species. J. Apic. Res. 2006, 45, 180–187.
- Peat, J.; Goulson, D. Effects of experience and weather on foraging rate and pollen versus nectar collection in the bumblebee, Bombus terrestris. Behav. Ecol. Sociobiol. 2005, 58, 152–156.
- Kovac, H.; Stabentheiner, A. Thermoregulation of foraging honeybees on flowering plants: Seasonal variability and influence of radiative heat gain. Ecol. Entomol. 2011, 36, 686–699.
- Stabentheiner, A.; Kovac, H. Honeybee economics: Optimisation of foraging in a variable world. Sci. Rep. 2016, 6, 1–7.
- Heinrich, B. Mechanisms of body temperature regulation in honeybees, Apis mellifera. I. Regulation of Head Temperature. J. Exp. Biol. 1980, 85, 61–72.
- Heinrich, B. Mechanisms of body-temperature regulation in honeybees, Apis mellifera. II. Regulation of thoracic temperature at high air temperatures. J. Exp. Biol. 1980, 85, 73–87.
- Attar, I.; Farhat, A. Efficiency evaluation of a solar water heating system applied to the greenhouse climate. Sol. Energy 2015, 119, 212–224.
- Vadiee, A.; Martin, V. Thermal energy storage strategies for effective closed greenhouse design. Appl. Energy 2013, 109, 337–343.
- Ghoulem, M.; El Moueddeb, K.; Nehdi, E.; Boukhanouf, R.; Calautit, J.K. Greenhouse design and cooling technologies for sustainable food cultivation in hot climates: Review of current practice and future status. Biosyst. Eng. 2019, 183, 121–150.
- Zhou, B.; Lin, S.; Jing, S.U.; Xue, F.; Jiang, T. Effects of temperature on the developments of honeybee oosperms and queen pupae. J. Fujian Agric. Univ. 2002, 31, 511–513.
- Zhu, X.; Zhou, B.; Luo, Q.; Luo, F.; Zhong, X.; Shi, M. The effect of temperature on the development of worker honey bee during sealed brood period. New Agric. Technol. 2006, 7, 57–60.
- Tautz, J.; Maier, S.; Groh, C.; Rossler, W.; Brockmann, A. Behavioral performance in adult honey bees is influenced by the temperature experienced during their pupal development. Proc. Natl. Acad. Sci. USA 2003, 100, 7343–7347.
- Groh, C.; Tautz, J.; Rossler, W. Synaptic organization in the adult honey bee brain is influenced by brood-temperature control during pupal development. Proc. Natl. Acad. Sci. USA 2004, 101, 4268–4273.
- Becher, M.A.; Scharpenberg, H.; Moritz, R.F.A. Pupal developmental temperature and behavioral specialization of honeybee workers (Apis mellifera L.). J. Comp. Physiol. A-Neuroethol. Sens. Neural Behav. Physiol. 2009, 195, 673–679.
- Doull, K.M. The effects of different humidities on the hatching of the eggs of honeybees. Apidologie 1976, 7, 61–66.
- Woodrow, W.A. Some Effects of Relative Humidity on the Length of Life and Food Consumption of Honeybees. J. Econ. Entomol. 1935, 28, 565–568.
- Jain, R.; Brockmann, A. Sex-specific molecular specialization and activity rhythm-dependent gene expression in honey bee antennae. J. Exp. Biol. 2020, 223, jeb217406.
- Lacher, V. Elektrophysiologische Untersuchungen an einzelnen Rezeptoren für Geruch, Kohlendioxyd, Luftfeuchtigkeit und Tempratur auf den Antennen der Arbeitsbiene und der Drohne (Apis mellifica L.). Z. Vgl. Physiol. 1964, 48, 587–623.
- Koeniger, N.; Veith, H.J. Glyceryl-1,2-dioleate-3-palmitate, a brood pheromone of the honey bee (Apis mellifera L.). Experientia 1983, 39, 1051–1052.
- Bujok, B.; Kleinhenz, M.; Fuchs, S.; Tautz, J. Hot spots in the bee hive. Naturwissenschaften 2002, 89, 299–301.
- Kleinhenz, M.; Bujok, B.; Fuchs, S.; Tautz, H. Hot bees in empty broodnest cells: Heating from within. J. Exp. Biol. 2003, 206, 4217–4231.
- Kronenberg, F.; Heller, H.C. Colonial thermoregulation in honey bees (Apis mellifera). J. Comp. Physiol. B 1982, 148, 65–76.
- Southwick, E.E. HOGG. Temperature Control in Honey Bee Colonies. BioScience 1987, 37, 395–399.
- Schneider, C.W.; Tautz, J.; Gruenewald, B.; Fuchs, S. RFID Tracking of Sublethal Effects of Two Neonicotinoid Insecticides on the Foraging Behavior of Apis mellifera. PLoS ONE 2012, 7, e30023.
- Henry, M.; Beguin, M.; Requier, F.; Rollin, O.; Odoux, J.-F.; Aupinel, P.; Aptel, J.; Tchamitchian, S.; Decourtye, A. A Common Pesticide Decreases Foraging Success and Survival in Honey Bees. Science 2012, 336, 348–350.
- Campbell, J.M.; Dahn, D.C.; Ryan, D.A.J. Capacitance-based sensor for monitoring bees passing through a tunnel. Meas. Sci. Technol. 2005, 16, 2503–2510.
- Jiang, J.-A.; Wang, C.-H.; Chen, C.-H.; Liao, M.-S.; Su, Y.-L.; Chen, W.-S.; Huang, C.-P.; Yang, E.-C.; Chuang, C.-L. A WSN-based automatic monitoring system for the foraging behavior of honey bees and environmental factors of beehives. Comput. Electron. Agric. 2016, 123, 304–318.
- BeeHero. Available online: https://www.beehero.io/ (accessed on 10 August 2022).
- Huimin, W.; Chuzhou, T.; Ming, L.; Zhongqiu, L.; Zhen, H.; Mingliang, W.; Juying, H.; Haiqing, Z. Effect of airflow speed on pollens distribution for hybrid rice breeding pollination. Trans. Chin. Soc. Agric. Eng. (Trans. CSAE) 2012, 28, 63–69.
- Sedgley, M.; Griffin, A.R. Sexual Reproduction of Tree Crops; Academic Press: Cambridge, MA, USA, 2013.
- Pinillos, V.; Cuevas, J. Artificial pollination in tree crop production. Hortic. Rev. 2008, 34, 239–276.
- Shu, Q.; Liu, Z.; Zhang, J.; Yun, J. Effects of different pollinators on the pollination effect of kiwifruit. J. Zhejiang Agric. Sci. 2015, 56, 1416–1417.
- Jianye, C.; Zhanhong, L.; Yuxia, N. Preparation method and biological effects of pollen suspension liquid forspraying pollination of Actinidia chinensis. J. Fruit Sci. 2014, 31, 1105–1109.
- Cerović, R.; Fotirić Akšić, M.; Meland, M. Success Rate of Individual Pollinizers for the Pear Cultivars “Ingeborg” and “Celina” in a Nordic Climate. Agronomy 2020, 10, 970.
- Santiago, J.P.; Sharkey, T.D. Pollen development at high temperature and role of carbon and nitrogen metabolites. Plant Cell Environ. 2019, 42, 2759–2775.
- Yi, W.; Law, S.E.; McCoy, D.; Wetzstein, H.Y. Stigma development and receptivity in almond (Prunus dulcis). Ann. Bot. 2006, 97, 57–63.
- Mahamad, H.K.; Ali, I. Effects of fungicides on pollen germination peach and nectarine in vitro. Afr. J. Plant Sci. 2011, 5, 643–647.
- Ascari, L.; Guastella, D.; Sigwebela, M.; Engelbrecht, G.; Stubbs, O.; Hills, D.; De Gregorio, T.; Siniscalco, C. Artificial pollination on hazelnut in South Africa: Preliminary data and perspectives. In Proceedings of the IX International Congress on Hazelnut 1226, Samsun, Turkey, 15–18 August 2017; pp. 141–148.
- Parker, A.J.; Tran, J.L.; Ison, J.L.; Bai, J.D.K.; Weis, A.E.; Thomson, J.D. Pollen packing affects the function of pollen on corbiculate bees but not non-corbiculate bees. Arthropod-Plant Interact. 2015, 9, 197–203.
- Wizenberg, S.B.; Weis, A.E.; Campbell, L.G. Comparing methods for controlled capture and quantification of pollen in Cannabis sativa. Appl. Plant Sci. 2020, 8, e11389.
- Vaknin, Y.; Gan-Mor, S.; Bechar, A.; Ronen, B.; Eisikowitch, D. Effects of desiccation and dilution on germinability of almond pollen. J. Hortic. Sci. Biotechnol. 1999, 74, 321–327.
- Buitink, J.; Claessens, M.M.; Hemminga, M.A.; Hoekstra, F.A. Influence of water content and temperature on molecular mobility and intracellular glasses in seeds and pollen. Plant Physiol. 1998, 118, 531–541.
- Payne, W.W. Structure and function in angiosperm pollen wall evolution. Rev. Palaeobot. Palynol. 1981, 35, 39–59.
- Jiayu, H.; Yigang, Z.; Yuanping, C.; Chunqing, W.; Youjin, L. A Strawberry Artificial Pollinator. No. ZL201920082178.3, 24 September 2019.
- Shuo, W.; Jizhan, L.; Jiangshan, W.; Meng, H. A Kind of Pneumatic Strawberry Pollinating Robot in Slightly Raised Greenhouse and Its Realization Method. No. CN109588305A, 9 April 2019.
